SATURATED HIDROCARBONS ALKANE Alkane paraffin saturated hidrocarbons hydrocarbons

SATURATED HIDROCARBONS ALKANE

Alkane = paraffin = saturated hidrocarbons = hydrocarbons with only single covalent bonds between the carbon atoms

Definition Acyclic branched or unbranched hydrocarbons having the general formula Cn. H 2 n+2 , and therefore consisting entirely of hydrogen atoms and saturated carbon atoms. IUPAC Gold Book

Sifat n n n Sukar bereaksi C 1 – C 4 pada tekanan & suhu normal fase gas C 5 – C 17 pada tekanan & suhu normal fase cair PC 18 pada tekanan & suhu normal fase padat Mudah larut pada pelarut non polar Berat jenis naik dengan penambahan atom C Pada alkana rantai lurus semakin panjang rantai maka titik didih & titik leleh semakin tinggi
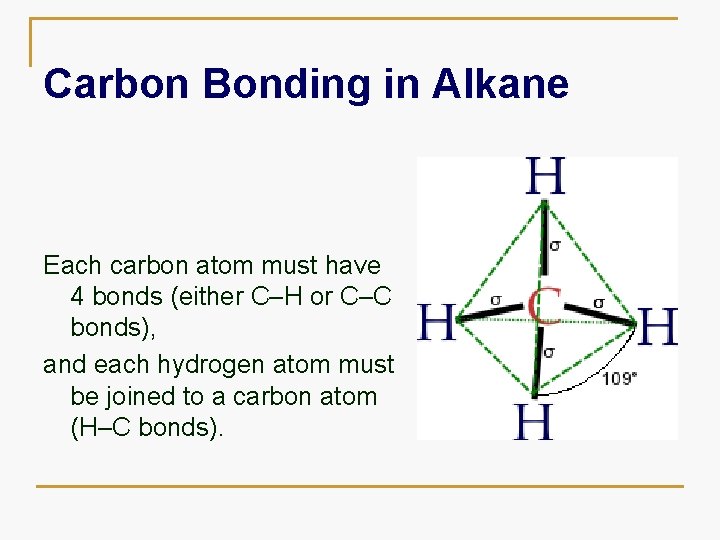
Carbon Bonding in Alkane Each carbon atom must have 4 bonds (either C–H or C–C bonds), and each hydrogen atom must be joined to a carbon atom (H–C bonds).
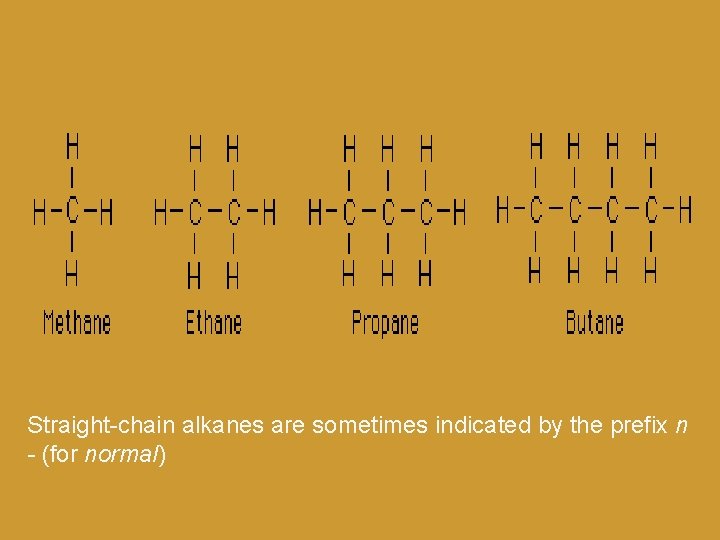
Straight-chain alkanes are sometimes indicated by the prefix n - (for normal)
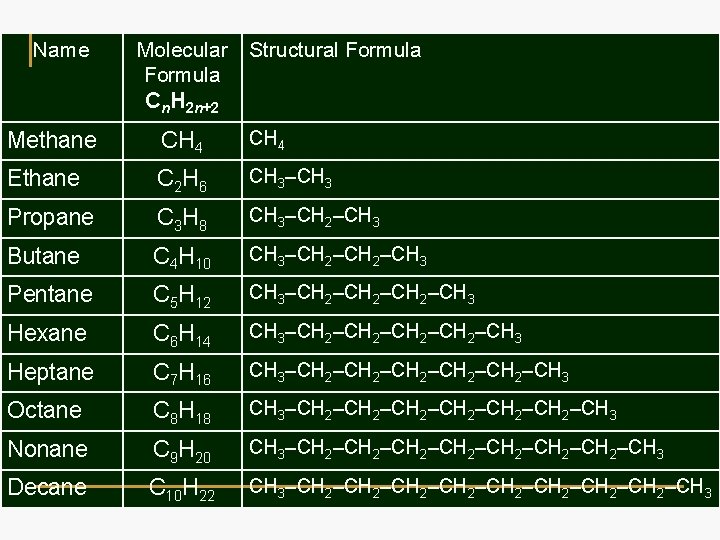
Name Molecular Formula Cn. H 2 n+2 Structural Formula Methane CH 4 Ethane C 2 H 6 CH 3–CH 3 Propane C 3 H 8 CH 3–CH 2–CH 3 Butane C 4 H 10 CH 3–CH 2–CH 3 Pentane C 5 H 12 CH 3–CH 2–CH 3 Hexane C 6 H 14 CH 3–CH 2–CH 3 Heptane C 7 H 16 CH 3–CH 2–CH 2–CH 3 Octane C 8 H 18 CH 3–CH 2–CH 2–CH 3 Nonane C 9 H 20 CH 3–CH 2–CH 2–CH 3 Decane C 10 H 22 CH 3–CH 2–CH 2–CH 3

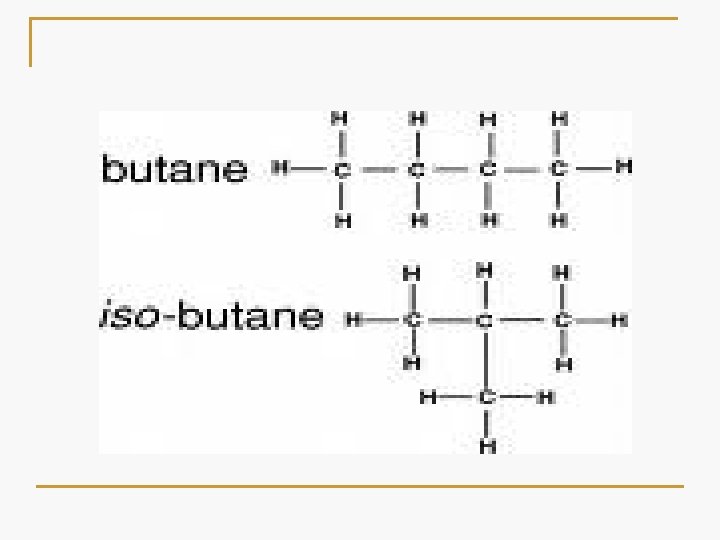
Isomerism Alkanes with more than three carbon atoms can be arranged in a multiple number of ways, forming different structural isomers. Isomers are compounds that have the same molecular formula but different structural formulas. College Chemistry_ch. 21. 8_p. 529

Practice 1 : Write structural formulas and condensed structural formulas of : 1. 2. Pentane C 5 H 12 Hexane C 6 H 14
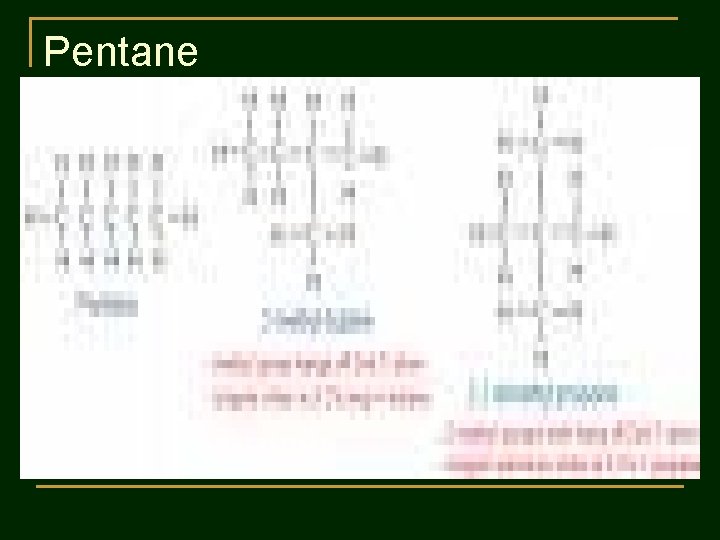
Pentane
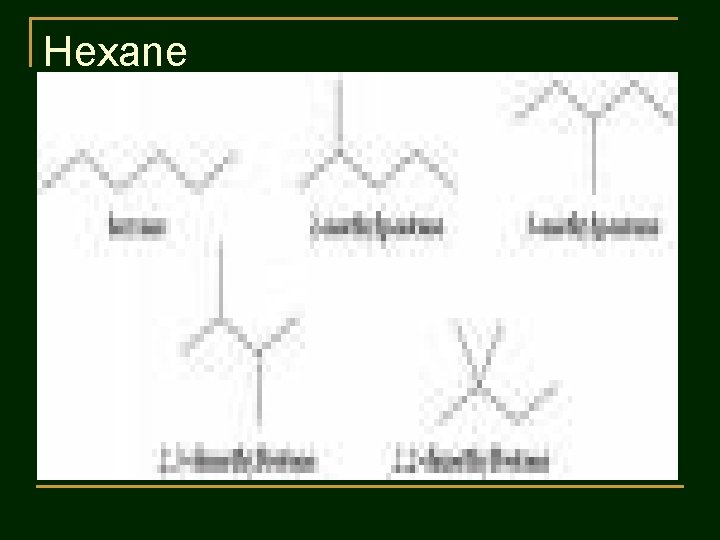
Hexane
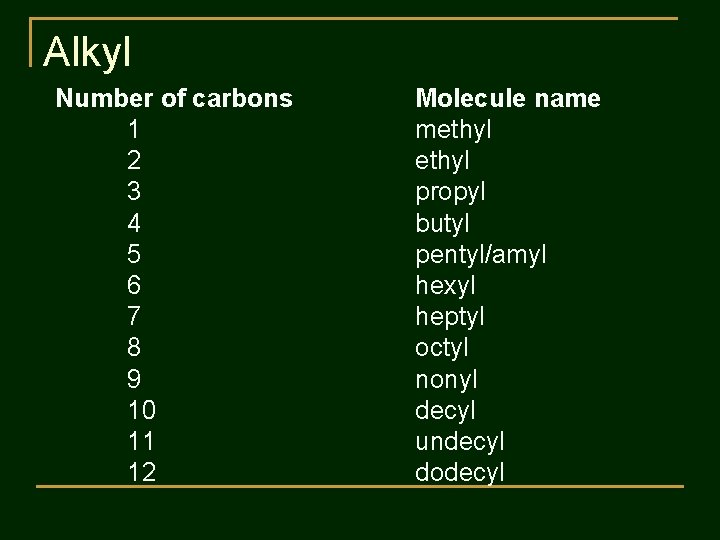
Alkyl Number of carbons 1 2 3 4 5 6 7 8 9 10 11 12 Molecule name methyl propyl butyl pentyl/amyl hexyl heptyl octyl nonyl decyl undecyl dodecyl
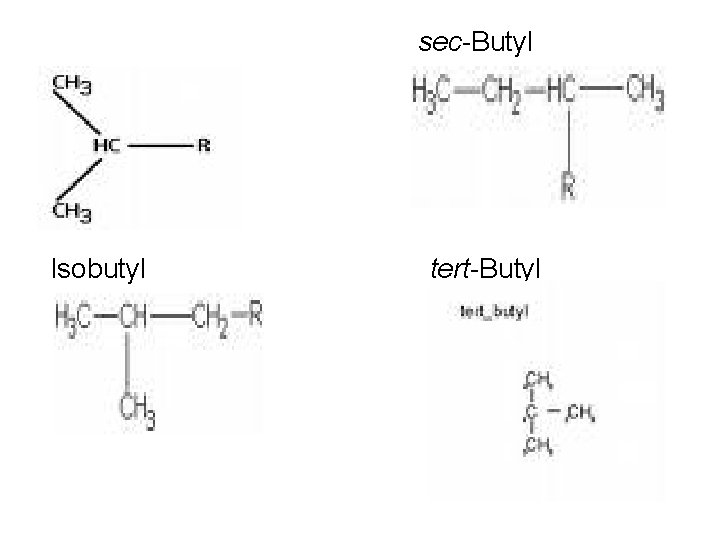
sec-Butyl Isopropyl Isobutyl tert-Butyl

IUPAC Nomenclature : Basic Principles (1) Select the longest continuous chain of carbon atoms as the parent compound, and consider all alkyl groups attached to it as side chains that have replaced hydrogen atoms of the parents hydrocarbon. The name of the alkane consists of the name of the parent compound prefixed by the names of the side-chain alkyl groups attached to it.

IUPAC Nomenclature : Basic Principles (2) Number the carbon atoms in the parent carbon chain starting from the end closest to the first carbon atom that has an alkyl or other group substitued for a hydrogen atom.

IUPAC Nomenclature : Basic Principles (3) Name each side-chain alkyl group and designate its position on the parent carbon chain by a number (for example, 2 -methyl means a methyl group attached to carbon number 2).

IUPAC Nomenclature : Basic Principles (4) When the same alkyl-group side chain occurs more than once, indicate this by a prefix (di-, tri-, tetra-, and si forth) written in front of the alkyl-group name (for example, dimethyl indicates two methyl groups). The numbers indicating the positions of these alkyl groups are separated by a comma and followed by a hypen and are placed in front of the name (for example, 2, 3 -dimethyl).

IUPAC Nomenclature : Basic Principles (5) When several different alkyl groups are attached to the parent compound, list them in alphabetical order; for example, ethyl before methyl in 3 -ethyl-4 -methyloctane. College Chemistry_ch. 21. 9_p. 533

Practice 2 : The following names are incorrect. Tell why the name is wrong and gave the correct name. 1. 3 -methylbutane 2. 2 -ethylbutane 3. 2 -dimethylpentane 4. 3 -methyl-5 -ethyloctane 5. 3, 5, 5 -triethylhexane
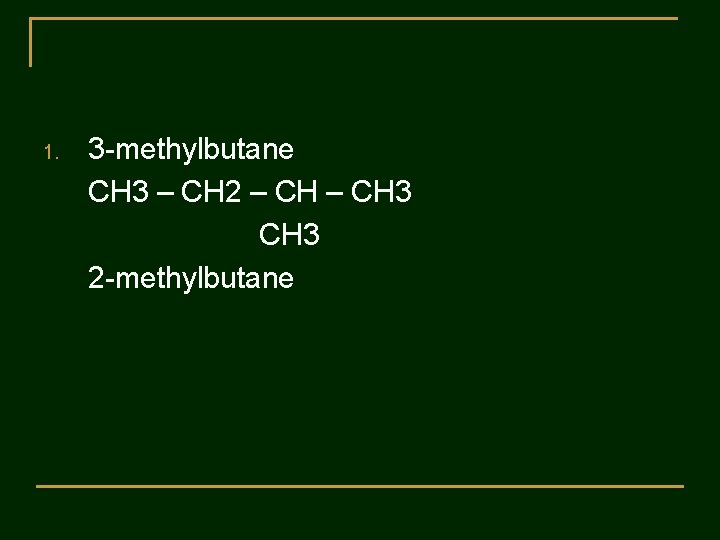
1. 3 -methylbutane CH 3 – CH 2 – CH 3 2 -methylbutane
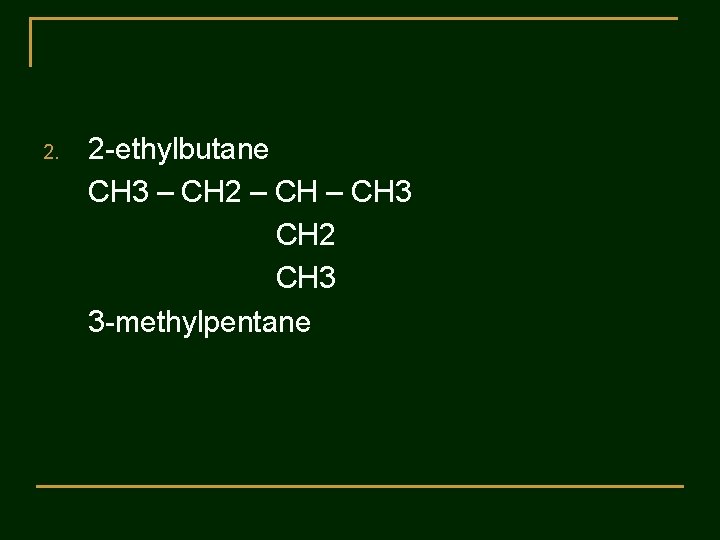
2. 2 -ethylbutane CH 3 – CH 2 – CH 3 CH 2 CH 3 3 -methylpentane
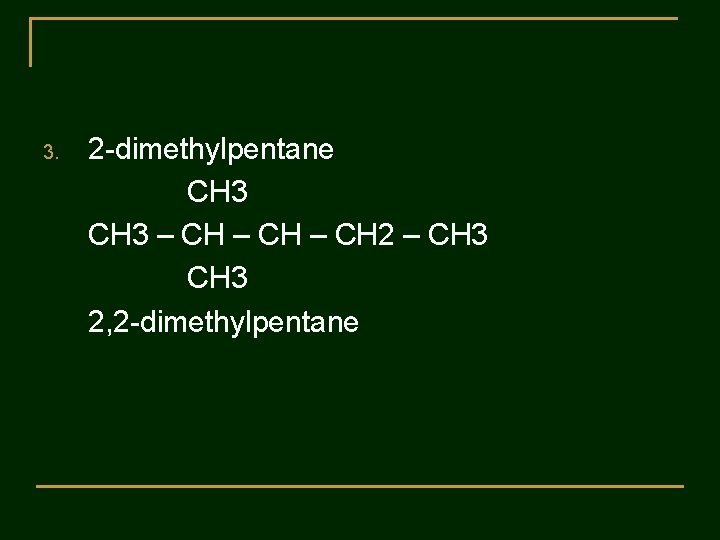
3. 2 -dimethylpentane CH 3 – CH 2 – CH 3 2, 2 -dimethylpentane

4. 3 -methyl-5 -ethyloctane CH 3 – CH 2 – CH 3 CH 2 CH 3 5 -ethyl-3 -methyloctane
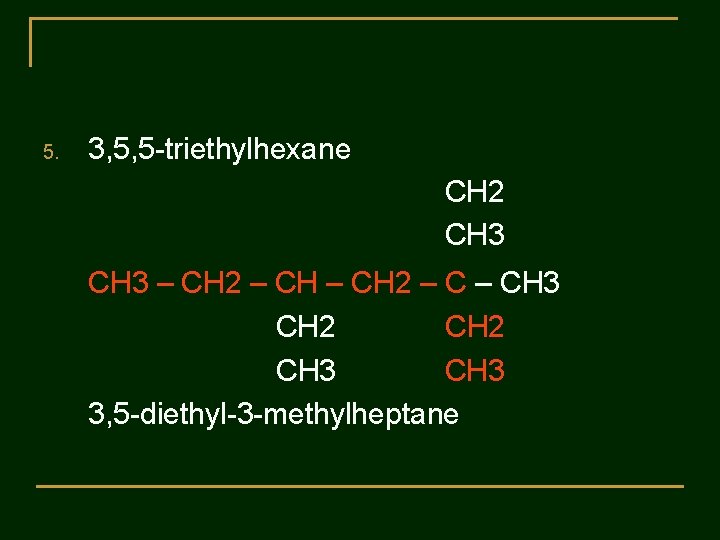
5. 3, 5, 5 -triethylhexane CH 2 CH 3 – CH 2 – CH 3 CH 2 CH 3 3, 5 -diethyl-3 -methylheptane

Reaction of Alkane : Halogenation means substitution of halogens for hydrogen. RH + X 2 RX + HX X = F, Cl, Br, I Example : CH 3 + Cl 2 CH 3 CH 2 Cl + HCl chloroethane
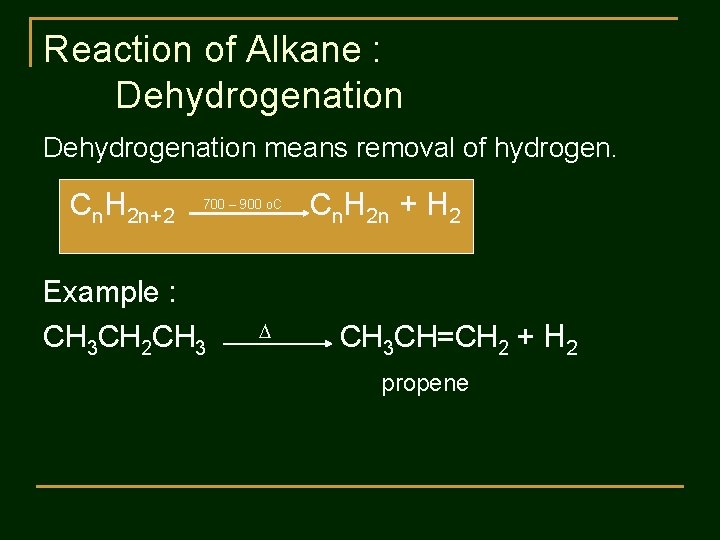
Reaction of Alkane : Dehydrogenation means removal of hydrogen. Cn. H 2 n+2 700 – 900 o. C Cn. H 2 n + H 2 Example : CH 3 CH 2 CH 3 CH=CH 2 + H 2 propene
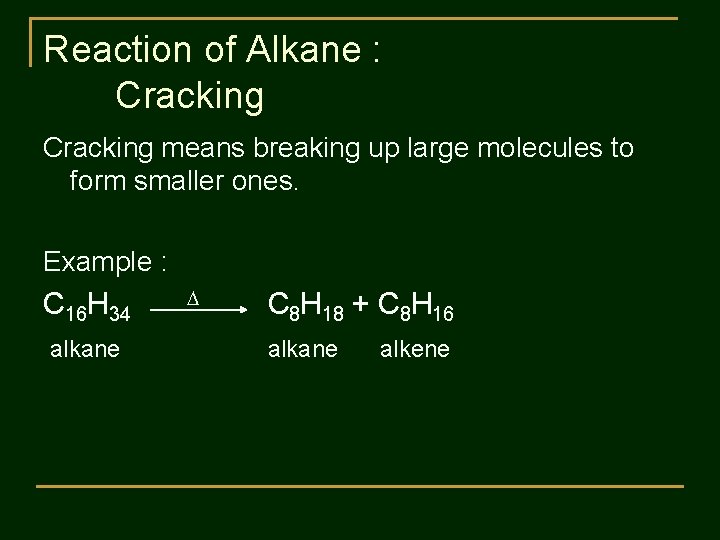
Reaction of Alkane : Cracking means breaking up large molecules to form smaller ones. Example : C 16 H 34 alkane C 8 H 18 + C 8 H 16 alkane alkene

Reaction of Alkane : Isomerization means rearrangement of molecular structures. Example :
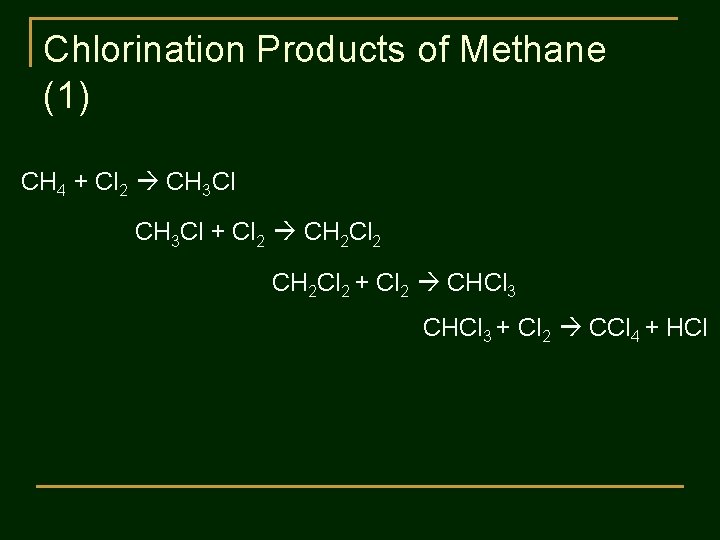
Chlorination Products of Methane (1) CH 4 + Cl 2 CH 3 Cl + Cl 2 CH 2 Cl 2 + Cl 2 CHCl 3 + Cl 2 CCl 4 + HCl

Chlorination Products of Methane (2) Formula IUPAC name Common name CH 3 Cl CH 2 Cl 2 CHCl 3 CCl 4 Chloromethane Dichloromethane Trichloromethane Tetrachloromethane Methyl chloryde Methylene chloryde Chloroform Carbon tetrachloride

Cycloalkane/cycloparaffin/naphta ne n n n Cycloalkanes are types of alkanes which have one or more rings of carbon atoms in the chemical structure of their molecules struktur alisiklik. Rumus: Cn. H 2 n Contoh:

Cyclopropane Cyclobutane C 3 H 6 C 4 H 8 Cyclopentane C 5 H 10 Cyclohexane C 6 H 12

Sources of Alkanes

The two main sources of alkanes are natural gas and petroleum / crude oil. Natural gas consists of : n Methane n Other hydrocarbons n Hydrogen n Nitrogen n Carbon monoxide n Carbon dioxide n Hydrogen sulfide (just in some location)

Petroleum is formed by the decomposition of plants and animals over million of years. Composition: n Carbon n Hydrogen n Nitrogen n Oxygen n Sulfur n Metals Four different types of hydrocarbon molecules appear in crude oil : n Paraffins n Naphthenes n Aromatics n Asphaltics

Fractional Distillation of Crude Oil
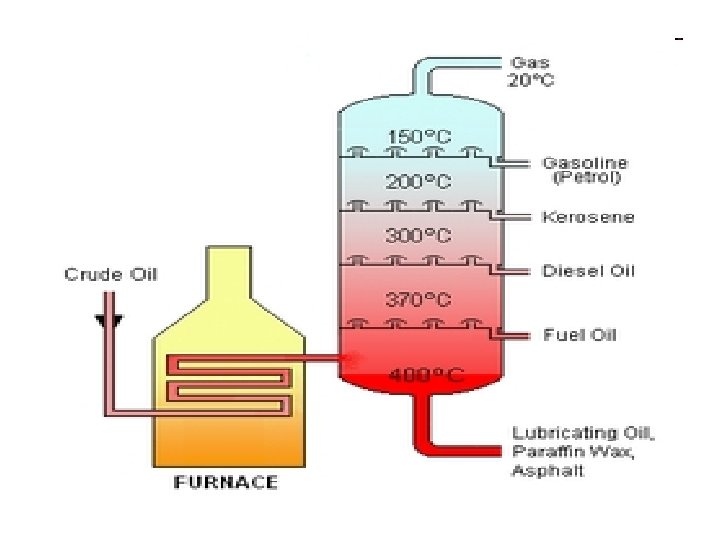

Crude oil is separated into fractions by fractional distillation. The fractions at the top of the fractional column have lower boiling points than the fractions at the bottom. The heavy bottom fractions are often cracked into lighter, more useful products. All of the fractions are processed further in other refining units.


Octane Rating
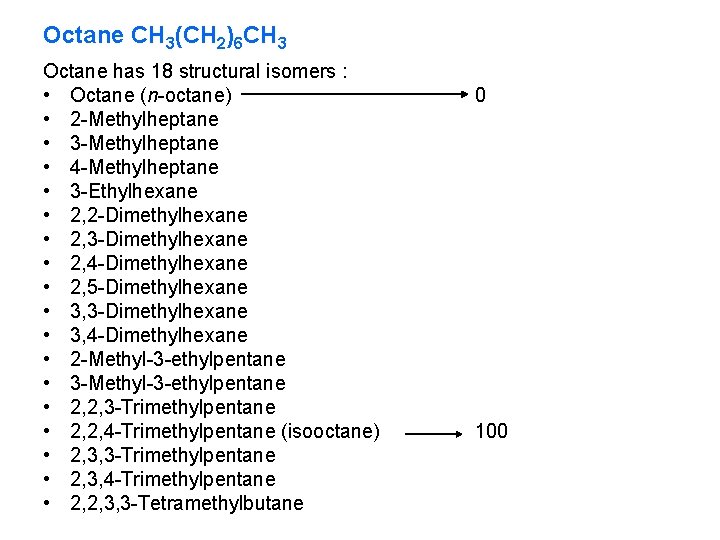
Octane CH 3(CH 2)6 CH 3 Octane has 18 structural isomers : • Octane (n-octane) • 2 -Methylheptane • 3 -Methylheptane • 4 -Methylheptane • 3 -Ethylhexane • 2, 2 -Dimethylhexane • 2, 3 -Dimethylhexane • 2, 4 -Dimethylhexane • 2, 5 -Dimethylhexane • 3, 3 -Dimethylhexane • 3, 4 -Dimethylhexane • 2 -Methyl-3 -ethylpentane • 3 -Methyl-3 -ethylpentane • 2, 2, 3 -Trimethylpentane • 2, 2, 4 -Trimethylpentane (isooctane) • 2, 3, 3 -Trimethylpentane • 2, 3, 4 -Trimethylpentane • 2, 2, 3, 3 -Tetramethylbutane 0 100

The octane rating was developed by chemist Russel Marker at the Ethyl Corporation at 1926. The selection of n-heptane as the zero point of the scale was due to the availability of very high purity n-heptane, not mixed with other isomers of heptane or octane, distilled from the resin of the Jeffrey Pine. Other sources of heptane produced from crude oil contain a mixture of different isomers with greatly differing ratings, which would not give a precise zero point. Higher octane ratings correlate to higher activation energies. Activation energy is the amount of energy necessary to start a chemical reaction. Since higher octane fuels have higher activation energies, it is less likely that a given compression will cause detonation.

The alternative method to boost octane rating and minimize engine knocking is too add small amounts of an additive to the fuel. One such additive commonly used in gasoline was tetraethyllead, (C 2 H 5)4 Pb. The function of tetraethyllead is to prevent the premature explosions that constitute knocking. Use of tetraetthyllead additives poses a serious enviromental hazard. Lead becomes pollutant in air, water, and sovent. Current additives in anleaded gasoline : • Toluene • Xylene • Methyl tert-butyl ether (MTBE)
- Slides: 44