Isotopes n Subatomic Particles protons atomic number Carbon

+ Isotopes

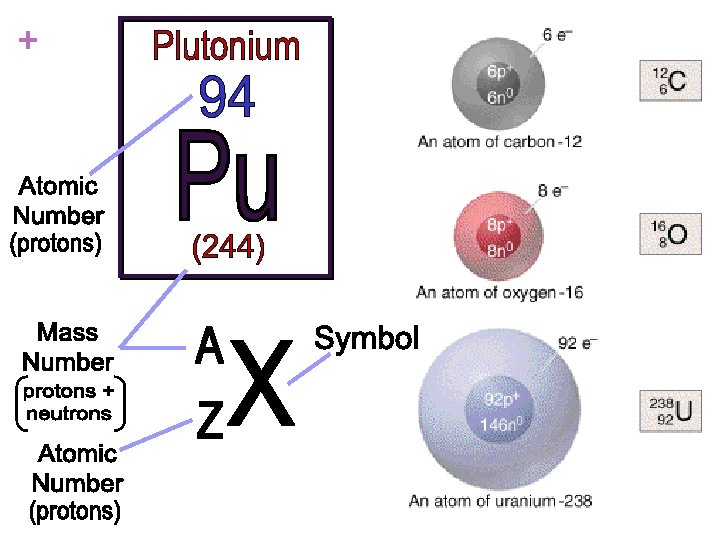
+ n# Subatomic Particles protons = atomic number Carbon - atomic number 6; # of protons 6 n# electrons (neutral atom) = # protons Carbon - # protons 6 = # electrons 6 n# neutrons = mass number – atomic number Carbon-12 6 neutrons = 12 (mass number) – 6 (atomic number)
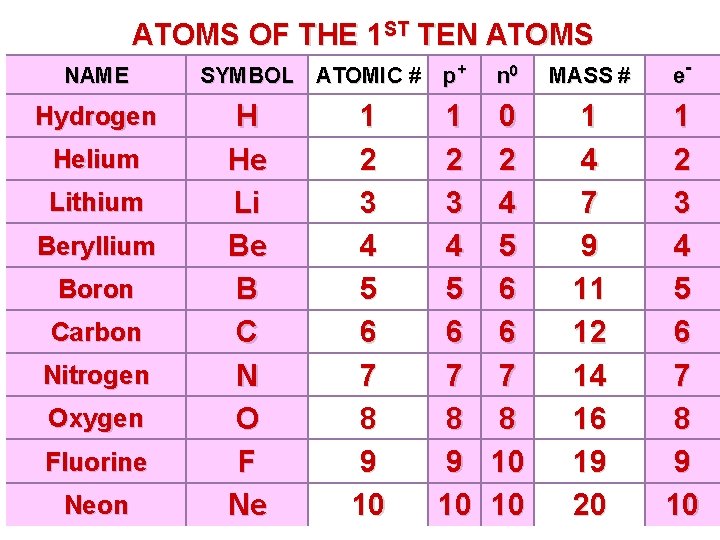
ATOMS OF THE 1 ST TEN ATOMS NAME Hydrogen Helium Lithium Beryllium Boron Carbon Nitrogen Oxygen Fluorine Neon SYMBOL ATOMIC # p+ H He Li Be B C N O F Ne 1 2 3 4 5 6 7 8 9 10 n 0 MASS # e- 0 2 4 5 6 6 7 8 10 10 1 4 7 9 11 12 14 16 19 20 1 2 3 4 5 6 7 8 9 10
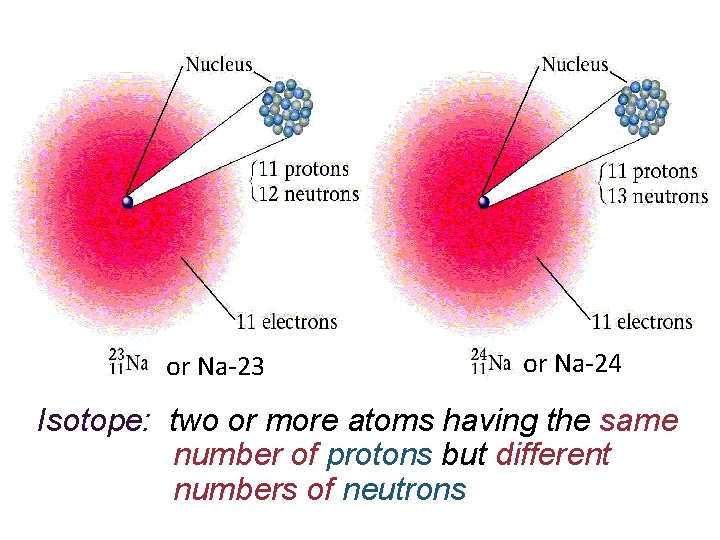
or Na-23 or Na-24 Isotope: two or more atoms having the same number of protons but different numbers of neutrons

+ n. Every Cl atom has 17 protons, w/o exception, but not every Cl atom has 18 neutrons. n. Atoms with the same # of protons but contain different #s of neutrons are called isotopes. n. Since isotopes of an element have different #s of neutrons they have different masses

+ n. Isotopes are chemically alike because they have identical numbers of protons and electrons n. It’s the electrons and protons that are responsible for chemical behavior n. Isotopes can be noted using hyphen notation (Cl-35 vs. Cl-37) or (Chlorine – 35 vs. Chlorine – 37) nelement symbol hyphen mass number nelement name hyphen mass number

+ Nuclide Protium Hydrogen Isotopes #p+ #n 0 # e- at. # mass number (#p+ and #n 0) 1 0 1 1 1 (1 + 0) Deuterium 1 1 2 (1 + 1) Tritium 2 1 1 3 (1 + 2) 1
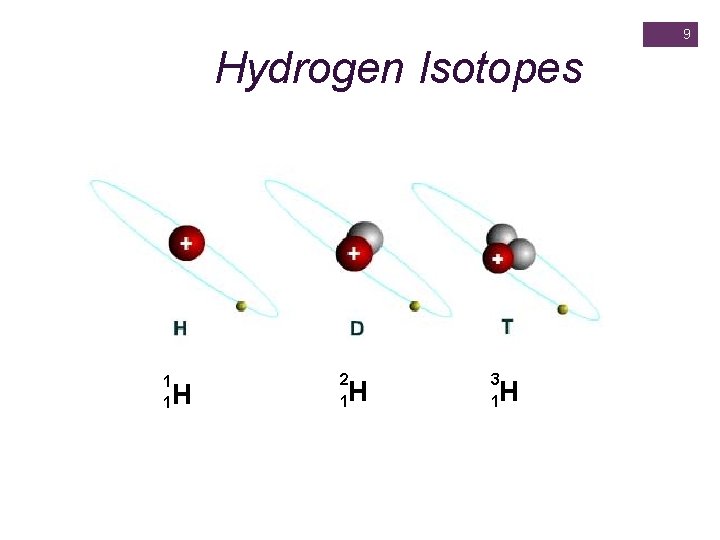
9 Hydrogen Isotopes 1 1 H 2 1 H 3 1 H
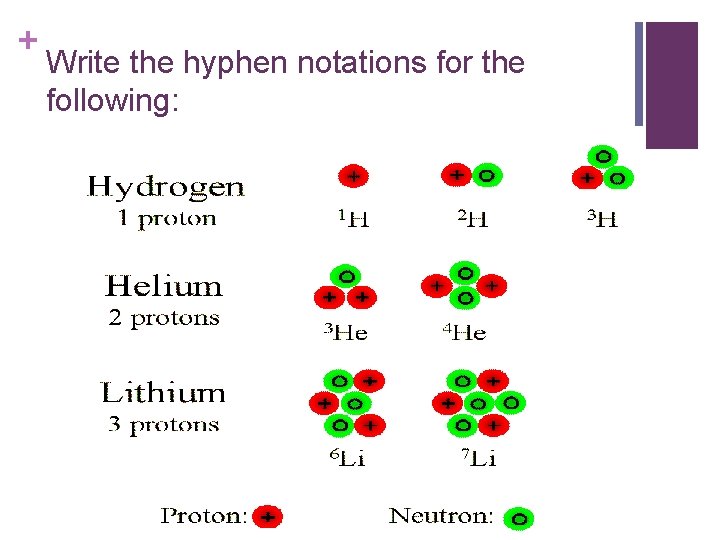
+ Write the hyphen notations for the following:

+ Average Atomic Mass n This is the mass that is on the periodic table. n It is a weighted average of the masses of all the isotopes of a particular element. n Atomic mass = (mass isotope 1)(% abundance) + (mass isotope 2)(% abundance) +. . . n This is what is located on the periodic table

+ 1. Practice Naturally occurring chlorine is 75. 53% chlorine-35 and 24. 47% chlorine-37. What is the average atomic mass? Ans. (0. 7553)(35) + (0. 2447)(37) = 2. The element Neon consists of three isotopes with masses 19. 99, 20. 99, and 21. 99 amu. These three isotopes are present in nature to the extent of 90. 51%, 0. 27%, and 9. 22% respectively. Calculate the atomic mass of neon.

Isotopes of Neon Isotopes % in Nature Atomic Number Atomic Mass (amu) 1 atom Mass Number Average Atomic Mass (amu) Ne-20 90. 51% 10 19. 99244 amu 20 20. 183 amu Ne-21 0. 27% 10 20. 99395 amu 21 Ne-22 9. 22% 10 21. 99138 amu 22

+ 3. The element silver exists in nature as two isotopes Ag-107 and Ag-109. The accepted atomic mass of silver is 107. 87 amu. What isotope is more abundant?

+ 4. Calculate the average atomic mass of an element given the following information: n. Nitrogen-15 is 30% abundant n. Nitrogen-13 is 20% abundant n. Nitrogen-14 is 50% abundant
- Slides: 15