Hepatic Physiology Assessment and interpretation of liver function









































- Slides: 60

Hepatic Physiology & Assessment and interpretation of liver function tests Dr. M Sabra

• Liver is the largest internal organ & largest gland in the human body. • Liver is at the epicenter of intermediary metabolism , performs versatile & massive biochemical pathways and destroys bacteria, inactivate antigens, detoxify harmful chemicals. • Dual blood supply: Portal vein = 75% of HBF (55% of 02) , Hepatic artery = 25% of HBF (45% of 02) – Total hepatic blood flow = 25% of CO • Surgery and anesthesia impact hepatic function primarily due to their impact on hepatic blood flow and not primarily as a result of the medications or

Physiological functions of liver • Intermediary metabolism Carbohydrate metabolism Bile metabolsim • • Coagulation Heme metabolism Bilirubin metabolism Xenobiotics metabolism ( a foreign chemical • • Storage Endocrine functions Immune & inflammatory response Blood reservoir Lipid metabolism Protein metabolism substance found within an oraganism , not normally naturally produced by organism. Eg drugs antibiotics

Carbohydrate metabolism • Liver is an important homeostatic regulator of blood glucose. It can either produce glucose or store glucose • In fed state- polymerize glucose to glycogen • In unfed state- depolymerize glycogen to glucose • Glucose → hepatocytes → glycogen ↑ →glucose Lactate Glycerol aminoacids

Carbohydrate metabolism • Glycogen metabolism • Regulation – 2 rate limiting enzymes 1. Glycogen synthase- synthesis of glycogen from monomers of UDP glucose. 2. Glycogen phosphorylase- clevage of glycogen to glucose-1 -phosphate.

Carbohydrate metabolism Gluconeogenesis • Liver glycogen stores depleted - hepatic gluconeogenesis to replenish blood glucose. • Substrates- lactate - glycerol from hydrolysis of triglycerides - gluconeogenic amino acid , alanine , glutamine

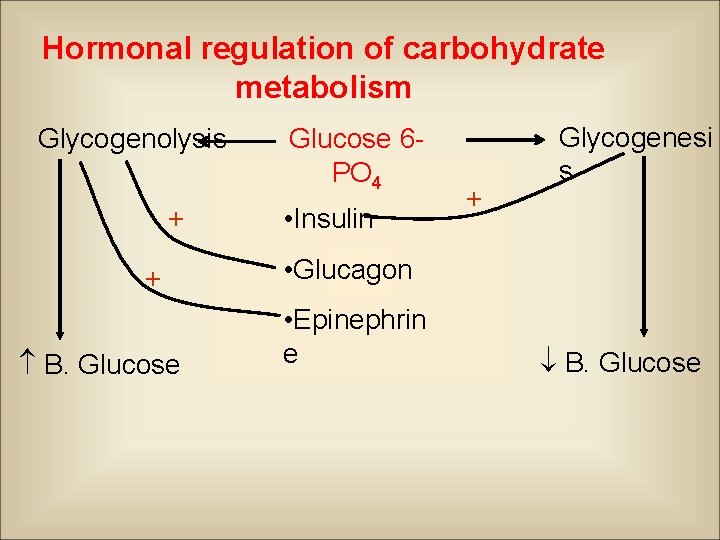
Hormonal regulation of carbohydrate metabolism Glycogenolysis + + B. Glucose 6 PO 4 • Insulin + Glycogenesi s • Glucagon • Epinephrin e B. Glucose

• Blood glucose regulation within a narrow limit (70100 mg/dl) not affected in liver disease due to large reserve of hepatic function q Effects of anaesthesia on carbohydrate metabolism • Halothane – release of insulin – rate of glycogenolysis – Inhibition of gluconeogenic response • Isoflurane – Impaired insulin secretion

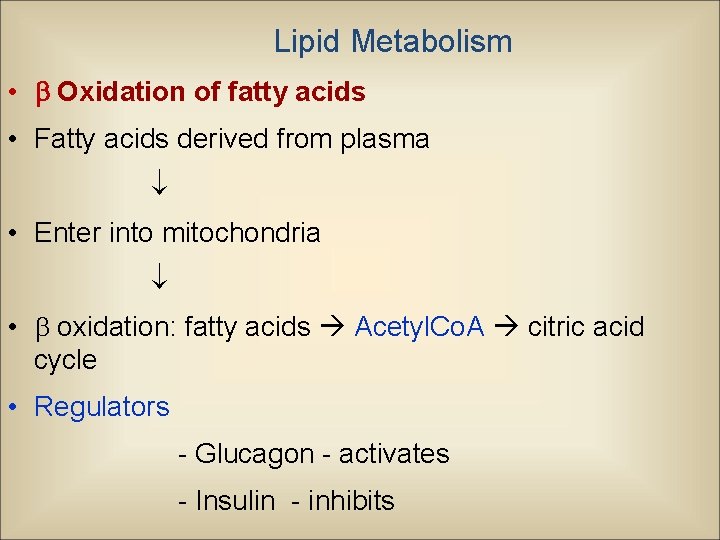
Lipid Metabolism • Oxidation of fatty acids • Fatty acids derived from plasma • Enter into mitochondria • oxidation: fatty acids Acetyl. Co. A citric acid cycle • Regulators - Glucagon - activates - Insulin - inhibits

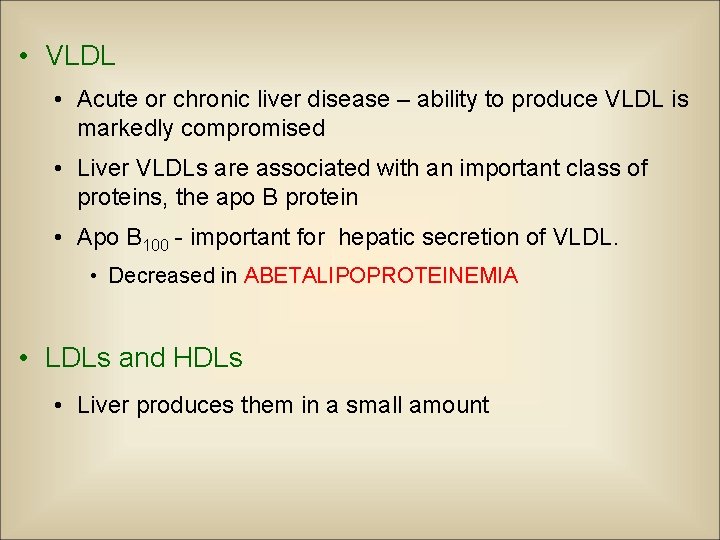
• Synthesis of lipoproteins • One of the major functions of the liver • Major classes – VLDL (Very low density lipoproteins ) – LDL ( low density lipoproteins ) – HDL (High density lipoproteins)

• VLDL • Acute or chronic liver disease – ability to produce VLDL is markedly compromised • Liver VLDLs are associated with an important class of proteins, the apo B protein • Apo B 100 - important for hepatic secretion of VLDL. • Decreased in ABETALIPOPROTEINEMIA • LDLs and HDLs • Liver produces them in a small amount

• Production of ketone bodies • Most organs except the liver- use ketone bodies as fuel • Ketone bodies – acetoacetic acid, acetone, hydroxybutyrate • Their formation by the liver is normal and physiologically important, e. g. – Fasting rapid depletion of glycogen stores in the liver shortage of substrates for citric acid cycle – Acetyl. Co. A formed from oxidation ketone bodies • Ketosis - conc. of ketone bodies in blood – Starvation – DM – After high fat diet

• Synthesis of cholesterol • Important role in cholesterol homeostasis • Liver cholesterol has both exogenous and endogenous source Ø Uses of hepatic cholesterol • Formation of bile acids- conjugated with other substances to form cholic acid. • Synthesis of VLDLs

Bile metabolism & enterohepatic circulation • Bile salts are end products of cholesterol synthesis • Daily production – 600 - 800 ml/d • Functions- activate lipase - promote micelle formation - intestinal uptake of fat soluble vitamins, cholesterol & lipids - facilitate excretion of xenobiotics, lipophillic substances, bilirubin, amphipathic steroid hormone

• Bile salts undergo enterohepatic circulation (20 -30 times/day) intrahepatic bile duct ↓ common hepatic duct cystic duct ↓ gall bladder CBD ↓ small intestine ( terminal ileum) • Clinical implication – Opioids can induce spasm of bile duct & spinter of oddi – Reversed by – glucagon, opioid antagonists ( naloxone), smooth muscle relaxant (NTG),

Protein and amino acid metabolism • Deamination of amino acids – Required before they can be used for energy or before they can be converted into carbohydrates or fats • Formation of urea for removal of ammonia from the body fluids • Production of proteins and peptides.

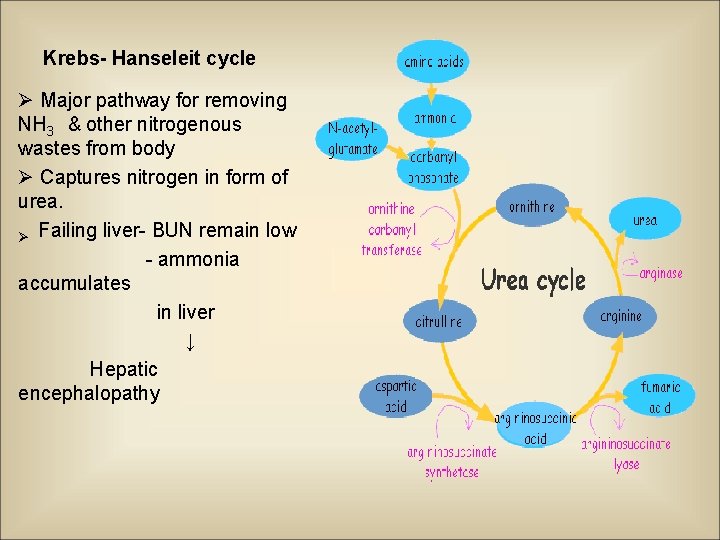
Krebs- Hanseleit cycle Ø Major pathway for removing NH 3 & other nitrogenous wastes from body Ø Captures nitrogen in form of urea. Ø Failing liver- BUN remain low - ammonia accumulates in liver ↓ Hepatic encephalopathy

Proteins & peptides Albumin • Most abundant protein • Normal plasma conc- 3 - 5 g% • Daily production -12 -15 g/d • Plasma half life – 15 -20 days • Functions – • maintains plasma oncotic pressure (80% by albumin) • binds ions, bilirubin, hormones & drugs • Hypoalbuminemia – Colloid oncotic pressure edema

ᾳ- feto protein • Resembles albumin genetically & functionally • Formation sites- yolksac, hepatocytes, enterocytes • Fetal & neonatal life- major determinant of plasma oncotic pressure • 1 year of age- albumin largely replaces AFP • ↑ ↑ AFP- HCC

Fibrinogen • Synthesized exclusively by hepatocytes • Plasma fibrinogen – 100 -700 mg/dl • Functions – polymerizes into long fibrin threads by the action of thrombin formation of clot

• Haptoglobins – Forms stable complexes with free Hb prevents loss of iron through urinary excretion, protects kidney from damage • Ceruloplasmin – binds with copper and helps in its transport and storage • Wilson’s disease – Deficiency of ceruloplasmin free Cu 2+ in circulation deposited in brain and liver

Coagulation • Synthesize most of the procoagulants excepta. factor III ( tissue thromboplastin) b. Factor IV ( calcium) c. Factor VIII ( von Willebrand factor ) • Produce protein regulators of coagulation & fibrinolytic pathways – Protein C, protein S ( protein C – inactivate F VIIIa- Va complex) – – protein Z ( degradation of Factor Xa ) plasminogen activator inhibitor (PAI) ( inhibits tissue plasminogen activators to convert plasminogen to plasmin ) – antithrombin III

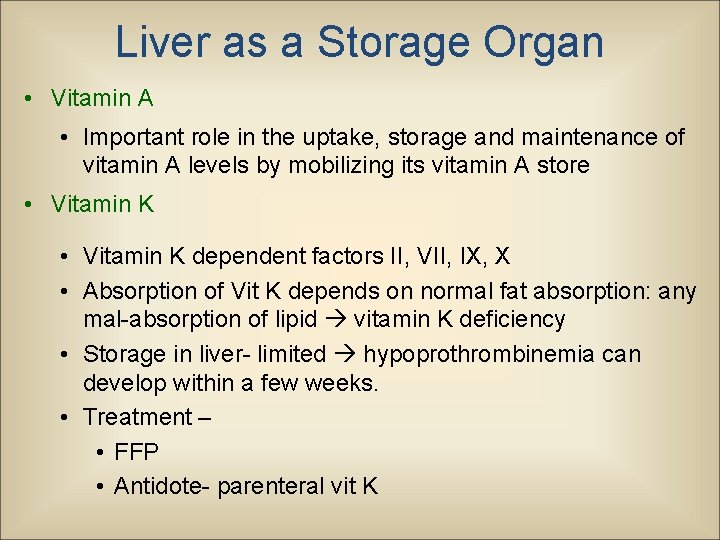
Liver as a Storage Organ • Vitamin A • Important role in the uptake, storage and maintenance of vitamin A levels by mobilizing its vitamin A store • Vitamin K dependent factors II, VII, IX, X • Absorption of Vit K depends on normal fat absorption: any mal-absorption of lipid vitamin K deficiency • Storage in liver- limited hypoprothrombinemia can develop within a few weeks. • Treatment – • FFP • Antidote- parenteral vit K

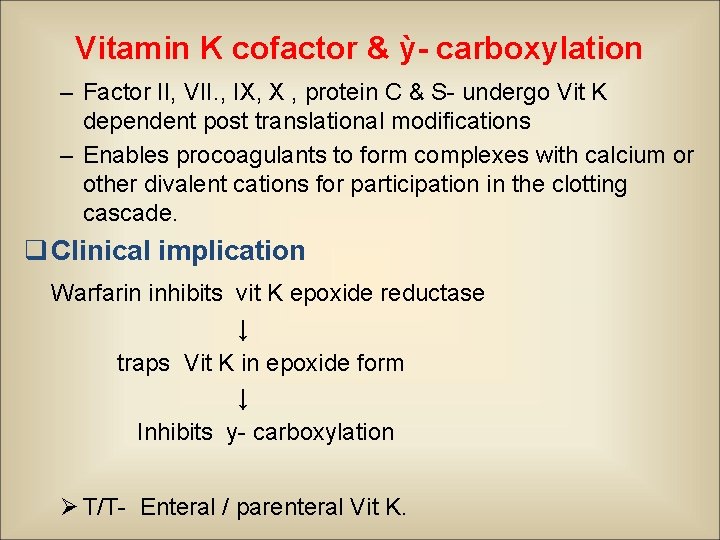
Vitamin K cofactor & ỳ- carboxylation – Factor II, VII. , IX, X , protein C & S- undergo Vit K dependent post translational modifications – Enables procoagulants to form complexes with calcium or other divalent cations for participation in the clotting cascade. q Clinical implication Warfarin inhibits vit K epoxide reductase ↓ traps Vit K in epoxide form ↓ Inhibits y- carboxylation Ø T/T- Enteral / parenteral Vit K.

Storage & Homeostasis of Iron • Major site of synthesis of proteins (Transferrin, Ferritin) involved in iron transport & metabolism. q Heme metabolism Clinical implication • Porphyrias • Acute Intermittent Porphyria – commonest – Defects in the heme pathway- accumulation of porphyrinogens – Trigger substances- barbiturates, sex hormones, glucocorticoides, cigarette smoke, CYP inducers.

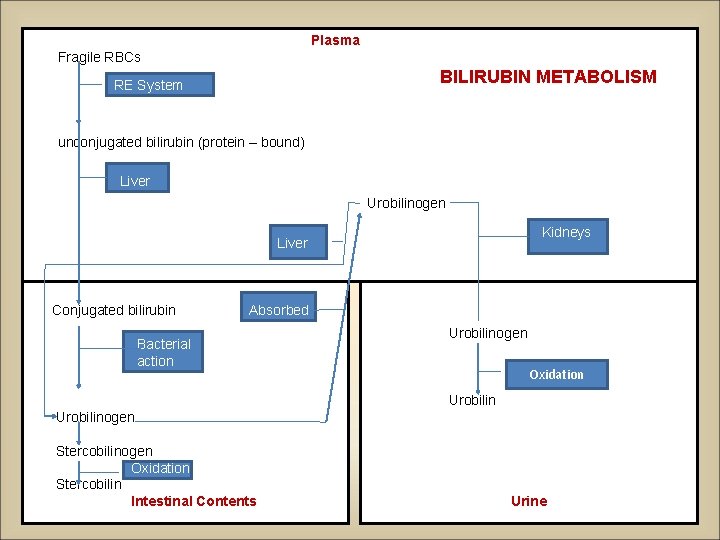
Bilirubin Metabolism • Main source of bilirubin is heme metabolism • Daily production- 300 mg • 80 % derived from senescent erythrocytes by macrophages in RE system. • Heme ↓ ( heme oxygenase + o 2 ) biliverdin IX + CO + free divalent iron ↓ ( biliverdin reductase) bilirubin

Plasma Fragile RBCs BILIRUBIN METABOLISM RE System unconjugated bilirubin (protein – bound) Liver Urobilinogen Kidneys Liver Conjugated bilirubin Absorbed Bacterial action Urobilinogen Oxidation Urobilinogen Stercobilinogen Oxidation Stercobilin Intestinal Contents Urine

• • • CO produced has many physiological roles Vasodilation ( regulation of vascular tone) Platelet aggregation Vascular myocyte proliferation Neurotransmitter release Cytoprotective , antiapoptotic, antioxidant effects • Biliverdin – confers protection from oxidative effects • rapidly converts to bilirubin

Metabolism of Drugs (Xenobiotics) Factor affecting drug metabolism: microsomal enzyme system liver blood flow route of administration competitive inhibition Ø Phase-I reactions • Alter the parent drug by inserting or unmasking a polar group • Converts drugs to more polar compounds • Reactions – oxidations, reduction, hydrolysis • Cytochrome P 450 – substrate binding site, located in the endoplasmic reticulum • Drugs– barbiturates, benzodiazepines, halogenated volatile anaesthetics, pethidine etc.

• Phase-II reactions • Creates conjugates of parent compound or its metabolite with endogenous hydrophilic substrate Ø Reactions Glucoronidation Methylation Sulphation Acetylation q Glucoronidation • Most common type • Hepatic microsomal enzyme, UDPglucuronyl transferase mediates the transfer of glucoronic acid from UDP glucuronic acid to the functional group on the xenobiotics • Drug handled by phase-II – morphine, propofol, thiopentone (initially oxidized subsequently conjugated)

Ø Phase-I reaction enzymes – more susceptible to destruction in cirrhosis Ø Phase-II reactions enzymes – more resistant, function even in advanced liver disease q Phase-III reactions • Involves ATP-binding cassette transport proteins (ABC) • These proteins use the energy of ATP hydrolysis to drive molecular transport • Dysfunction of ABC proteins hinders flow of bile predisposing to drug accumulation and cholestatic liver injury

Microsomal enzyme induction • Anticonvulsants, rifampicin, isoniazid, glucocorticoids, chronic alcohol consumption Consequences of enzyme induction § duration of action of drugs that are inactivated by metabolism § intensity of action of drugs that are activated by metabolism

Endocrine functions • Liver can modify or amplify hormone action • Metabolic conversion of Vitamin D to form 25(OH)D • 25(OH)D 1, 25(OH)2 D in kidney • Peripheral conversion of T 4 to T 3 Ø Pseudocholinesterase • Hydrolysis of succinylcholine • Plasma t½ - 14 days • Severe liver disease duration of action of succinylcholine

• Insulin-like growth factors or somatomedins – growth hormone like action • Important role in cartilage function by promoting uptake of sulphate and synthesis of collagen Ø Removes circulating hormones • Insulin, glucagon, growth hormone, gastrointestinal hormones, e. g. gastrin

• Blood reservoir – Liver is an expandable organ – 10 -15 % of total blood volume can be sequestered and quickly released after sympathetic stimulation. • Immune & inflammatory responses – kuffer cells protect against foreign intrutions, degrade toxins, process antigens, and phagocytose bacteria. – Induce & intensify inflammation by recruiting neutrophils – Release proinflammatory mediators

Liver Function Tests • Uses • To detect the presence of liver disease • To distinguish among different type of liver disorders • To guage the extent of known liver damage • To follow the response to treatment • Shortcomings of LFTs Can be normal in pts with serious liver disease and abnormal in pts with diseases that do not affect the liver Only categorises into hepatocellular or cholestatic

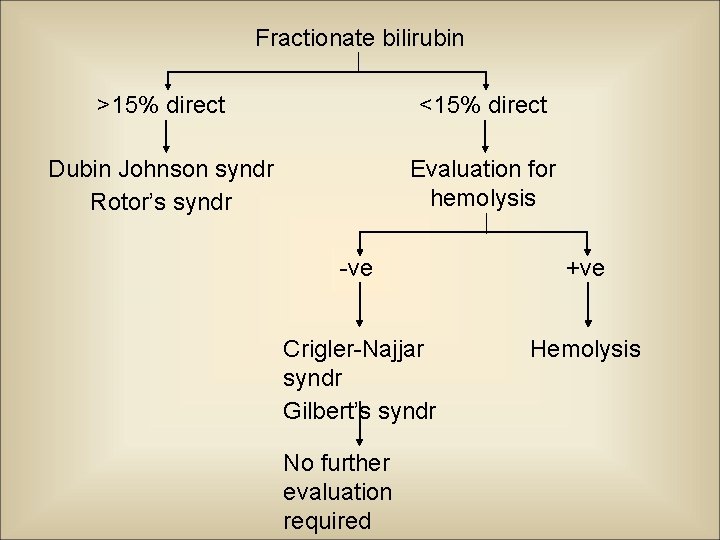
• Classification of LFTs q Tests based on detoxification and excretory functions • Serum bilirubin • Breakdown product of porphyrin ring of heme containing proteins • 2 fractions - conjugated (direct – 30%) unconjugated (indirect – 70%) • Normal total serum bilirubin 1 mg/dl • in unconjugated fraction is rarely due to liver disease

Fractionate bilirubin >15% direct <15% direct Dubin Johnson syndr Rotor’s syndr Evaluation for hemolysis -ve +ve Crigler-Najjar syndr Gilbert’s syndr Hemolysis No further evaluation required

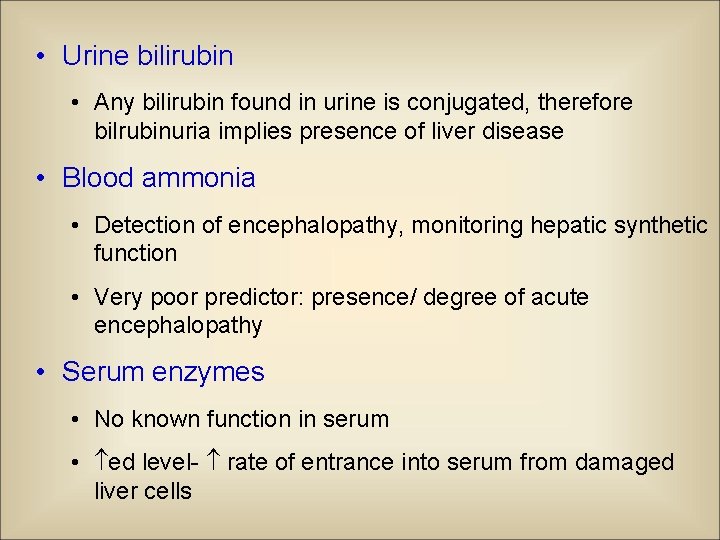
• Urine bilirubin • Any bilirubin found in urine is conjugated, therefore bilrubinuria implies presence of liver disease • Blood ammonia • Detection of encephalopathy, monitoring hepatic synthetic function • Very poor predictor: presence/ degree of acute encephalopathy • Serum enzymes • No known function in serum • ed level- rate of entrance into serum from damaged liver cells

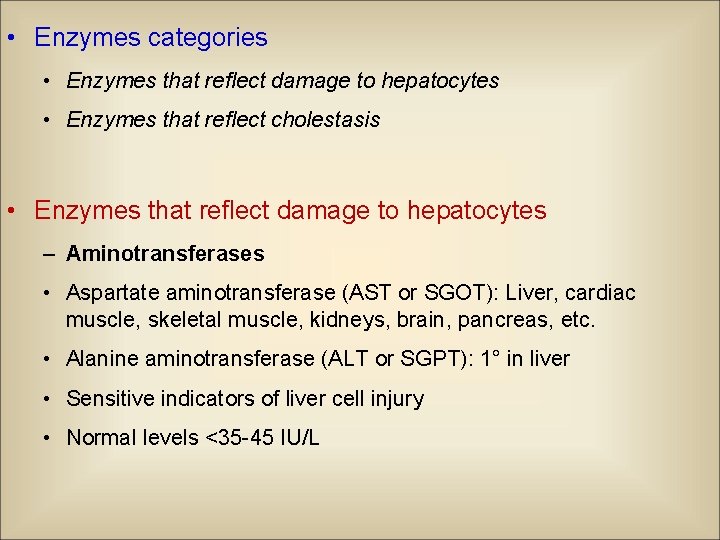
• Enzymes categories • Enzymes that reflect damage to hepatocytes • Enzymes that reflect cholestasis • Enzymes that reflect damage to hepatocytes – Aminotransferases • Aspartate aminotransferase (AST or SGOT): Liver, cardiac muscle, skeletal muscle, kidneys, brain, pancreas, etc. • Alanine aminotransferase (ALT or SGPT): 1° in liver • Sensitive indicators of liver cell injury • Normal levels <35 -45 IU/L

• in aminotransferases • Mild - <250 IU/l – Any pathologic process that causes hepatocellular injury, e. g. hepatic steatosis, alcohol or drug induced liver disease, chronic viral hepatitis, cirrhosis, hemachromatosis • Moderate – 250 -1000 IU/l – Disorder that produces hepatocellular necrosis – e. g. Acute viral hepatitis, drug induced hepatitis, exacerbation of chronic hepatitis (alcoholic) • Large - >1000 IU/l – Viral or drug induced liver damage superimposed on ALD, autoimmune hepatitis

• Extreme - >2000 IU/l • Massive hepatic necrosis, usually from drugs (acetaminophan), halothane hepatitis, toxins, ischemic hepatitis (shock liver), acute viral hepatitis • AST/ALT ratio – DERITIS QUOTIENT – Normal - 1 or slightly > 1 • <1 – non-alcoholic steatosis or hepatitis without cirrhosis • 2 -4 – ALD • >4 – Wilsonian hepatitis AST/ ALT not ↑ in purely obstructive disorder except Acute biliary obstruction caused by passage of gallstones to CBD

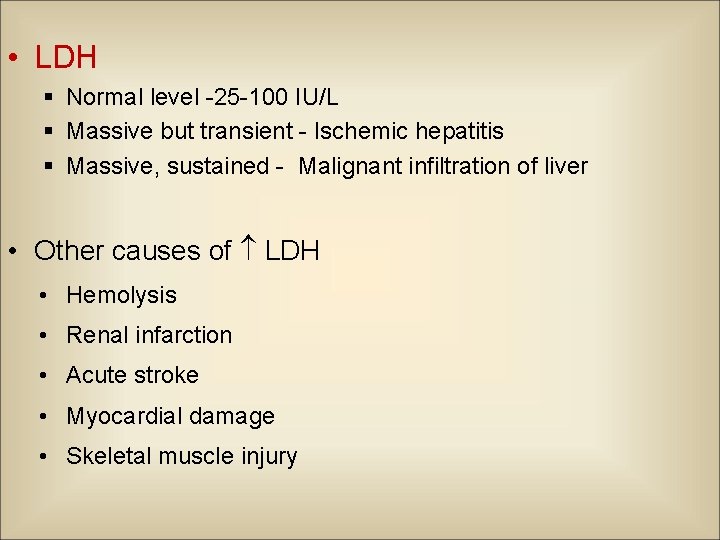
• LDH § Normal level -25 -100 IU/L § Massive but transient - Ischemic hepatitis § Massive, sustained - Malignant infiltration of liver • Other causes of LDH • Hemolysis • Renal infarction • Acute stroke • Myocardial damage • Skeletal muscle injury

• Glutathione S transferase • Relatively sensitive and specific test for detecting druginduced hepatocellular injury • Plasma t½ 90 min, rapidly released into the circulation following hepatocellular injury • Plasma GST ( isoenzyme B ) – reveal time course of hepatocellular injury from onset to resolution • GST – located in the centrilobular region (zone 3), where hepatocytes are most susceptible to injuries from hypoxia and reactive drug metabolism

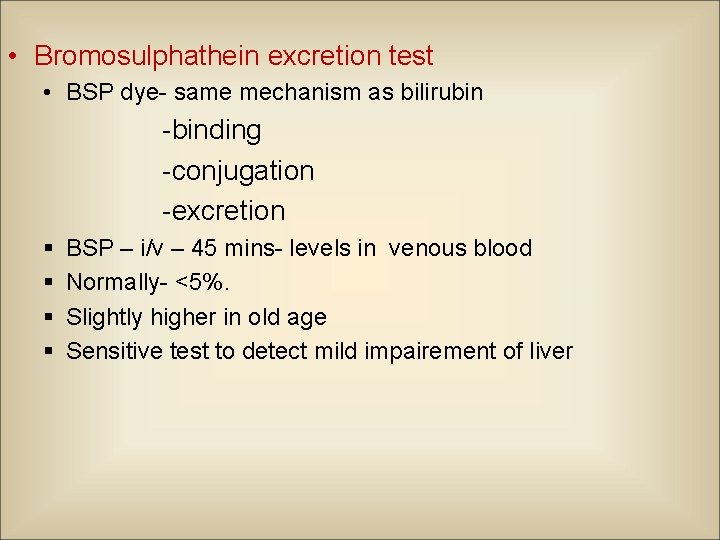
• Bromosulphathein excretion test • BSP dye- same mechanism as bilirubin -binding -conjugation -excretion § § BSP – i/v – 45 mins- levels in venous blood Normally- <5%. Slightly higher in old age Sensitive test to detect mild impairement of liver

Enzymes reflecting cholestasis • Alkaline phosphatase- present in cells of the bile duct § Isoenzymes- bone , liver, intestine, placenta , kidney , leukocytes. § Normal levels- 42 -122 IU/L - 3 -13 KA units/dl Non-pathological Age >60 yrs Bld group – O & B Growing children & adolescents Late in normal pregnancy Pathological 1° biliary cirrhosis Choledocholithiasis Hepatic malignancy 1° & 2° Paget’s disease

§ in serum ALP in an apparently healthy pt. § Fractionate the ALP to identify source of isoenzyme § ALP from different tissues differ in susceptibility inactivation by heat § Measure - 5' NT, GGT

• 5' NT • Sensitive and specific for hepatobiliary disorders (HBD) • Normal pregnancy, bone growth and bone diseases do not affect 5' NT • In pts with HBD, changes in ALP are usually followed by similar changes in 5' NT • GGT • Inducible microsomal enzyme. N levels – 5 - 40 IU/L. • Less specific than 5' NT as a marker for HBD • Unlike 5' NT, GGT may be released from many sites beside the hepatobiliary tree Ø Bone – important source of ALP, has little GGT thus GGT useful for differentiating hepatic & osseous sources of ALP

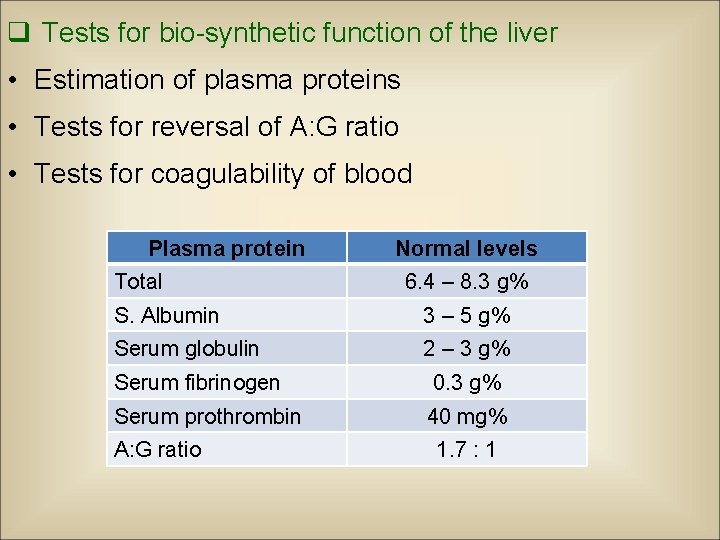
q Tests for bio-synthetic function of the liver • Estimation of plasma proteins • Tests for reversal of A: G ratio • Tests for coagulability of blood Plasma protein Total Normal levels 6. 4 – 8. 3 g% S. Albumin 3 – 5 g% Serum globulin 2 – 3 g% Serum fibrinogen 0. 3 g% Serum prothrombin 40 mg% A: G ratio 1. 7 : 1

• Serum albumin • S. albumin <3 g/dl suspect chronic liver disease • Hypoalbuminemia – not specific for liver disease – Protein malnutrition of any cause – Protein losing enteropathies – Nephrotic syndrome – Chronic infections – Burns • Reversal of A : G ratio chronic liver dysfunction.

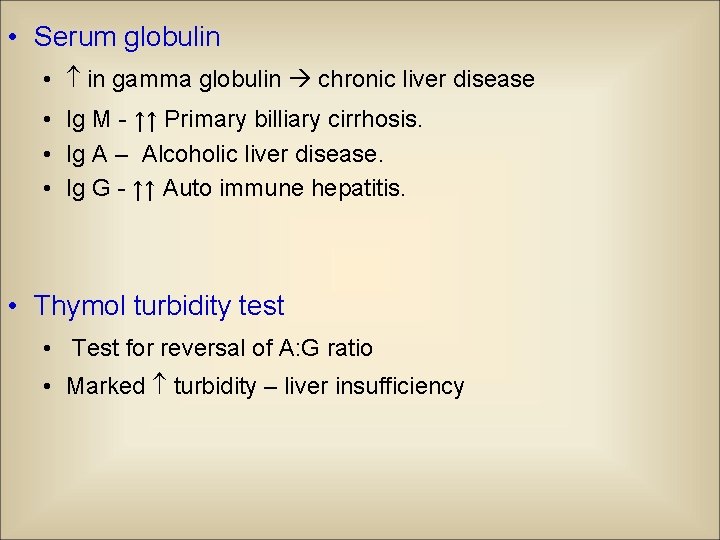
• Serum globulin • in gamma globulin chronic liver disease • Ig M - ↑↑ Primary billiary cirrhosis. • Ig A – Alcoholic liver disease. • Ig G - ↑↑ Auto immune hepatitis. • Thymol turbidity test • Test for reversal of A: G ratio • Marked turbidity – liver insufficiency

• Coagulation factors § Factor I, II, V, VII § Short t 1/2 – single best measure of acute hepatic synthetic function § Tests –PT- N 11 -16 sec - PTTK – N 30 - 40 sec • Prognostic value§ PT > 5 sec above control – indicative of poor prognostic sign in acute viral hepatitis. § in hepatitis, cirrhosis, disorders leading to vit K deficiency such as obstructive jaundice or fat malabsorption

q Immunological tests • Antibodies to specific etiologic agents– – – • HBV- HBs. Ag , HBc. Ag, HBe. Ag Antibody to Entamoeba histolytica Antibody to CMV, HCV, EBV Non specific antibodies – – – Antimitochondrial antibody- PBC Antismooth muscle antibody- Auto immune hepatitis p. ANCA- Primary sclerosing cholangitis

• Serum tumor markers • feto-protein - ↑ in HCC. • Hepatobiliary imaging • USG, CT scan - 1 st line investigation • ERCP, PTC- visualization of biliary tract • Doppler USG& MRI- hepatic vasculature & heamodynamics • CT & MRI- hepatic masses & tumours

• Others • FNAC • Biopsy – percutaneous needle liver biopsy a) VIM –SILVERMAN ( cutting ) needle b) MENGHINI’S ( aspiration ) needle • Indications – – – Unexplained hepatomegaly Cholestasis of unknown cause Persistent abnormal LFTs Infiltrative disorders- sarcoidosis, tuberculosis Pyrexia of unknown origin Primary/ metastatic liver diseases

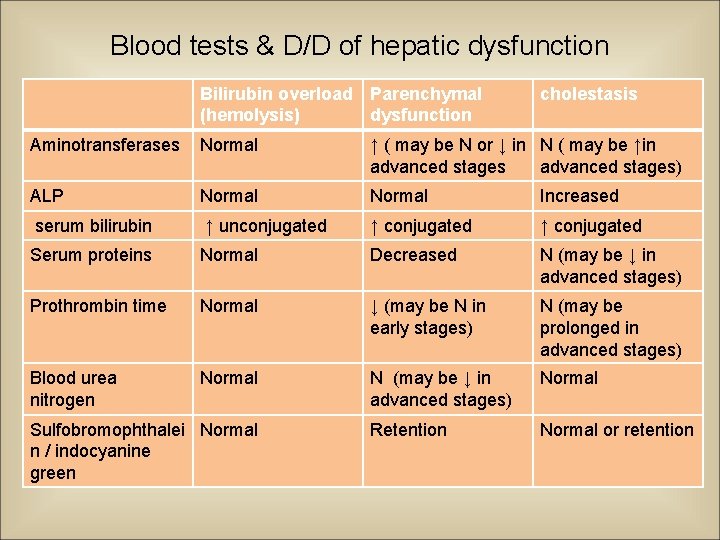
Blood tests & D/D of hepatic dysfunction Bilirubin overload Parenchymal (hemolysis) dysfunction cholestasis Aminotransferases Normal ↑ ( may be N or ↓ in N ( may be ↑in advanced stages) ALP Normal Increased serum bilirubin ↑ unconjugated ↑ conjugated Serum proteins Normal Decreased N (may be ↓ in advanced stages) Prothrombin time Normal ↓ (may be N in early stages) N (may be prolonged in advanced stages) Blood urea nitrogen Normal N (may be ↓ in advanced stages) Normal Retention Normal or retention Sulfobromophthalei Normal n / indocyanine green


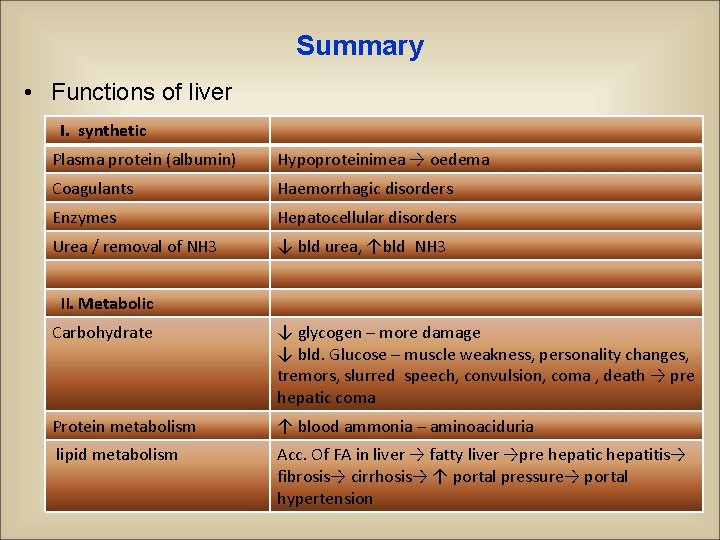
Summary • Functions of liver I. synthetic Plasma protein (albumin) Hypoproteinimea → oedema Coagulants Haemorrhagic disorders Enzymes Hepatocellular disorders Urea / removal of NH 3 ↓ bld urea, ↑bld NH 3 II. Metabolic Carbohydrate ↓ glycogen – more damage ↓ bld. Glucose – muscle weakness, personality changes, tremors, slurred speech, convulsion, coma , death → pre hepatic coma Protein metabolism ↑ blood ammonia – aminoaciduria lipid metabolism Acc. Of FA in liver → fatty liver →pre hepatic hepatitis→ fibrosis→ cirrhosis→ ↑ portal pressure→ portal hypertension

III. Bile secretion Bile salts & acids steatorrhea Conjugation of bilirubin Hepatocellular jaundice IV. Miscellaneous Vit A, K Deficiency- ↓vit A , K Antibacterial action Prevent infections Destruction of RBCs Anemia , ↑ bilirubin

THANK YOU