lithium nitrate lead II sulfide lithium nitride barium
lithium nitrate lead (II) sulfide lithium nitride barium sulfide lithium nitrite Chemistry sulfur dioxide Unit 5: Bonding and Inorganic Nomenclature Na. Cl. O 3 NO 2 Fe(Cl. O 3)2 N 2 O 4 Fe(Cl. O 3)3 N 2 O 5
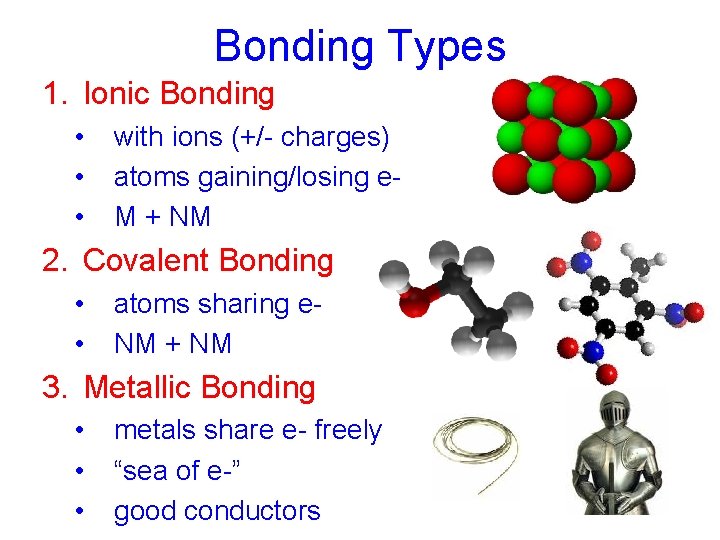
Bonding Types 1. Ionic Bonding • • • with ions (+/- charges) atoms gaining/losing e. M + NM 2. Covalent Bonding • • atoms sharing e. NM + NM 3. Metallic Bonding • • • metals share e- freely “sea of e-” good conductors
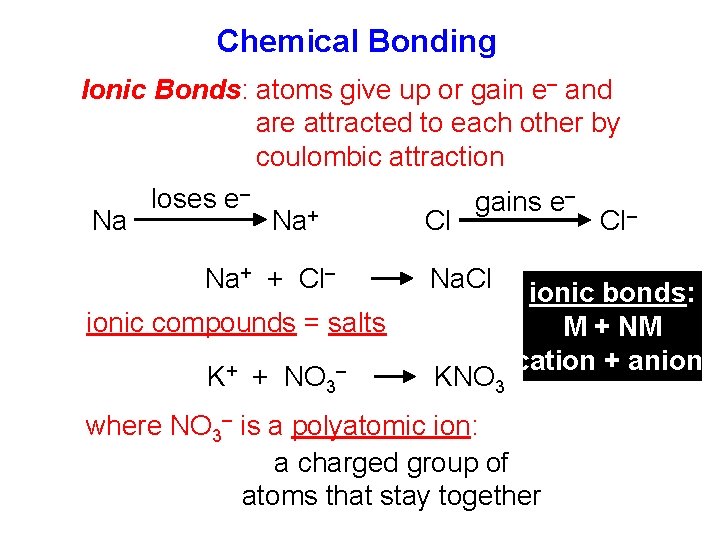
Chemical Bonding Ionic Bonds: atoms give up or gain e– and are attracted to each other by coulombic attraction Na loses e– Na+ + Cl– Cl gains e– Na. Cl ionic compounds = salts K+ + NO 3– KNO 3 Cl– ionic bonds: M + NM cation + anion where NO 3– is a polyatomic ion: a charged group of atoms that stay together

Common Polyatomic Ions Names of Common Polyatomic Ions Ion Name NH 4 + ammonium *NO 2 - *nitrite NO 3 nitrate 2 *SO 3 *sulfite SO 4 2 sulfate HSO 4 - hydrogen sulfate OH hydroxide CN cyanide C 2 H 3 O 2 acetate Mn. O 4 permanganate PO 4 3 phosphate *PO 3 3 - *phosphite H 2 PO 4 - dihydrogen phosphate Ion *Cl. O 2 - Memorize the BOLD ions Zumdahl, De. Coste, World of Chemistry 2002, page 100 Name CO 3 2 carbonate HCO 3 - bicarbonate *IO 3*iodate *IO 4 *periodate *Cl. O *hypochlorite *chlorite Cl. O 3 chlorate *Cl. O 4 - *perchlorate *Br. O 3*bromate 2 C 2 O 4 oxalate Cr 2 O 7 2 dichromate Cr. O 4 2 chromate O 2 2 - peroxide * = you will also be responsible for knowing these

Polyatomic Ion Trends *Charges NEVER change between different forms +1 oxygen “Per____ate” Br. O 4 - perbromate Cl. O 4 - perchlorate IO 4 - periodate “Normal” “–ate” form Br. O 3 bromate Cl. O 3 chlorate -1 oxygen “-ite” form -2 oxygens “hypo___ite” Br. O 2 - Br. O- chlorite hypochlorite IO 3 iodate NO 3 nitrate CO 32 carbonate SO 42 sulfate PO 43 phosphate IO 2 - IO- bromite hypobromite Cl. O 2 - Cl. O- iodite NO 2 - nitrite CO 22 - carbonite SO 32 - sulfite PO 33 - phosphite hypoiodite

Properties of Ionic Salts 1. very hard each ion is bonded to several oppositely charged ions 2. high melting points 3. brittle many bonds must be broken with sufficient force, like atoms are brought next to each other and repel calcite

Writing Formulas of Ionic Compounds chemical formula: has neutral charge; shows types of atoms and how many of each To write an ionic compound’s formula, we need: 1. the two types of ions (i. e. , cations and anions) 2. the charge on each ion Na+ and F– Na. F Ba 2+ and O 2– Ba. O Na+ and O 2– Na 2 O Ba 2+ and F– Ba. F 2
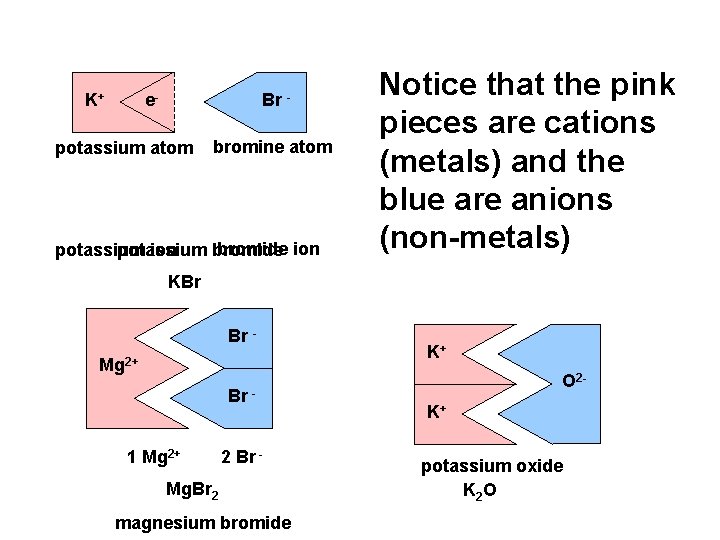
K+ e- e- potassium atom Br. Br bromine atom bromide ion potassium ion bromide Notice that the pink pieces are cations (metals) and the blue are anions (non-metals) KBr Br Mg 2+ Br 1 Mg 2+ 2 Br - Mg. Br 2 magnesium bromide K+ O 2 K+ potassium oxide K 2 O
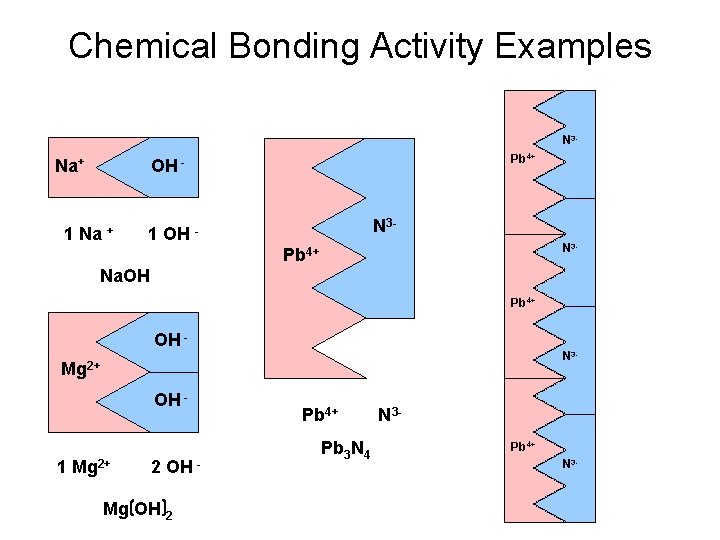
Chemical Bonding Activity Examples N 3 - Na+ Pb 4+ OH - 1 Na + N 3 - 1 OH - N 3 - Pb 4+ Na. OH Pb 4+ OH - N 3 - Mg 2+ OH - 1 Mg 2+ 2 OH - Mg OH 2 Pb 4+ Pb 3 N 4 N 3 Pb 4+ N 3 -
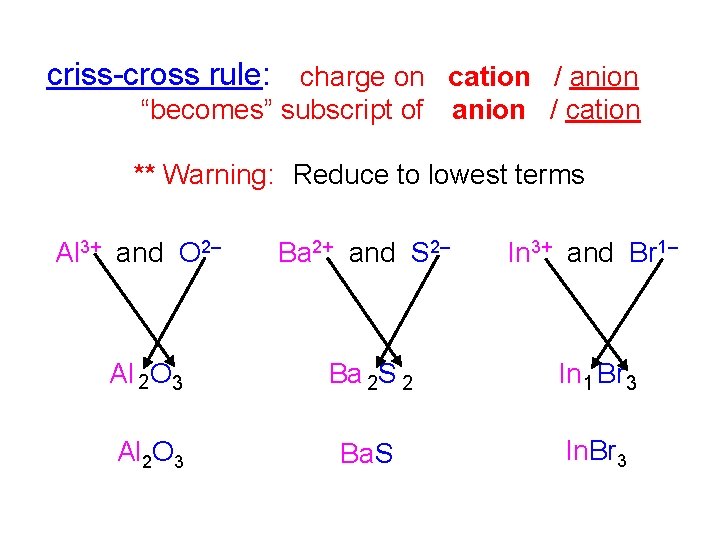
criss-cross rule: charge on cation / anion “becomes” subscript of anion / cation ** Warning: Reduce to lowest terms Al 3+ and O 2– Ba 2+ and S 2– In 3+ and Br 1– Al O 2 3 Ba S 2 2 In Br 1 3 Al 2 O 3 Ba. S In. Br 3

Writing Formulas w/Polyatomic Ions Parentheses are required only when you need more than one “bunch” of a particular polyatomic ion Ba 2+ and SO 42– Ba. SO 4 Mg 2+ and NO 2– Mg(NO 2)2 NH 4+ and Cl. O 3– NH 4 Cl. O 3 Sn 4+ and SO 42– Sn(SO 4)2 Fe 3+ and Cr 2 O 72– Fe 2(Cr 2 O 7)3 NH 4+ and N 3– (NH 4)3 N
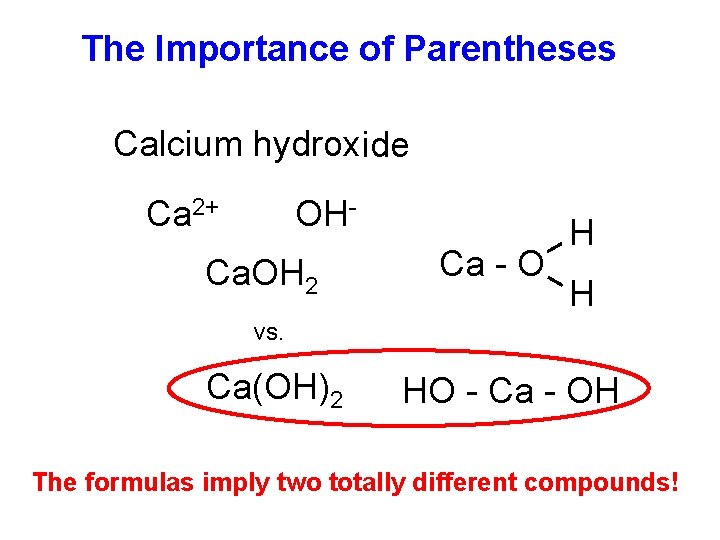
The Importance of Parentheses Calcium hydroxide Ca 2+ OH- Ca. OH 2 Ca - O H H vs. Ca(OH)2 HO - Ca - OH The formulas imply two totally different compounds!
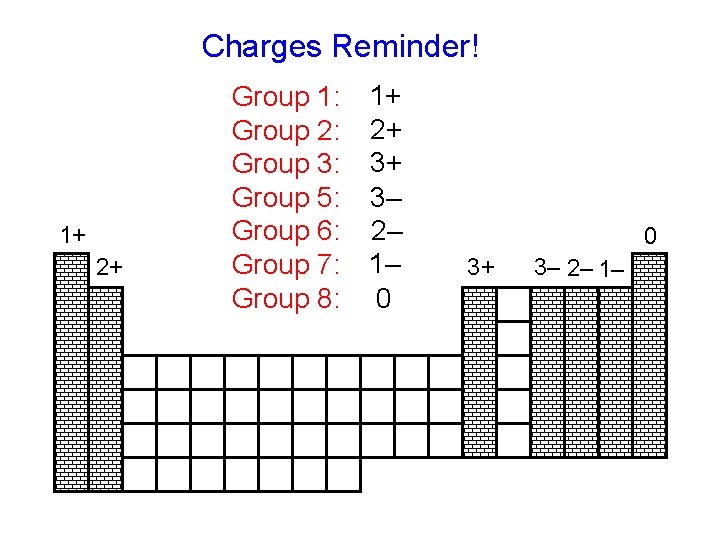
Charges Reminder! 1+ 2+ Group 1: Group 2: Group 3: Group 5: Group 6: Group 7: Group 8: 1+ 2+ 3+ 3– 2– 1– 0 0 3+ 3– 2– 1–

Inorganic Nomenclature potassium nitrate KNO 3 copper (II) sulfate Cu. SO 4 dinitrogen monoxide N 2 O sodium hydroxide Na. OH
Nomenclature - Humor Fe 2+ Fe 2+ “Ferrous Wheel” Fe = iron (Latin = ferrum) Fe 2+ = lower oxidation state = ferrous Fe 3+ = higher oxidation state = ferric Ba. Na 2 “Ba. Na” What weapon can you make from the elements nickel, potassium and iron? A KNi. Fe

Teacher: What is the formula for water? Student: H, I, J, K, L, M, N, O Teacher: That’s not what I taught you. Student: But you said the formula for water was…H to O. "H-O-H"? ! WHAT'S THAT SPELL? ! mis WATER? “Little Johnny took a drink, Now he shall drink no more. For what he thought was H 2 O, Was H 2 SO 4. ” Under aged Pb walks into a bar and the bartender turns to the gold Bouncer and says, “Au, get the lead out!”

“Perhaps one of you gentlemen would mind telling me just what is outside the window that you find so attractive. . ? ” Image courtesy Nearing. Zero. net

Ionic Compounds (cation/anion combos) Fixed-Charge Cations with Elemental Anions i. e. , “pulled off the The fixed-charge cations are: Table” anions groups 1, 2, 13, Ag+ and Zn 2+ 1+ 2+ 3+ 3– 2– 1–
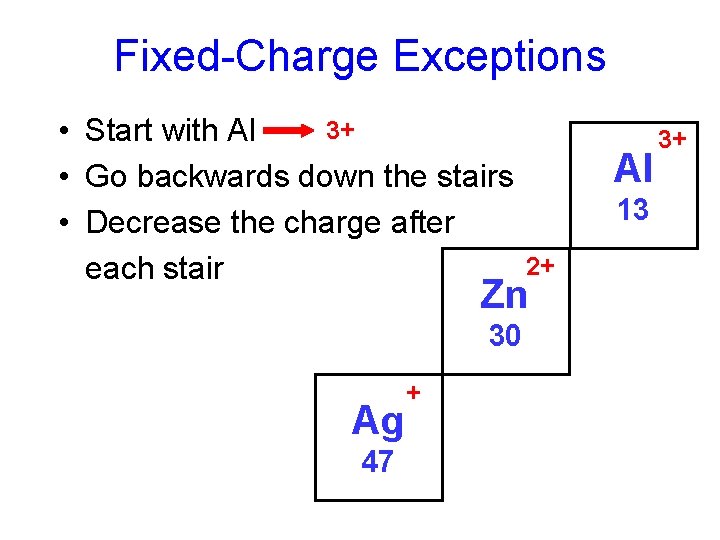
Fixed-Charge Exceptions 3+ • Start with Al • Go backwards down the stairs • Decrease the charge after each stair Al 13 2+ Zn 30 Ag 47 + 3+
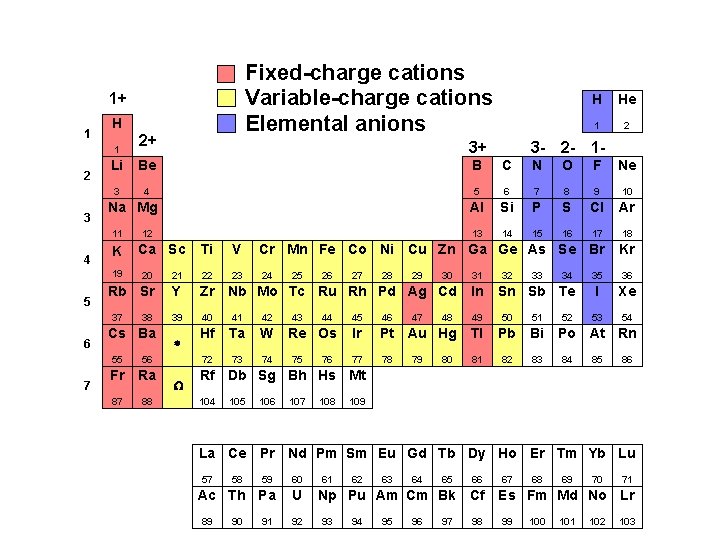
Fixed-charge cations Variable-charge cations Elemental anions 1+ 1 H 1 2 3 7 2 3 - 2 - 1 - B C N O F Ne 3 4 5 6 7 8 9 10 Al Si P S Cl Ar 13 14 15 16 17 18 Na Mg K 12 Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 1 Be 19 5 3+ He Li 11 4 2+ H 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 43 26 44 Re Os 75 76 27 28 29 47 30 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103
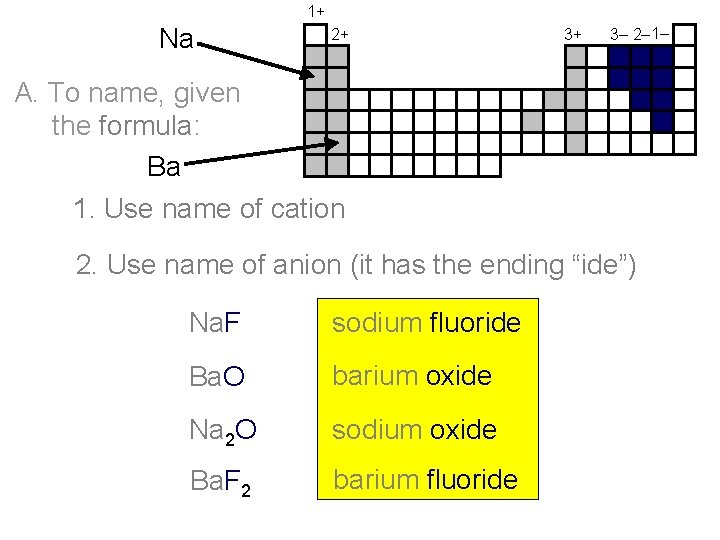
1+ Na 2+ 3+ 3– 2– 1– A. To name, given the formula: Ba 1. Use name of cation 2. Use name of anion (it has the ending “ide”) Na. F sodium fluoride Ba. O barium oxide Na 2 O sodium oxide Ba. F 2 barium fluoride
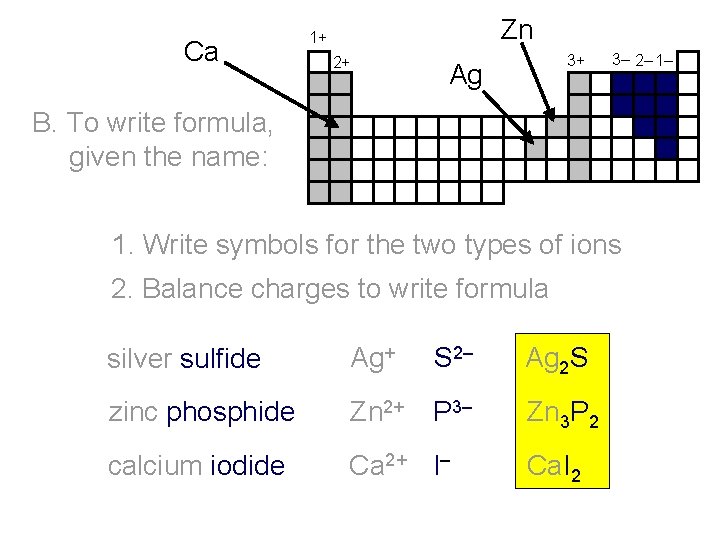
Ca Zn 1+ 2+ 3+ Ag 3– 2– 1– B. To write formula, given the name: 1. Write symbols for the two types of ions 2. Balance charges to write formula silver sulfide Ag+ zinc phosphide Zn 2+ P 3– Zn 3 P 2 calcium iodide Ca 2+ I– Ca. I 2 S 2– Ag 2 S

Variable-Charge Cations with Elemental Anions i. e. , “pulled off the Table” anions The variable-charge cations are: Pb, Sn, and the transition metals (but – of course! – not Ag or Zn)
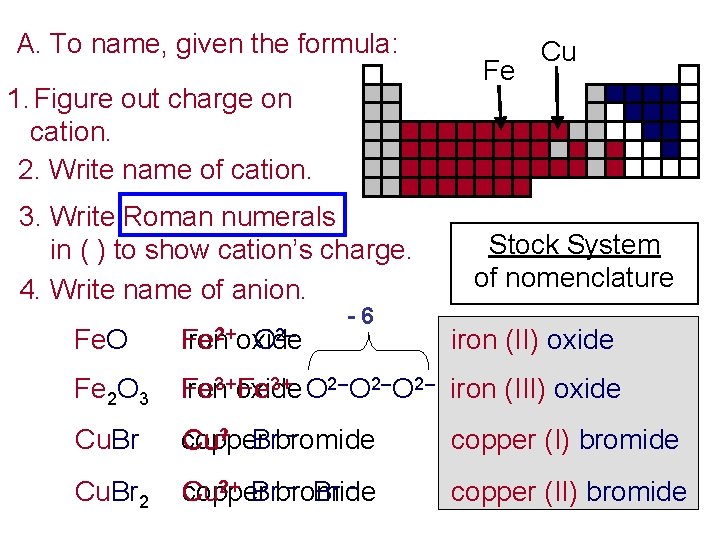
A. To name, given the formula: 1. Figure out charge on cation. 2. Write name of cation. 3. Write Roman numerals in ( ) to show cation’s charge. 4. Write name of anion. Fe. O 2+ ? iron oxide Fe O 2– -6 Fe Cu Stock System of nomenclature iron (II) oxide ? 3+ iron oxide Fe? 3+ O 2– iron (III) oxide Fe 2 O 3 Fe copper bromide Cu+? Br – copper (I) bromide Cu. Br 2 copper bromide Cu? 2+ Br – copper (II) bromide Cu. Br
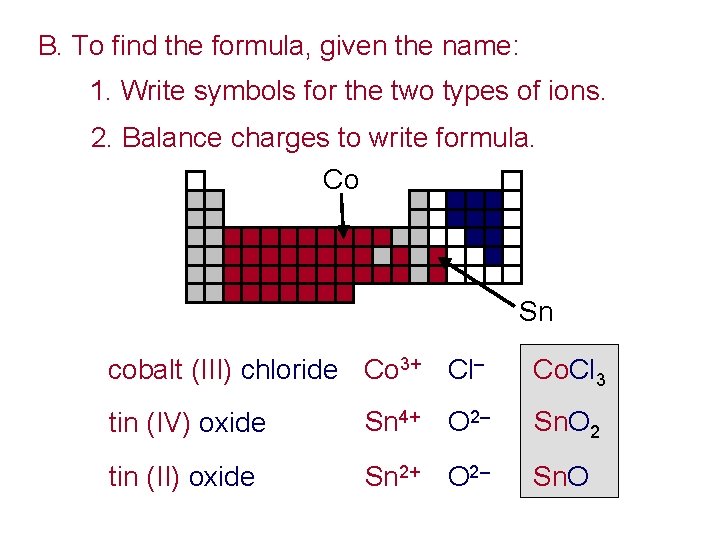
B. To find the formula, given the name: 1. Write symbols for the two types of ions. 2. Balance charges to write formula. Co Sn cobalt (III) chloride Co 3+ Cl– Co. Cl 3 tin (IV) oxide Sn 4+ O 2– Sn. O 2 tin (II) oxide Sn 2+ O 2– Sn. O

Ionic Nomenclature Practice 1. sodium hydroxide Na. OH 2. Hg. SO 4 Fe 2 O 3 mercury (II) sulfate 10. Mg(NO 3)2 magnesium nitrate 11. calcium sulfide 3. lead (II) phosphate 4. (NH 4)2 S Pb 3(PO 4)2 ammonium sulfide 6. Cu 2 CO 3 12. K 2 O Ca. S potassium oxide 13. magnesium chloride 5. aluminum chlorate Al(Cl. O 3)3 copper (I) carbonate 7. manganese (IV) oxide 8. Mn. SO 4 9. iron (III) oxide Mn. O 2 manganese (II) sulfate 14. Cr 2 O 3 Mg. Cl 2 chromium (III) oxide 15. gold (III) bromide 16. Zn(NO 2)2 Au. Br 3 zinc nitrite
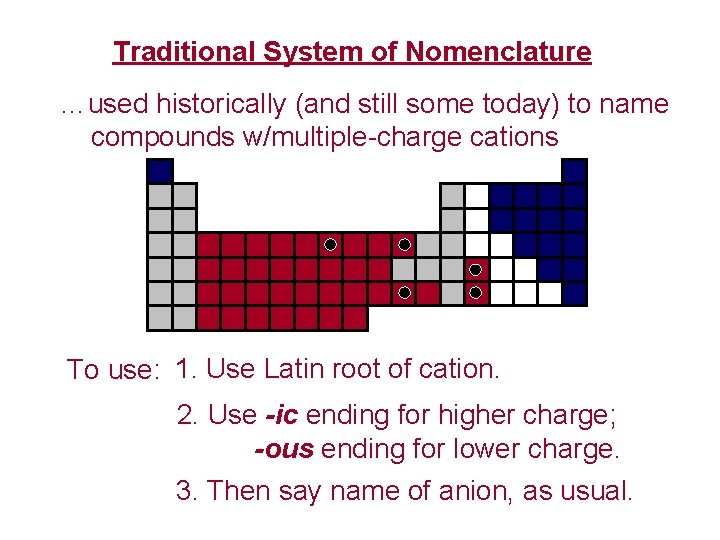
Traditional System of Nomenclature …used historically (and still some today) to name compounds w/multiple-charge cations To use: 1. Use Latin root of cation. 2. Use -ic ending for higher charge; -ous ending for lower charge. 3. Then say name of anion, as usual.

Element gold, Au lead, Pb tin, Sn copper, Cu iron, Fe Latin root aurplumbstanncuprferr- Write formulas: cuprous sulfide Cu+ S 2– Cu 2 S auric nitride Au 3+ N 3 Au. N ferrous fluoride Fe. F 2 Fe 2+ F– -ic Au 3+ Pb 4+ Sn 4+ Cu 2+ Fe 3+ -ous Au+ Pb 2+ Sn 2+ Cu+ Fe 2+ Write names: ? P 3– Pb 4+ Pb 3 P 4 plumbic phosphide Pb? 2+ P 3– Pb 3 P 2 plumbous phosphide Sn? 4+ Cl– Sn Sn. Cl 4 stannic chloride
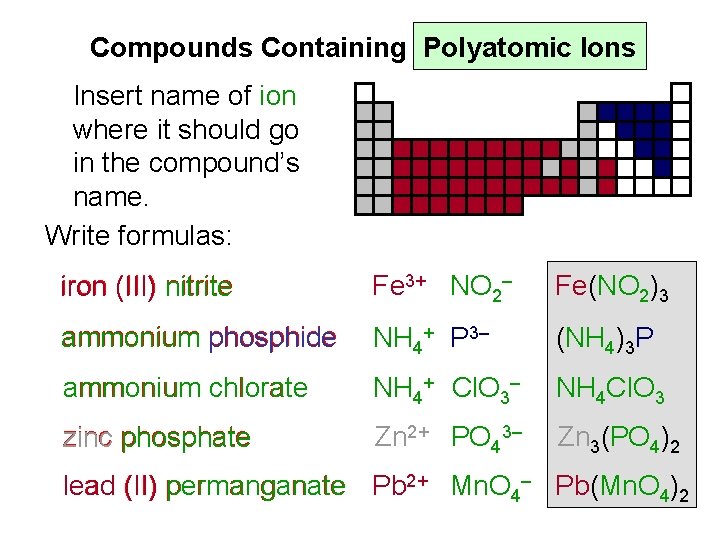
Compounds Containing Polyatomic Ions Insert name of ion where it should go in the compound’s name. Write formulas: iron (III) nitrite Fe 3+ NO 2– Fe(NO 2)3 ammonium phosphide NH 4+ P 3– (NH 4)3 P ammonium chlorate NH 4+ Cl. O 3– NH 4 Cl. O 3 zinc phosphate Zn 2+ PO 43– Zn 3(PO 4)2 lead (II) permanganate Pb 2+ Mn. O 4– Pb(Mn. O 4)2
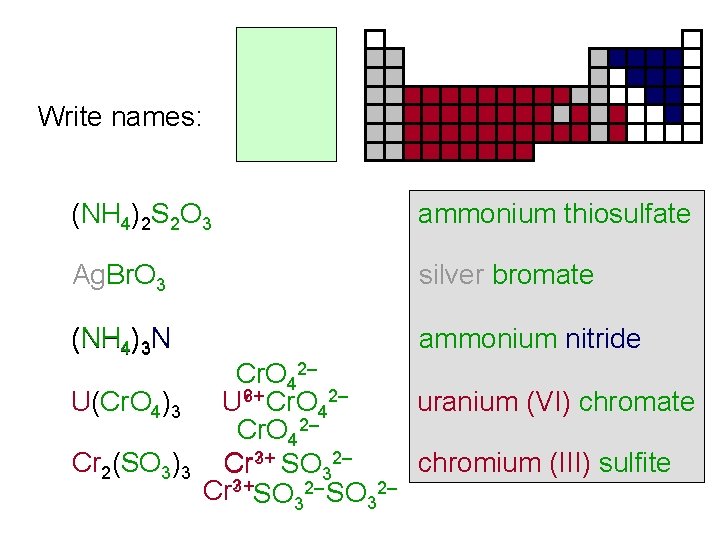
Write names: (NH 4)2 S 2 O 3 ammonium thiosulfate Ag. Br. O 3 silver bromate (NH 4)3 N ammonium nitride Cr. O 42– ? Cr. O 2– uranium (VI) chromate U(Cr. O 4)3 U 6+ 4 Cr. O 42– ? SO 2– Cr 2(SO 3)3 Cr 3+ chromium (III) sulfite 3 Cr? 3+SO 32–

Self-Test Quiz 1. lithium nitrate 2. lithium nitride 3. lithium nitrite 4. Na. Cl. O 3 5. Fe(Cl. O 3)2 6. Fe(Cl. O 3)3 7. lead(II) sulfide 8. barium sulfide 9. Never show charges in a compound’s formula. (T/F) 10. When writing names with Group 9 cations, you need to use Roman numerals. (T/F) 11. When using an anion off the polyatomic ion sheet, change name’s ending to “-ide. ” (T/F) 12. A Roman numeral indicates the charge on the anion. (T/F)

Self-Test Quiz Answers 1. lithium nitrate Li. NO 3 2. lithium nitride Li 3 N 3. lithium nitrite Li. NO 2 4. Na. Cl. O 3 sodium chlorate 5. Fe(Cl. O 3)2 iron(II) chlorate 6. Fe(Cl. O 3)3 iron(III) chlorate 7. lead(II) sulfide Pb. S 8. barium sulfide Ba. S

Self-Test Quiz Answers T 9. Never show charges in a compound’s formula. T 10. When writing names with Group 9 cations, you need to use Roman numerals. 11. When using an anion off the polyatomic ion sheet, change the name’s ending to “-ide. ” F F 12. A Roman numeral indicates the charge on the anion.

Covalent Bonds (2 nonmetals) …atoms share e– to get a full valence shell C 1 s 2 2 p 2 4 valence e. F 1 s 2 2 p 5 7 valence e- Both need 8 valence e- for a full outer shell… otherwise known as the octet rule o o C o xx o x x F xx x

Draw the Lewis dot structure for the following elements: Si 1 s 2 2 p 6 3 s 2 3 p 2 4 valence e- O 1 s 2 2 p 4 6 valence e- P 1 s 2 2 p 6 3 s 2 3 p 3 5 valence e- B 1 s 2 2 p 1 3 valence e- Ar 1 s 2 2 p 6 3 s 2 3 p 6 8 valence e- Br 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 5 7 valence e-

Notice any trends…?

Drawing Lewis Structures Lewis structure: a model of a covalent molecule that shows all of the valence e– 1. Two shared e– make a single covalent bond, four make a double bond, etc. 2. unshared pairs: pairs of unbonded valence e– 3. Each atom needs a full outer shell, i. e. , 8 e–. Exception: H needs 2 e–
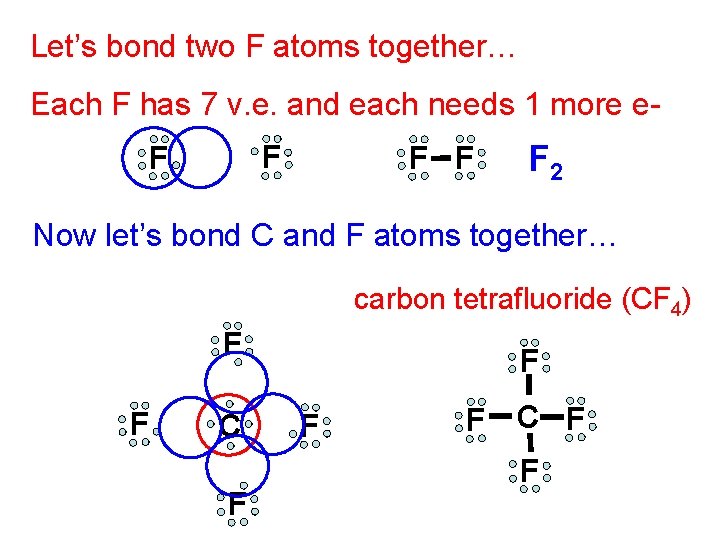
Let’s bond two F atoms together… Each F has 7 v. e. and each needs 1 more e. F F F 2 Now let’s bond C and F atoms together… carbon tetrafluoride (CF 4) F F C F F

And now, a video break…
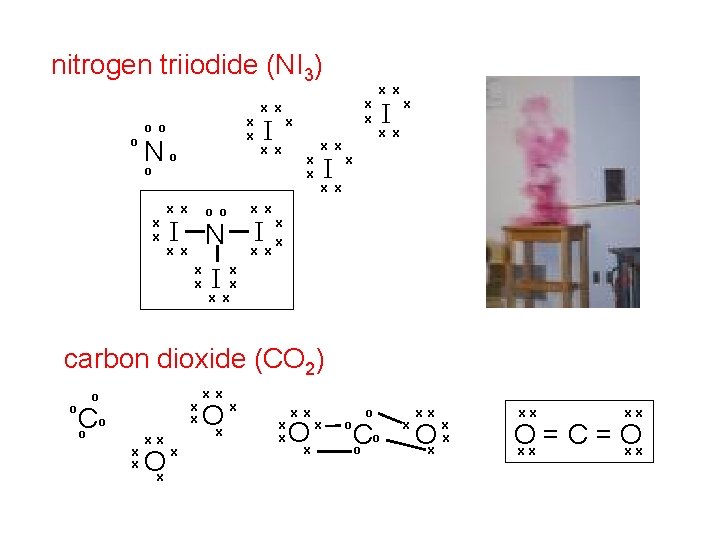
nitrogen triiodide (NI 3) o x x o o N o o x x x Ix x I Ix x xo N x x I x x x o x x x o o x x I carbon dioxide (CO 2) o o x x o C o x x Ox x x Ox x o o o C o x xx Ox x x xx xx O = C = O xx xx
covalent compounds = molecular compounds -- have lower melting points than do ionic compounds (consist of two or more nonmetal elements) butter

Dihydrogen Monoxide: A Tale of Danger and Irresponsibility major component of acid rain found in all cancer cells inhalation can be deadly excessive ingestion results in acute physical symptoms: e. g. , frequent urination, bloated sensation, profuse sweating often an industrial byproduct of chemical reactions; dumped wholesale into rivers and lakes
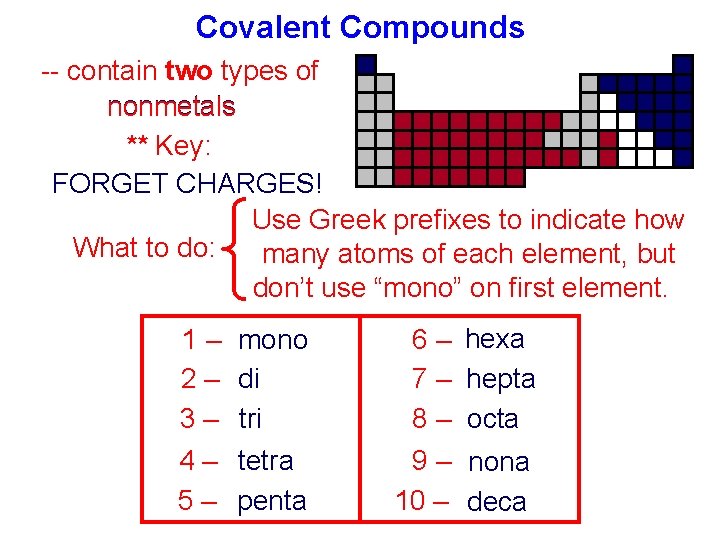
Covalent Compounds -- contain two types of nonmetals ** Key: FORGET CHARGES! Use Greek prefixes to indicate how What to do: many atoms of each element, but don’t use “mono” on first element. 1 – mono 2 – di 3 – tri 6 – hexa 7 – hepta 8 – octa 4 – tetra 5 – penta 9 – nona 10 – deca
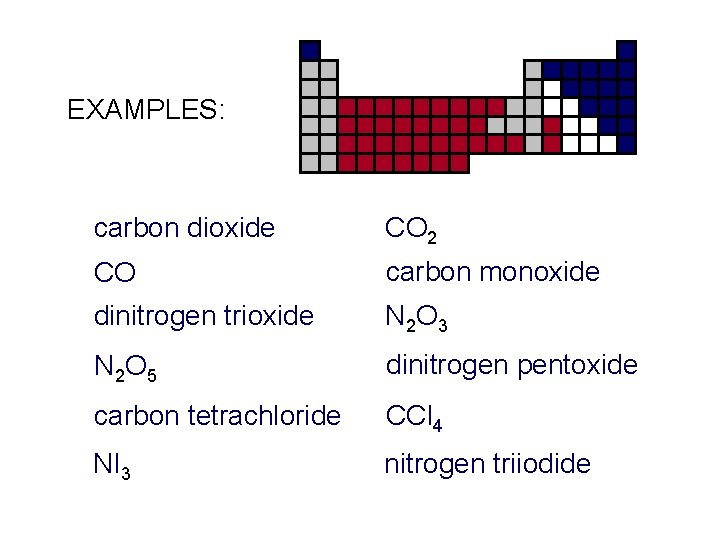
EXAMPLES: carbon dioxide CO 2 CO carbon monoxide dinitrogen trioxide N 2 O 3 N 2 O 5 dinitrogen pentoxide carbon tetrachloride CCl 4 NI 3 nitrogen triiodide
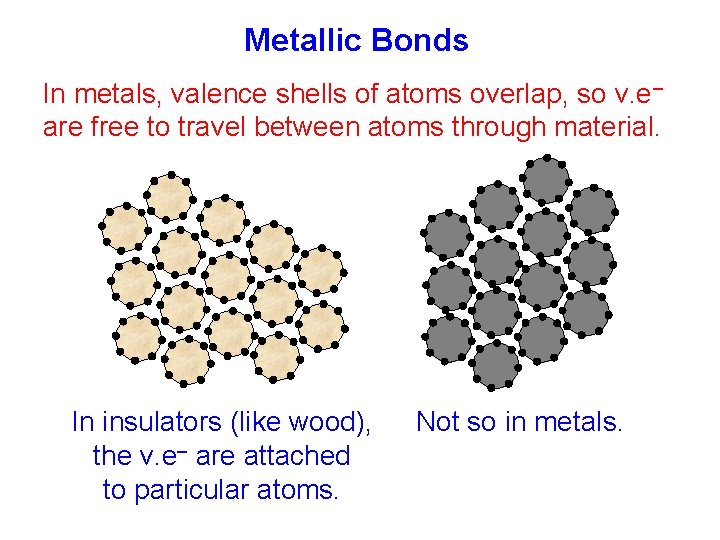
Metallic Bonds In metals, valence shells of atoms overlap, so v. e– are free to travel between atoms through material. In insulators (like wood), the v. e– are attached to particular atoms. Not so in metals.

Properties of Metals ductile malleable conduct heat and electricity All due to free-moving v. e–.
Other Types of Forces/Attractions dipole-dipole forces hydrogen bonds London dispersion forces ion-dipole forces boiling H 2 O DNA These are much weaker than ionic, covalent, or metallic bonds, but very important in determining states of matter, boiling and melting points, and molecular shape (among other things).

Empirical Formula and Molecular Formula lowest-terms formula shows the true number and type of atoms in a m’cule Compound Molecular Formula Empirical Formula glucose C 6 H 12 O 6 CH 2 O propane C 3 H 8 butane C 4 H 10 C 2 H 5 naphthalene C 10 H 8 C 5 H 4 sucrose C 12 H 22 O 11 octane C 8 H 18 C 4 H 9

Nomenclature Review Flow Chart
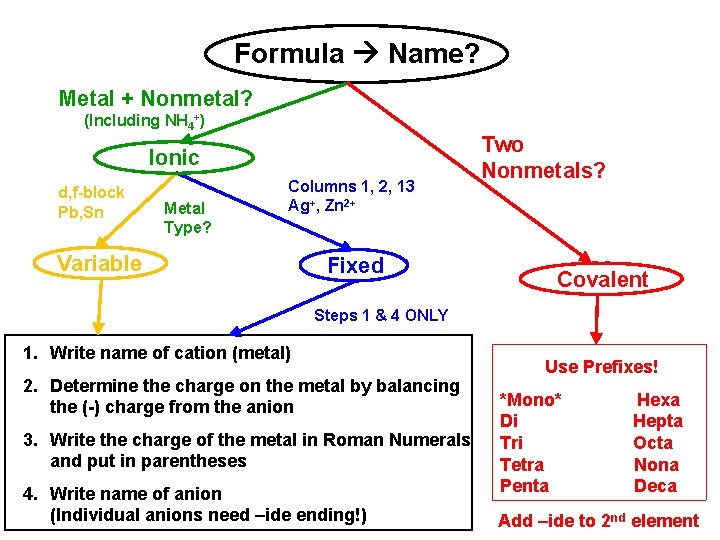
Formula Name? Metal + Nonmetal? (Including NH 4+) Ionic d, f-block Pb, Sn Metal Type? Columns 1, 2, 13 Ag+, Zn 2+ Variable Fixed Two Nonmetals? Covalent Steps 1 & 4 ONLY 1. Write name of cation (metal) 2. Determine the charge on the metal by balancing the (-) charge from the anion 3. Write the charge of the metal in Roman Numerals and put in parentheses 4. Write name of anion (Individual anions need –ide ending!) Use Prefixes! *Mono* Di Tri Tetra Penta Hexa Hepta Octa Nona Deca Add –ide to 2 nd element
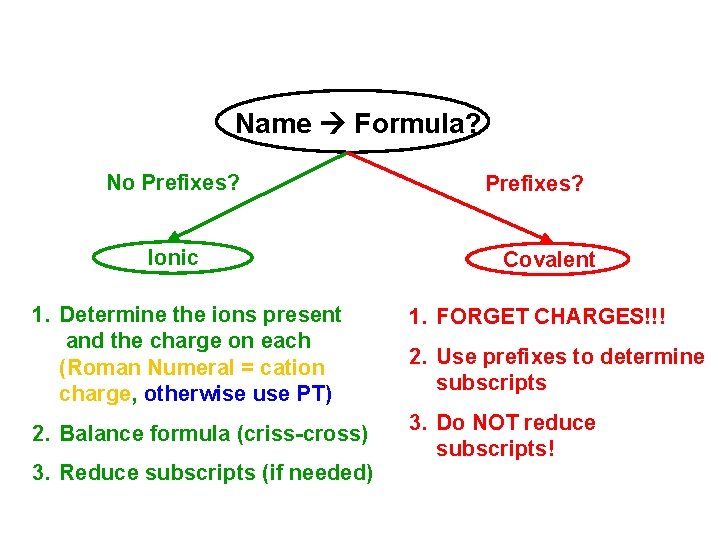
Name Formula? No Prefixes? Ionic Prefixes? Covalent 1. Determine the ions present and the charge on each (Roman Numeral = cation charge, otherwise use PT) 1. FORGET CHARGES!!! 2. Balance formula (criss-cross) 3. Do NOT reduce subscripts! 3. Reduce subscripts (if needed) 2. Use prefixes to determine subscripts
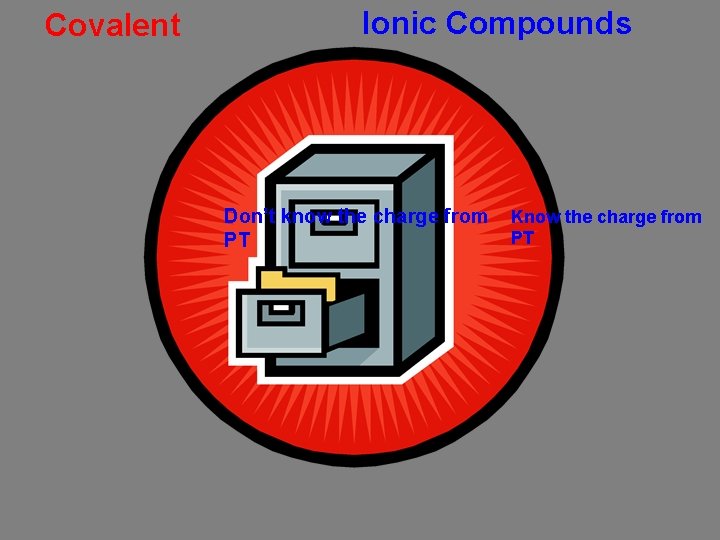
Covalent Ionic Compounds Two nonmetals Variable-charge cation carbon sulfur tetrabromide N NCl O 35 2 dichloride vanadium niobium. Mn Pt(IO (V) (II) Sperchlorate 2 53)chromate 4 rubidium sulfate NH 4 KI Cl. Ooxide barium 3 Roman numeral for name only Roman numeral Greek prefixes Charge Criss. Cross Rule Roman numeral Fixed-charge Don’t know the charge from Know the charge from PT Polyatomic ions OK Where would you file this? VCr. O 4 dinitrogen pentoxide Ba. O platinum (IV) iodate CBr 4 ammonium chlorate Nb(Cl. O 4)5 potassium iodide SCl 2 nitrogen trichloride Rb 2 SO 4 manganese (V) sulfide

Overall Nomenclature Practice Ionic/variable Ir F 3 fluoride iridium (III) Ca S sulfide calcium Ti S 2 sulfide titanium (IV) Ionic/variable HCl(aq) hydrochloric acid Acid Ba Br 2 bromide barium Ionic/fixed N 3 P 2 diphosphide trinitrogen Covalent HF(aq) hydrofluoric acid Zn I 2 zinc iodide Ionic/fixed NCl 4 tetrachloride nitrogen Covalent Au 2 O 3 oxide gold (III) Na 3 P phosphide sodium Ionic/fixed Acid Ionic/variable Ionic/fixed

Ionic Nomenclature Practice Ionic/variable Ir 2 (Cr 2 O 7)3 iridium (III) dichromate Ca (OH)2 calcium hydroxide HNO 3 (aq) nitric acid Pt (CH 3 COO)2 platinum (II) acetate HBr. O 2(aq) bromous acid Sr SO 4 strontium sulfate Ionic/fixed KCN potassium cyanide Ionic/fixed Zn (NO 2)2 zinc nitrite Ionic/fixed Mn (Cl. O 3)4 manganese (IV) chlorate Ionic/variable Au PO 4 gold (III) phosphate Ionic/variable H SO acid 2 3 (aq) sulfurous Ionic/fixed Acid Ionic/variable Acid

Write the compound formed by the following ions: 1) Al 3+ S 22) Mg 2+ PO 43 - When a formula is given…write the proper name. When a name is given…write the proper formula. 3) Ba. O 4) lithium bromide 5) Ni 2 S 3 6) triphosphorus heptoxide 7) N 2 O 5 8) molybdenum (VI) nitride Write the total number of atoms that make up each compound. 9) trinitrotoluene (TNT)… CH 3 C 6 H 2(NO 2)3 10) phosphoric acid H 3 PO 4 Extra credit: What is the formula for plumbic iodide? (Hint: lead is Pb 2+ or Pb 4+)

Write the compound formed by the following ions: 1) Al 3+ S 22) Mg 2+ PO 43 - When a formula is given…write the proper name. When a name is given…write the proper formula. 3) Ba. O POP QUIZ 4) lithium bromide 5) Ni 2 S 3 6) triphosphorus heptoxide 7) N 2 O 5 8) molybdenum (VI) nitride Write the total number of atoms that make up each compound. 9) trinitrotoluene (TNT)… CH 3 C 6 H 2(NO 2)3 10) phosphoric acid H 3 PO 4 Extra credit: What is the formula for plumbic iodide? (Hint: lead is Pb 2+ or Pb 4+)

Answer Key Write the compound formed by the following ions: 1) Al 3+ S 22) Mg 2+ Al 2 S 3 Mg 3(PO 4)2 PO 43 - When a formula is given…write the proper name. When a name is given…write the proper formula. 3) Ba. O barium oxide Li. Br 4) lithium bromide 5) Ni 2 S 3 nickel (III) sulfide P 3 O 7 6) triphosphorus heptoxide 7) N 2 O 5 dinitrogen pentoxide 8) molybdenum (VI) nitride Write the total number of atoms that make up each compound. 9) trinitrotoluene (TNT)… CH 3 C 6 H 2(NO 2)3 10) phosphoric acid H 3 PO 4 Mo. N 2 21 8 Extra credit: What is the formula for plumbic iodide? (Hint: lead is Pb 2+ or Pb 4+) Pb. I 4
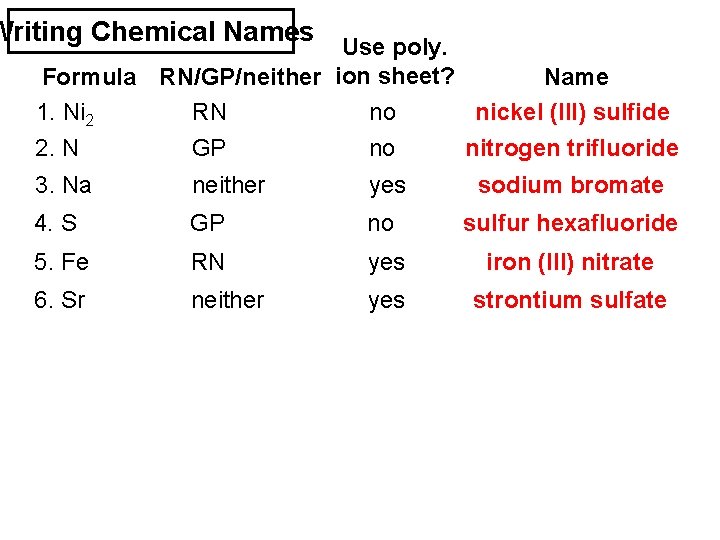
Writing Chemical Names Use poly. Formula RN/GP/neither ion sheet? Name 1. Ni 2 S 3 RN no nickel (III) sulfide 2. N F 3 GP no nitrogen trifluoride 3. Na Br. O 3 neither yes sodium bromate 4. S F 6 GP no sulfur hexafluoride 5. Fe (NO 3)3 RN yes iron (III) nitrate 6. Sr SO 4 neither yes strontium sulfate

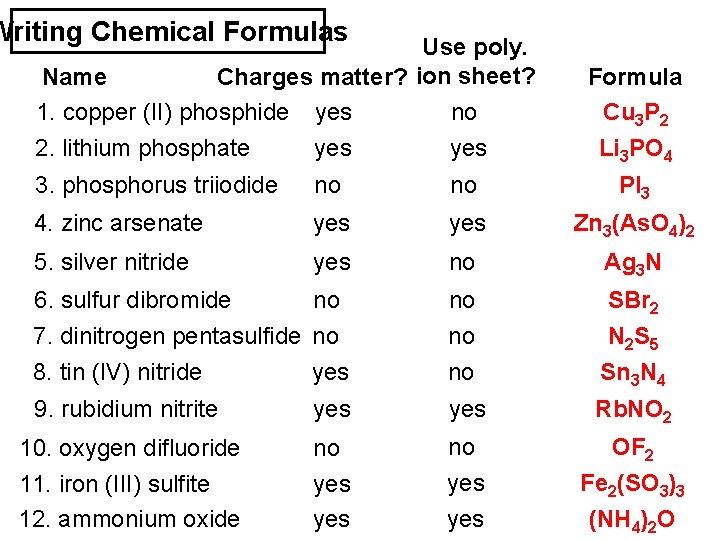
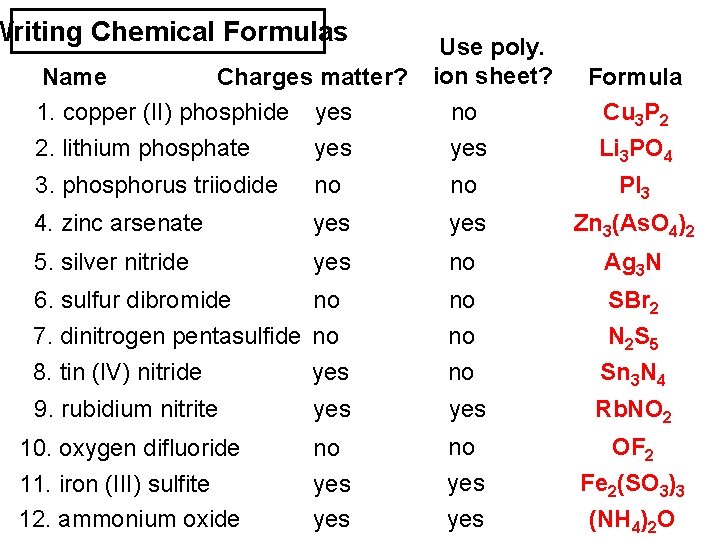
Writing Chemical Formulas Use poly. Name Charges matter? ion sheet? 1. copper (II) phosphide yes no 2. lithium phosphate yes Formula Cu 3 P 2 Li 3 PO 4 3. phosphorus triiodide no no PI 3 4. zinc arsenate yes Zn 3(As. O 4)2 5. silver nitride yes no Ag 3 N 6. sulfur dibromide no no SBr 2 7. dinitrogen pentasulfide no 8. tin (IV) nitride yes no no N 2 S 5 Sn 3 N 4 9. rubidium nitrite yes Rb. NO 2 10. oxygen difluoride no no OF 2 11. iron (III) sulfite 12. ammonium oxide yes yes Fe 2(SO 3)3 (NH 4)2 O
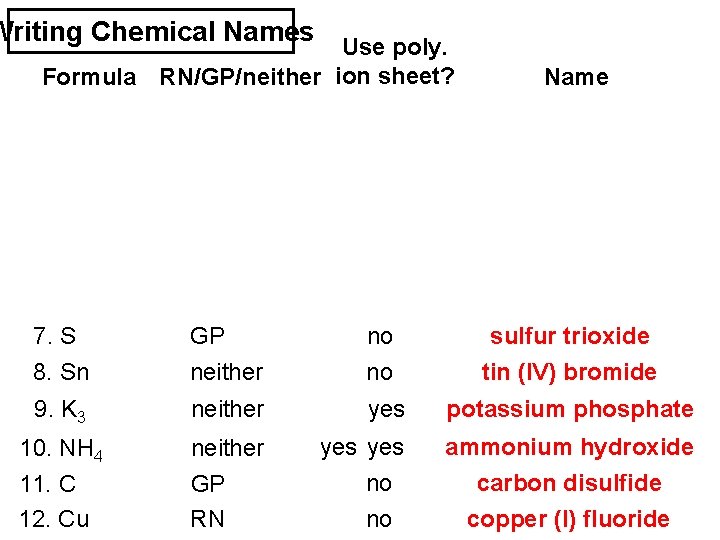
Writing Chemical Names Use poly. Formula RN/GP/neither ion sheet? Name 7. S O 3 8. Sn Br 4 GP neither no no sulfur trioxide tin (IV) bromide 9. K 3 PO 4 neither yes potassium phosphate 10. NH 4 OH neither yes ammonium hydroxide 11. C S 2 12. Cu F GP RN no no carbon disulfide copper (I) fluoride

Writing Chemical Formulas Use poly. Name Charges matter? ion sheet? 1. copper (II) phosphide yes no 2. lithium phosphate yes Formula Cu 3 P 2 Li 3 PO 4 3. phosphorus triiodide no no PI 3 4. zinc arsenate yes Zn 3(As. O 4)2 5. silver nitride yes no Ag 3 N 6. sulfur dibromide no no SBr 2 7. dinitrogen pentasulfide no 8. tin (IV) nitride yes no no N 2 S 5 Sn 3 N 4 9. rubidium nitrite yes Rb. NO 2 10. oxygen difluoride no no OF 2 11. iron (III) sulfite 12. ammonium oxide yes yes Fe 2(SO 3)3 (NH 4)2 O
Writing Chemical Formulas Use poly. Name Charges matter? ion sheet? 1. copper (II) phosphide yes no 2. lithium phosphate yes Formula Cu 3 P 2 Li 3 PO 4 3. phosphorus triiodide no no PI 3 4. zinc arsenate yes Zn 3(As. O 4)2 5. silver nitride yes no Ag 3 N 6. sulfur dibromide no no SBr 2 7. dinitrogen pentasulfide no 8. tin (IV) nitride yes no no N 2 S 5 Sn 3 N 4 9. rubidium nitrite yes Rb. NO 2 10. oxygen difluoride no no OF 2 11. iron (III) sulfite 12. ammonium oxide yes yes Fe 2(SO 3)3 (NH 4)2 O
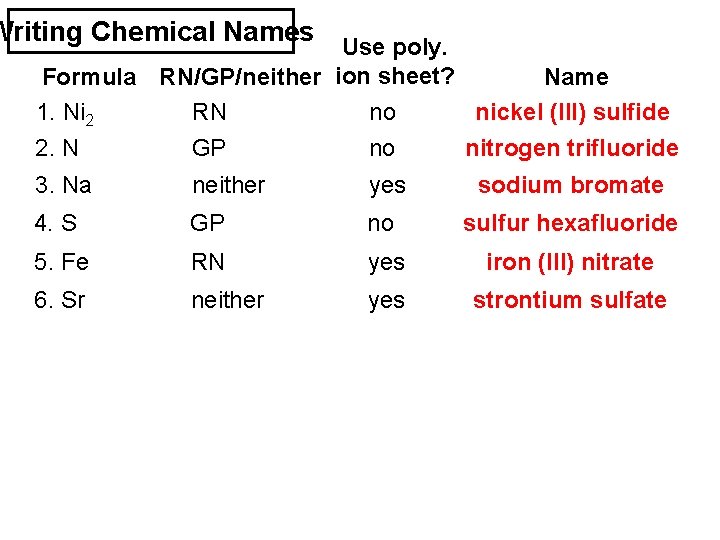
Writing Chemical Names Use poly. Formula RN/GP/neither ion sheet? Name 1. Ni 2 S 3 RN no nickel (III) sulfide 2. N F 3 GP no nitrogen trifluoride 3. Na Br. O 3 neither yes sodium bromate 4. S F 6 GP no sulfur hexafluoride 5. Fe (NO 3)3 RN yes iron (III) nitrate 6. Sr SO 4 neither yes strontium sulfate 7. S O 3 8. Sn Br 4 GP neither no no sulfur trioxide tin (IV) bromide 9. K 3 PO 4 neither yes potassium phosphate 10. NH 4 OH neither yes ammonium hydroxide 11. C S 2 12. Cu F GP RN no no carbon disulfide copper (I) fluoride
Writing Chemical Formulas Name Charges matter? 1. copper (II) phosphide yes 2. lithium phosphate yes Use poly. ion sheet? no yes Formula Cu 3 P 2 Li 3 PO 4 3. phosphorus triiodide no no PI 3 4. zinc arsenate yes Zn 3(As. O 4)2 5. silver nitride yes no Ag 3 N 6. sulfur dibromide no no SBr 2 7. dinitrogen pentasulfide no 8. tin (IV) nitride yes no no N 2 S 5 Sn 3 N 4 9. rubidium nitrite yes Rb. NO 2 10. oxygen difluoride no no OF 2 11. iron (III) sulfite 12. ammonium oxide yes yes Fe 2(SO 3)3 (NH 4)2 O
- Slides: 64