Empirical and Molecular Formulas Definitions n Empirical formula





















- Slides: 24

Empirical and Molecular Formulas

Definitions n Empirical formula – the lowest whole-number ratio of the atoms of the elements in a compound. n Molecular formula – chemical formula that shows the actual number of atoms in a compound.

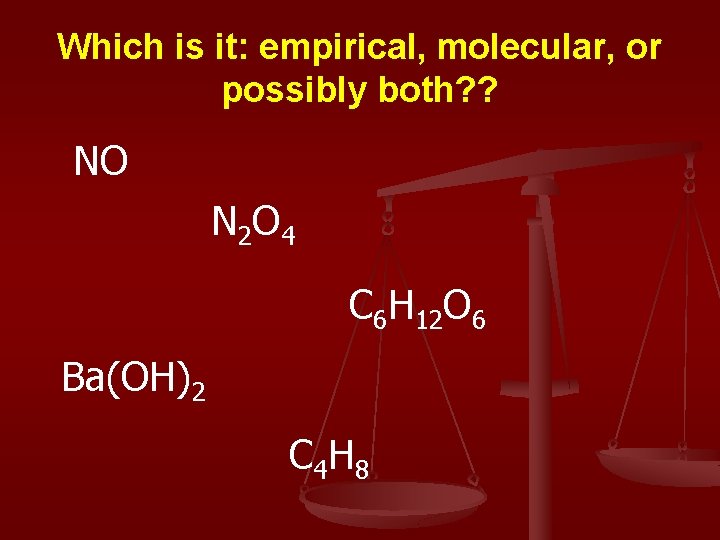
Which is it: empirical, molecular, or possibly both? ? NO N 2 O 4 C 6 H 12 O 6 Ba(OH)2 C 4 H 8

Empirical Formulas n The simplest whole-number ratio of atoms in a compound. H 2 O 2 Both are divisible by 2. H 2 O Already simplified C 6 H 12 O 6 Ca. Cl 2 All are divisible by 6. Correct ionic formulas are always empirical. HO H 2 O Ca. Cl 2

Finding an Empirical Formula 1. Grams mole 2. Divide by smallest 3. Round ‘til whole An unknown compound contains: 0. 0806 grams of C 0. 01353 grams of H 0. 1074 grams of O What is the empirical formula of the

Step 1: Change grams to moles 0. 0806 g C 1 mol C 12. 01 g C 0. 0068 mol C 0. 01353 g H 0. 1074 g O 0. 01353 g H 1 mol H 0. 1074 g O 1 mol O 1. 01 g H 16. 00 g O 0. 01340 mol H 0. 0067 mol O

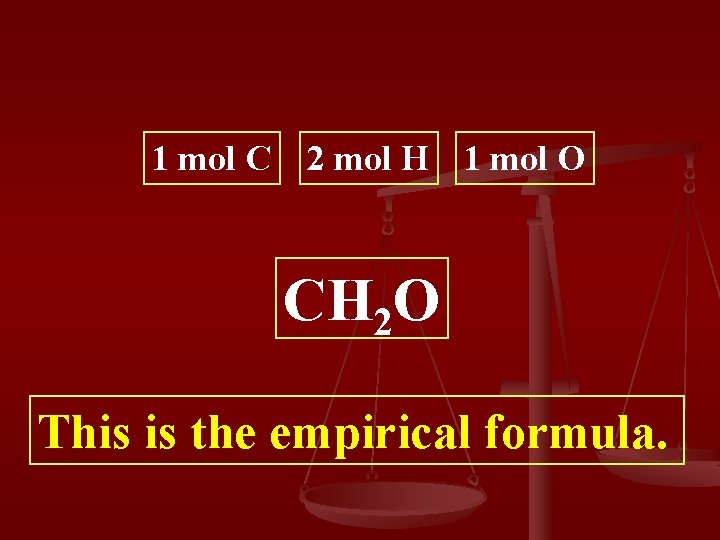
Step 2: Divide by smallest mole value Step 3: Round to the whole 0. 0068 mol C 0. 0067 0. 01340 mol H 0. 0067 mol O 0. 0067 1. 0149 mol C 2 mol H 1 mol O 1 mol C 2 mol H 1 mol O

1 mol C 2 mol H 1 mol O CH 2 O This is the empirical formula.

n Let’s try another – This time, using percent composition!! n

An unknown compound is known to have the following composition: 11. 11% hydrogen 88. 89% oxygen What is the empirical formula of the compound? In these problems we will choose to use a 100 gram sample. So, in this sample we have 11. 11 grams H and 88. 89 grams O.

Step 1: Change grams to moles. 11 g H 88. 89 g O 1 mol H = 11. 00 mol H 1. 01 g H 1 mol O 16. 00 g O = 5. 56 mol O

Step 2: Divide by smallest mole value. Step 3: Round to a whole number. 11. 00 mol H 5. 56 2 5. 56 mol O 5. 56 1

The ratio of moles of hydrogen to moles of oxygen is 2: 1 H 2 O

Going one step further… Finding molecular formulas from empirical formulas. Remember: The empirical formula is the simplest whole number ratio; whereas, the molecular formula shows the actual number of each atom. Step 1: Find molar mass of empirical formu Step 2: Divide the molar mass of the compound by the molar mass of the empirical formula. Step 3: Multiply the empirical formula by that number.

Practice Problem CH 2 O is the empirical formula. The molar mass of the actual compound is 180 g/mol. What is the molecular formula?

Step 1: Find molar mass of empirical form (1 x 12. 01) + (2 x 1. 01) + (1 x 16. 00) = 30. 03 g/mol Step 2: Divide the molar mass of the compound by the molar mass of the empirical formula. 180 g / 30 g = 6 Step 3: Multiply the empirical formula by that number. 6 (CH 2 O) = C 6 H 12 O 6 The Molecular Formula

You try one… n n Empirical formula – CH 4 The actual molecular mass is 64 g. Find the molar mass of empirical formula C + 4(H) = 16 g Divide molecular mass by empirical mass 64 g / 16 g = 4 Multiply the empirical formula by that number 4 (CH 4) C 4 H 16 molecular formula

One more thing to mention… n Hydrates vs. Anhydrates n Hydrates attached – salts with water molecules n Ba. Cl 2 • 2 H 2 O n Name the salt, then use a prefix + word “hydrate”—Barium Chloride Dihydrate n Anhydrates n Ba. Cl 2 – the salt without its water

Finding the Formulas of Hydrates --Similar to Finding an Empirical Formula n 1. Find the mass of the water by subtraction. n n The mass of the hydrate and the anhydrate will be given. 2. Change g to mol Mass of water to mol n Mass of salt to mol n n n 3. Divide by the smallest mole value 4. Round to a whole # if necessary.

Finding the Formulas of Hydrates Ba. Cl 2 • X H 2 O Hydrate mass (mass with water) – 24. 40 g Anhydrate mass (mass w/o water) - 20. 80 g Step 1: Find the mass of the water. Mass H 2 O = 24. 40 g – 20. 80 g = 3. 60 g H 2 O For the formula we want the ratio of moles of water to moles of anhydrate.

Step 2: Change grams to moles Grams of Salt 20. 80 g Ba. Cl 2 1 mol = 0. 1 mol Ba. Cl 2 208 g Ba. Cl 2 Grams of Water 3. 60 g H 2 O 1 mol 18 g H 2 O = 0. 2 mol H 2 O

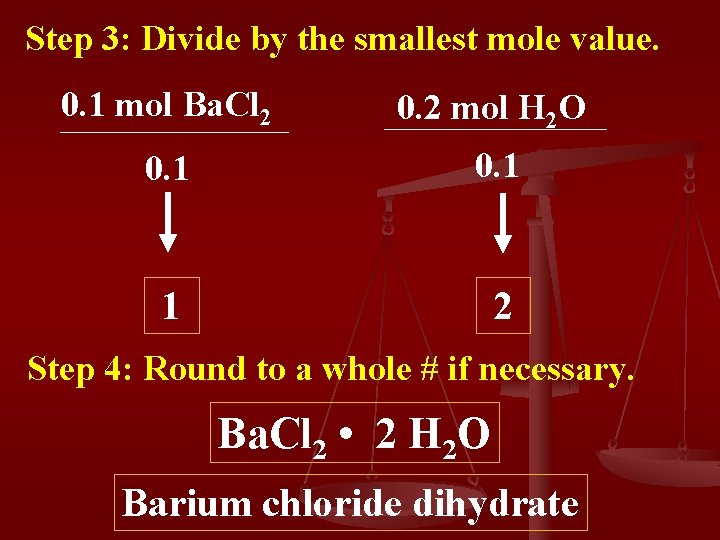
Step 3: Divide by the smallest mole value. 0. 1 mol Ba. Cl 2 0. 2 mol H 2 O 0. 1 1 2 Step 4: Round to a whole # if necessary. Ba. Cl 2 • 2 H 2 O Barium chloride dihydrate

Calculating Percent Water in a Hydrate Calculating the percent water in a hydrate is similar to calculating percent composition: % H 2 O = mass of the water mass of the hydrate x 100

Example Problem In the lab, a five gram sample of hydrous copper (II) nitrate is heated. If 3. 9 grams of the anhydrous salt remains, what is the percent water in the hydrate? Mass of water: 5 g – 3. 9 g = 1. 1 g H 2 O Mass of hydrate: 5 g (1. 1 g H 2 O / 5 g total) x 100 = 22 % H 2 O