GENERAL CHEMISTRY PRINCIPLES AND MODERN APPLICATIONS ELEVENTH EDITION










- Slides: 56

GENERAL CHEMISTRY PRINCIPLES AND MODERN APPLICATIONS ELEVENTH EDITION PETRUCCI HERRING MADURA BISSONNETTE Chemical Bonding II: Valence Bond and Molecular Orbital Theories 11 PHILIP DUTTON UNIVERSITY OF WINDSOR DEPARTMENT OF CHEMISTRY AND BIOCHEMISTRY Slide 11 - 1 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

Chemical Bonding II: Valence Bond and Molecular Orbital Theories Slide 11 - 2 CONTENTS 11 -1 What a Bonding Theory Should Do 11 -2 Introduction to the Valence-Bond Method 11 -3 Hybridization of Atomic Orbitals 11 -4 Multiple Covalent Bonds 11 -5 Molecular Orbital Theory 11 -6 Delocalized Electrons: An Explanation Based on Molecular Orbital Theory 11 -7 Some Unresolved Issues: Can Electron Charge-Density Plots Help? General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

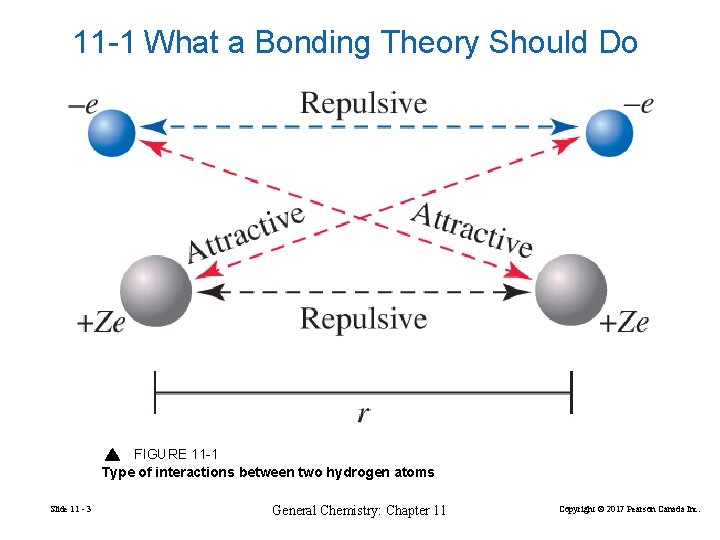
11 -1 What a Bonding Theory Should Do FIGURE 11 -1 Type of interactions between two hydrogen atoms Slide 11 - 3 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

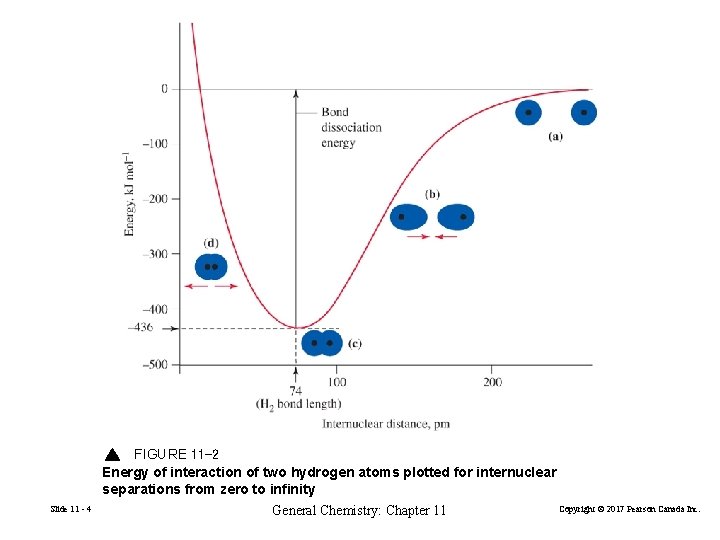
FIGURE 11 -2 Energy of interaction of two hydrogen atoms plotted for internuclear separations from zero to infinity Slide 11 - 4 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

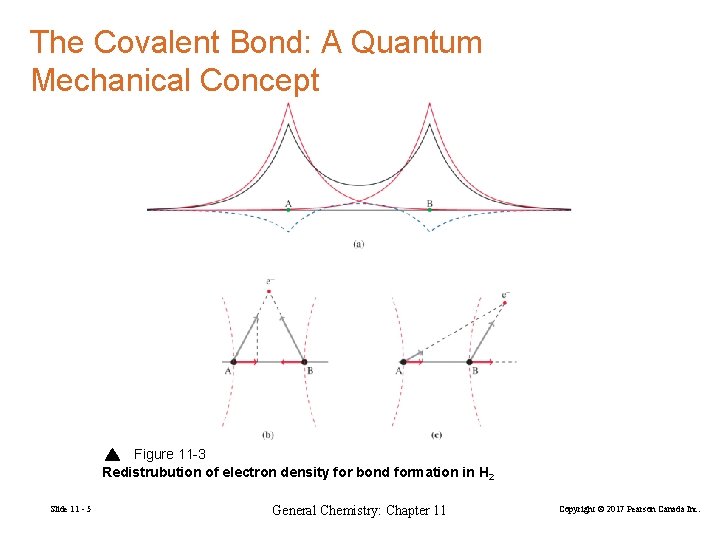
The Covalent Bond: A Quantum Mechanical Concept Figure 11 -3 Redistrubution of electron density for bond formation in H 2 Slide 11 - 5 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

11 -2 Introduction to the Valence-Bond Method Overlap of half-filled atomic orbital overlap normally describes covalent bonding. Sometimes involves the overlap of a filled orbital on one atom with an empty orbital on another. A localized electron model of bonding. Slide 11 - 6 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

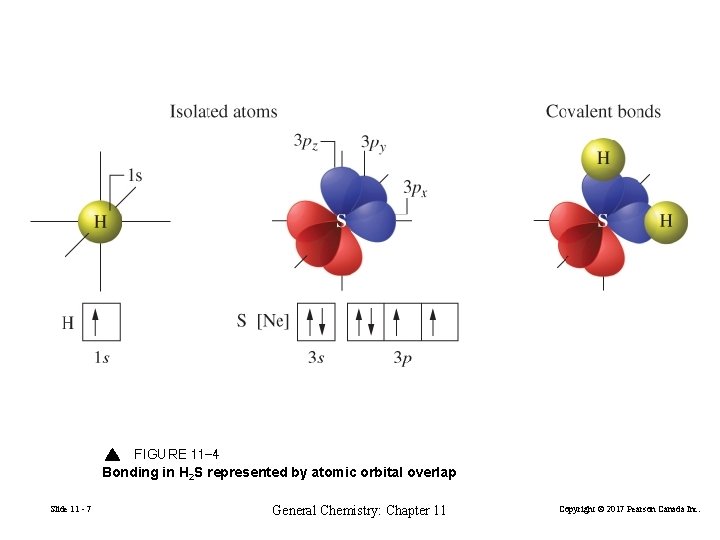
FIGURE 11 -4 Bonding in H 2 S represented by atomic orbital overlap Slide 11 - 7 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

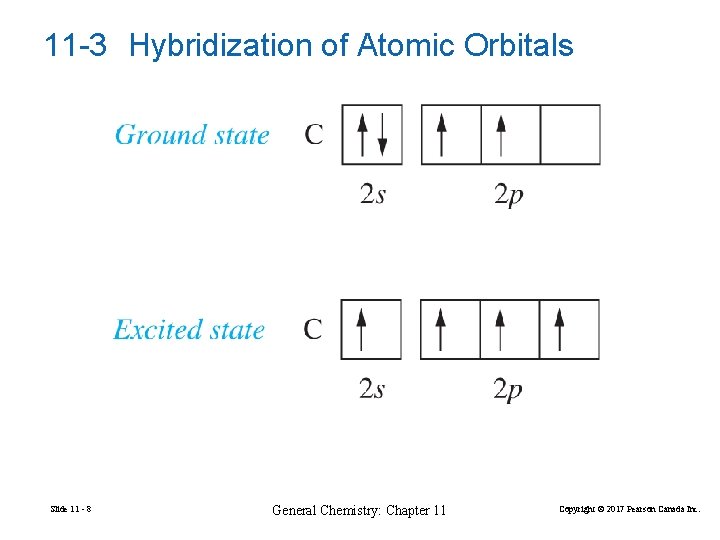
11 -3 Hybridization of Atomic Orbitals Slide 11 - 8 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

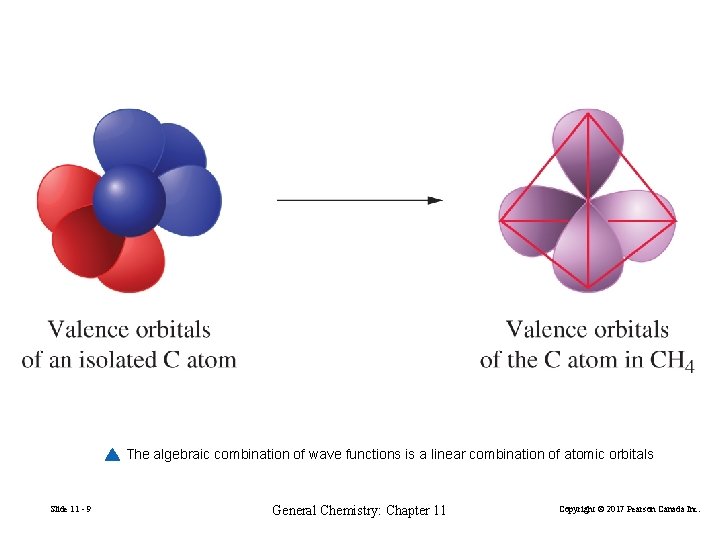
The algebraic combination of wave functions is a linear combination of atomic orbitals Slide 11 - 9 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

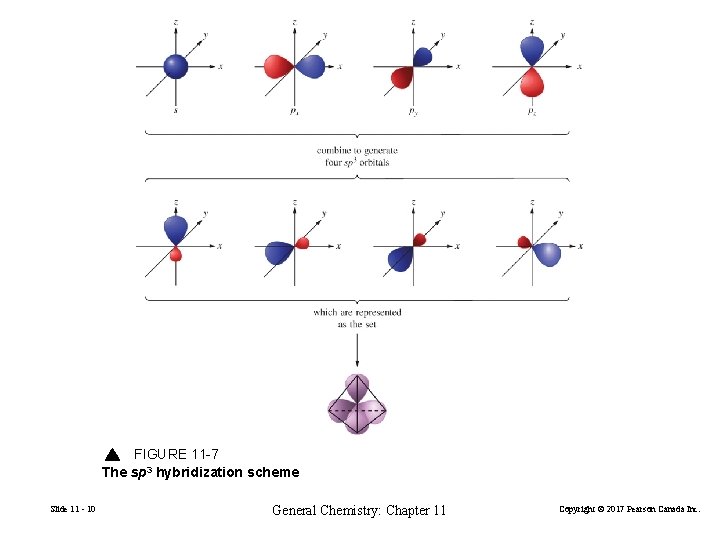
FIGURE 11 -7 The sp 3 hybridization scheme Slide 11 - 10 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

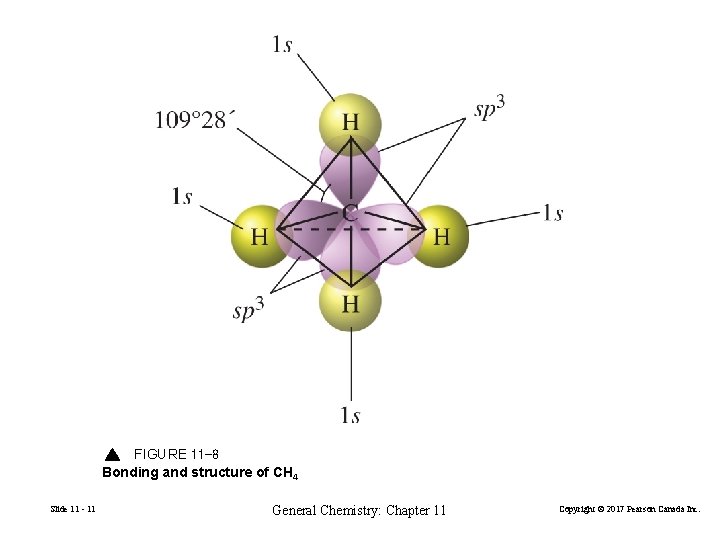
FIGURE 11 -8 Bonding and structure of CH 4 Slide 11 - 11 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

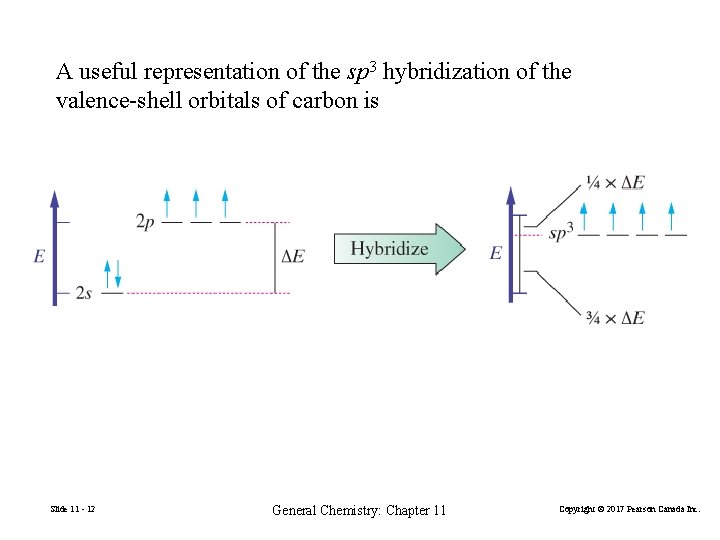
A useful representation of the sp 3 hybridization of the valence-shell orbitals of carbon is Slide 11 - 12 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

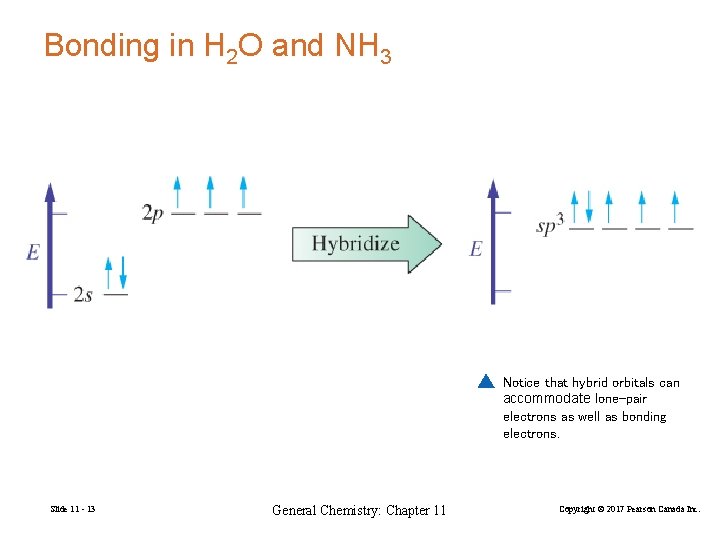
Bonding in H 2 O and NH 3 Notice that hybrid orbitals can accommodate lone-pair electrons as well as bonding electrons. Slide 11 - 13 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

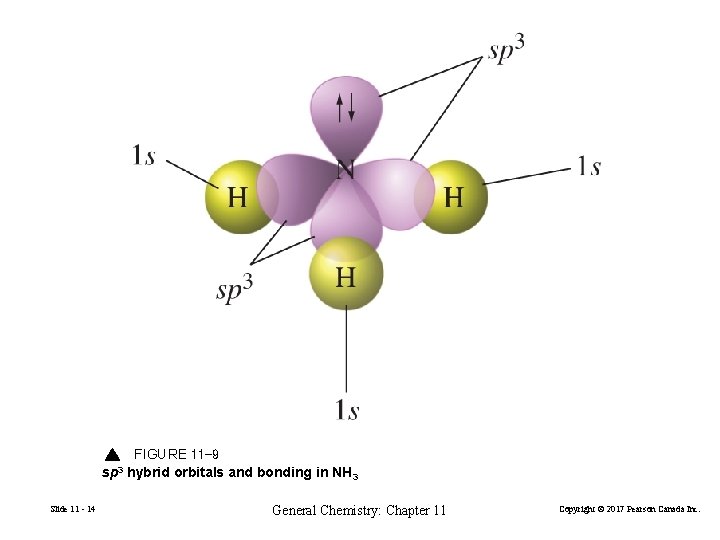
FIGURE 11 -9 sp 3 hybrid orbitals and bonding in NH 3 Slide 11 - 14 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

sp 2 Hybrid Orbitals Slide 11 - 15 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

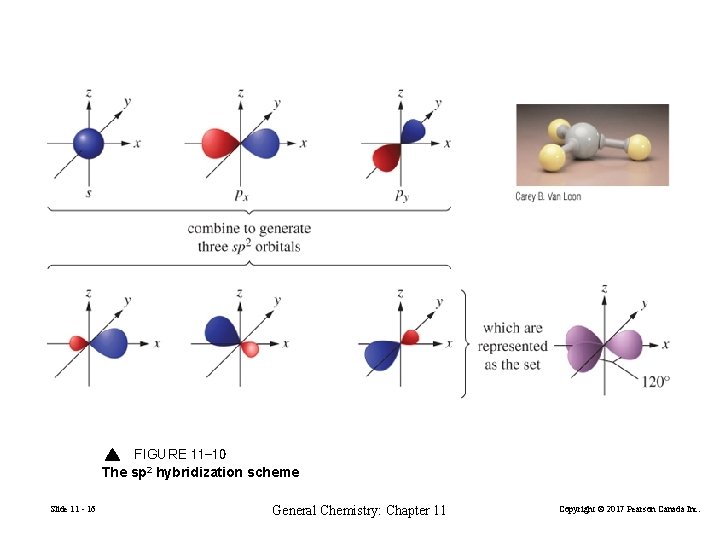
FIGURE 11 -10 The sp 2 hybridization scheme Slide 11 - 16 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

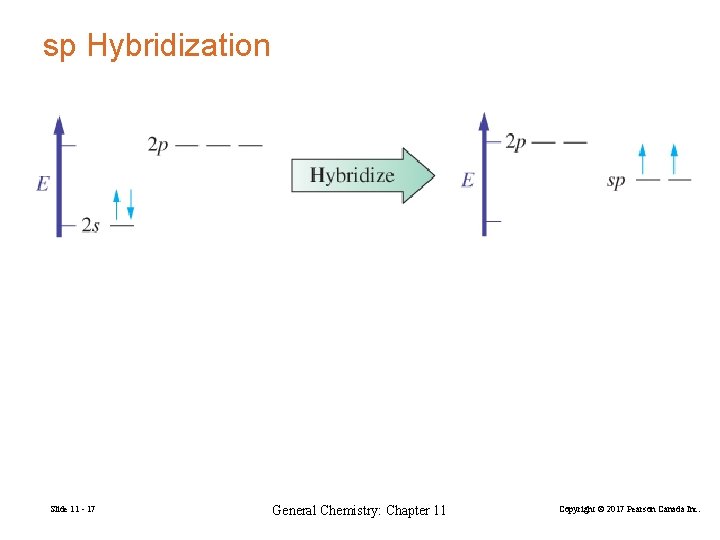
sp Hybridization Slide 11 - 17 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

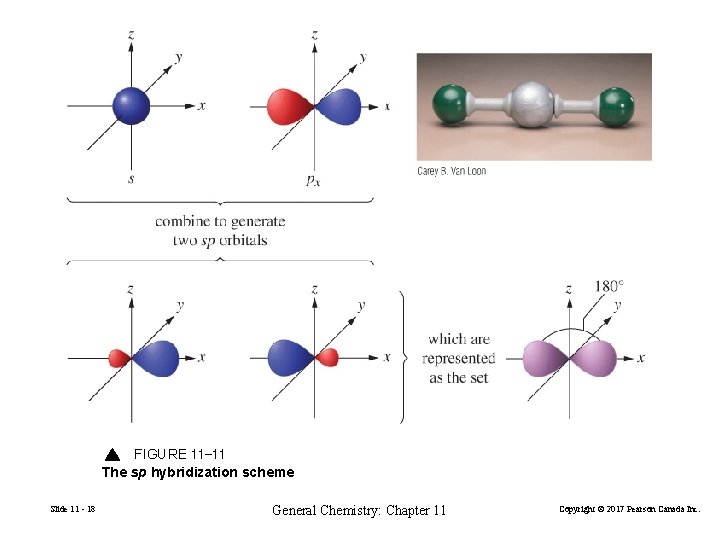
FIGURE 11 -11 The sp hybridization scheme Slide 11 - 18 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

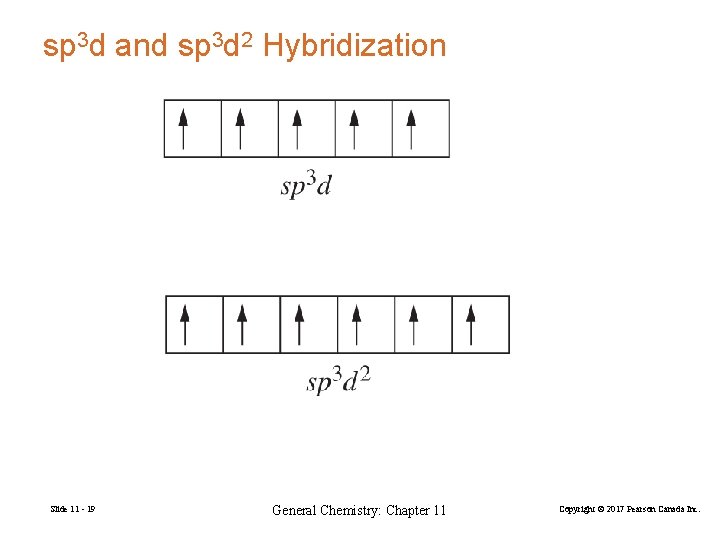
sp 3 d and sp 3 d 2 Hybridization Slide 11 - 19 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

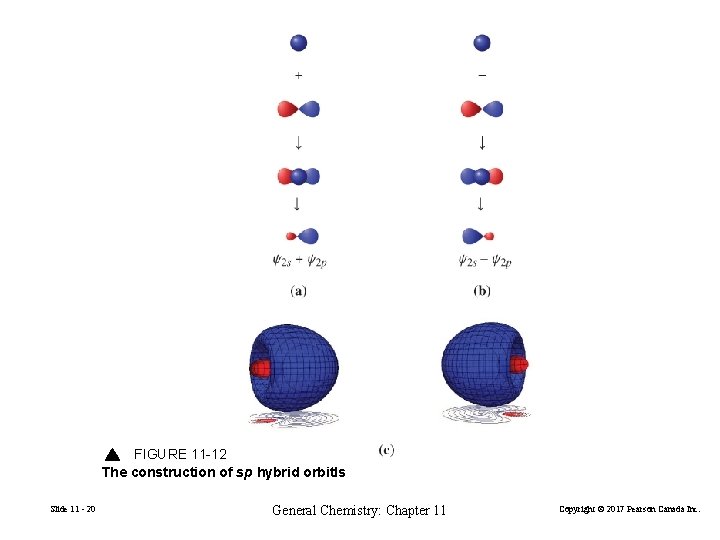
FIGURE 11 -12 The construction of sp hybrid orbitls Slide 11 - 20 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

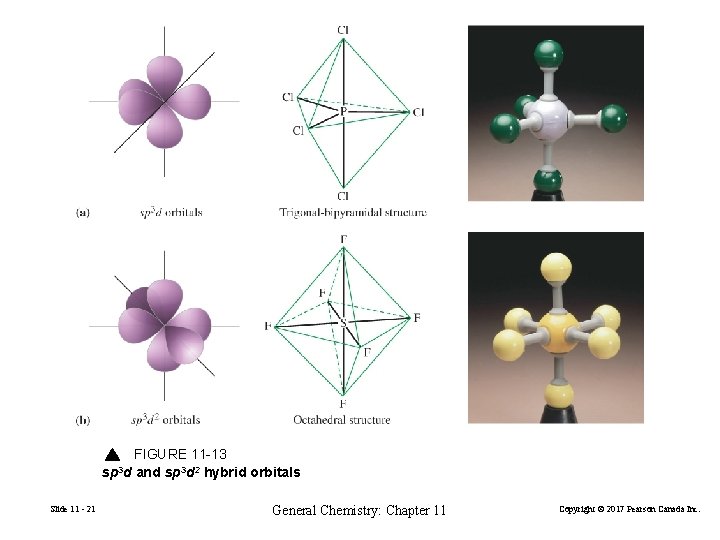
FIGURE 11 -13 sp 3 d and sp 3 d 2 hybrid orbitals Slide 11 - 21 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

Hybrid Orbitals and Valence-Shell Electron-Pair Repulsion (VSEPR) Theory • • • Write a plausible Lewis structure. Use VSEPR to predict electrongroup geometry. Select the corresponding hybridization scheme. FIGURE 11 -14 Using electron-group geometry to determine hybrid orbitals Slide 11 - 22 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

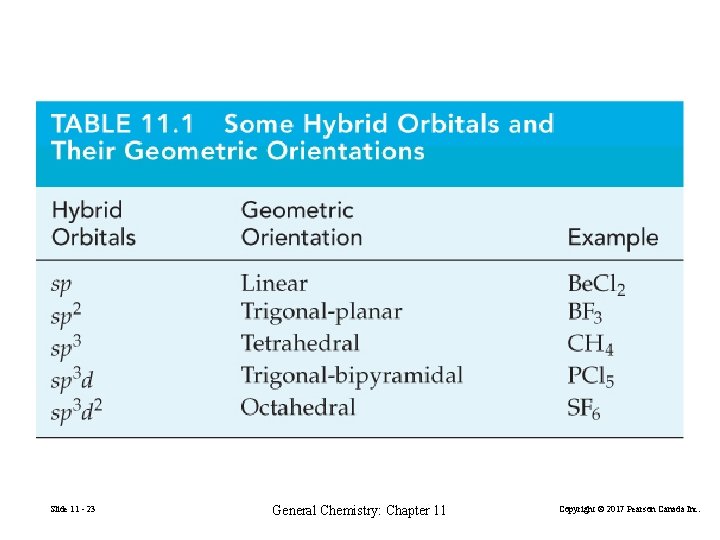
Slide 11 - 23 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

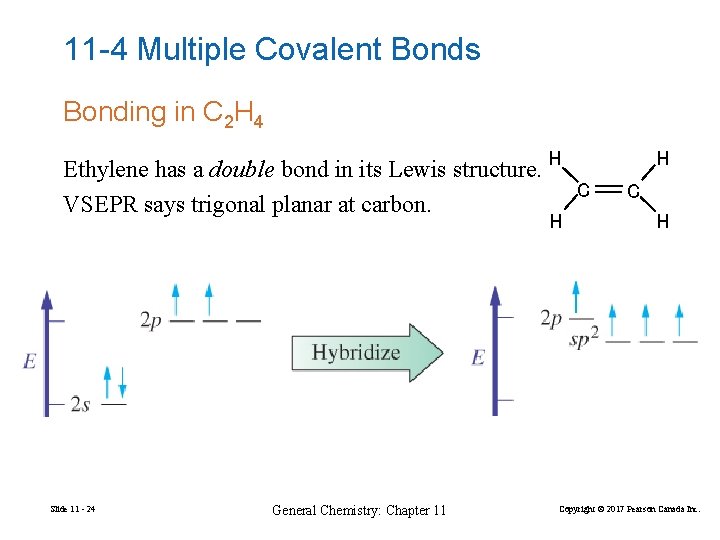
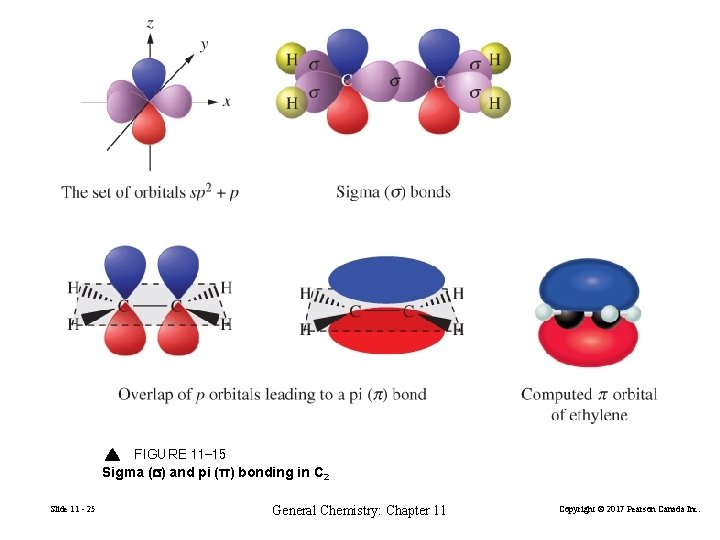
11 -4 Multiple Covalent Bonds Bonding in C 2 H 4 Ethylene has a double bond in its Lewis structure. H C VSEPR says trigonal planar at carbon. H Slide 11 - 24 General Chemistry: Chapter 11 H Copyright © 2017 Pearson Canada Inc.

FIGURE 11 -15 Sigma (s) and pi (π) bonding in C 2 Slide 11 - 25 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

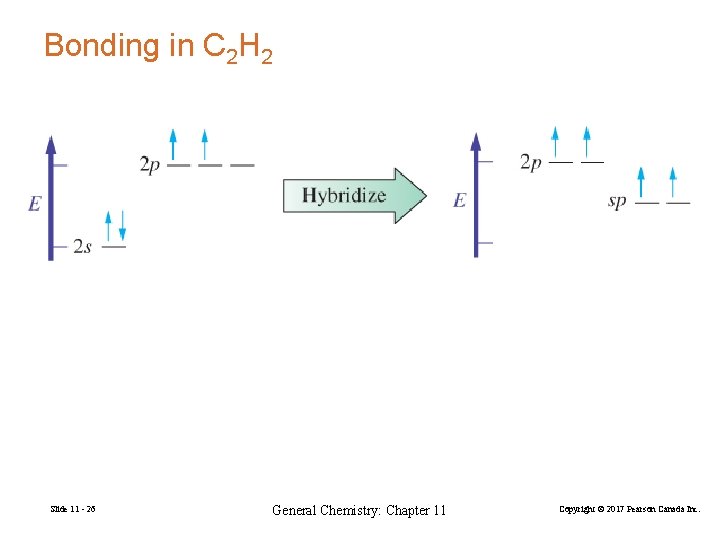
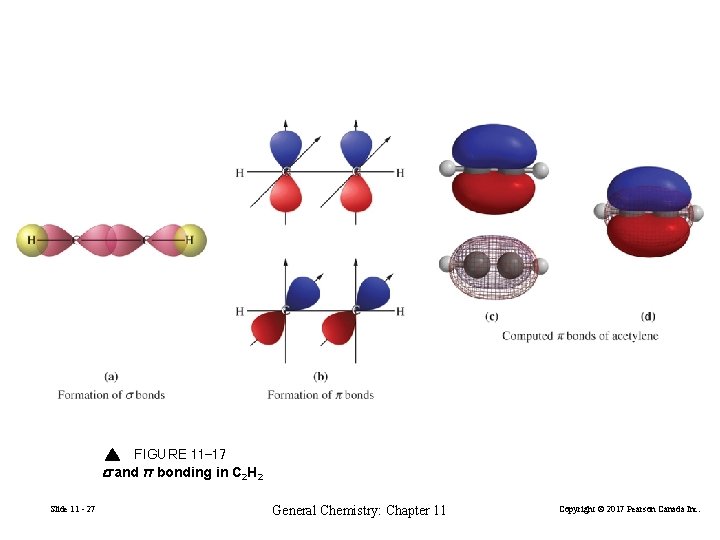
Bonding in C 2 H 2 Slide 11 - 26 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

FIGURE 11 -17 s and π bonding in C 2 H 2 Slide 11 - 27 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

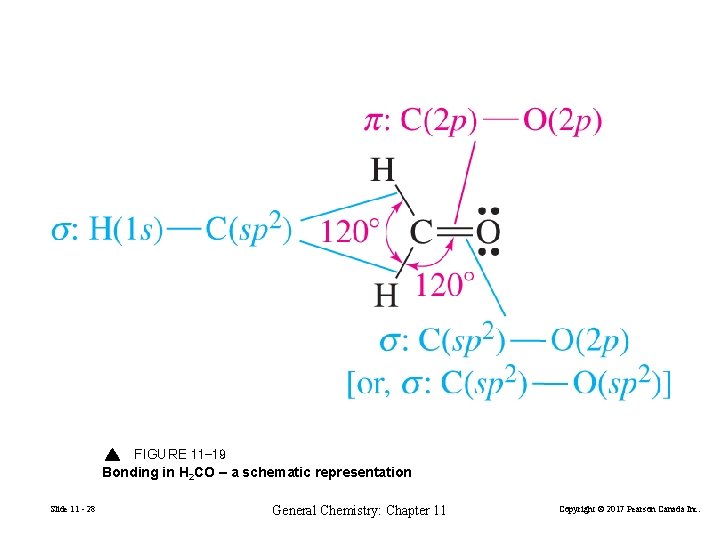
FIGURE 11 -19 Bonding in H 2 CO – a schematic representation Slide 11 - 28 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

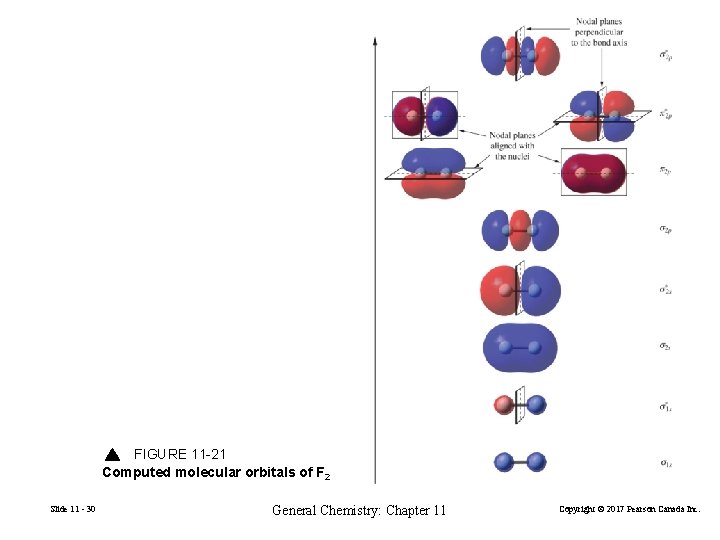
11 -5 Molecular Orbital Theory Atomic orbitals are isolated on atoms. Molecular orbitals span two or more atoms. Calculated orbitals for F 2 Analysis of the total wave function describing F 2 in its lowest energy state MO’s are distributed around both nuclei Linear Combination of Atomic Orbitals Slide 11 - 29 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

FIGURE 11 -21 Computed molecular orbitals of F 2 Slide 11 - 30 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

LCAO Linear Combination of Atomic Orbitals. Ψ 1 = φ1 + φ2 Ψ 2 = φ 1− φ 2 • Each MO can be represented mathematically as a (linear) combination of AO’s • The total number of MOs is equal to the total number of Aos • MOs can be classified as bonding, antibonding, or nonbonding • The electron configuration for the molecule is obtained by placing the electrons into the MOs in a particular order Slide 11 - 31 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

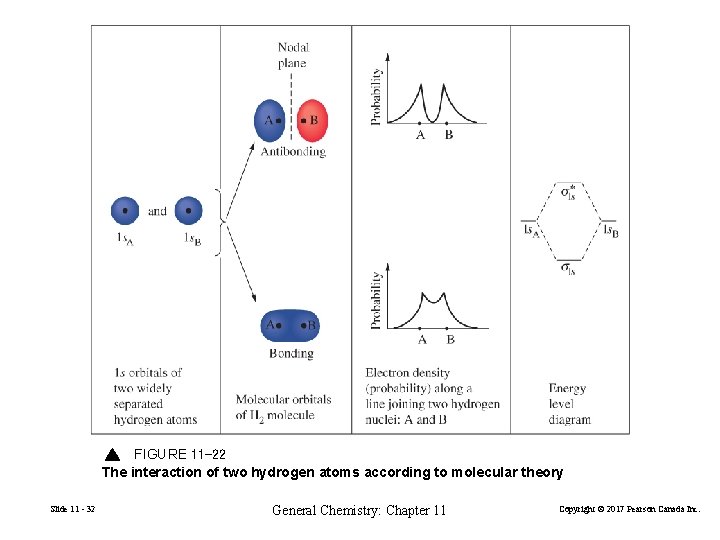
FIGURE 11 -22 The interaction of two hydrogen atoms according to molecular theory Slide 11 - 32 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

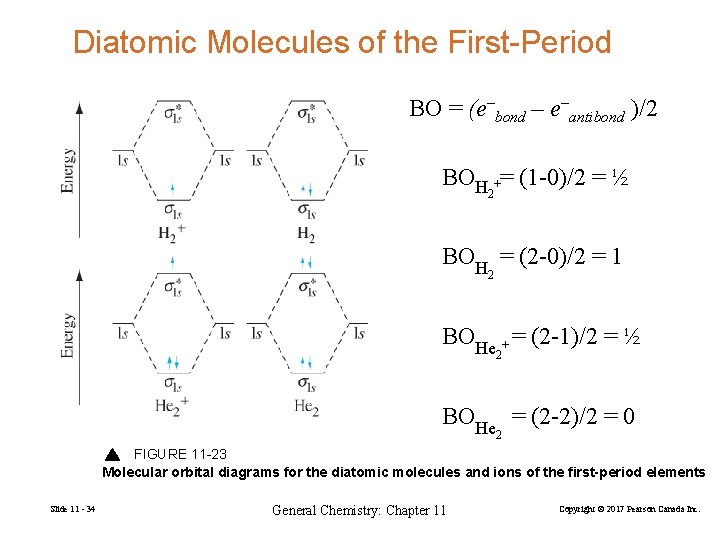
Bond Order Stable species have more electrons in bonding orbitals than antibonding. No. e− in bonding MOs − No. e− in antibonding MOs Bond Order = 2 Slide 11 - 33 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

Diatomic Molecules of the First-Period BO = (e–bond – e–antibond )/2 BOH += (1 -0)/2 = ½ 2 BOH = (2 -0)/2 = 1 2 BOHe + = (2 -1)/2 = ½ 2 BOHe = (2 -2)/2 = 0 2 FIGURE 11 -23 Molecular orbital diagrams for the diatomic molecules and ions of the first-period elements Slide 11 - 34 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

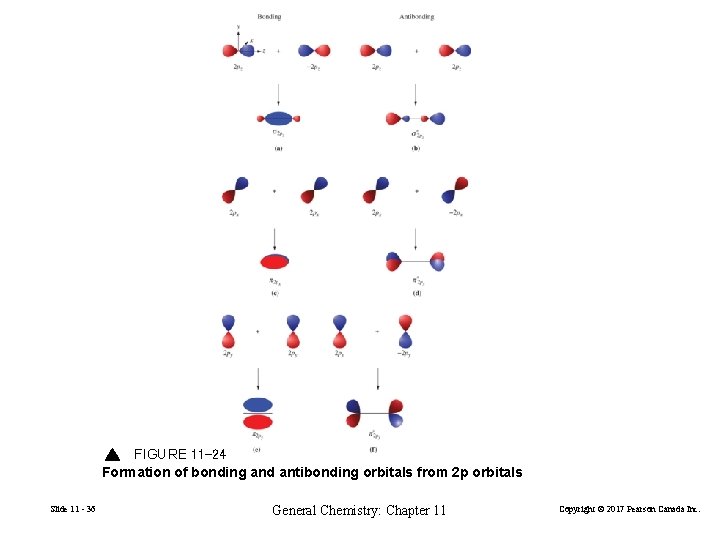
Molecular Orbitals of the Second Period Elements First period use only 1 s orbitals. Second period have 2 s and 2 p orbitals available. p orbital overlap: End-to end overlap – sigma bond (σ). Side-to-side overlap– pi bond (π). Slide 11 - 35 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

FIGURE 11 -24 Formation of bonding and antibonding orbitals from 2 p orbitals Slide 11 - 36 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

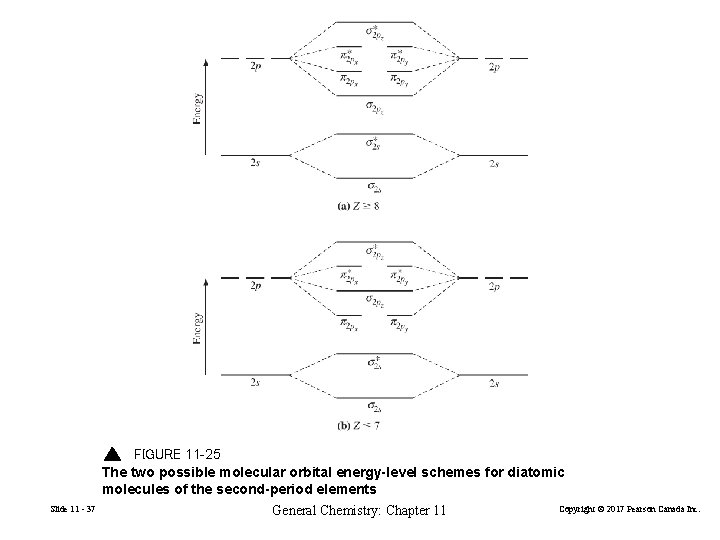
FIGURE 11 -25 The two possible molecular orbital energy-level schemes for diatomic molecules of the second-period elements Slide 11 - 37 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

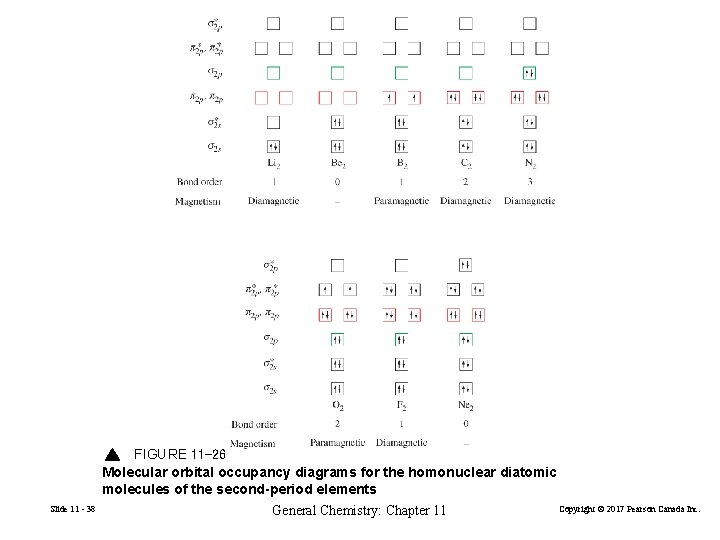
FIGURE 11 -26 Molecular orbital occupancy diagrams for the homonuclear diatomic molecules of the second-period elements Slide 11 - 38 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

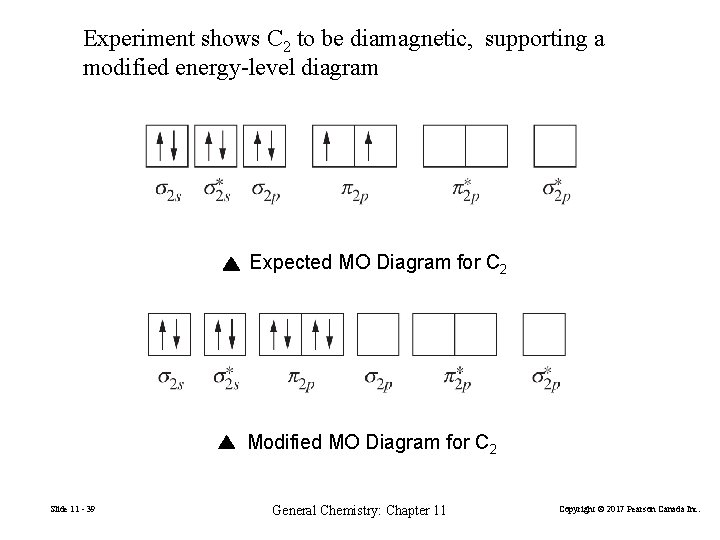
Experiment shows C 2 to be diamagnetic, supporting a modified energy-level diagram Expected MO Diagram for C 2 Modified MO Diagram for C 2 Slide 11 - 39 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

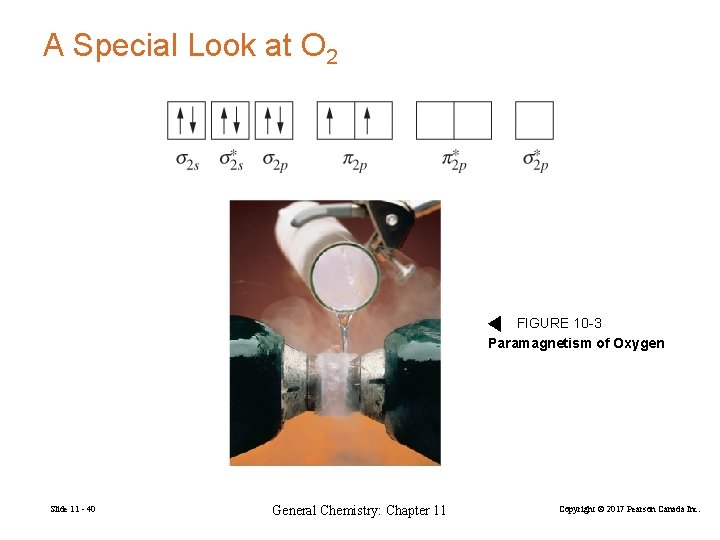
A Special Look at O 2 FIGURE 10 -3 Paramagnetism of Oxygen Slide 11 - 40 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

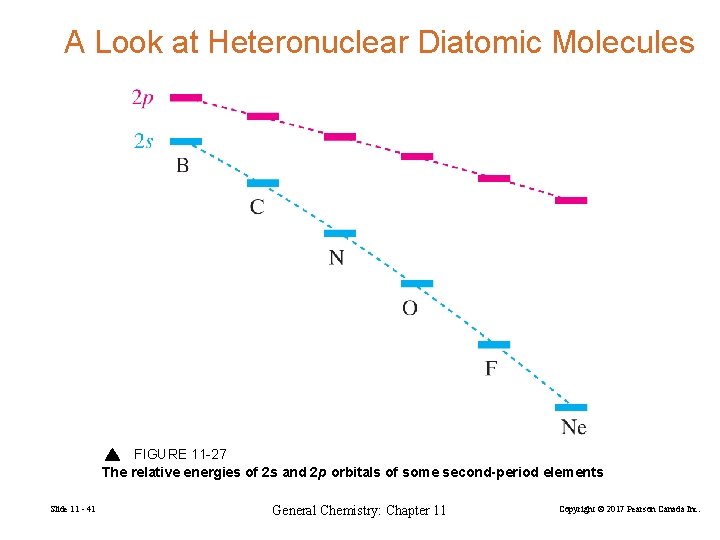
A Look at Heteronuclear Diatomic Molecules FIGURE 11 -27 The relative energies of 2 s and 2 p orbitals of some second-period elements Slide 11 - 41 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

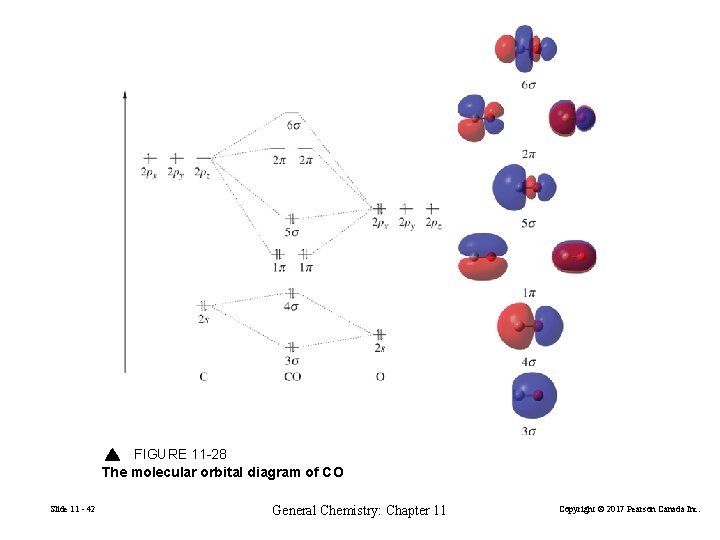
FIGURE 11 -28 The molecular orbital diagram of CO Slide 11 - 42 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

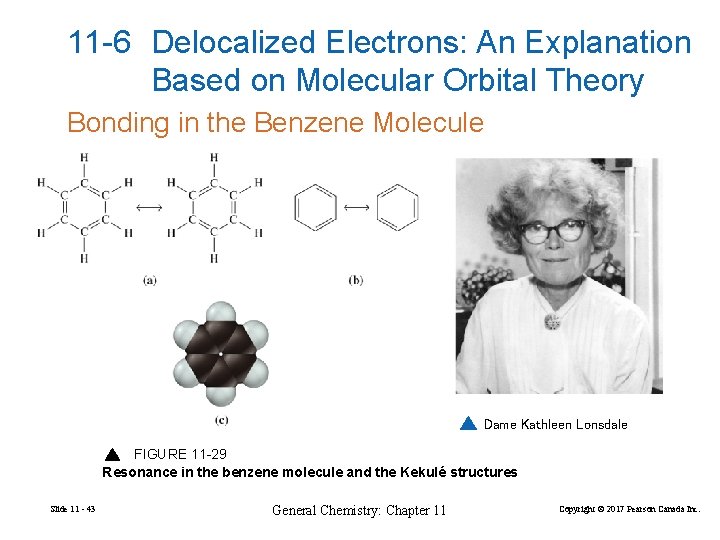
11 -6 Delocalized Electrons: An Explanation Based on Molecular Orbital Theory Bonding in the Benzene Molecule Dame Kathleen Lonsdale FIGURE 11 -29 Resonance in the benzene molecule and the Kekulé structures Slide 11 - 43 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

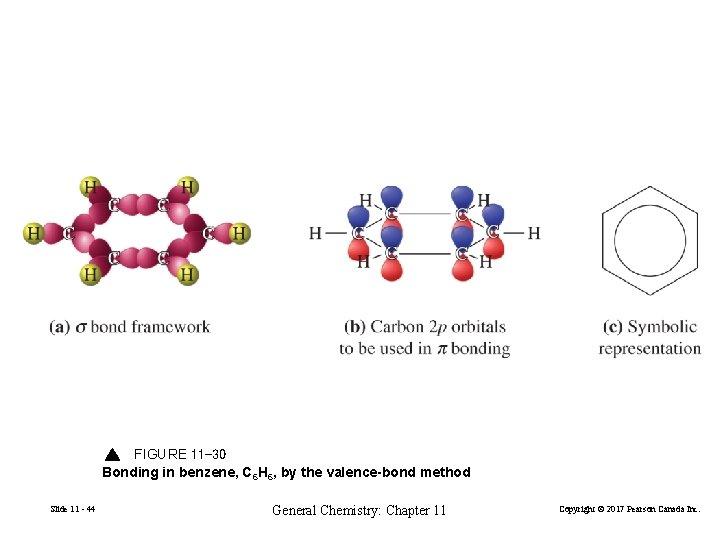
FIGURE 11 -30 Bonding in benzene, C 6 H 6, by the valence-bond method Slide 11 - 44 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

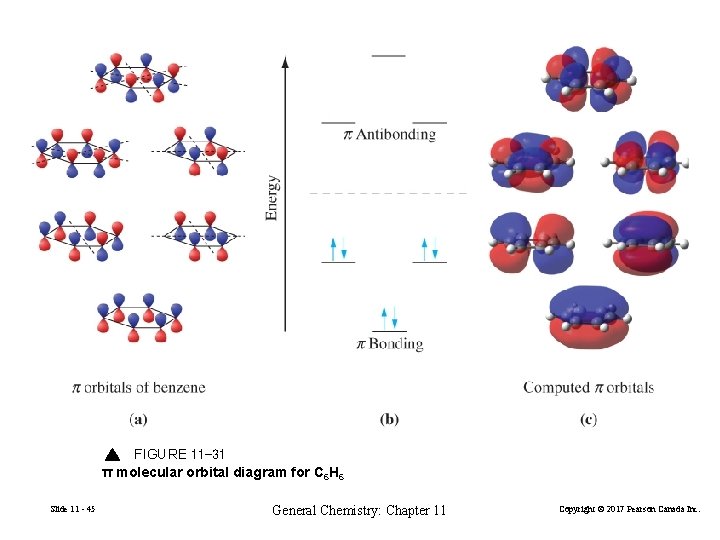
FIGURE 11 -31 π molecular orbital diagram for C 6 H 6 Slide 11 - 45 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

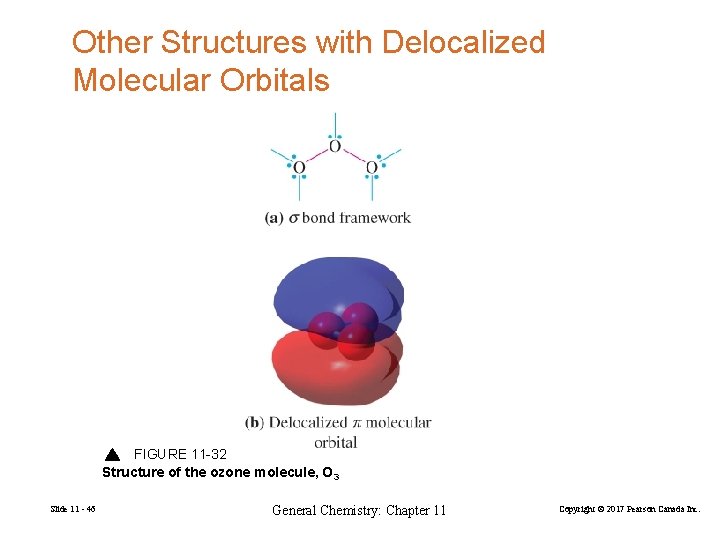
Other Structures with Delocalized Molecular Orbitals FIGURE 11 -32 Structure of the ozone molecule, O 3 Slide 11 - 46 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

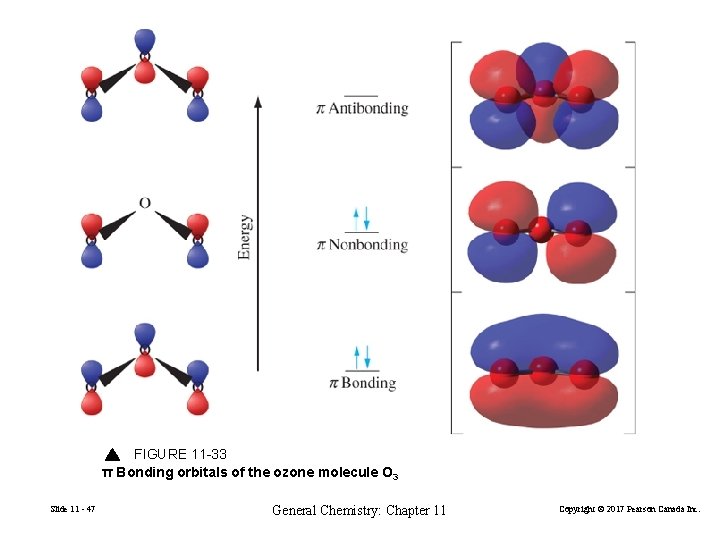
FIGURE 11 -33 π Bonding orbitals of the ozone molecule O 3 Slide 11 - 47 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

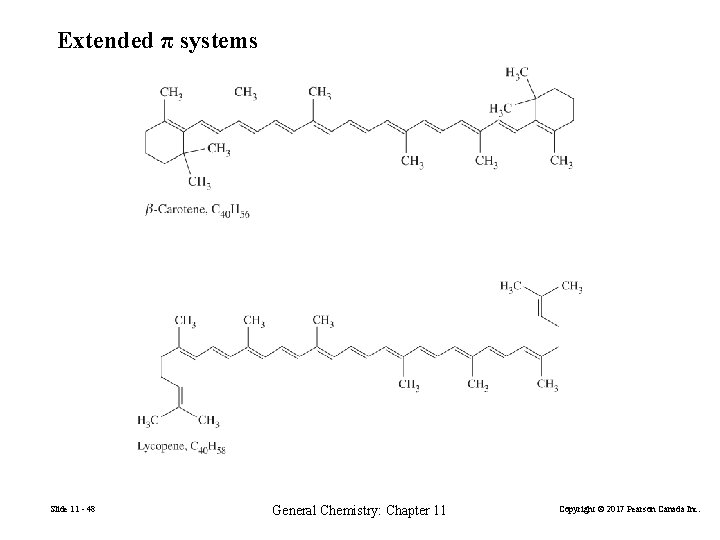
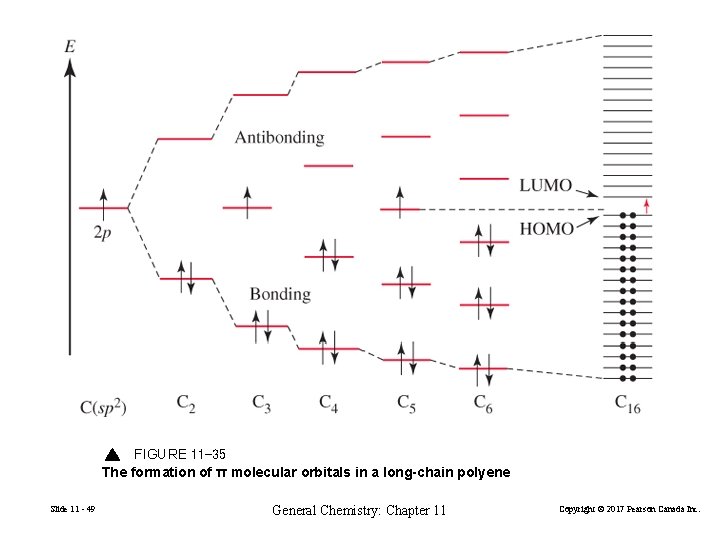
Extended π systems Slide 11 - 48 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

FIGURE 11 -35 The formation of π molecular orbitals in a long-chain polyene Slide 11 - 49 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

11 -7 Some Unresolved Issues: Can Electron Charge-Density Plots Help? Bonding in SF 6 Slide 11 - 50 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

Hyperconjugation or hypervalent? Compare to quantum mechanical calculations: S +3. 17 F – 0. 53 Expanded octet is questionable since wave function calculations show very little contribution from d-orbitals. Slide 11 - 51 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

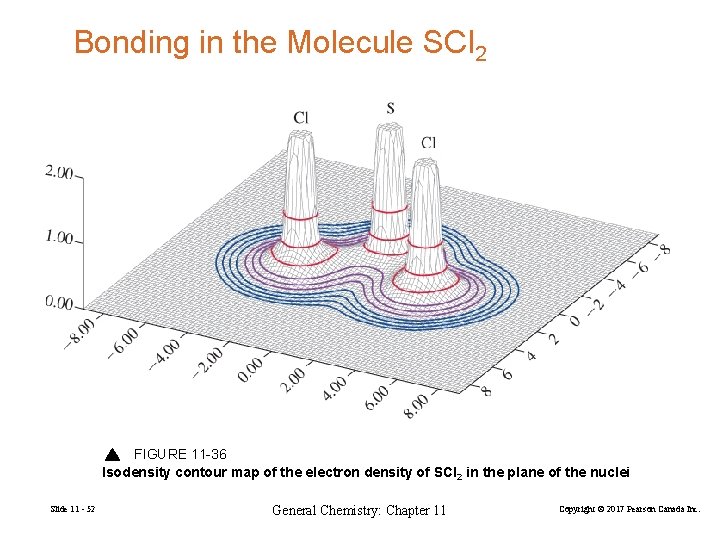
Bonding in the Molecule SCl 2 FIGURE 11 -36 Isodensity contour map of the electron density of SCl 2 in the plane of the nuclei Slide 11 - 52 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

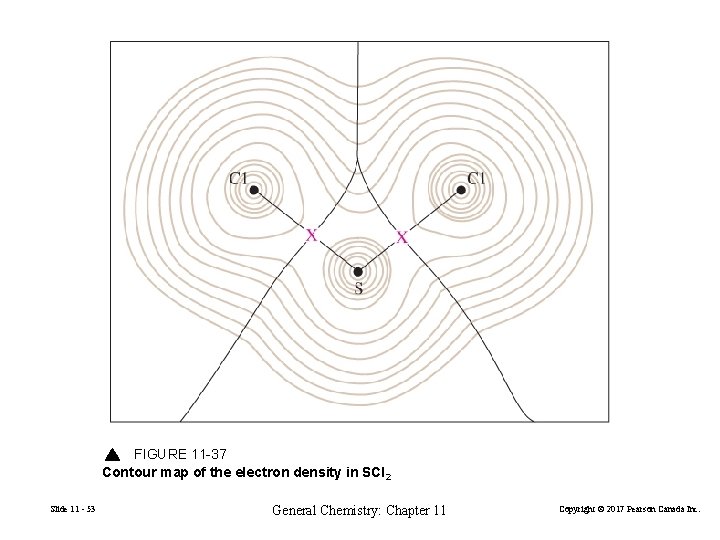
FIGURE 11 -37 Contour map of the electron density in SCl 2 Slide 11 - 53 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

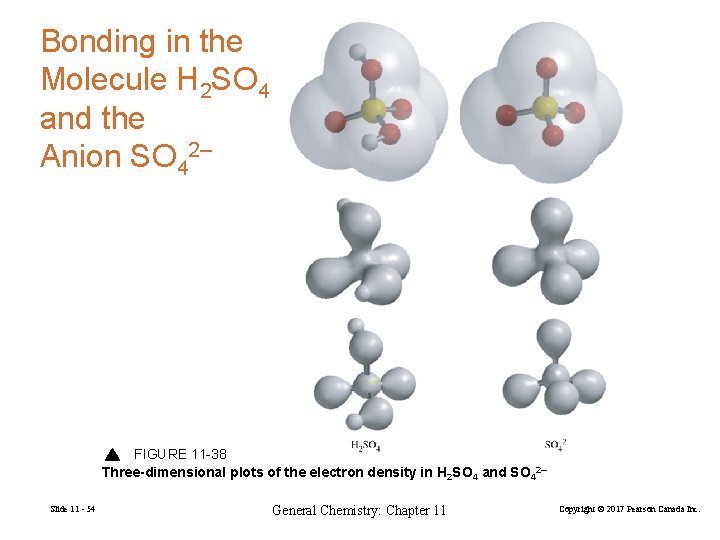
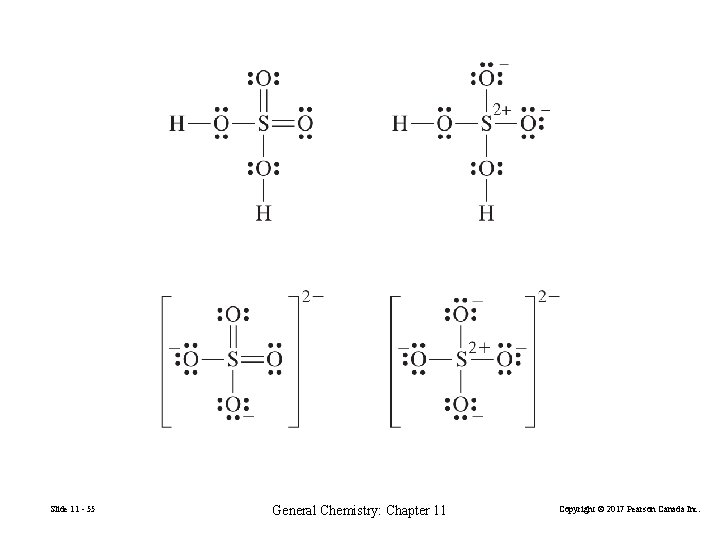
Bonding in the Molecule H 2 SO 4 and the Anion SO 42– FIGURE 11 -38 Three-dimensional plots of the electron density in H 2 SO 4 and SO 42– Slide 11 - 54 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

Slide 11 - 55 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.

End of Chapter Slide 11 - 56 General Chemistry: Chapter 11 Copyright © 2017 Pearson Canada Inc.