Empirical and Molecular Formulas Empirical vs Molecular Formula


































- Slides: 36

Empirical and Molecular Formulas

Empirical vs Molecular Formula • The Molecular Formula (MF) gives the actual number of each type of atom present. • The Empirical Formula (EF) gives the lowest wholenumber ratio of the atoms present. • Example: C 2 H 6 and C 3 H 9 – they have the same EF CH 3 yet have very different MF

Types of Formulas The formulas for compounds can be expressed as an empirical formula and as a molecular(true) formula. Empirical Molecular (true) Name CH C 2 H 2 acetylene CH C 6 H 6 benzene CO 2 CH 2 O C 5 H 10 O 5 Timberlake Lecture. PLUS carbon dioxide ribose 3

• An empirical formula represents the simplest whole number ratio of the atoms in a compound. • The molecular formula is the true or actual ratio of the atoms in a compound. Timberlake Lecture. PLUS 4

EF vs. MF When you have an ionic compound: the EF = MF For some molecular compounds: the EF = MF, but not always

Determining the EF Remember: The EF is the lowest whole-number ratio of the moles of each atom present. Example: CH 4 has 1 mol C atoms 4 mol H atoms

Determining the EF If a compound consists of: 62. 1% C 13. 8% H 24. 1% N The percentages are based on MASS not MOLES We can compare moles, not masses

Determining the EF In order to determine the ratio of C: H: N, we need to know the mole ratio Step 1: Convert % of each into grams Make it easy on yourself, assume a sample size of 100. 00 g 62. 1 g C 13. 8 g H 24. 1 g N

Determining the EF Now that you know how many grams of each atom you have: Step 2: Convert grams to moles using the molar mass of each 62. 1 g C 13. 8 g H 24. 1 g N

Determining the EF Now that you have the mol ratio, you need to make them Whole-Numbers. Step 3: Divide each mol by the smallest mol value from step #2 5. 17 mol C 13. 7 mol H 1. 72 mol N

Determining the EF This results in 3 mol C, 8 mol H and 1 mol N therefore the EF = C 3 H 8 N

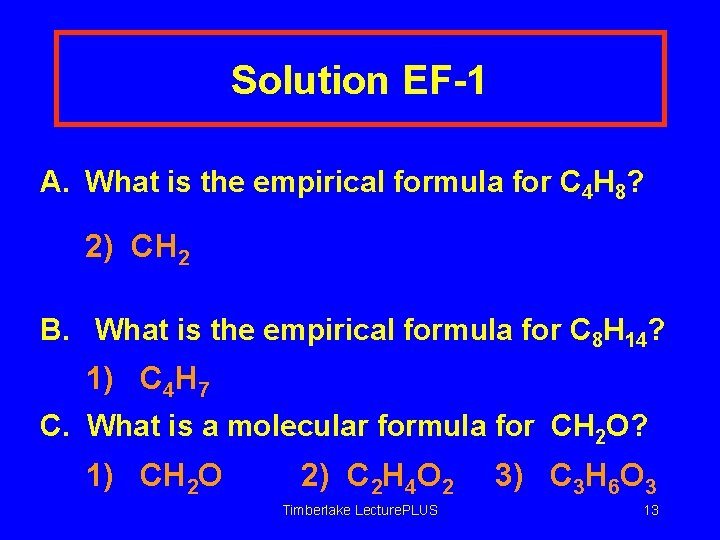
Learning Check EF-1 A. What is the empirical formula for C 4 H 8? 1) C 2 H 4 2) CH 2 3) CH B. What is the empirical formula for C 8 H 14? 1) C 4 H 7 2) C 6 H 12 3) C 8 H 14 C. What is a molecular formula for CH 2 O? 1) CH 2 O 2) C 2 H 4 O 2 Timberlake Lecture. PLUS 3) C 3 H 6 O 3 12

Solution EF-1 A. What is the empirical formula for C 4 H 8? 2) CH 2 B. What is the empirical formula for C 8 H 14? 1) C 4 H 7 C. What is a molecular formula for CH 2 O? 1) CH 2 O 2) C 2 H 4 O 2 Timberlake Lecture. PLUS 3) C 3 H 6 O 3 13

Learning Check EF-2 If the molecular formula has 4 atoms of N, what is the molecular formula if SN is the empirical formula? Explain. 1) SN 2) SN 4 3) S 4 N 4 Timberlake Lecture. PLUS 14

Solution EF-2 If the molecular formula has 4 atoms of N, what is the molecular formula if SN is the empirical formula? Explain. 3) S 4 N 4 If the actual formula has 4 atoms of N, and S is related 1: 1, then there must also be 4 atoms of S. Timberlake Lecture. PLUS 15

Empirical and Molecular Formulas molar mass = simplest mass a whole number = n n = 1 molar mass = empirical mass molecular formula = empirical formula n = 2 molar mass = 2 x empirical mass molecular formula = 2 x empirical formula molecular formula = or > empirical formula 16 Timberlake Lecture. PLUS

EF to MF To go from the EF to the MF, you need two additional pieces of information: 1 – calculate the mass from your EF 2 – You must be given the mass of the MF (X) EFmass = MFmass X = MFmass / EFmass

EF to MF Example: You found the EF to be HO, and the MFmass = 34. 02 g 1. 2. 3. 4. Calculate the EFmass = 17. 01 g Calculate X = 34. 02 g / 17. 01 g X=2 EF = HO MF = H 2 O 2

Learning Check EF-3 A compound has a formula mass of 176. 0 and an empirical formula of C 3 H 4 O 3. What is the molecular formula? 1) C 3 H 4 O 3 2) C 6 H 8 O 6 3) C 9 H 12 O 9 Timberlake Lecture. PLUS 19

Solution EF-3 A compound has a formula mass of 176. 0 and an empirical formula of C 3 H 4 O 3. What is the molecular formula? 2) C 6 H 8 O 6 C 3 H 4 O 3 = 88. 0 g/EF 176. 0 g = 2. 00 88. 0 Timberlake Lecture. PLUS 20

Learning Check EF-4 If there are 192. 0 g of O in the molecular formula, what is the true formula if the EF is C 7 H 6 O 4? 1) C 7 H 6 O 4 2) C 14 H 12 O 8 3) C 21 H 18 O 12 Timberlake Lecture. PLUS 21

Solution EF-4 If there are 192. 0 g of O in the molecular formula, what is the true formula if the EF is C 7 H 6 O 4? 3) C 21 H 18 O 12 192 g O = 3 x O 4 or 3 x C 7 H 6 O 4 Timberlake Lecture. PLUS 64. 0 g O in EF 22

Finding the Molecular Formula A compound is Cl 71. 65%, C 24. 27%, and H 4. 07%. What are the empirical and molecular formulas? The molar mass is known to be 99. 0 g/mol. 1. State mass percents as grams in a 100. 00 -g sample of the compound. Cl 71. 65 g C 24. 27 g H 4. 07 g Timberlake Lecture. PLUS 23

2. Calculate the number of moles of each element. 71. 65 g Cl x 1 mol Cl = 2. 02 mol Cl 35. 5 g Cl 24. 27 g C x 1 mol C 12. 0 g C = 2. 02 mol C 4. 07 g H x 1 mol H 1. 01 g H = Timberlake Lecture. PLUS 4. 04 mol H 24

Why moles? Why do you need the number of moles of each element in the compound? Timberlake Lecture. PLUS 25

3. Find the smallest whole number ratio by dividing each mole value by the smallest mole values: Cl: 2. 02 = 1 Cl 2. 02 C: H: 2. 02 = 1 C 4. 04 = 2 H 2. 02 4. Write the simplest or empirical formula Timberlake Lecture. PLUS 26 CH 2 Cl

5. EM (empirical mass) = 1(C) + 2(H) + 1(Cl) = 49. 5 6. n = molar mass/empirical mass Molar mass EM = 99. 0 g/mol 49. 5 g/EM = n=2 7. Molecular formula (CH 2 Cl)2 = C 2 H 4 Cl 2 Timberlake Lecture. PLUS 27

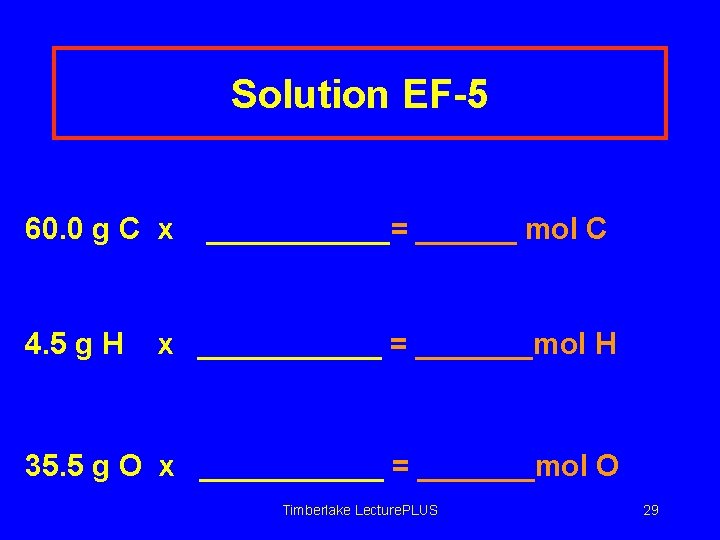
Learning Check EF-5 Aspirin is 60. 0% C, 4. 5 % H and 35. 5 O. Calculate its simplest formula. In 100 g of aspirin, there are 60. 0 g C, 4. 5 g H, and 35. 5 g O. Timberlake Lecture. PLUS 28

Solution EF-5 60. 0 g C x 4. 5 g H ______= ______ mol C x ______ = _______mol H 35. 5 g O x ______ = _______mol O Timberlake Lecture. PLUS 29

Solution EF-5 60. 0 g C x 1 mol C = 5. 00 mol C = 4. 5 mol H = 2. 22 mol O 12. 0 g C 4. 5 g H x 1 mol H 1. 01 g H 35. 5 g O x 1 mol O 16. 0 g O Timberlake Lecture. PLUS 30

Divide by the smallest # of moles. 5. 00 mol C = ________ mol O 4. 5 mol H = ______ mol O ________ 2. 22 mol O = ________ mol O Are are the results whole numbers? _____ Timberlake Lecture. PLUS 31

Divide by the smallest # of moles. 5. 00 mol C = ___2. 25__ 2. 22 mol O 4. 5 mol H 2. 22 mol O = ___2. 00__ 2. 22 mol O = ___1. 00__ 2. 22 mol O Are are the results whole numbers? _____ Timberlake Lecture. PLUS 32

Finding Subscripts A fraction between 0. 1 and 0. 9 must not be rounded. Multiply all results by an integer to give whole numbers for subscripts. (1/2) (1/3) (1/4) (3/4) 0. 5 0. 333 0. 25 0. 75 x 2 x 3 x 4 = = Timberlake Lecture. PLUS 1 1 1 3 33

Multiply everything x 4 C: 2. 25 mol C x 4 = 9 mol C H: 2. 0 mol H x 4 = 8 mol H O: 1. 00 mol O x 4 = 4 mol O Use the whole numbers of mols as the subscripts in the simplest formula C 9 H 8 O 4 Timberlake Lecture. PLUS 34

Learning Check EF-6 A compound is 27. 4% S, 12. 0% N and 60. 6 % Cl. If the compound has a molar mass of 351 g/mol, what is the molecular formula? Timberlake Lecture. PLUS 35

Solution EF 6 0. 853 mol S /0. 853 = 1 S 0. 857 mol N /0. 853 = 1 N 1. 71 mol Cl /0. 853 = 2 Cl Empirical formula = SNCl 2 = 117. 1 g/EF Mol. Mass/ Empirical mass 351/117. 1 = 3 Molecular formula = S 3 N 3 Cl 6 Timberlake Lecture. PLUS 36