Lab Oreo Lab 1 6 3 Limiting Reagents

Lab “Oreo Lab” 1

6. 3 Limiting Reagents Caution: this stuff can be difficult to follow at first. Be patient.

Limiting Reagents and Reagents in Excess • So far we have assumed that all of the reactants taking part in the reaction have been transformed into products. (No left-overs) • Not usually the case. 3

Ex. Wood burning – Unlimited oxygen in the air for the reaction available (oxygen = reagent in excess) – When all wood is burned, the reaction stops, no longer using oxygen from the air, and making no more water or CO 2. -The amount of wood limits the amount of products, therefor, it is the limiting reagent 4

Ex. Cake baking -The recipe calls for 2 eggs to combine with 1 cup of flour and this make 1 cake. -How many cakes could you bake if you have 4 eggs in your fridge, and 4 cups of flour in your pantry? -To use up all 4 eggs, you would only need 2 cups of flour. (2 cups left over). -Flour = reagent in excess -Eggs = limiting reagent 5
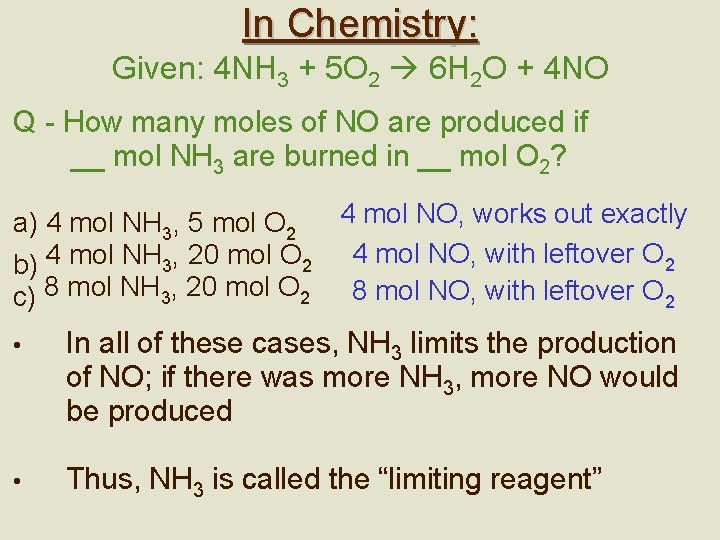
In Chemistry: Given: 4 NH 3 + 5 O 2 6 H 2 O + 4 NO Q - How many moles of NO are produced if __ mol NH 3 are burned in __ mol O 2? 4 mol NO, works out exactly a) 4 mol NH 3, 5 mol O 2 b) 4 mol NH 3, 20 mol O 2 4 mol NO, with leftover O 2 c) 8 mol NH 3, 20 mol O 2 8 mol NO, with leftover O 2 • In all of these cases, NH 3 limits the production of NO; if there was more NH 3, more NO would be produced • Thus, NH 3 is called the “limiting reagent”

In Chemistry: Given: 4 NH 3 + 5 O 2 6 H 2 O + 4 NO Q - How many moles of NO are produced if __ mol NH 3 are burned in __ mol O 2? (d) 4 mol NH 3, 2. 5 mol O 2 • • • 2 mol NO, leftover NH 3 Here, O 2 limits the production of NO; if there was more O 2, more NO would be produced Thus, O 2 is called the “limiting reagent” Sometimes the question is more complicated. For example, if grams of the two reactants are given instead of moles.

Procedure for solving limiting reagent problems 1. If not already, balance the chemical equation. 2. Convert information given into units of moles. 3. Divide the mole amount of each reactant by its stoichiometric coefficient from the balanced chemical equation. 4. The reactant with the smallest quotient from step #3 will be the limiting reagent. 5. Work as typical stoichiometry problem using the limiting reagent. 8

Example #1 2 Ca + O 2 → 2 Ca. O How many moles of calcium oxide can you form starting with 15 moles of calcium and 12 moles of oxygen? 1. If not already, balance the chemical equation. Balanced. 2. Convert information given into units of moles. Already in units of moles. 3. Divide the mole amount of each reactant by its stoichiometric ratio from the balanced chemical equation. 15 moles Ca = 7. 5 12 moles O 2 = 12 2 moles Ca 1 mol O 2 9

Example #1 (con’t) 2 Ca + O 2 → 2 Ca. O How many moles of calcium oxide can you form starting with 15 moles of calcium and 12 moles of oxygen? 4. The reactant with the smallest quotient from step #3 will be the limiting reagent. Calcium will be the limiting reagent in this problem. 5. Work as typical stoichiometry problem using the limiting reagent. 15 mol Ca x 2 mol Ca. O = 15 mol Ca. O 2 mol Ca 10

Example #2 What mass of CO 2 can be produced by the reaction of 8. 0 grams of CH 4 with 48 grams of O 2 according to the equation below? CH 4 + 2 O 2 → CO 2 + 2 H 2 O 1. If not already, balance the chemical equation. Balanced. 2. Convert information given into units of moles. Information given is in units of grams. We need to use the molar mass of each reactant to find moles of each reactant. 8. 0 g CH 4 x 1 mol CH 4 = 0. 50 mol CH 4 16. 04 g CH 4 48 g O 2 x 1 mol O 2 = 1. 50 mol O 2 32. 0 g O 2 11

Example #2 (con’t) What mass of CO 2 can be produced by the reaction of 8. 0 grams of CH 4 with 48 grams of O 2 according to the equation below? CH 4 + 2 O 2 → CO 2 + 2 H 2 O 3. Divide the mole amount of each reactant by its stoichiometric ratio from the balanced chemical equation. 0. 5 mol CH 4 = 0. 5 1. 5 mol O 2 = 0. 75 1 mol CH 4 2 mol O 2 4. The reactant with the smallest quotient from step #3 will be the limiting reagent. CH 4 12

Example #2 (con’t) What mass of CO 2 can be produced by the reaction of 8. 0 grams of CH 4 with 48 grams of O 2 according to the equation below? CH 4 + 2 O 2 → CO 2 + 2 H 2 O 4. Work as typical stoichiometry problem using the limiting reagent. 0. 5 mol CH 4 x 1 mol CO 2 x 44 g CO 2 = 22 g CO 2 1 mol CH 4 1 mol CO 2 13

Limiting Reagents: shortcut • Do two separate calculations using both given quantities. The smaller answer is correct. Ex 3. How many g NO are produced if 20 g NH 3 is burned in 30 g O 2? 4 NH 3 + 5 O 2 6 H 2 O+ 4 NO # g NO= 20 g NH 3 x 1 mol NH 3 x 4 mol NO 17. 0 g NH 3 4 mol NH 3 = 30 g O 2 x 1 mol O 2 x 4 mol NO 32. 0 g O 2 5 mol O 2 = 30. 0 g NO x 1 mol NO 35. 3 g NO 30. 0 g NO x 1 mol NO 22. 5 g NO

Assignment 1. 2. 3. 4. 5. 2 Al + 6 HCl 2 Al. Cl 3 + 3 H 2 If 25 g of aluminum was added to 90 g of HCl, what mass of H 2 will be produced (try this two ways – with the steps & using the shortcut)? N 2 + 3 H 2 2 NH 3: If you have 20 g of N 2 and 5. 0 g of H 2, which is the limiting reagent? What mass of aluminum oxide is formed when 10. 0 g of Al is burned in 20. 0 g of O 2? When C 3 H 8 burns in oxygen, CO 2 and H 2 O are produced. If 15. 0 g of C 3 H 8 reacts with 60. 0 g of O 2, how much CO 2 is produced? How can you tell if a question is a limiting reagent question vs. typical stoichiometry?
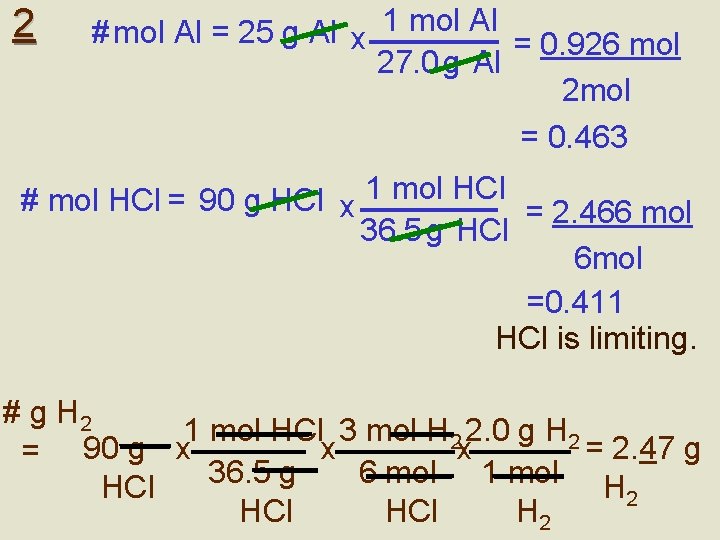
2 # mol Al = 25 g Al x 1 mol Al = 0. 926 mol 27. 0 g Al 2 mol = 0. 463 # mol HCl = 90 g HCl x 1 mol HCl = 2. 466 mol 36. 5 g HCl 6 mol =0. 411 HCl is limiting. # g H 2 1 mol HCl 3 mol H 2 2. 0 g H 2 x x = 2. 47 g = 90 g x 36. 5 g 6 mol 1 mol HCl H 2
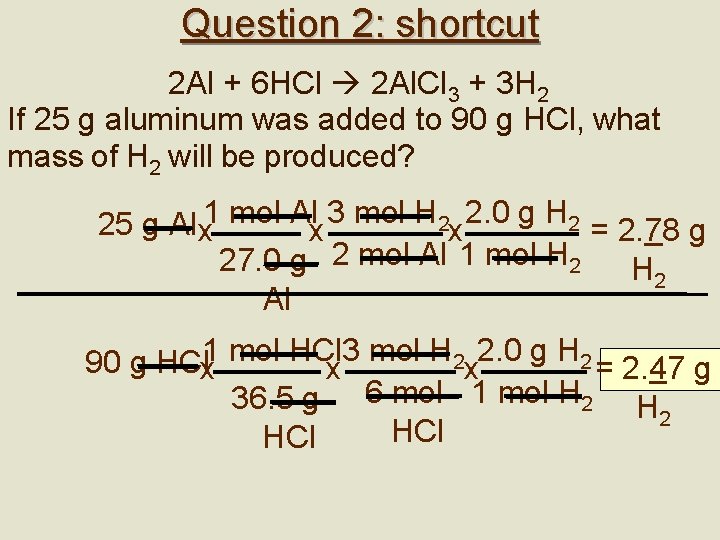
Question 2: shortcut 2 Al + 6 HCl 2 Al. Cl 3 + 3 H 2 If 25 g aluminum was added to 90 g HCl, what mass of H 2 will be produced? 25 g Al x 1 mol Alx 3 mol H 2 x 2. 0 g H 2 = 2. 78 g 27. 0 g 2 mol Al 1 mol H 2 Al 3 mol H 2 2. 0 g H 2 90 g HClx 1 mol HCl x x = 2. 47 g 36. 5 g 6 mol 1 mol H 2 HCl
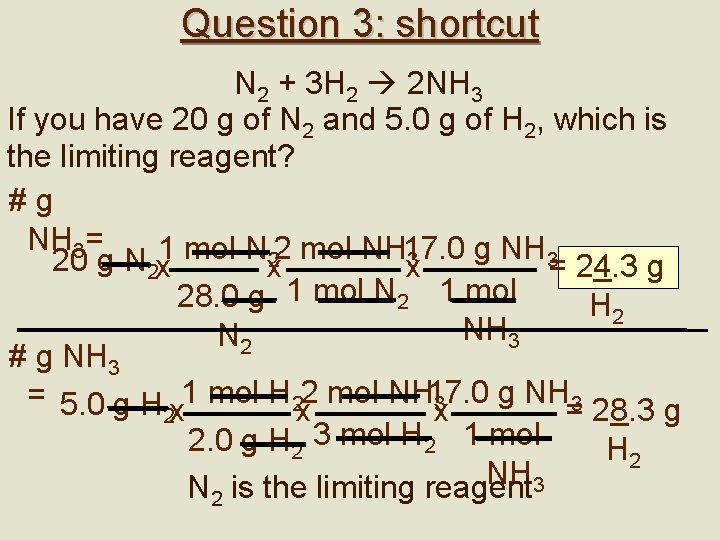
Question 3: shortcut N 2 + 3 H 2 2 NH 3 If you have 20 g of N 2 and 5. 0 g of H 2, which is the limiting reagent? #g NH 3= g NH 3 3 20 g N 2 x 1 mol Nx 22 mol NH 17. 0 x = 24. 3 g 28. 0 g 1 mol N 2 1 mol H 2 NH 3 N 2 # g NH 3 = 5. 0 g H x 1 mol H 2 x 2 mol NH 17. 0 3 x = 28. 3 g 2 2. 0 g H 2 3 mol H 2 1 mol H 2 NH N 2 is the limiting reagent 3

Question 4: shortcut 4 Al + 3 O 2 2 Al 2 O 3 What mass of aluminum oxide is formed when 10. 0 g of Al is burned in 20. 0 g of O 2? #g Al 2 O 3= 10. 0 g Alx 1 mol Alx 2 mol Al 2 O 3 x 102. 0 g = 18. 9 g Al 2 O H 32 27. 0 g 4 mol Al 1 mol Al 2 O 3 Al #g Al O = 2 3 g Al 2 O 3 = 42. 5 g 20. 0 g Ox 21 mol O 2 x 2 mol 102. 0 x Al 2 OO 32 1 mol H 32. 0 g O 2 3 mol 2 Al O 2 3

Question 5: shortcut C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O When C 3 H 8 burns in oxygen, CO 2 and H 2 O are produced. If 15. 0 g of C 3 H 8 reacts with 60. 0 g of O 2, how much CO 2 is produced? #g g CO 2 2=g x 1 mol x 3 mol CO 44. 0 15. 0 x = 45. 0 g C 3 Hg 8 1 mol C 3 H 8 44. 0 CO 2 C 3 H 8 CO 2 C H 3 8 #g COg 2=Ox 21 mol O 2 x 3 mol CO 2 x 44. 0 g CO 2 = 49. 5 g 60. 0 32. 0 g O 2 5 mol O 2 1 mol CO 2 5. Limiting reagent questions give values for two or more reagents (not just one)

6. 4 Assignment Con’t 6. How many grams of NH 3 can be produced from the reaction of 28 g of N 2 and 25 g of H 2? 34 g NH 3 7. How much of the excess reagent in Problem 6 is left over? 19 g H 2 excess 8. What volume of hydrogen at STP is produced from the reaction of 50. 0 g of Mg and the equivalent of 75 g of HCl? 23. 0 L H 2 9. How much of the excess reagent in Problem 3 is left over? 25 g Mg excess 10. Silver nitrate and sodium phosphate are reacted in equal amounts of 200 g each. How many grams of silver phosphate are produced? 164. 3 g Ag 3 PO 4 11. How much of the excess reagent in problem 5 is left? 135. 6 g 21 Na 3 PO 4

6. 4 Assignment 12. Iron reacts with sulfur to form iron(II) sulfide. You have 32. 0 g of sulfur and 100 g of iron. Calculate the number of grams of iron(II) sulfide that will form. 87. 9 g Fe. S 13. Zinc reacts with sulfuric acid in a single replacement reaction. You have 40 g of zinc and 57 g of sulfuric acid. What volume of hydrogen gas will form? 13. 0 L H 2 14. Silver nitrate and sodium bromide will react in a double replacement reaction. You have 24 g of silver nitrate and 39 g of sodium bromide. How many grams of sodium nitrate will form? 11. 9 g Na. NO 3 15. When hydrochloric acid reacts with iron(II) sulfide, a double replacement reaction is to be expected. You have 67 g of hydrochloric acid and 58 of iron(II) sulfide. How many grams of hydrogen sulfide will form? 22. 5 g H 2 S 16. When aluminum is heated in oxygen, aluminum oxide is formed. You have 384 g of each reactant. How many moles of aluminum oxide will 22 form? 6. 45 mol Al 2 O 3

6. 4 Limiting Reagents C. P. 1. A mixture of 49. 1 g of tin(II) nitrate and 81. 2 g of sodium chloride react. How many grams of tin(II) chloride are produced? How much excess is left over? 2. Silicon monocarbide, commonly known as carborundum, is prepared by heating silicon dioxide in the presence of carbon. How many grams of silicon monocarbide can be formed 23 from 100. 0 g of graphite and 100. 0 g of silicon

6. 4 Limiting Reagents C. P. 1. Silver nitrate and sodium phosphate are reacted in equal amounts of 137 g each. How many grams of silver phosphate are produced? 2. How much of the excess reagent in #1 is left? 24

What volume of hydrogen at STP is produced from the reaction of 50. 0 g of Mg and the equivalent of 75 g of HCl? How much of the excess reagent in Problem 3 is left over? 25

Check Point #2 A mixture of 49. 1 g of tin(II) nitrate and 81. 2 g of sodium chloride react. How many grams of tin(II) chloride are produced? How much excess is left over? 26
- Slides: 26