BY NICOLE STEVENS HYPOXIC ISCHAEMIC ENCEPHALOPATHY HIE HIE

BY: NICOLE STEVENS HYPOXIC ISCHAEMIC ENCEPHALOPATHY (HIE)

HIE � � � Definition & Diagnosis Etiology & Risk Factors Incidence Pathophysiology Diagnosis Assessment Multisystem involvement Hypothermia management Nursing management Outcomes Case scenarios & Questions

DEFINITIONS Hypoxia or Anoxia: A partial (hypoxia) or complete (anoxia) lack of oxygen in the brain or blood � Asphyxia: The state in which placental or pulmonary gas exchange is compromised or ceases altogether � Ischemia: The reduction or cessation of bloodflow to an organ which compromises both oxygen and substrate delivery to the tissue � Hypoxic-Ischemic Encephalopathy: Abnormal neurologic behavior in the neonatal period arising as a result of a hypoxic-ischemic event. �
ETIOLOGY OF HIE Maternal: Cardiac arrest Asphyxiation Severe anaphylaxis Status epilepticus Hypovolemic shock Uteroplacental: Placental abruption Cord prolapse Uterine rupture Hyperstimulation with oxytocic agents Fetal: Fetomaternal hemorrhage Twin to twin transfusion Severe isoimmune hemolytic disease Cardiac arrhythmia

INCIDENCE OF HIE � Occurs in 1 -6 per 1000 live term births in developed countries � 25% die or have multiple disabilities � 4% have mild to moderate forms of cerebral palsy � 10% have developmental delay (this is similar to the control population!)
PATHOPHYSIOLOGY � Gestational age plays an important role in the susceptibility of CNS structures � � � < 20 weeks: Insult leads to neuronal heterotopia or polymicrogyria 26 -36 weeks: Insult affects white matter, leading to periventricular leukomalacia Term: Insult affects primarily gray matter
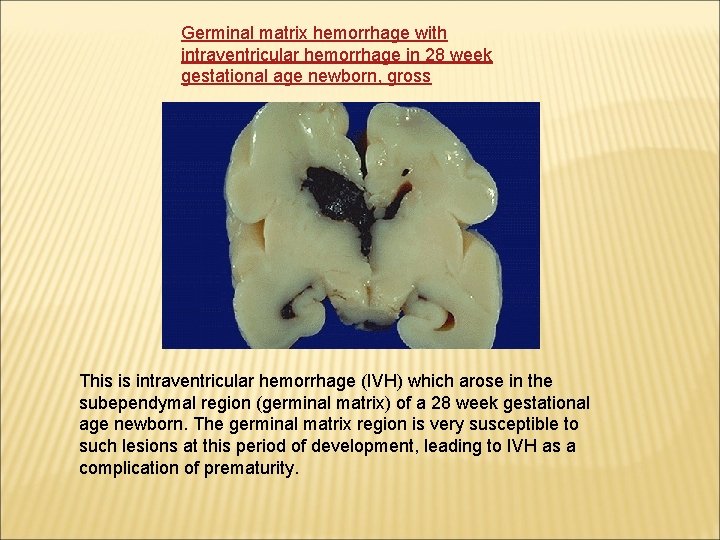
Germinal matrix hemorrhage with intraventricular hemorrhage in 28 week gestational age newborn, gross This is intraventricular hemorrhage (IVH) which arose in the subependymal region (germinal matrix) of a 28 week gestational age newborn. The germinal matrix region is very susceptible to such lesions at this period of development, leading to IVH as a complication of prematurity.

PATHOPHYSIOLOGY � Acute HIE leads to primary and secondary events: Primary neuronal damage: cytotoxic changes due to failure of microcirculation inhibition of energyproducing molecular processes ATPase membrane pump failure cytotoxic edema and free radical formation compromised cellular integrity � Secondary neuronal damage: May extend up to 72 hours or more after the acute insult and results in an inflammatory response and cell necrosis or apoptosis (fueled by reperfusion) �

DIAGNOSIS There is no clear diagnostic test for HIE � Abnormal findings on the neurologic exam in the first few days after birth is the single most useful predictor that brain insult has occurred in the perinatal period � � Essential Criteria for Diagnosis of HIE: Metabolic acidosis (cord p. H <7 or base deficit of >12) � Early onset of encephalopathy � Multisystem organ dysfunction �

CLINICAL STAGING OF HIE (SARNAT AND SARNAT, 1976) HIE can be divided into Mild, Moderate, and Severe

ASSESSMENT TOOLS IN HIE � Amplitude-integrated EEG (a. EEG) � When performed early, it may reflect dysfunction rather than permanent injury � Most useful in infants who have moderate to severe encephalopathy � Marginally abnormal or normal a. EEG is very reassuring of good outcome � Severely abnormal a. EEG in infants with moderate HIE raises the probability of death or severe disability from 25% to 75%

ASSESSMENT TOOLS IN HIE � Neuroimaging Cranial ultrasound: Not the best in assessing abnormalities in term infants. Echogenicity develops gradually over days � CT: Less sensitive than MRI for detecting changes in the central gray nuclei � MRI: Most appropriate technique and is able to show different patterns of injury. Presence of signal abnormality in the internal capsule later in the first week has a very high predictive value for neurodevelopmental outcome �

CNS COMPLICATIONS � Most widely reported and best known sequelae � Major findings include: intracranial haemorrhage, cerebral infarction, cerebral oedema & gross brain swelling � Clinical manifestations: abnormal states of consciousness and tone, full or tense anterior fontanel, irritability, tremors, convulsions and poor suck

Cerebrum, coronal section, acute watershed infarct, gross The bilaterally symmetric dark discolored areas seen superiorly and just lateral to the midline represent recent infarction in the watershed zone between anterior and middle cerebral arterial circulations. Such watershed infarctions can occur with relative or absolute hypoperfusion of the brain.
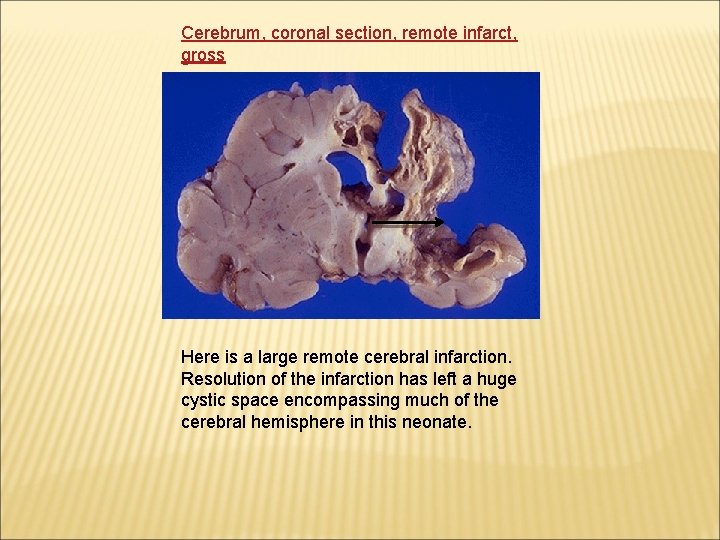
Cerebrum, coronal section, remote infarct, gross Here is a large remote cerebral infarction. Resolution of the infarction has left a huge cystic space encompassing much of the cerebral hemisphere in this neonate.
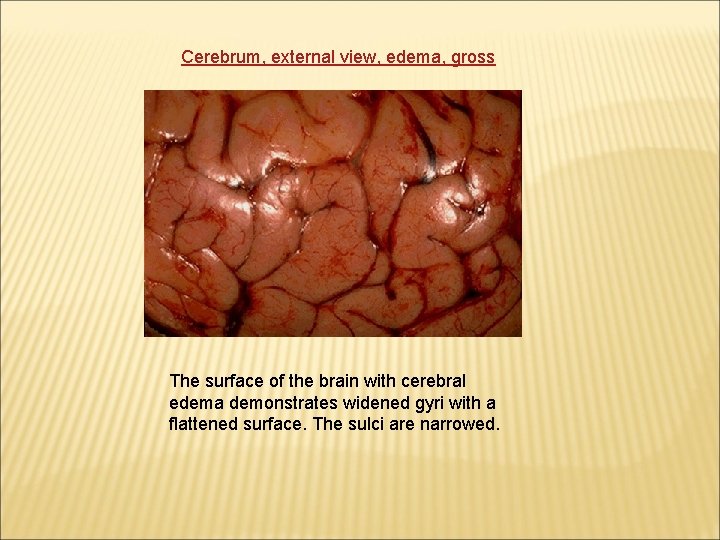
Cerebrum, external view, edema, gross The surface of the brain with cerebral edema demonstrates widened gyri with a flattened surface. The sulci are narrowed.

CNS COMPLICATIONS � Management/monitoring: monitor systemic blood pressure, intracranial pressure (by feel of fontanel), and structural changes/damage (usually MRI) � Pharmacologically management: anticonvulsants (phenobarbitone, phenytoin); sedation, especially if ventilated and cooled (morphine, midazolam) � Prevent fluid overloading

CARDIOVASCULAR & RENAL COMPLICATIONS � Hypovolaemia, hypotension, shock and renal failure are the most serious sequelae of HIE � Need to monitor: urine output (weigh nappies or measure hrly if a catheter is insitu), monitor electolytes, urea & creatinine � Have to find the balance between adequate hydration for renal function but not fluid overloading (somewhere between 60 – 150 m. Ls/kg/day)

CARDIOVASCULAR AND RENAL COMPLICATIONS � Hypovolaemia & hypotension may require management with volume expanders, blood transfusion and/or inotropes (improve cardiac contractility and cardiac output; therefore good for improving renal function as well) � If there is hypovoleamia/hypotension the bodies response is to vasoconstrict (peripheral and splanchnic), hence, there is poor perfusion to gut and kidneys. Shock response/maintain function of vital organs

PULMONARY COMPLICATIONS � Provide respiratory support if required (CPAP or ventilation) � Monitor respiratory status by observation and blood gases � Treat underlying pathology, ie. Sepsis (antibiotics), meconium aspiration/PPHN (ventilation/nitric oxide), poor or no respiratory drive due to CNS complications (ventilation)

METABOLIC COMPLICATIONS Monitor and manage sequelae such as: � Metabolic acidosis (ventilate and use sodium bicarbonate corrections) � Hypoglycaemia (initial infusion of 10% dextrose, but more concentrated solutions may be required if fluid restriction is required) � Hypocalcaemia (calcium corrections) � Hyponatraemia: dilutional or actual? (Fluid restrict for dilutional or replace Na if losing it through the kidneys) � Monitor blood pressure, iatrogenic blood loss (from mulitiple tests) and haematocrit level (will give you an indication of degree of haemodilution or concentration)

ICE (INFANT COOLING EVALUATION) TRIAL � Conducted throughout 28 centres in Australia, New Zealand, Canada and the USA (2001 2007) � Cooling is started within 6 hrs of birth � Cooling can be started at the hospital of birth and continued throughout transport, by using simple equipment (gel ice packs) � The aim is to lower the brain temperature to protect it from damage � This treatment could prevent as many as 1 in 7 from dying or surviving with a significant disability

MANAGEMENT - HYPOTHERMIA � Aim for body temperature 33 – 34 degrees � Treatment continues for 72 hrs � Babies are rewarmed gradually over a 12 – 24 hr period after this time � Supportive management continues throughout treatment � EEG monitoring is performed continuously � Blood tests are performed to monitor electrolytes, liver function, renal function etc

HYPOTHERMIA - MECHANISM OF ACTION Reduces cerebral metabolism, prevents edema � Decreases energy utilization � Reduces/suppresses cytotoxic amino acid accumulation and nitric oxide � Inhibits platelet-activating factor, inflammatory cascade � Suppresses free radical activity � Attenuates secondary neuronal damage � Inhibits cell death � Reduces extent of brain damage � � DEATH OR SEVERE DISABILITY AT 18 MONTHS OF AGE SIGNIFICANTLY REDUCED!!

CRITERIA FOR HYPOTHERMIA � Generally only used on term infants � Must be commenced within 6 hrs � Infants must meet at least 2 of following criteria: 1. 2. 3. � Apgar score of 5 or less at 10 minutes Mechanical ventilation or resuscitation still required at 10 mins Cord or arterial p. H of <7, or base deficit of 12 or more within 60 minutes of birth Core goal temperature of 33 – 34 degrees C for 72 hrs

OUTCOMES � The location and extent of damage, and the immediate medical management, will determine the short and long term outcomes � It is possible to have no long term complications as a result of a brain injury at birth � But complications that can occur are delayed, or failure to reach, milestones (as determined by developmental assessments); cerebral palsy; death

NURSING MANAGEMENT � � � � Initial resuscitation Transfer to SCN/NICU for ongoing management & observation Baseline observations Consider need for cooling (discuss with paediatricians/NETS) Observe for seizure activity Intravenous access & bloods to be taken Medications & fluids as ordered Measure urine output (m. Ls/kg/hr): weigh nappy, divide weight of urine by weight of baby and no. of hrs since last measured. Eg: 20 m. Ls/3. 3 kgs/4 hrs = 1. 5 m. Ls/kg/hr

CASE SCENARIO � 32 year old G 2 P 1 woman whose last baby was delivered via LUSCS for breech presentation, but who wished to attempt a vaginal birth for this pregnancy. The woman, who is at term and had been in labor for the past 4 hours, suddenly complained of excruciating abdominal pain, became hypotensive, and fetal heart rate was undetectable. An EM LUSCS is performed under GA.

CASE SCENARIO � What is the differential diagnosis? � What are the implications of the maternal condition on the fetus? � What management might you anticipate being required in the initial resuscitation of this newborn

CASE SCENARIO � The baby is delivered via LUSCS and is brought to the resuscitaire. Initial assessment reveals an unresponsive floppy infant with no respiratory effort and a heart rate of 80 bpm. Resuscitation is undertaken. Subsequent Apgar scores are 1, 4, and 7 at 1, 5 and 10 minutes, respectively. The baby is transferred to the SCN for further care.

CASE SCENARIO � What complications should you expect from this delivery and what is the underlying pathophysiology? � What diagnostic and prognostic studies should be done in the NICU? � What treatments are available for this baby and what are the criteria to treat?

QUESTIONS 1. The mother of a baby with suspected HIE inquires about the possibility of a brain insult in her infant. Of the following, the single most useful predictor of brain insult in this infant is the evidence of: A. Abnormal neurologic exam findings B. Cerebral edema on cranial US C. Elevated creatinine phosphokinase D. Hemodynamic and pulmonary imbalance E. Multisystem organ dysfunction
QUESTIONS 1. The mother of a baby with suspected HIE inquires about the possibility of a brain insult in her infant. Of the following, the single most useful predictor of brain insult in this infant is the evidence of: A. Abnormal neurologic exam findings B. Cerebral edema on cranial US C. Elevated creatinine phosphokinase D. Hemodynamic and pulmonary imbalance E. Multisystem organ dysfunction

QUESTIONS 2. The severity of HIE can be graded as mild, moderate, or severe, using a classification proposed by Sarnat and Sarnat. Of the following, the criterion most consistent with the diagnosis of mild HIE is: A. Absence of seizures B. Low Apgar scores C. Need for assisted ventilation D. Proximal muscle weakness E. Obtunded state of consciousness

QUESTIONS 2. The severity of HIE can be graded as mild, moderate, or severe, using a classification proposed by Sarnat and Sarnat. Of the following, the criterion most consistent with the diagnosis of mild HIE is: A. Absence of seizures B. Low Apgar scores C. Need for assisted ventilation D. Proximal muscle weakness E. Obtunded state of consciousness

QUESTIONS 3. Several ancillary tests have been proposed to improve the prediction of long-term outcome of infants who have suffered from HIE. Of the following, the most useful and practical test for determining the prognosis of HIE is: A. Cranial ultrasound B. MRI C. EEG D. Near-infrared spectroscopy E. Somatosensory evoked potentials

QUESTIONS 3. Several ancillary tests have been proposed to improve the prediction of long-term outcome of infants who have suffered from HIE. Of the following, the most useful and practical test for determining the prognosis of HIE is: A. Cranial ultrasound B. MRI C. EEG D. Near-infrared spectroscopy E. Somatosensory evoked potentials

REFERENCES � � � Allan WC. The clinical spectrum and prediction of outcome in hypoxic-ischemic encephalopathy. Neoreviews 2002; 3; e 108 -e 115 Delivoria-Papadopoulos M, et al. Biochemical basis of hypoxicischemic encephalopathy. Neoreviews 2010; 11; e 184 -e 193 Fanaroff and Martin’s Neonatal-Perinatal Medicine: Diseases of the Fetus and Infant, 9 th edition. 2011, p 952 -976 Marro, PJ, et al. Pharmacology review: Neuroprotective treatments for hypoxic-ischemic injury. Neoreviews 2010; 11; e 311 -e 315 Shankaran S. Neonatal encephalopathy: Treatment with hypothermia. Neoreviews 2010; 11; e 85 -e 92 Fomufod, A. K. , White, P. L Perinatal asphyxia II: Clinical management of multisystemic sequelae. Journal of the National Medical Association (1979), Vol. 71, No. 11 pg 1065 -1066
- Slides: 38