Empirical and Molecular Formulas Empirical vs Molecular Formulas














- Slides: 15

Empirical and Molecular Formulas

Empirical vs. Molecular Formulas n n Empirical Formula – the simplest formula. A formula with the smallest whole-number ratio of the elements that make up a compound. Molecular Formula – the true formula. A formula that specifies the actual number of atoms of each element in one molecule of a compound.

Empirical Formula n n May or may not be the same as the molecular formula Molecular formula is always a simple multiple (ratio) of the empirical formula n ex. H O 2 2 n n Empirical formula is HO Molecular formula is TWO times the empirical formula

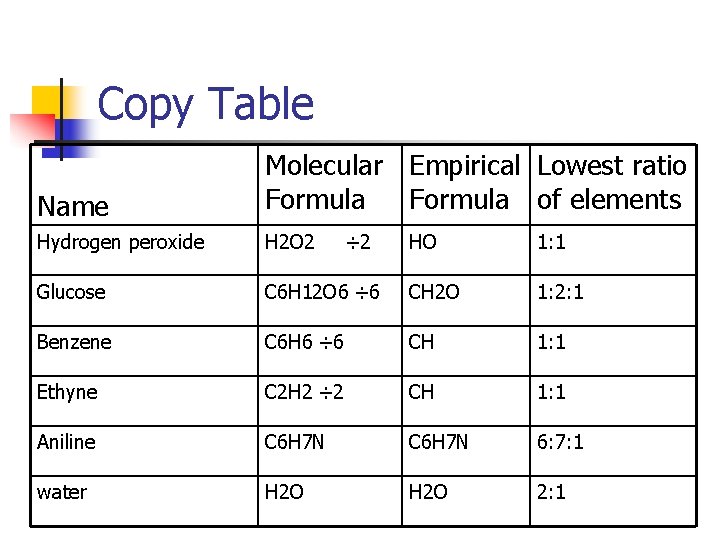
Copy Table Name Molecular Empirical Lowest ratio Formula of elements Hydrogen peroxide H 2 O 2 Glucose ÷ 2 HO 1: 1 C 6 H 12 O 6 ÷ 6 CH 2 O 1: 2: 1 Benzene C 6 H 6 ÷ 6 CH 1: 1 Ethyne C 2 H 2 ÷ 2 CH 1: 1 Aniline C 6 H 7 N 6: 7: 1 water H 2 O 2: 1

How to calculate an empirical formula STEP 1: You will be given either masses or percent composition. n STEP 2: If you are given % composition, turn it into grams by assuming a 100. 0 g sample. NOTE: If you are given mass, you do not need to do this step. n STEP 3: Convert the masses to the number of moles of each element. n

n n STEP 4: Figure out the proportion of moles of each element in the compound by dividing each by the smallest number of moles. STEP 5: If step 4 resulted in whole numbers, you are done! However, if there were decimals, you will need to multiply by small, whole numbers until you have whole numbers.

A way to remember those steps: COPY THIS DOWN A Poem by Joel Thompson: n n Percent to mass Mass to mole Divide by small Multiply ‘til whole

An example: n STEP 1: n n STEP 2: n n Compound is 40. 05% S and 59. 95% O I assume 100 g of the compound, so it is: n 40. 05 g S and 59. 95 g O STEP 3: n n 40. 05 g S • (1 mol S/32. 07 g S) = 1. 249 mol S 59. 95 g O • (1 mol O/16. 00 g O) = 3. 747 mol O

Continued… n STEP 4: n n STEP 5: n n 1. 249 mol S : 3. 747 mol O Divide each by 1. 249 (smallest number in ratio) 1 mol S : 3 mol O SO 3 You are done! The compound is sulfur trioxide.

n Molecular Formula

Molecular Formula n n Molecular Formula – this tells us how many atoms of each type there really are in the compound. Can two substances have the same empirical formula but be different? n n YES! Benzene vs. acetylene: C 6 H 6 vs. C 2 H 2 What is their empirical formula? How is this different from ionic compounds?

Calculating Molecular Formula n STEP 1: n n STEP 2: n n Calculate the empirical mass (mass of the empirical formula). STEP 3: n n You will be given the molar mass of the compound and the empirical formula. Divide the given molar mass by the empirical mass. You should get a small whole number. STEP 4: n Multiply the subscripts of the empirical formula with the number obtained.

Step #3 in more detail Ratio = Molar Mass (Actual) Molar Mass (EF) Your ratio will give you a whole number ratio.

Example n The empirical formula of a compound is found to be CH 2 O. After careful analysis the molar mass is found to be 180. 18 g. Determine the molecular formula of this compound.

Molecular Formula Example n STEP 1: n n STEP 2: n n The empirical mass is 12. 01 g + 2*1. 01 g + 16. 00 g = 30. 03 g STEP 3: n n The empirical formula is CH 2 O and the molar mass is 180. 18 g. Ratio= 180. 18 g/ 30. 03 g = 6 STEP 4: n CH 2 O becomes C 6 H 12 O 6