14 11 Alkane Synthesis Using Organocopper Reagents Lithium









![Example CO OC Ni CO CO Nickel carbonyl Ni has the electron configuration [Ar]4 Example CO OC Ni CO CO Nickel carbonyl Ni has the electron configuration [Ar]4](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-22.jpg)
![(Benzene)tricarbonylchromium OC Cr CO CO Cr has the electron configuration [Ar]4 s 23 d (Benzene)tricarbonylchromium OC Cr CO CO Cr has the electron configuration [Ar]4 s 23 d](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-23.jpg)
![Ferrocene Fe Fe 2+ has the electron configuration [Ar]3 d 6 Each cyclopentadienide anion Ferrocene Fe Fe 2+ has the electron configuration [Ar]3 d 6 Each cyclopentadienide anion](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-24.jpg)













- Slides: 48

14. 11 Alkane Synthesis Using Organocopper Reagents

Lithium Dialkylcuprates Lithium dialkylcuprates are useful synthetic reagents. They are prepared from alkyllithiums and a copper(I) halide. 2 RLi + Cu. X R 2 Cu. Li + Li. X [customary solvents are diethyl ether and tetrahydrofuran (THF)]

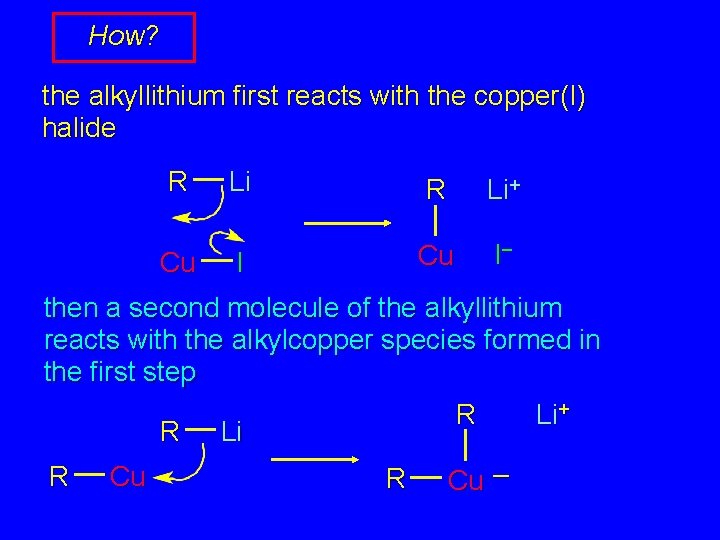
How? the alkyllithium first reacts with the copper(I) halide R Cu Li R Li+ I Cu I– then a second molecule of the alkyllithium reacts with the alkylcopper species formed in the first step R Li+ R Li R Cu –

Lithium diorganocuprates are used to form C—C bonds R 2 Cu. Li + R'X R R' + RCu + Li. X Ar 2 Cu. Li + R'X Ar R' + Ar. Cu + Li. X

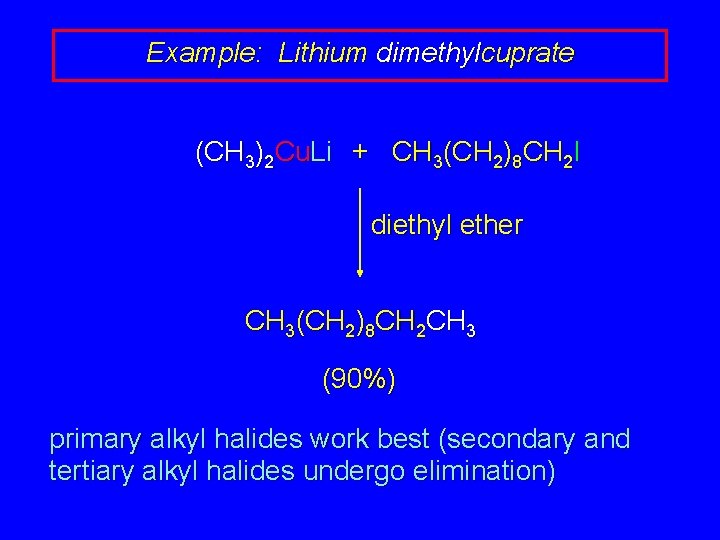
Example: Lithium dimethylcuprate (CH 3)2 Cu. Li + CH 3(CH 2)8 CH 2 I diethyl ether CH 3(CH 2)8 CH 2 CH 3 (90%) primary alkyl halides work best (secondary and tertiary alkyl halides undergo elimination)

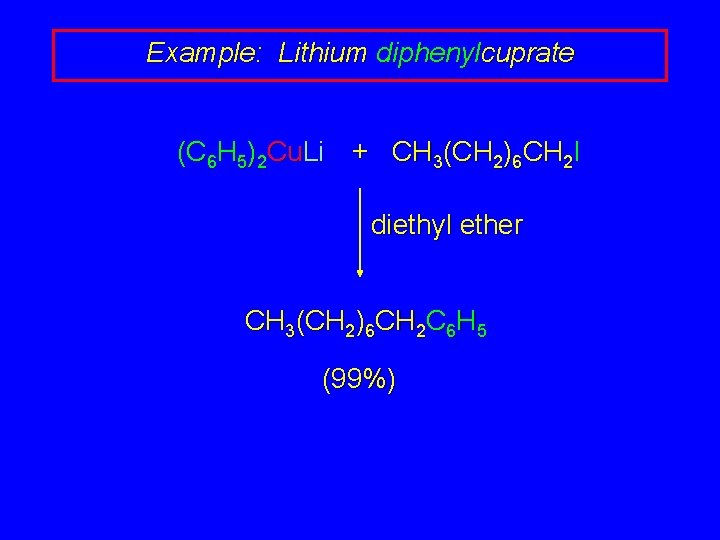
Example: Lithium diphenylcuprate (C 6 H 5)2 Cu. Li + CH 3(CH 2)6 CH 2 I diethyl ether CH 3(CH 2)6 CH 2 C 6 H 5 (99%)

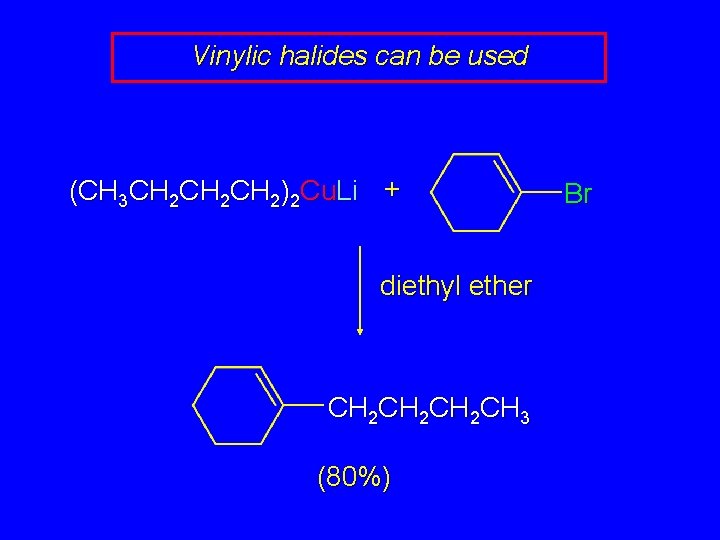
Vinylic halides can be used (CH 3 CH 2 CH 2)2 Cu. Li + diethyl ether CH 2 CH 2 CH 3 (80%) Br

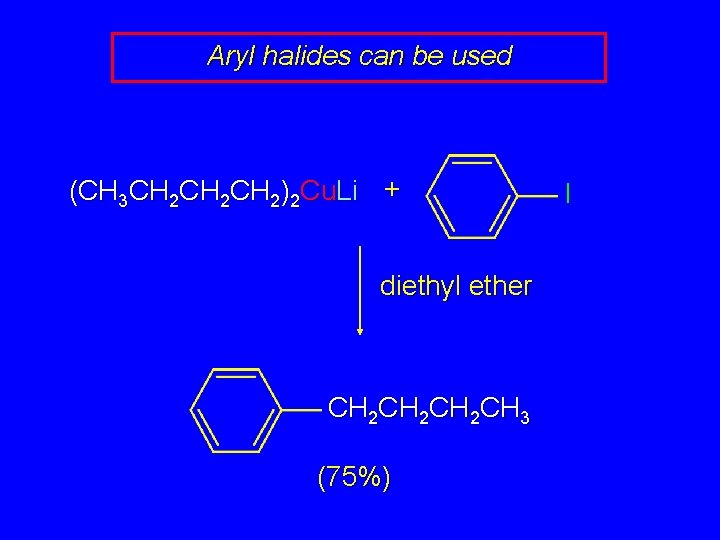
Aryl halides can be used (CH 3 CH 2 CH 2)2 Cu. Li + diethyl ether CH 2 CH 2 CH 3 (75%) I

14. 12 An Organozinc Reagent for Cyclopropane Synthesis

Iodomethylzinc iodide formed by reaction of diiodomethane with zinc that has been coated with copper (called zinc-copper couple) CH 2 I 2 + Zn Cu ICH 2 Zn. I reacts with alkenes to form cyclopropanes reaction with alkenes is called the Simmons-Smith reaction

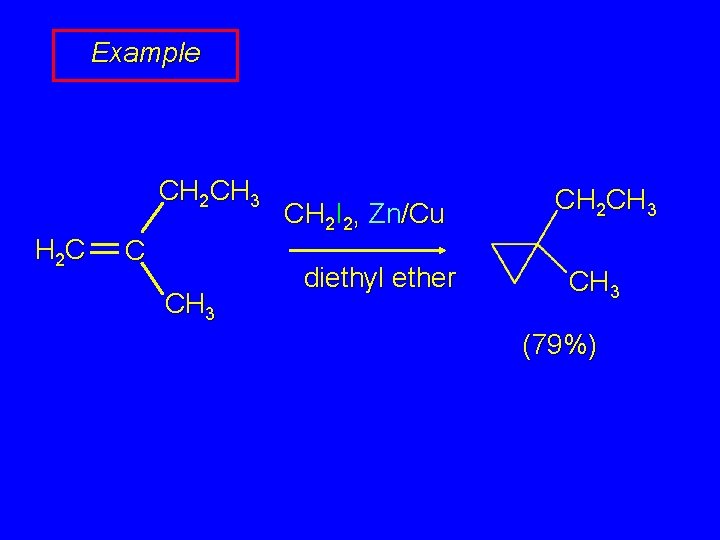
Example CH 2 CH 3 H 2 C C CH 3 CH 2 I 2, Zn/Cu diethyl ether CH 2 CH 3 (79%)

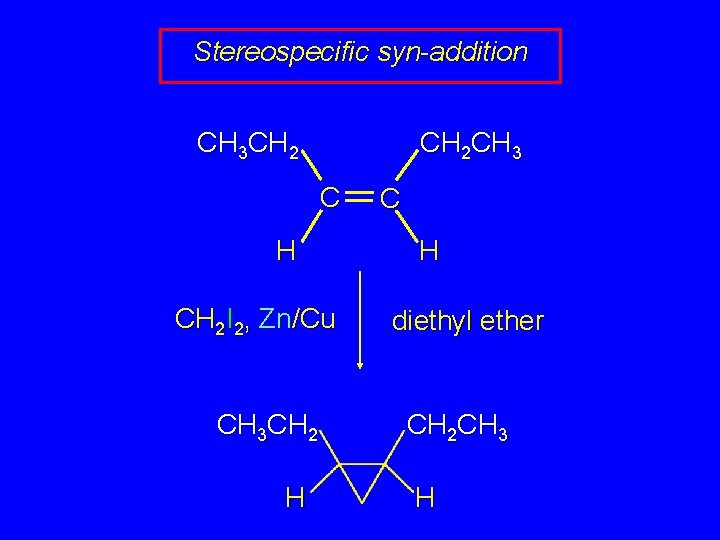
Stereospecific syn-addition CH 3 CH 2 CH 3 C H CH 2 I 2, Zn/Cu CH 3 CH 2 H C H diethyl ether CH 2 CH 3 H

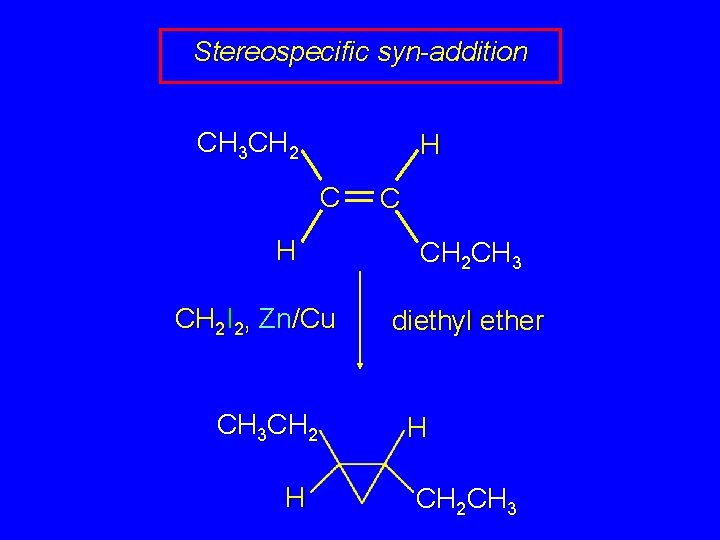
Stereospecific syn-addition CH 3 CH 2 H CH 2 I 2, Zn/Cu CH 3 CH 2 H C CH 2 CH 3 diethyl ether H CH 2 CH 3

14. 13 Carbenes and Carbenoids

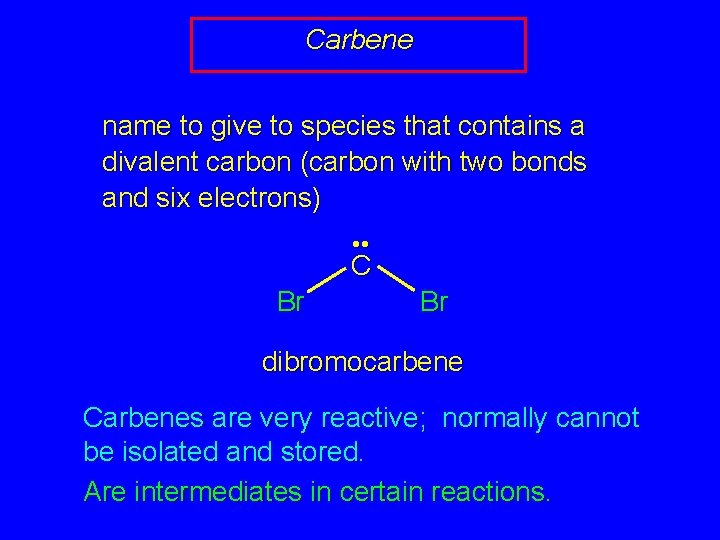
Carbene name to give to species that contains a divalent carbon (carbon with two bonds and six electrons) • • C Br Br dibromocarbene Carbenes are very reactive; normally cannot be isolated and stored. Are intermediates in certain reactions.

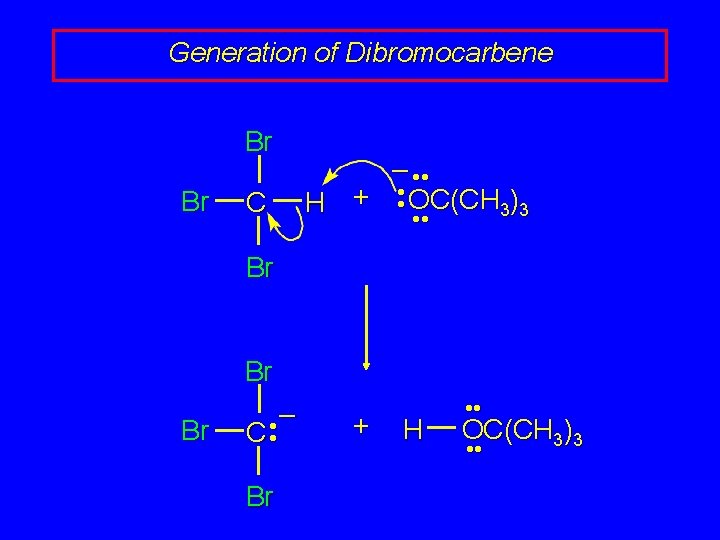
Generation of Dibromocarbene Br Br C – • • OC(CH ) + H 3 3 • • Br Br Br • – C • Br + H • • OC(CH 3)3 • •

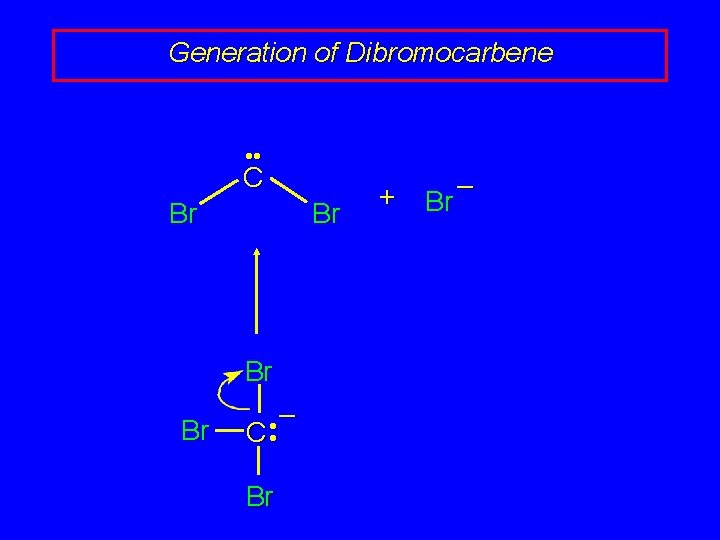
Generation of Dibromocarbene • • C Br Br – • C • Br + Br –

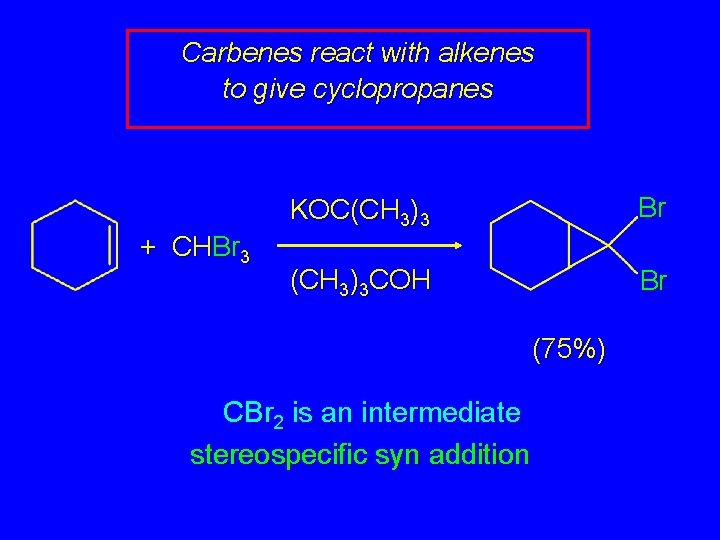
Carbenes react with alkenes to give cyclopropanes + CHBr 3 KOC(CH 3)3 Br (CH 3)3 COH Br (75%) CBr 2 is an intermediate stereospecific syn addition

14. 14 Transition-Metal Organic Compounds

Introduction Many organometallic compounds derived from transition metals have useful properties. Typical transition metals are iron, nickel, chromium, platinum, and rhodium.

18 -Electron Rule The number of ligands attached to a metal will be such that the sum of the electrons brought by the ligands plus the valence electrons of the metal equals 18. When the electron-count is less than 18, metal is said to be coordinatively unsaturated and can take on additional ligands. 18 -Electron rule is to transition metals as the octet rule is to second-row elements.
![Example CO OC Ni CO CO Nickel carbonyl Ni has the electron configuration Ar4 Example CO OC Ni CO CO Nickel carbonyl Ni has the electron configuration [Ar]4](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-22.jpg)
Example CO OC Ni CO CO Nickel carbonyl Ni has the electron configuration [Ar]4 s 23 d 8 Ni has 10 valence electrons Each CO uses 2 electrons to bond to Ni 4 CO contribute 8 valence electrons 10 + 8 = 18
![Benzenetricarbonylchromium OC Cr CO CO Cr has the electron configuration Ar4 s 23 d (Benzene)tricarbonylchromium OC Cr CO CO Cr has the electron configuration [Ar]4 s 23 d](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-23.jpg)
(Benzene)tricarbonylchromium OC Cr CO CO Cr has the electron configuration [Ar]4 s 23 d 4 Cr has 6 valence electrons Each CO uses 2 electrons to bond to Cr 3 CO contribute 6 valence electrons benzene uses its 6 electrons to bind to Cr.
![Ferrocene Fe Fe 2 has the electron configuration Ar3 d 6 Each cyclopentadienide anion Ferrocene Fe Fe 2+ has the electron configuration [Ar]3 d 6 Each cyclopentadienide anion](https://slidetodoc.com/presentation_image/6abb3973b223001135385a3ef81313da/image-24.jpg)
Ferrocene Fe Fe 2+ has the electron configuration [Ar]3 d 6 Each cyclopentadienide anion contributes 6 electrons Total 6 + 6 = 18 Organometallic compounds with cyclopentadienide ligands are called metallocenes.

14. 15 Homogeneous Catalytic Hydrogenation Wilkinson’s Catalyst

Wilkinson’s Catalyst Ni, Pt, Pd, and Rh can act as a heterogeneous catalyst in the hydrogenation of alkenes. However, tris(triphenylphosphine)rhodium chloride was found to be soluble in organic solvents. This catalyst was developed by Sir Geoffrey Wilkinson, who received a Nobel Prize in 1973.

Mechanism of Homogeneous Hydrogenation Steps 1 and 2: Catalyst is converted to the active form. This is the active form of the catalyst.

Mechanism of Homogeneous Hydrogenation Step 3: Alkene bonds to rhodium through electrons.

Mechanism of Homogeneous Hydrogenation Step 4: Rhodium-alkene complex rearranges.

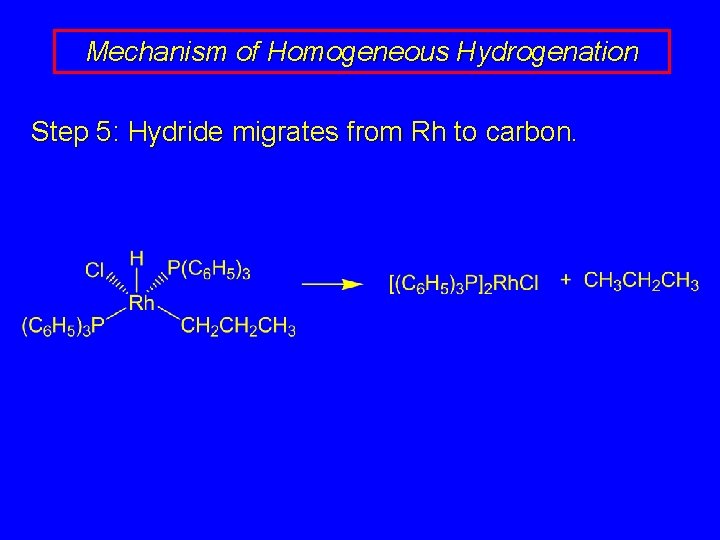
Mechanism of Homogeneous Hydrogenation Step 5: Hydride migrates from Rh to carbon.

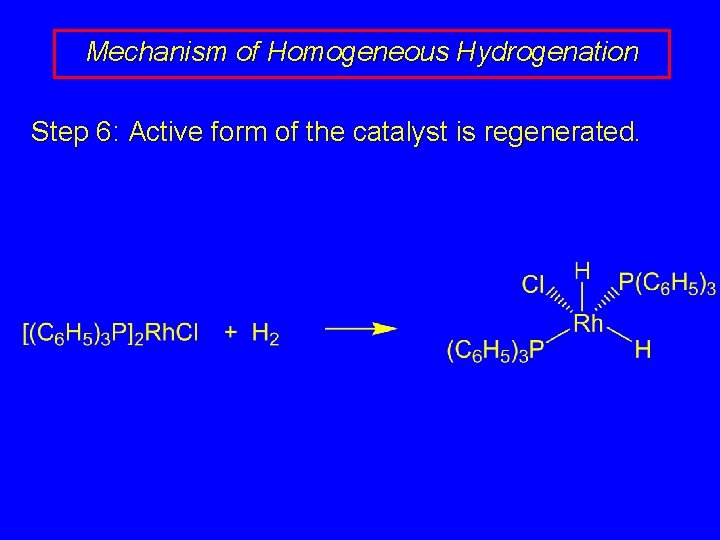
Mechanism of Homogeneous Hydrogenation Step 6: Active form of the catalyst is regenerated.

14. 16 Olefin Metathesis

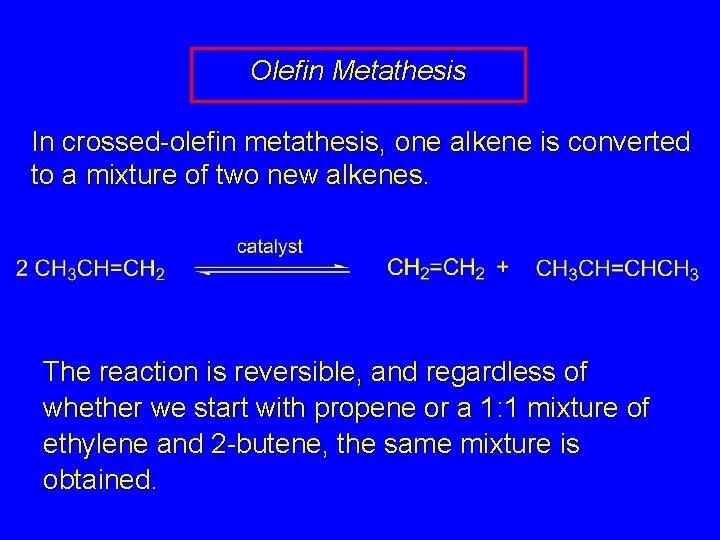
Olefin Metathesis In crossed-olefin metathesis, one alkene is converted to a mixture of two new alkenes. The reaction is reversible, and regardless of whether we start with propene or a 1: 1 mixture of ethylene and 2 -butene, the same mixture is obtained.

Olefin Metathesis The reaction is generally catalyzed a transition metal complex. Typically Ru, W, or Mo are used. Shown below is Grubb’s catalyst.

Ring-Opening Metathesis Ring-opening metathesis is used as a method of polymerization. Usually, it is applied most often when ring opening creates a relief of strain, as in some bicyclic alkenes.

14. 17 Ziegler-Natta Catalysis of Alkene Polymerization The catalysts used in coordination polymerization are transition-metal organic compounds.

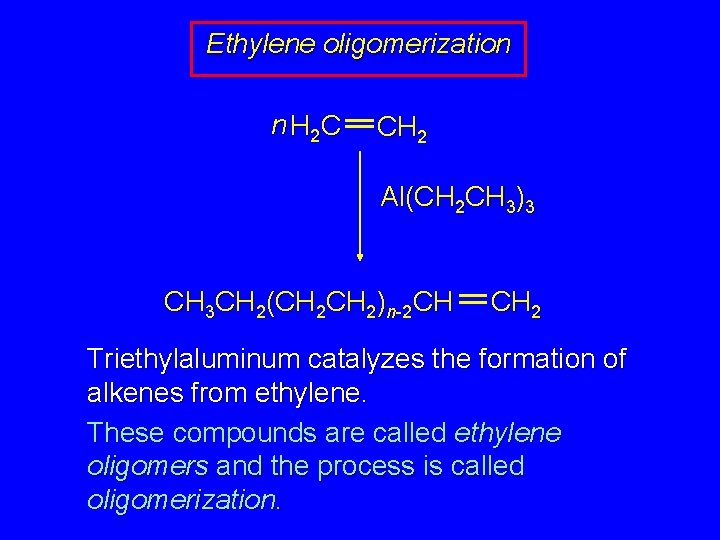
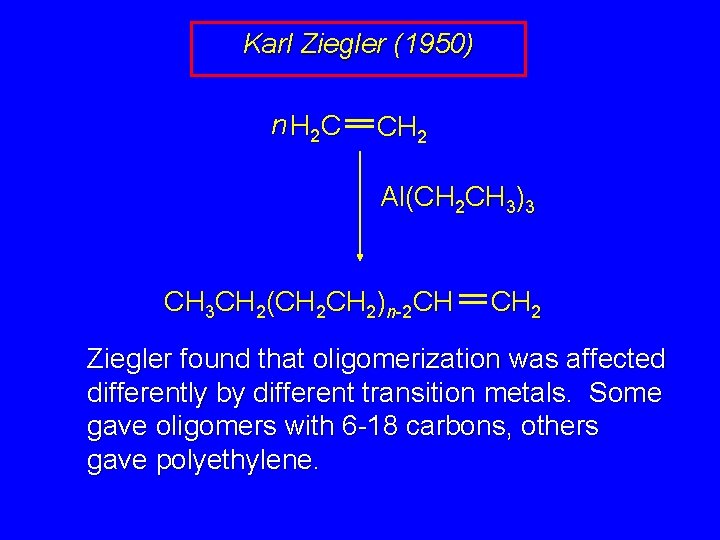
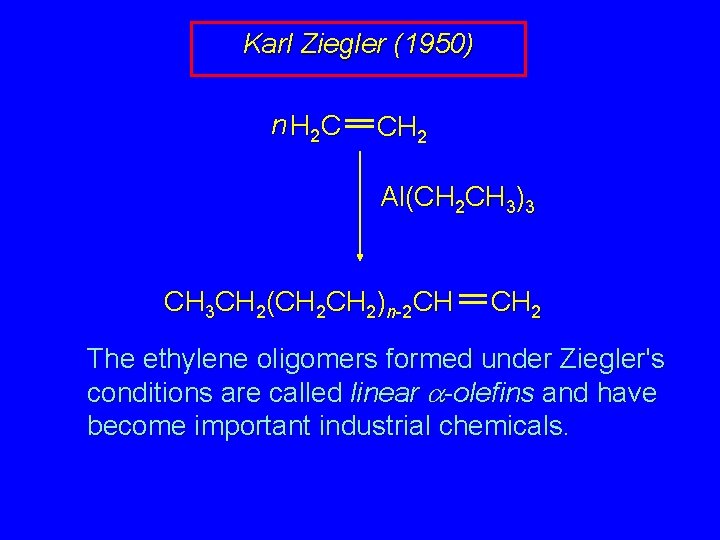
Ethylene oligomerization n H 2 C CH 2 Al(CH 2 CH 3)3 CH 3 CH 2(CH 2)n-2 CH CH 2 Triethylaluminum catalyzes the formation of alkenes from ethylene. These compounds are called ethylene oligomers and the process is called oligomerization.

Karl Ziegler (1950) n H 2 C CH 2 Al(CH 2 CH 3)3 CH 3 CH 2(CH 2)n-2 CH CH 2 Ziegler found that oligomerization was affected differently by different transition metals. Some gave oligomers with 6 -18 carbons, others gave polyethylene.

Giulio Natta n H 2 C CHCH 3 Al(CH 2 CH 3)3 polypropylene Natta found that polymerization of propene under Ziegler's conditions gave mainly isotactic polypropylene. This discovery made it possible to produce polypropylene having useful properties.

Karl Ziegler (1950) n H 2 C CH 2 Al(CH 2 CH 3)3 CH 3 CH 2(CH 2)n-2 CH CH 2 The ethylene oligomers formed under Ziegler's conditions are called linear -olefins and have become important industrial chemicals.

Karl Ziegler (1950) n H 2 C CH 2 Al(CH 2 CH 3)3 CH 3 CH 2(CH 2)n-2 CH CH 2 The polyethylene formed under Ziegler's conditions is called high-density polyethylene and has, in many ways, more desirable properties than the polyethylene formed by free-radical polymerization.

Ziegler-Natta Catalysts Early Ziegler-Natta catalyst were a combination of Ti. Cl 4 and (CH 3 CH 2)2 Al. Cl, or Ti. Cl 3 and (CH 3 CH 2)3 Al. Currently used Ziegler-Natta catalyst combinations include a metallocene such as bis(cyclopentadienyl)zirconium dichloride. Cl Zr Cl

Ziegler-Natta Catalysts Early Ziegler-Natta catalyst were a combination of Ti. Cl 4 and (CH 3 CH 2)2 Al. Cl, or Ti. Cl 3 and (CH 3 CH 2)3 Al. Currently used Ziegler-Natta catalyst combinations include a metallocene such as bis(cyclopentadienyl)zirconium dichloride.

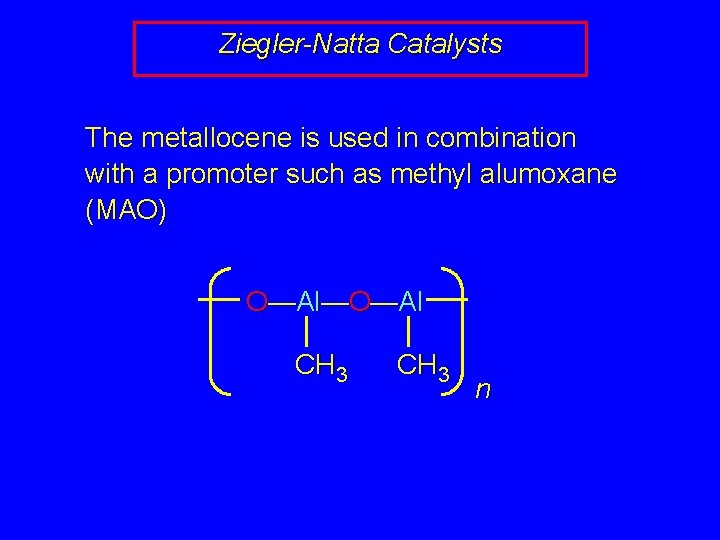
Ziegler-Natta Catalysts The metallocene is used in combination with a promoter such as methyl alumoxane (MAO) O—Al—O—Al CH 3 n

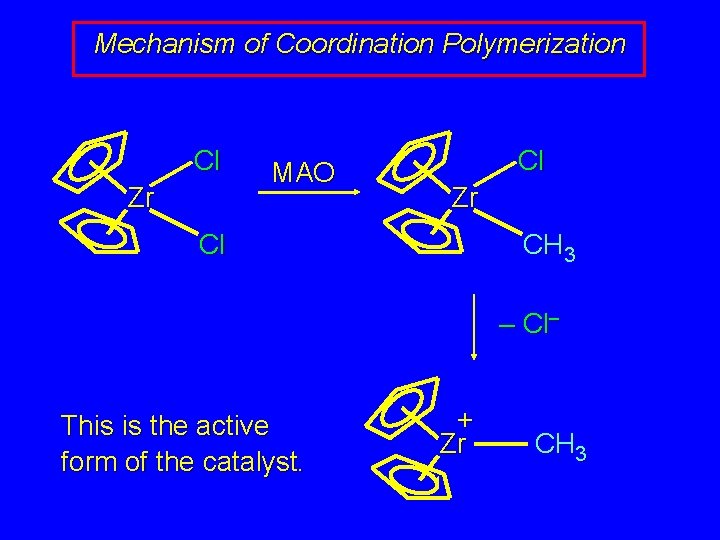
Mechanism of Coordination Polymerization Cl Zr MAO Cl Zr Cl CH 3 – Cl– This is the active form of the catalyst. + Zr CH 3

Mechanism of Coordination Polymerization CH 3 + Zr CH 2 H 2 C CH 3 + Zr CH 2 CH 3

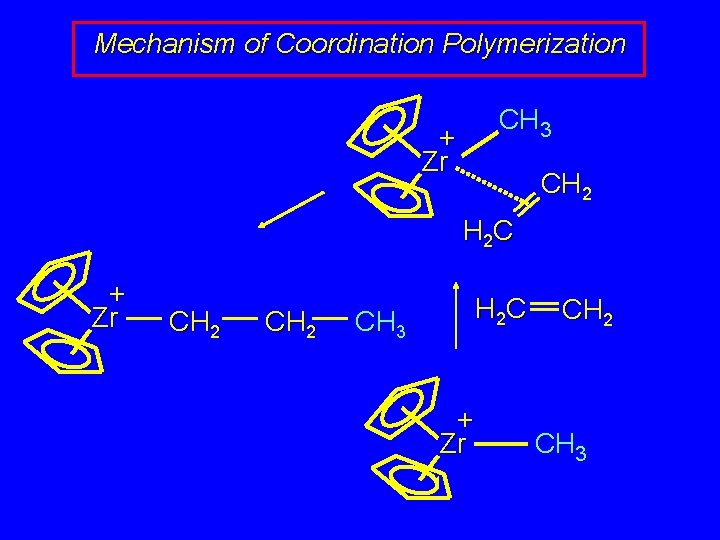
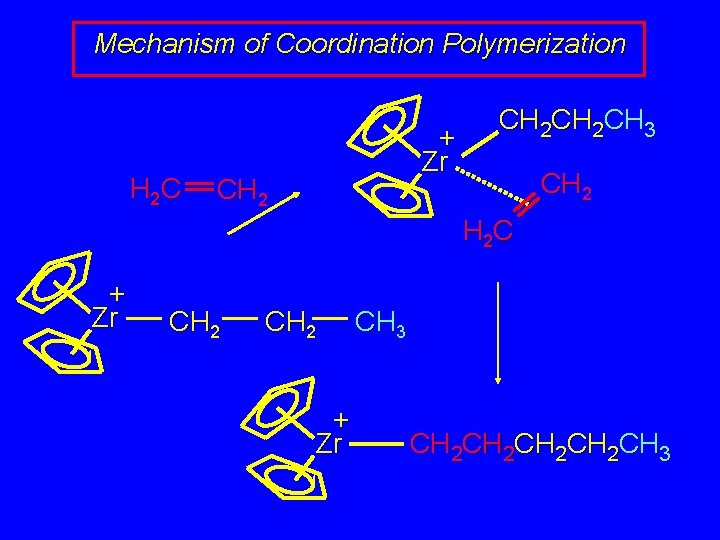
Mechanism of Coordination Polymerization H 2 C + Zr CH 2 CH 2 CH 3 CH 2 H 2 C + Zr CH 2 + Zr CH 3 CH 2 CH 2 CH 3

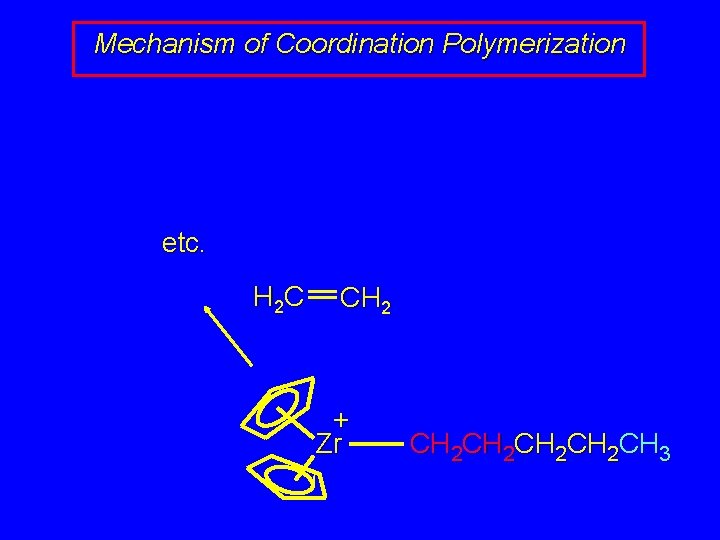
Mechanism of Coordination Polymerization etc. H 2 C CH 2 + Zr CH 2 CH 2 CH 3