Disease Modifying Treatments Joseph R Berger M D

Disease Modifying Treatments Joseph R. Berger, M. D. Professor of Neurology Chief of the MS Division Perelman School of Medicine University of Pennsylvania

What is MS? • A chronic disease that affects the central nervous system (CNS-brain and spinal cord) • MS is thought to be immune mediated – The Immune system becomes activated and incorrectly targets the CNS • Associated with relapses and remissions – Relapses: new or returning neurological symptoms that last at least 24 hours and are not associated with another cause such as a fever. • Worsening or progression of disability is possible over time • The cause is unknown but research strongly suggests that certain factors increase the risk of developing MS
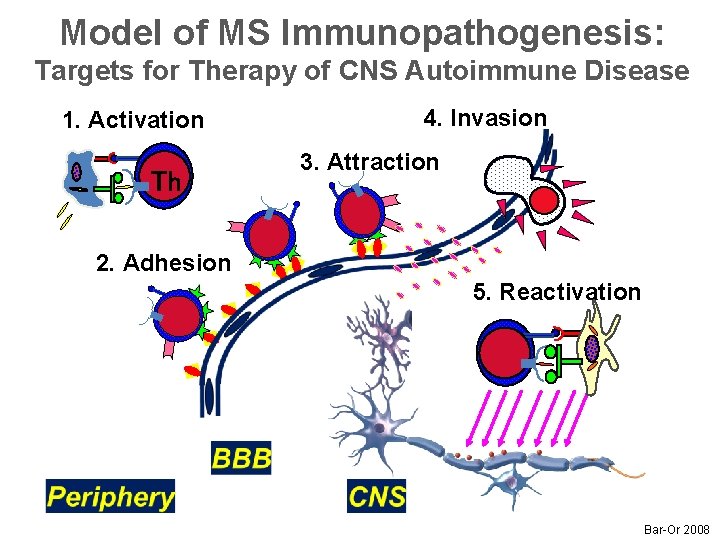
Model of MS Immunopathogenesis: Targets for Therapy of CNS Autoimmune Disease 1. Activation Th 4. Invasion 3. Attraction 2. Adhesion 5. Reactivation Bar-Or 2008
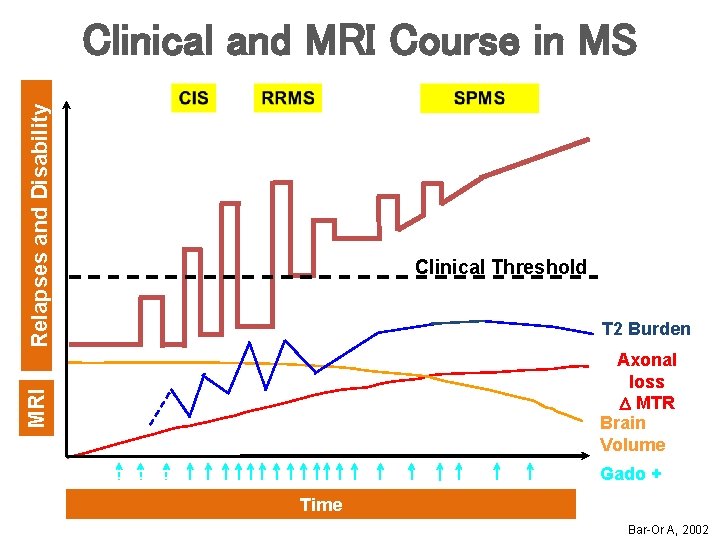
Relapses and Disability Clinical and MRI Course in MS Clinical Threshold T 2 Burden MRI Axonal loss D MTR Brain Volume Gado + Time Bar-Or A, 2002
Evolution of MS lesions over time
Brain Atrophy (Shrinkage) in Untreated MS
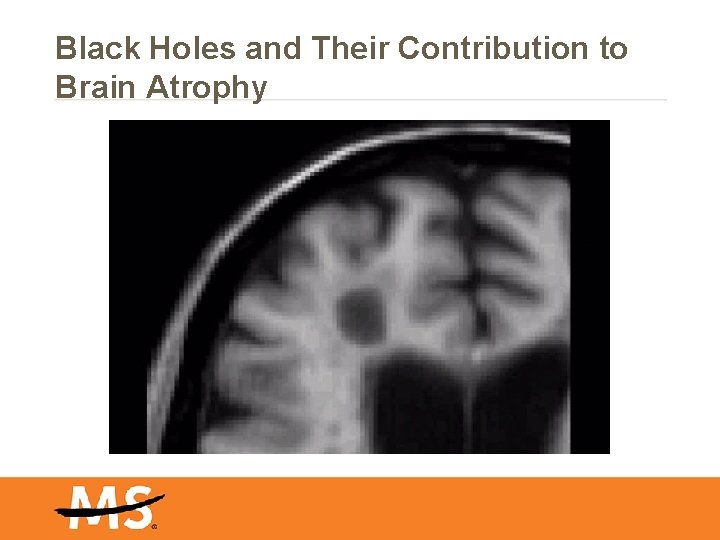
Black Holes and Their Contribution to Brain Atrophy

Possible MS Risk factors • • Genetic susceptibility Smoking Childhood and adolescent obesity Low Vitamin D History of Epstein Barr infection Other close family members with MS Living in a temperate climate However, having these risk factors does not guarantee that a person will develop MS

MS Epidemiology - Genetics • Family history as a risk factor • Higher rates if index case is female • Relationship – General population 0. 1% – Sibling 2. 0% (20 X) – Fraternal twin 2 -12% – Identical twin 21 -70% – Child of MS patient 1. 0% – Aunt/Uncle 2. 0% – Niece/Nephew 0. 4% – Conjugal rare
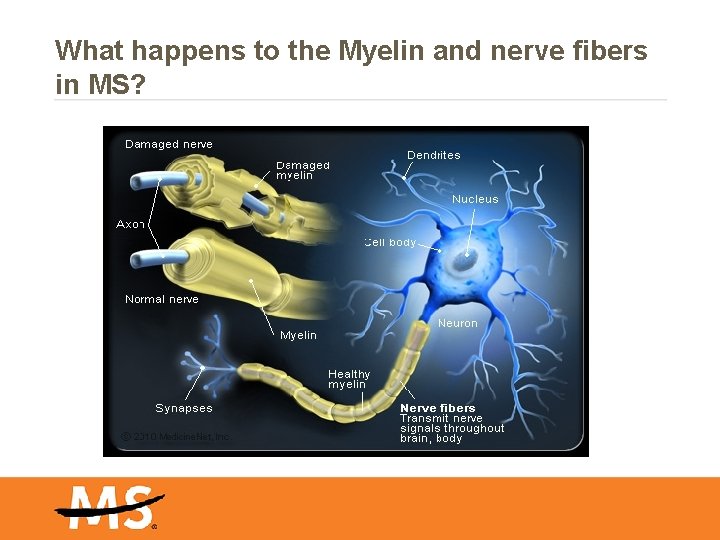
What happens to the Myelin and nerve fibers in MS?

What Causes MS? Exact cause is not known, but is thought to be a complex interplay of genetic predisposition and environmental factors Genetic Predisposition Environmental Trigger Immune mediated response Inflammation/Damage/destruction of myelin and axons

Who gets MS? • Usually diagnosed between 20 and 50 – Less frequently diagnosed in young children and older adults • More common in women than men (3: 1) • More common in temperate areas (further from the equator) although in some recent studies the geographic gradient is becoming less apparent.
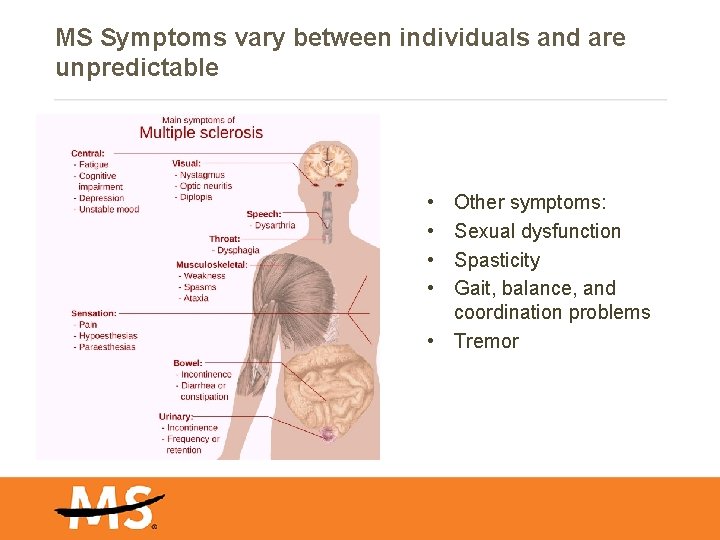
MS Symptoms vary between individuals and are unpredictable • • Other symptoms: Sexual dysfunction Spasticity Gait, balance, and coordination problems • Tremor

How is MS diagnosed? • MS is a clinical diagnosis: – Medical history of symptoms referable to the CNS – Neurological findings on examination – Laboratory tests: MRI, lumbar puncture, serum analysis, evoked potentials • Requires dissemination in time and space – Space: Objective evidence of lesions, typical of MS, in two or more distinct areas of the CNS – Time: Two clinical events that occurred at different points in time – No better explanation for the symptoms and findings
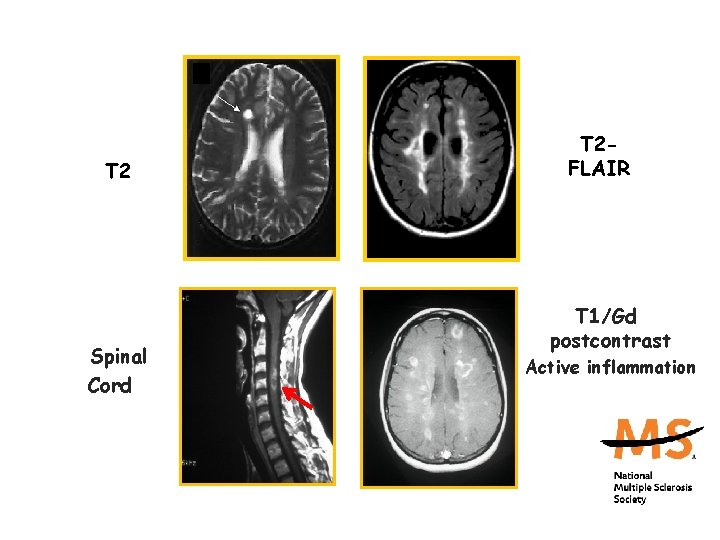
MRI Findings in MS T 2 Spinal Cord T 2 FLAIR T 1/Gd postcontrast Active inflammation

Goals of MS Management • • Optimal treatment of relapses Effective symptom management Disease modification Ability to live well with MS

Who is on the MS “Management Team”? • • Neurologist Urologist Nurse Advanced Practice Nurse Physician Assistant Physical therapist Occupational therapist Speech/language pathologist • • • Physiatrist Psychotherapist Neuropsychologist Social worker/Care manager • Primary care physician • Pharmacist
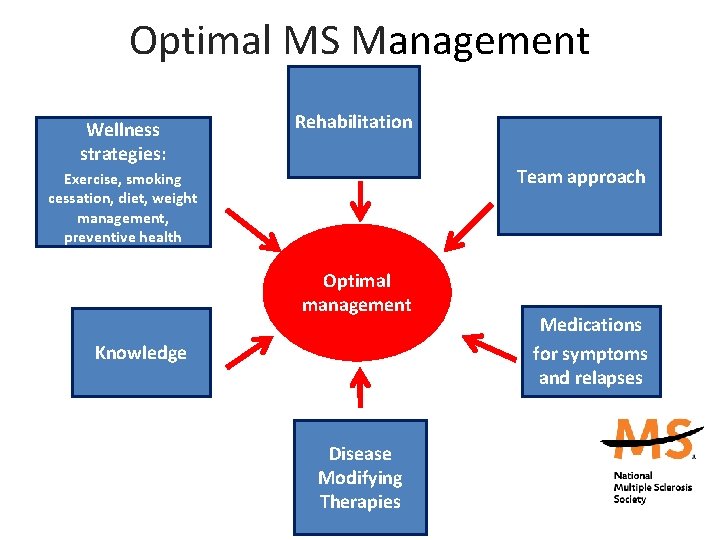
Optimal MS Management Wellness strategies: Rehabilitation Team approach Exercise, smoking cessation, diet, weight management, preventive health Optimal management Knowledge Disease Modifying Therapies Medications for symptoms and relapses

How are relapses treated? • Not all relapses require treatment – Symptoms that interfere with function (e. g. , visual or motor) are usually treated • High dose IV glucocorticoids are treatment of choice for acute relapses – Length of treatment varies – High-dose oral steroids are used by some neurologists • When needed, rehabilitation to help restore/maintain function

Association for Research in Nervous and Mental Disease December 10 th-11 th, 1948 Robert Foster Kennedy One should no more tell our patients they have multiple sclerosis than we should tell them they have inoperable cancer…When I have to make the diagnosis of multiple sclerosis, I make it to the relations, not to the patient; and I try to defend the patient from hearing the name because once the name is heard, it is vested with lamentable result. Robert Foster Kennedy, M. D.

Association for Research in Nervous and Mental Disease December 10 th-11 th, 1948 And I certainly agree with him that the diagnosis should never be told to the patient unless it is absolutely essential to that patient for the arrangement of his life, and that the facts should be explained to the family. H. Houston Merritt, M. D.
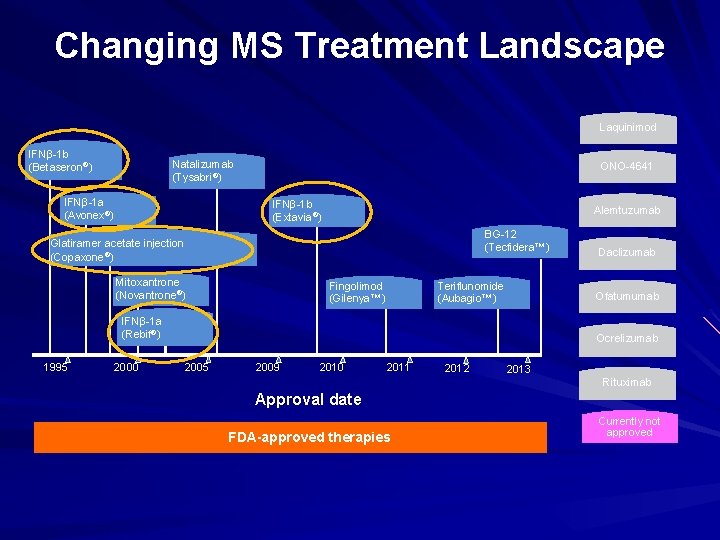
Changing MS Treatment Landscape Laquinimod IFNβ-1 b (Betaseron®) Natalizumab (Tysabri®) IFNβ-1 a (Avonex®) ONO-4641 IFNβ-1 b (Extavia®) Alemtuzumab BG-12 (Tecfidera™) Glatiramer acetate injection (Copaxone®) Mitoxantrone (Novantrone®) Teriflunomide (Aubagio™) Fingolimod (Gilenya™) IFNβ-1 a (Rebif®) 1995 2000 Daclizumab Ofatumumab Ocrelizumab 2005 2009 2010 2011 2012 2013 Rituximab Approval date FDA-approved therapies Currently not approved
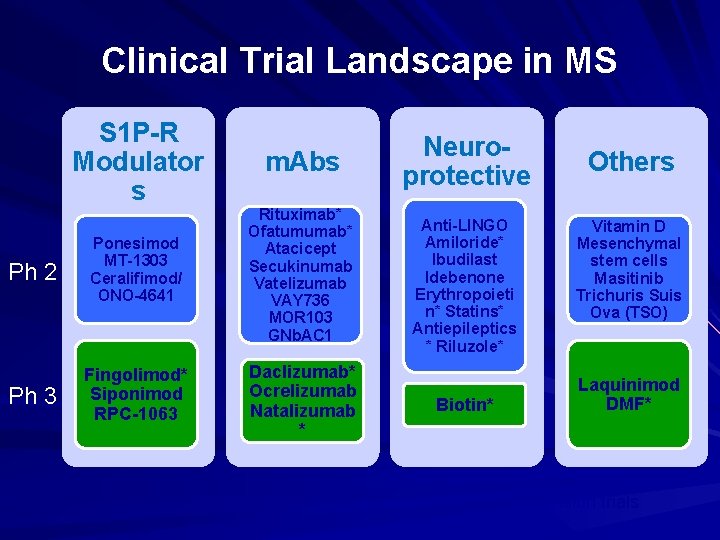
Clinical Trial Landscape in MS S 1 P-R Modulator s m. Abs Ph 2 Ponesimod MT-1303 Ceralifimod/ ONO-4641 Rituximab* Ofatumumab* Atacicept Secukinumab Vatelizumab VAY 736 MOR 103 GNb. AC 1 Ph 3 Fingolimod* Siponimod RPC-1063 Daclizumab* Ocrelizumab Natalizumab * Neuroprotective Anti-LINGO Amiloride* Ibudilast Idebenone Erythropoieti n* Statins* Antiepileptics * Riluzole* Biotin* Others Vitamin D Mesenchymal stem cells Masitinib Trichuris Suis Ova (TSO) Laquinimod DMF* * Extension trials
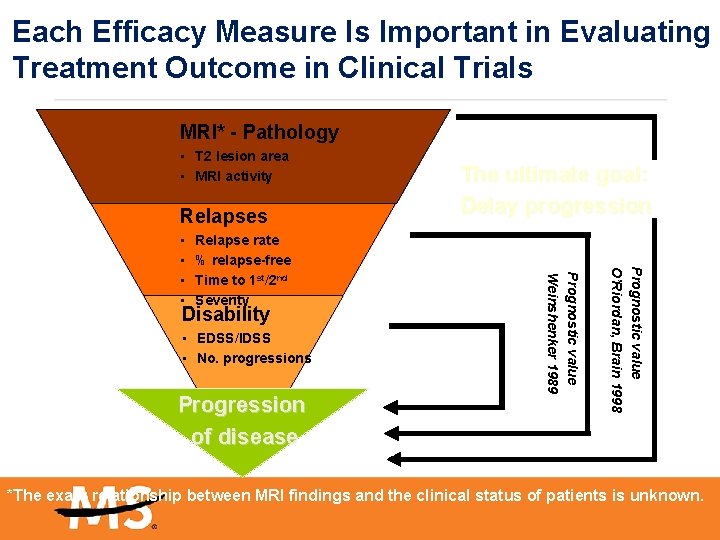
Each Efficacy Measure Is Important in Evaluating Treatment Outcome in Clinical Trials MRI* - Pathology • T 2 lesion area • MRI activity Relapses Disability • EDSS/IDSS • No. progressions Progression of disease Prognostic value O’Riordan, Brain 1998 Relapse rate % relapse-free Time to 1 st/2 nd Severity Prognostic value Weinshenker 1989 • • The ultimate goal: Delay progression *The exact relationship between MRI findings and the clinical status of patients is unknown.

How important is early treatment? • The MS Coalition recommends that treatment with a disease modifying agent initiated as early as CIS confirmed. – Irreversible axonal damage occurs even in the earliest stages of the illness. – Tx is most effective when started early when inflammation is most likely to occur • No treatment has been approved for primary-progressive MS. Approximately 60% of people with MS are on a DMT
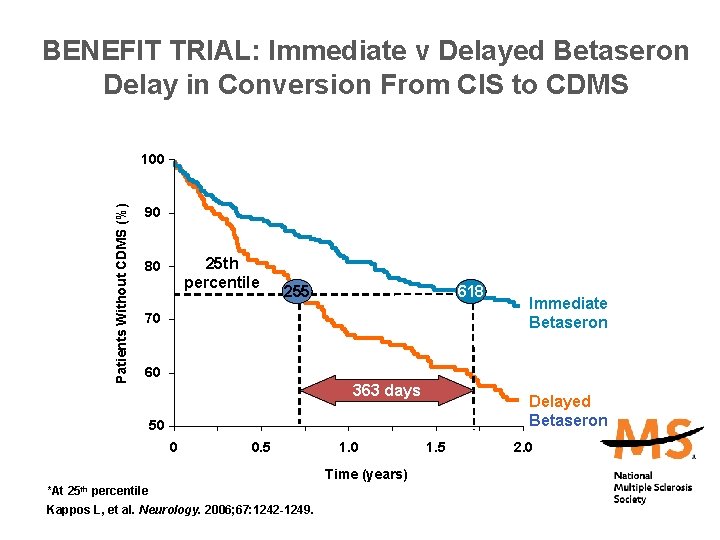
BENEFIT TRIAL: Immediate v Delayed Betaseron Delay in Conversion From CIS to CDMS Patients Without CDMS (%) 100 90 25 th percentile 80 255 618 70 60 363 days Delayed Betaseron 50 0 0. 5 1. 0 Time (years) *At 25 th Immediate Betaseron percentile Kappos L, et al. Neurology. 2006; 67: 1242 -1249. 1. 5 2. 0
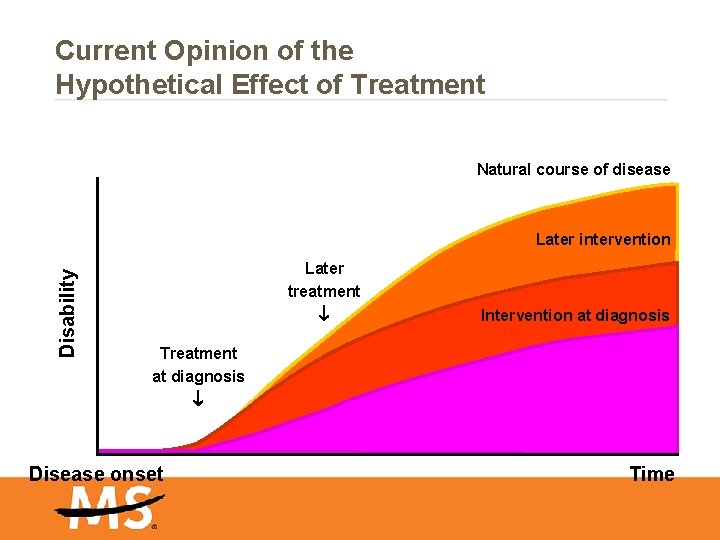
Current Opinion of the Hypothetical Effect of Treatment Natural course of disease Disability Later intervention Later treatment Intervention at diagnosis Treatment at diagnosis Disease onset Time

Disease Modifying Treatments (DMT’s) • Modify the inflammatory immune activity – Reduce the number of new relapses – Delay worsening (progression) – Limit new areas of damage in the CNS • 12 FDA approved DMT’s – 6 injectable treatments – 3 oral treatments – 3 infusion treatments • 11 are approved for relapsing forms of MS; • Novantrone is approved for secondary progressive MS and worsening relapsing MS • Lemtrada is recommended for those who have had insufficient benefit from at least two other medications. • Certain interferons and Copaxone are approved for people who have had one neurological event suggestive of MS

Injectable DMT’s • Interferon beta’s – Betaseron – Extavia Interferon beta -1 b – Rebif – Avonex Interferon beta -1 a – Plegridy Pegylated Interferon beta -1 a

Interferon beta-1 a and 1 b • Reduce the ability of certain immune system cells from becoming activated • Reduce the ability of certain immune system cells from entering the CNS • Reduces relapse rate • Reduce new CNS inflammation • Delay progression

Interferon Administration • Betaseron and Extavia – Subcutaneous injection (under the skin) every other day • Rebif – Subcutaneous injection given three times/week • Avonex – Intramuscularly (into a large muscle) once weekly • Plegridty – Subcutaneous injection once every 2 weeks • Auto – injector devices are available for each of the interferon DMT’s

Interferon Side Effects Side effects are similar among the interferons – Flu-like symptoms following injection, which lessen over time for many. – Injection site reactions. (subcutaneous injections) – Less common: • Allergic reactions • Depression • Liver abnormalities • Low white blood cell counts. • Seizure • Heart problems

Interferon beta Warnings/Precautions • Individuals with a history of depression or a seizure disorder should be closely monitored while on this medication • This medication should be used with caution in people with depression • Rare but significant allergic reactions have been reported with this medications • Skin infections or areas of severe skin damage can occur with subcutaneous injections; injection sites should be rotated on a regular basis • People with cardiac problems should be monitored closely • Blood counts and liver function should be monitored

Copaxone – glatiramer acetates • A mixture of 4 protein building blocks (amino acids) • Copaxone shifts the immune system response to one that is less inflammatory • Reduces relapse rate • Reduces new CNS inflammation • 2 formulations – 20 mg given daily by subcutaneous injection – 40 mg given three times per week by subcutaneous injection

Copaxone Side Effects • Injection site reactions • Less common: – Immediate post injection reaction that can occur immediately following the injection ands lasts 15 -30 minutes. This may consist of: • Vasodilation (dilation of blood vessels • Chest pain • Heart palpitations • Shortness of breath, • Flushing

Copaxone Warnings/Precautions • Immediate post-injection reaction – Generally occurs after the first few months of treatment and may occur more than once in a given individual. • vasodilation (dilation of blood vessels), chest pain, heart palpitations, shortness of breath, flushing • Transient chest pain — without any long-term effects — may also occur one or more times, either as part of the post -injection reaction or separately. • Permanent depressions under the skin at injection sites can occur because of destruction of the fatty tissue. – In addition, areas of severe skin damage can occur. – Careful rotation of injection sites is recommended so that no single area is injected more than one time per week.

Oral DMT’s • Gilenya (fingolimod) – 0. 5 mg capsule taken once daily • Aubagio (teriflunomide) – 7 mg or 14 mg tablets taken once daily • Tecfidera (dimethyl fumarate) – 240 mg capsule taken twice daily

Gilenya • Restricts certain immune system cells called lymphocytes from exiting the lymph nodes and thus keeps them out of the bloodstream. • Reduces relapse rate • Delays worsening • Limits new inflammation in the CNS

Gilenya Side Effects • • Headache Flu Diarrhea Back pain Liver enzyme elevations Cough Less common – Slowed heart rate following first dose (requires monitoring for 6 or more hours after the first dose – Infections – Macular edema

Gilenya Warnings • Slowed heart rate for several hours after the first dose: – All patients should have an electrocardiogram (ECG) prior to the first dose, – Monitoring required for six hours after the first dose with hourly pulse and blood pressure measurement, and then given a repeat ECG. • Reduction in the number of white blood cell • Possible respiratory difficulties • A vision test is recommended prior to starting treatment and about 3 months later • Possible liver enzyme abnormalities • Potential for reactivation of herpes viruses including VZV • One case of PML

Aubagio • Reduces the number of white blood cells – some which are considered to cause MS inflammation • Reduces relapse rate • Delays worsening • Reduces new CNS inflammation

Aubagio Side Effects • • • Hair thinning (tends to be temporary) Diarrhea Flu Nausea Abnormal liver tests (must be monitored closely) Unusual numbness or tingling in the hands or feet (paresthesias). • Less common: lowered levels of white blood cells, which can increase the risk of infections; increase in blood pressure; severe liver damage

Aubagio Warnings • Aubagio can cause liver damage. – Blood tests are needed on a regular basis including before and monthly after starting treatment. • Aubagio can cause major birth defects for up to two years after the medication is discontinued. – Pregnancy test before starting the medication – Use of effective birth control while taking Aubagio – If pregnancy occurs while on Aubagio, stop taking Aubagio immediately and undergo treatment to remove the medication rapidly from the body. – Men who plan to father a child should stop taking the medication and undergo treatment to remove the medication rapidly from the body before they and their partners try to conceive.

Aubagio Warnings/Precautions • Aubagio can increase a person’s risk of infections. – Complete blood count prior to starting treatment – Monitor for infection while on treatment. – TB test before starting treatment. – If positive TB test, TB treatment before taking Aubagio – Aubagio can cause damage to peripheral – Aubagio can cause acute kidney failure and elevated potassium in the blood. • Kidney function should be monitored in anyone who experiences symptoms of renal failure or elevated potassium levels. • Aubagio can cause elevations in blood pressure; blood pressure should monitored and managed during treatment.

Tecfidera • Shifts the immune system to a less inflammatory response • May reduce oxidative stress, which is known to cause damage in MS • Reduces relapse rate • Delays worsening • Reduces new CNS inflammation

Tecfidera Side Effects • Flushing (sensation of heat or itching and a blush on the skin) • Gastrointestinal issues (nausea, diarrhea, abdominal pain) • Rash • Protein in the urine • Elevated liver enzymes • Reduction in blood lymphocyte (white blood cell) counts.

Tecfidera Warnings/Precautions • Tecfidera® (dimethyl fumarate) can cause a reduction in blood lymphocyte (white blood cell) counts. – Check blood cell count before starting Tecfidera – A CBC including lymphocyte count should also be obtained after 6 months of treatment, every 6 to 12 months thereafter, and as clinically indicated. • PML (progressive multifocal leukoencephalopathy – Occurred in one person who had been on Tecfidera for 4 years and a very low white blood cell count • Severe allergic reactions – Difficulty breathing, hives and itching, swelling of the throat and tongue.

Infused DMT’s • Tysabri (natalizumab) – 300 mg intravenously (IV) every 4 weeks • Novantrone (mitoxantrone) – 12 mg/m 2 IV every 3 months (with a lifetime maximum dose of 140 mg/m 2 • Lemtrada (alemtuzumab) – 12 mg IV daily on 5 consecutive days and then 12 mg IV on three consecutive days one year later.

Tysabri • Blocks lymphocytes (a type of white blood cell) from entering the CNS • Reduces relapse rate • Delays disability • Reduces new CNS inflammation

Tysabri Warnings • Tysabri must be infused in an approved infusion center. • PML (progressive multifocal leukoencephalopathy), which is caused by the common JC virus. PML is a serious CNS infection that can cause significant disability and may cause death • Liver damage • Herpes infections

Novantrone • An immunosuppressant medication that suppresses the production of white blood cells • Delays worsening • Reduces relapse rate • Reduces new CNS inflammation

Novantrone Warnings/Precautions • The total lifetime dose is limited in order to avoid possible heart damage. • People taking Novantrone should have tests of their heart function before each dose and periodically after treatment has ended. • It cannot be used in people with pre-existing heart problems, liver disease, and certain blood disorders. • Acute myelogenous leukemia (AML), a type of cancer

Lemtrada • Depletes the numbers of certain immune system cells that a molecule on their cell surface known as CD 52 • Reduces relapses rate • Delays worsening • Reduces new CNS inflammation

Lemtrada Warnings/Precautions • ~25% develop autoimmune conditions, e. g, immune thrombocytopenia (ITP, a rare bleeding condition) and antiglomerular basement membrane disease (which impacts the kidneys) • Possible serious and life-threatening infusion reactions (while the medication is being given and for 24 hours after each infusion) • Increased risk of malignancies (thyroid cancer, melanoma, and blood cancers). • A person with an active infection should not start treatment until the infection is controlled. Because of the risks associated with Lemtrada, this treatment is only available from certified prescribers and pharmacies, and people taking the medication, as well as the healthcare facility administering the medication, must be enrolled
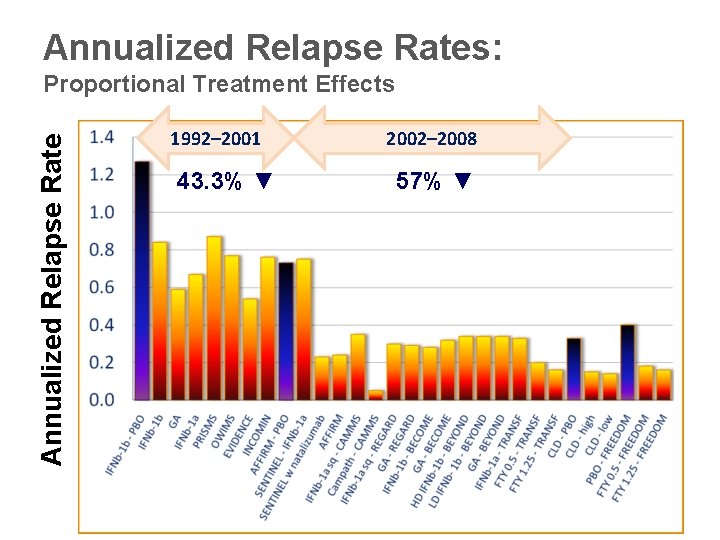
Annualized Relapse Rates: Annualized Relapse Rate Proportional Treatment Effects 1992– 2001 43. 3% ▼ 2002– 2008 57% ▼

Summary • The FDA has approved 12 medications for the treatment of relapsing MS • All treatments have been found to be effective • Starting treatment early in the disease process has been found to be most effective • DMT’s should be continued unless there is continued MS activity or intolerable side effects • Stopping and/or switching a DMT should occur only for medical reasons and after consultation with your neurology provider

More Research Needed
- Slides: 57