Empirical and Molecular Formulas Empirical vs Molecular Empirical














- Slides: 15

Empirical and Molecular Formulas

Empirical vs. Molecular �Empirical formula: a formula that shows the simplest ratio of elements present in the compound �Molecular formula: formula that states the number of atoms in a particular molecule (can be reduced further)

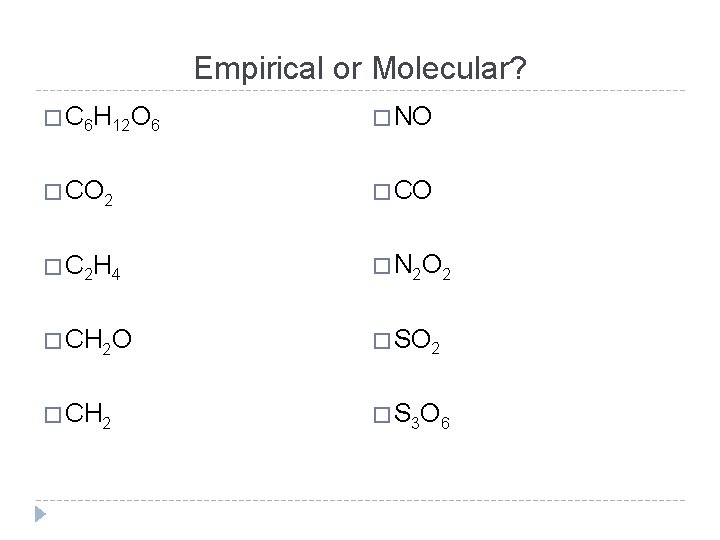
Empirical or Molecular? � C 6 H 12 O 6 � NO � CO 2 � CO � C 2 H 4 � N 2 O 2 � CH 2 O � SO 2 � CH 2 � S 3 O 6

Question 1 � Analysis of a sample of a covalent compound showed that it contained 85. 6% carbon and 14. 4% hydrogen. What is the empirical formula for this compound?

Question 2 � What is the empirical formula for a compound containing 68. 3% lead, 10. 6% sulfur and the remainder oxygen?

Question 3 �A compound contains sulfur, oxygen, and chlorine. Analysis shows that it contains 26. 95% sulfur, and 59. 61% chlorine. What is the simplest formula for this compound?

Question 4 �A compound contains carbon, oxygen, and hydrogen. Analysis of a sample showed that it contained by mass 68. 83% carbon and 4. 96% hydrogen. What is the simplest formula for this compound?

A compound with an empirical formula of CO 2 and a molar mass of 132 grams per mole. �What is the molecular formula of this compound?

Question 5 �A compound contains 87. 5% nitrogen and 12. 5% hydrogen. Its molecular weight is found to be 32 g/mol. What is its molecular formula?

Question 6 �A compound contains only carbon, hydrogen, and oxygen. Analysis of a sample showed that it contained 54. 52% C and 9. 15% H. Its molecular weight was determined to be approximately 88 g/mol. What is its molecular formula?

Empirical Formulas w/ Mass � Determine the empirical formula for a hydrocarbon if the complete combustion of a sample produces 5. 28 g of CO 2 and 1. 62 g of H 2 O � C 2 H 3

Practice 1 � Determine the simplest formula for a hydrocarbon if the complete combustion of a sample produces 3. 96 g of CO 2 and 2. 16 g of H 2 O. � C 3 H 8

Practice 2 �A compound is known to contain only carbon, hydrogen, and oxygen. If the complete combustion of a 0. 150 g sample of this compound produces 0. 225 g of CO 2 and 0. 0614 g of H 2 O, what is the empirical formula of this compound? � C 3 H 4 O 3

Practice 3 � The complete combustion of a 0. 2864 g sample of a compound yielded 0. 420 g of CO 2 and 0. 172 g of H 2 O. The molecular weight is determined to be approximately 60 g/mol. What is the molecular formula of this compound if it contains only carbon, hydrogen, and oxygen? � C 2 H 4 O 2

Practice 4 � 41 g of CO 2, 25 g of H 2 O, 29. 90 g of Total Compound containing C, H, and O � CH 3 O