Lithium By Jessica Samantha Madeline Lithium Lithium comes

Lithium By: Jessica, Samantha, Madeline

Lithium ¡ ¡ ¡ Lithium comes from the Greek word for stone, “lithos” Symbol – Li Atomic Number – 3 Atomic Mass – 6. 941 Category – Alkali metal Its density is half of water’s (. 5 grams per cubic cm)
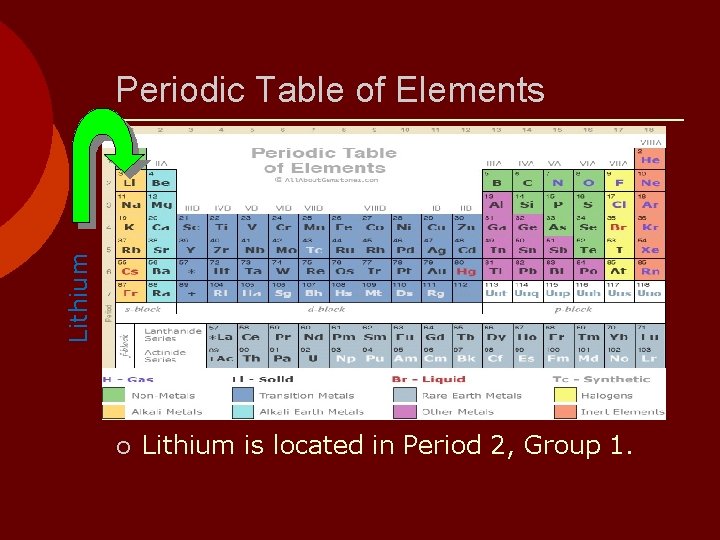
Lithium Periodic Table of Elements ¡ Lithium is located in Period 2, Group 1.

Lithium o o o Solid at room temperature Melting point-180. 5 degrees Celsius Boiling point-1342 degrees Celsius Not found in pure form in nature, it can be found in compounds normally in almost all igneous rocks and brines Lithium is a metal

Who discovered Lithium? Johann August Arfvedson ¡ Discovered in 1817 ¡ Isolated by William Thomas Brande +Sir Humphrey Davis ¡ Was discovered in Stockholm, Sweden ¡

Bohr Model of LITHIUM Description: Crimson to flame, white to strong flames, soft silvery – white, light, has a metallic smell to it, and is 0. 6 on Moh’s scale. Lithium is toxic and targets the central nervous system.

Lewis Dot Structure for LITHIUM ¡ Because Lithium is in Group 1, it only has one valence electron

Lithium Uses Alloying agent ¡ Battery anode material (in pacemaker batteries) ¡ Special glasses and ceramics ¡ Heat transfer applications ¡ Lubricants ¡

Lithium Uses (Cont. ) Lithium carbonate-is used in a drug to treat maniac depression disorder ¡ Lithium hydroxide-is used to remove carbonate from the atmosphere/spacecraft ¡
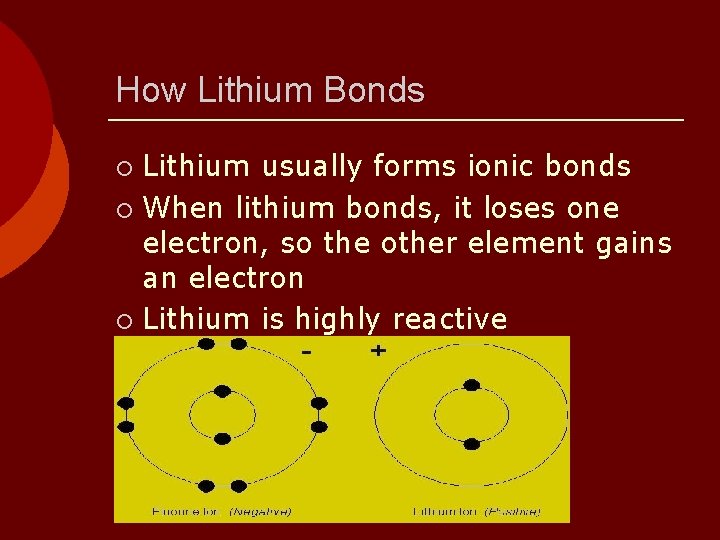
How Lithium Bonds Lithium usually forms ionic bonds ¡ When lithium bonds, it loses one electron, so the other element gains an electron ¡ Lithium is highly reactive ¡

Fun Facts Is $300/pound ¡ Is the lightest of all metals with the density of about half of water ¡ Can be discovered in California in Searles Lake and Nevada ¡ Has the highest specific heat of all solid elements ¡

Work Cited Ø Web. Elements: the periodic table on the WWW [http: //www. webelements. com/] Ø Chemicool Periodic Table. 11/4/2013 <http: //www. chemicool. com/
- Slides: 12