Empirical Molecular Formulas composition Empirical Formulas H 2


![Step 4] If not whole #s, multiply by integer. Ca 1. 5 P Step 4] If not whole #s, multiply by integer. Ca 1. 5 P](https://slidetodoc.com/presentation_image/586fb35a2b899627e016effd3986def3/image-4.jpg)


![Step 3] Calculate the mole ratio by dividing by the smallest number of moles: Step 3] Calculate the mole ratio by dividing by the smallest number of moles:](https://slidetodoc.com/presentation_image/586fb35a2b899627e016effd3986def3/image-7.jpg)










- Slides: 17

Empirical & Molecular Formulas % composition

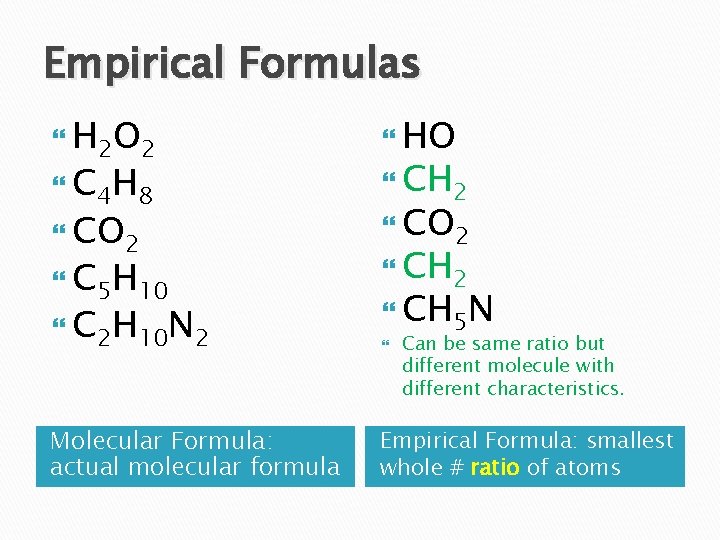
Empirical Formulas H 2 O 2 HO CO 2 C 4 H 8 C 5 H 10 C 2 H 10 N 2 Molecular Formula: actual molecular formula CH 2 CH 5 N Can be same ratio but different molecule with different characteristics. Empirical Formula: smallest whole # ratio of atoms

Finding Empirical Formulas What is the EF of a compound if a 100. 0 g sample contains 66. 0 g Ca and 34. 0 g P? Step 1] Find mass of each element in compound. Ca = 66. 0 g P = 34. 0 g Step 2] Convert grams to moles Ca: 66. 0 g x 1 mole = 1. 65 moles /1. 10 = 1. 5 40. 08 g P: 34. 0 g x 1 mole = 1. 10 moles /1. 10 = 1. 0 30. 97 g Step 3] Divide all mole values by smallest # Ca 1. 5 P 1
![Step 4 If not whole s multiply by integer Ca 1 5 P Step 4] If not whole #s, multiply by integer. Ca 1. 5 P](https://slidetodoc.com/presentation_image/586fb35a2b899627e016effd3986def3/image-4.jpg)
Step 4] If not whole #s, multiply by integer. Ca 1. 5 P 1 x 2 = Ca 3 P 2 Unless it is almost a whole # you need to find the best representative ratio.

Calculating Empirical Formulas One can calculate the empirical formula from the percent composition

compound para-aminobenzoic acid (you may have seen it listed as PABA on your bottle of sunscreen) is composed of carbon (61. 31%), hydrogen (5. 14%), nitrogen (10. 21%), and oxygen (23. 33%). Find the empirical formula of PABA. � The � Step 1 -2] Assuming 100. 00 g of para-aminobenzoic acid, 61. 31 g x 1 mol 12. 01 g � C: � H: 5. 14 g x 1 mole 1. 01 g � N: 10. 21 g x 1 mole 14. 01 g = 0. 7288 mol N � O: 23. 33 g x 1 mole = 1. 456 mol O 16. 0 g = 5. 105 mol C = 5. 09 mol H
![Step 3 Calculate the mole ratio by dividing by the smallest number of moles Step 3] Calculate the mole ratio by dividing by the smallest number of moles:](https://slidetodoc.com/presentation_image/586fb35a2b899627e016effd3986def3/image-7.jpg)
Step 3] Calculate the mole ratio by dividing by the smallest number of moles: C: 5. 105/0. 7288 = 7. 005 7 H: 5. 09/0. 7288 = 6. 984 7 N: 0. 7288/0. 7288 = 1. 000 O: 1. 456/0. 7288 = 2. 001 2

Calculating Empirical Formulas These are the subscripts for the EF: C 7 H 7 NO 2

Combustion Analysis Compounds containing C, H and O are routinely analyzed through combustion in a chamber like this ◦ C is determined from the mass of CO 2 produced ◦ H is determined from the mass of H 2 O produced ◦ O is determined by difference after the C and H have been determined

SAMPLE EXERCISE 3. 13 Calculating an Empirical Formula Ascorbic acid (vitamin C) contains 40. 92% C, 4. 58% H, and 54. 50% O by mass. What is the empirical formula of ascorbic acid? Solve: We first assume, for simplicity, that we have exactly 100 g of material (although any mass can be used). In 100 g of ascorbic acid, we have Second, we calculate the number of moles of each element:

SAMPLE EXERCISE 3. 13 continued Third, we determine the simplest whole-number ratio of moles by dividing each number of moles by the smallest number of moles, 3. 406: The ratio for H is too far from 1 to attribute the difference to experimental error; in fact, 1 it is 3 quite close to 1–. This suggests that if we multiply the ratio by 3, we will obtain whole numbers: The whole-number mole ratio gives us the subscripts for the empirical formula: Check: It is reassuring that the subscripts are moderately sized whole numbers. Otherwise, we have little by which to judge the reasonableness of our answer.

Email from NAU 9/24/12 Hey Ms. Brunet, Things are going pretty well here in college. I really love it here. Expect for the weather, which is quite unpredictable at times. Currently I have decided to be major in Electrical Engineering and I'm taking 15 units. Three of those units includes a Chemistry lecture class. This class is seriously a joke and I honestly don't know how this professor has a doctorate. I learned more in one week in your class then I have in 5 weeks of this class. Everything that has been taught in this class I already know. We only had to memorize 7 polyatomic ions and we don't even have to memorize the solubility rules. I can truly say that AP Chem was about 10 times harder than this class. But this professor makes the class so confusing. I got my test back tomorrow and I started realizing that three of the questions that were marked wrong for the multiple choice were actually correct. For example, this question. . The percent composition by mass of a compound is 76. 0% C, 12. 8% H, and 11. 2% O. The molar mass of this composition is 284. 5 g/mol. What is the molecular formula of the compound? a. C 10 H 6 O b. C 9 H 18 O c. C 16 H 28 O 4 d. C 20 H 12 O 2 e. C 18 H 36 O 2 I worked it out and got the answer e, which is right, but the correct answer was b. I even asked him after class but he just got frustrated and told us to leave. This guy makes no sense. I hope it gets better some how. Well anyways, how is your AP class going? How is Cypress this year? Robert Kasper Class of 2011

SAMPLE EXERCISE 3. 15 Determing Empirical Formula by Combustion Analysis Isopropyl alcohol, a substance sold as rubbing alcohol, is composed of C, H, and O. Combustion of 0. 255 g of isopropyl alcohol produces 0. 561 g of CO 2 and 0. 306 g of H 2 O. Determine the empirical formula of isopropyl alcohol. The total mass of the sample, 0. 255 g, is the sum of the masses of the C, H, and O. Thus, we can calculate the mass of O as follows:

SAMPLE EXERCISE 3. 15 continued We then calculate the number of moles of C, H, and O in the sample: To find the empirical formula, we must compare the relative number of moles of each element in the sample. The relative number of moles of each element is found by dividing each number by the smallest number, 0. 0043. The mole ratio of C : H : O so obtained is 2. 98 : 7. 91 : 1. 00. The first two numbers are very close to the whole numbers 3 and 8, giving the empirical formula C 3 H 8 O.

Percent Composition One can find the percentage of the mass of a compound that comes from each of the elements in the compound by using this equation: % element = (number of atoms)(atomic weight) (FW of the compound) x 100

Percent Composition So the percentage of carbon in ethane (C 2 H 6) is… (2)(12. 0 amu) %C = (30. 0 amu) = 24. 0 amu 30. 0 amu = 80. 0% x 100

SAMPLE EXERCISE 3. 6 Calculating Percentage Composition Calculate the percentage of carbon, hydrogen, and oxygen (by mass) in C 12 H 22 O 11. Check: The percentages of the individual elements must add up to 100%, which they do in this case. We could have used more significant figures for our atomic weights, giving more significant figures for our percentage composition, but we have adhered to our suggested guideline of rounding atomic weights to one digit beyond the decimal point.