Organic Chemistry The chemistry of carbon and carbonbased

Organic Chemistry The chemistry of carbon and carbon-based compounds
Why is carbon so special? Carbon is unique among the elements because: -- it can have up to four bonds per C atom single, double, or triple bonds –C–C– C=C –C=C– -- it can form REALLY long chains of C atoms -- its bonds are strong
Organic Chemistry is everywhere! Smells & tastes fruits, chocolate, fish, mint Medications Aspirin, Tylenol, Decongestants, Sedatives Addictive substances Caffeine, Nicotine, Alcohol, Narcotics Hormones/Neurotransmitters Adrenaline, Epinephrine Food/Nutrients Carbohydrates, Protein, Fat, Vitamins Genetics DNA, RNA Consumer products Plastics, Nylon, Rayon, Polyester
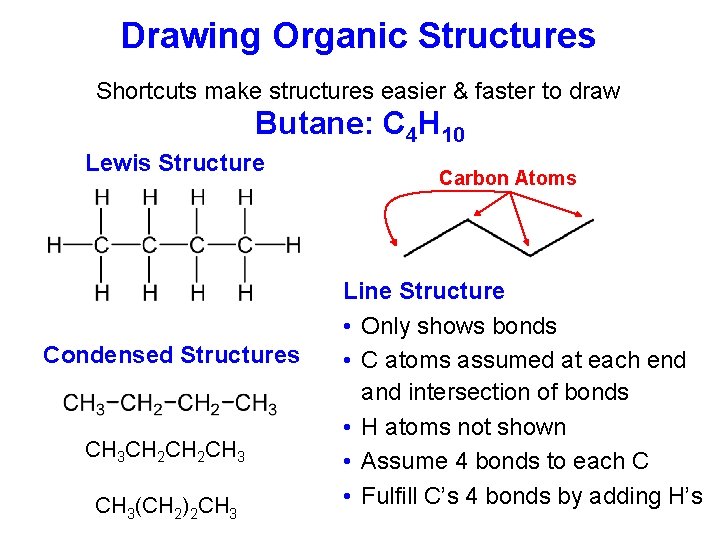
Drawing Organic Structures Shortcuts make structures easier & faster to draw Butane: C 4 H 10 Lewis Structure Condensed Structures CH 3 CH 2 CH 3(CH 2)2 CH 3 Carbon Atoms Line Structure • Only shows bonds • C atoms assumed at each end and intersection of bonds • H atoms not shown • Assume 4 bonds to each C • Fulfill C’s 4 bonds by adding H’s
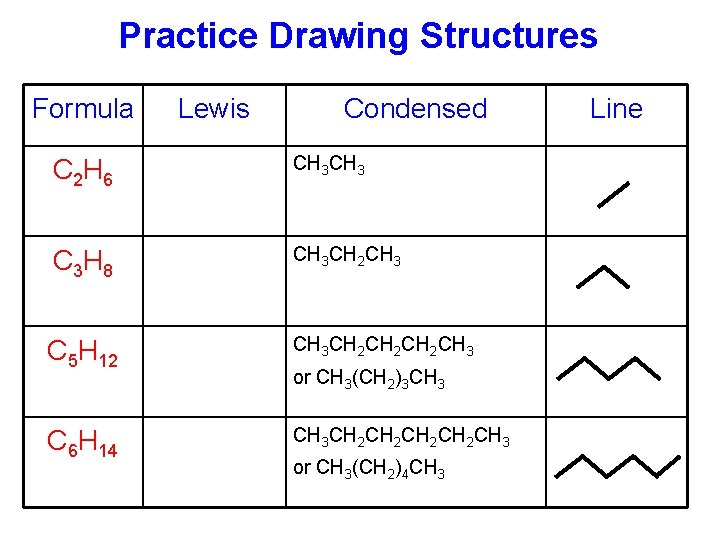
Practice Drawing Structures Formula Lewis Condensed C 2 H 6 CH 3 C 3 H 8 CH 3 CH 2 CH 3 C 5 H 12 CH 3 CH 2 CH 2 CH 3 C 6 H 14 CH 3 CH 2 CH 2 CH 3 or CH 3(CH 2)3 CH 3 or CH 3(CH 2)4 CH 3 Line
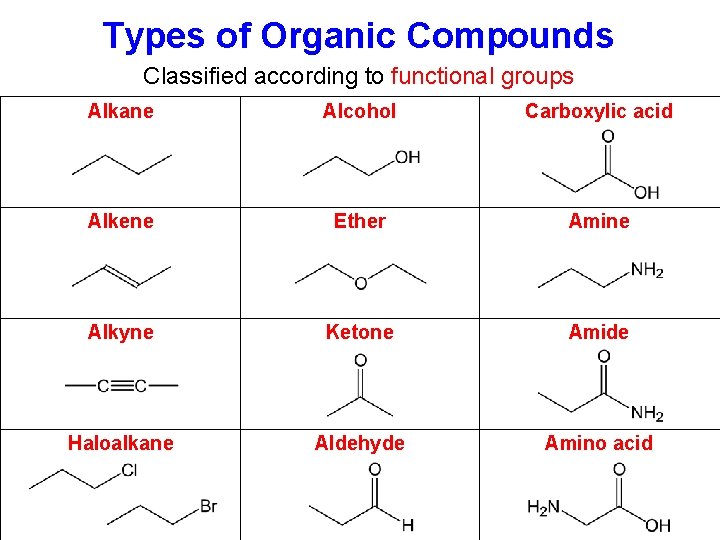
Types of Organic Compounds Classified according to functional groups Alkane Alcohol Carboxylic acid Alkene Ether Amine Alkyne Ketone Amide Haloalkane Aldehyde Amino acid

Big Idea in Organic Chemistry Structure controls Function Each functional group has predictable reactivity
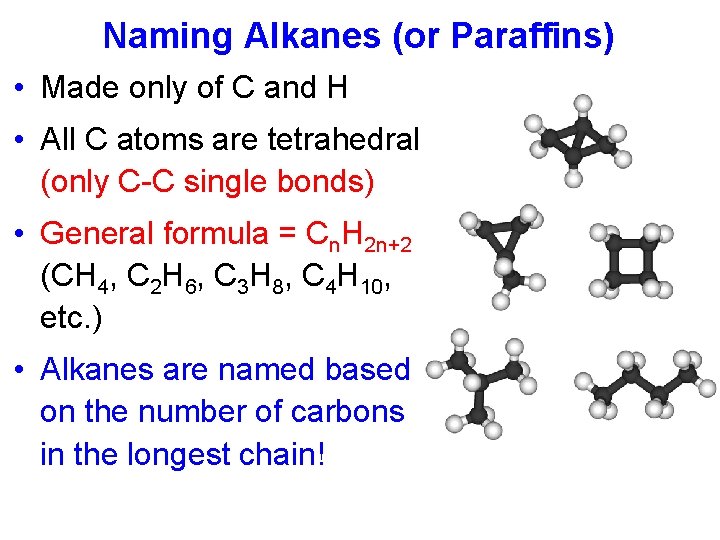
Naming Alkanes (or Paraffins) • Made only of C and H • All C atoms are tetrahedral (only C-C single bonds) • General formula = Cn. H 2 n+2 (CH 4, C 2 H 6, C 3 H 8, C 4 H 10, etc. ) • Alkanes are named based on the number of carbons in the longest chain!
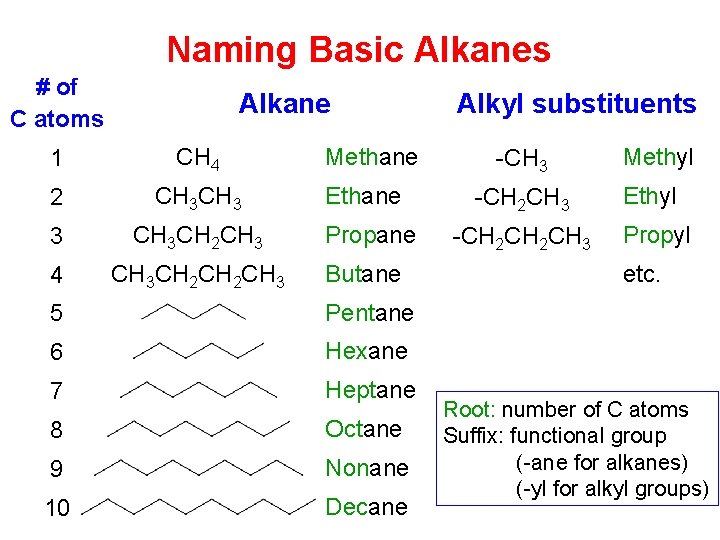
Naming Basic Alkanes # of C atoms Alkane 1 CH 4 2 CH 3 3 CH 3 CH 2 CH 3 4 CH 3 CH 2 CH 3 Methane Ethane Propane Butane 5 Pentane 6 Hexane 7 Heptane 8 Octane 9 Nonane 10 Decane Alkyl substituents -CH 3 -CH 2 CH 2 CH 3 Methyl Ethyl Propyl etc. Root: number of C atoms Suffix: functional group (-ane for alkanes) (-yl for alkyl groups)
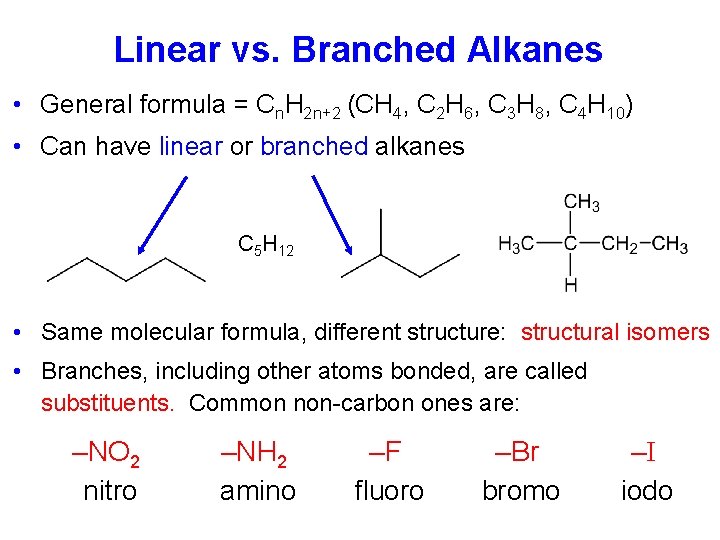
Linear vs. Branched Alkanes • General formula = Cn. H 2 n+2 (CH 4, C 2 H 6, C 3 H 8, C 4 H 10) • Can have linear or branched alkanes C 5 H 12 • Same molecular formula, different structure: structural isomers • Branches, including other atoms bonded, are called substituents. Common non-carbon ones are: –NO 2 nitro –NH 2 amino –F fluoro –Br bromo –I iodo
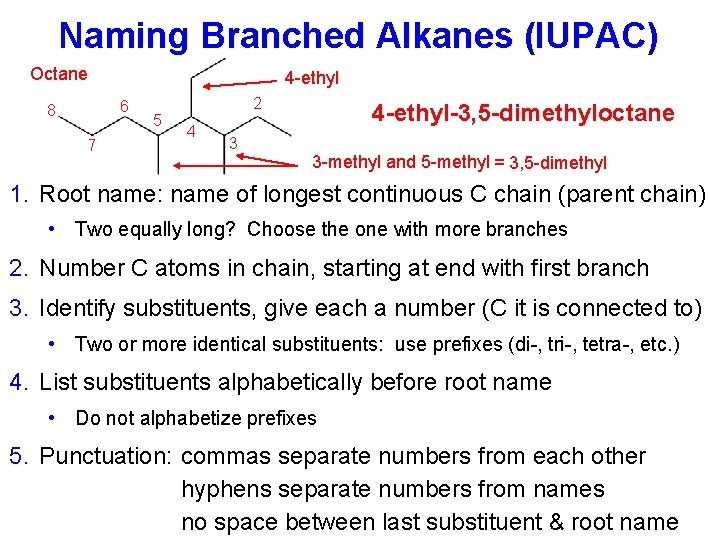
Naming Branched Alkanes (IUPAC) Octane 4 -ethyl 6 8 7 5 2 4 4 -ethyl-3, 5 -dimethyloctane 3 3 -methyl and 5 -methyl = 3, 5 -dimethyl 1. Root name: name of longest continuous C chain (parent chain) • Two equally long? Choose the one with more branches 2. Number C atoms in chain, starting at end with first branch 3. Identify substituents, give each a number (C it is connected to) • Two or more identical substituents: use prefixes (di-, tri-, tetra-, etc. ) 4. List substituents alphabetically before root name • Do not alphabetize prefixes 5. Punctuation: commas separate numbers from each other hyphens separate numbers from names no space between last substituent & root name
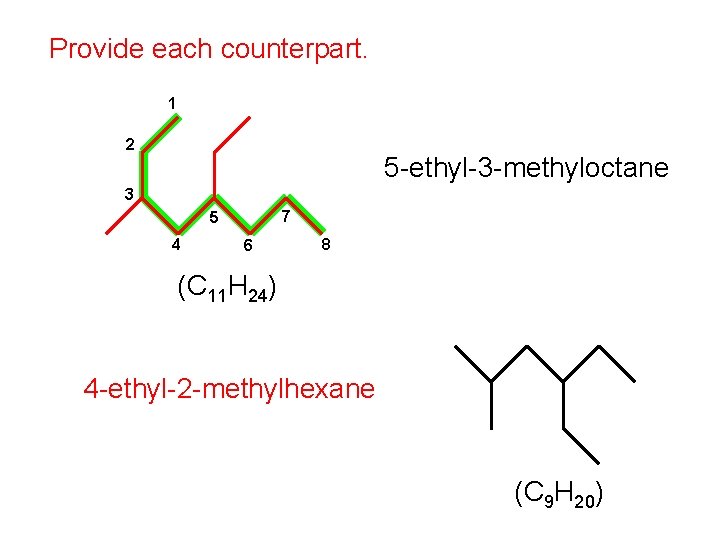
Provide each counterpart. 1 2 5 -ethyl-3 -methyloctane 3 7 5 4 6 8 (C 11 H 24) 4 -ethyl-2 -methylhexane (C 9 H 20)
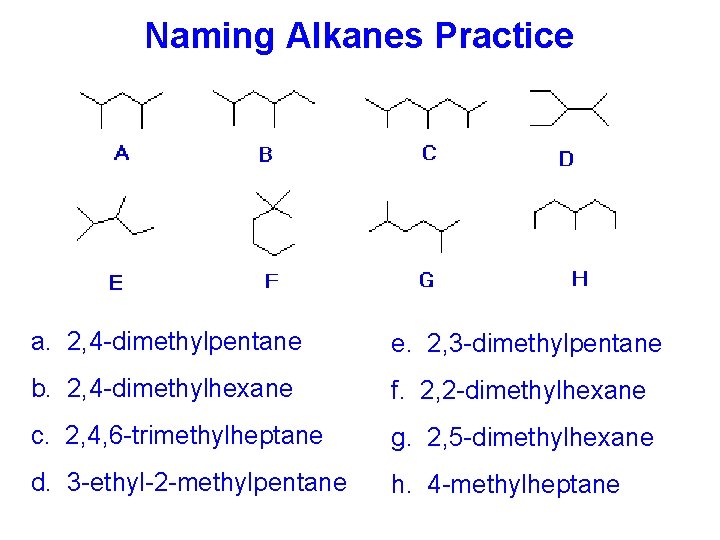
Naming Alkanes Practice a. 2, 4 -dimethylpentane e. 2, 3 -dimethylpentane b. 2, 4 -dimethylhexane f. 2, 2 -dimethylhexane c. 2, 4, 6 -trimethylheptane g. 2, 5 -dimethylhexane d. 3 -ethyl-2 -methylpentane h. 4 -methylheptane

Isomers The fat dog shook himself, and then rolled over on the wet rug. OR The dog shook the fat rug, then rolled over and wet on himself. These two statements use the same words. . . but have very different meanings! Likewise, isomers may have the same formula, but have very different structures…

Structural Isomer Practice • On piece of your own paper, draw AND name ALL of the isomers for the following alkanes: # isomers Formulas C 5 H 12 3 C 6 H 14 5 C 7 H 16 9 Some of your drawings may look different, but they are only different structures (isomers) if they also have different names If you complete that, try to draw and name all of the isomers for octane (C 8 H 18) in your HW. There are 18 of them!
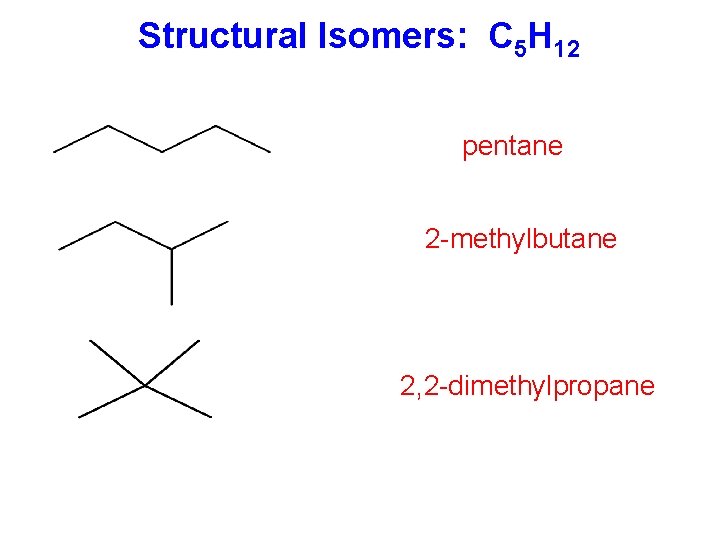
Structural Isomers: C 5 H 12 pentane 2 -methylbutane 2, 2 -dimethylpropane
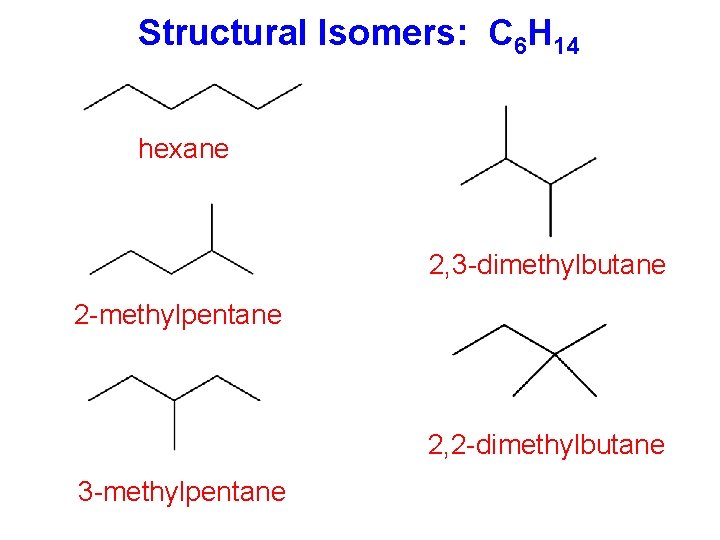
Structural Isomers: C 6 H 14 hexane 2, 3 -dimethylbutane 2 -methylpentane 2, 2 -dimethylbutane 3 -methylpentane
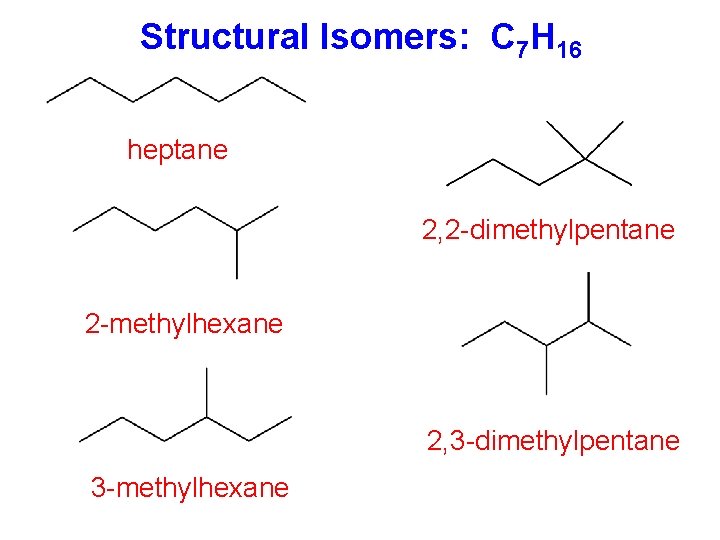
Structural Isomers: C 7 H 16 heptane 2, 2 -dimethylpentane 2 -methylhexane 2, 3 -dimethylpentane 3 -methylhexane
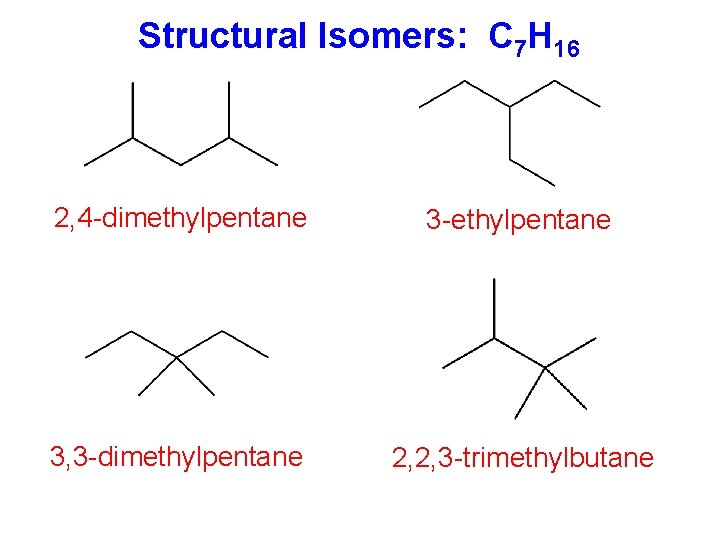
Structural Isomers: C 7 H 16 2, 4 -dimethylpentane 3 -ethylpentane 3, 3 -dimethylpentane 2, 2, 3 -trimethylbutane
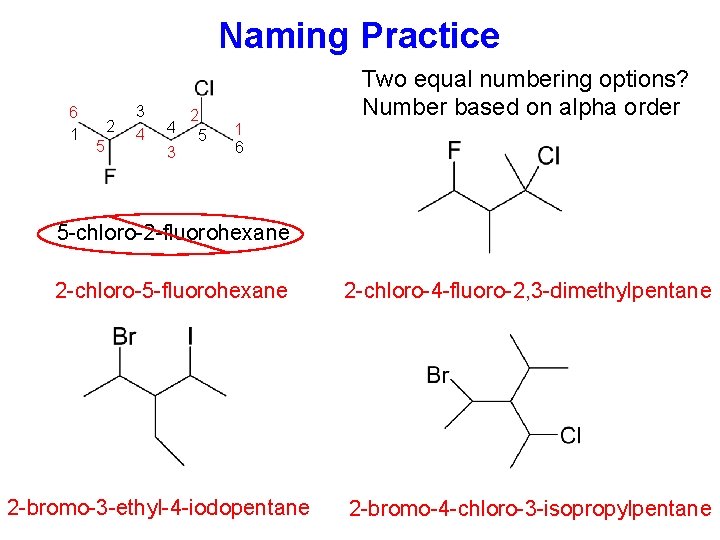
Naming Practice 6 1 2 5 3 4 4 3 2 5 1 6 Two equal numbering options? Number based on alpha order 5 -chloro-2 -fluorohexane 2 -chloro-5 -fluorohexane 2 -bromo-3 -ethyl-4 -iodopentane 2 -chloro-4 -fluoro-2, 3 -dimethylpentane 2 -bromo-4 -chloro-3 -isopropylpentane
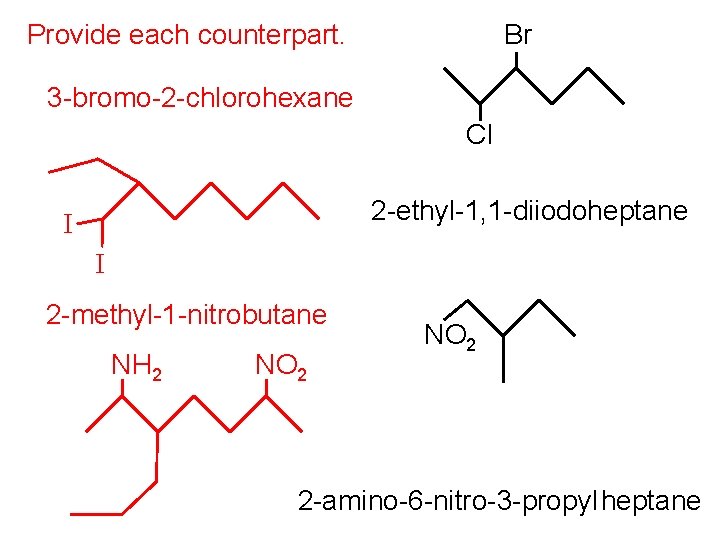
Provide each counterpart. Br 3 -bromo-2 -chlorohexane Cl 2 -ethyl-1, 1 -diiodoheptane I I 2 -methyl-1 -nitrobutane NH 2 NO 2 2 -amino-6 -nitro-3 -propyl heptane
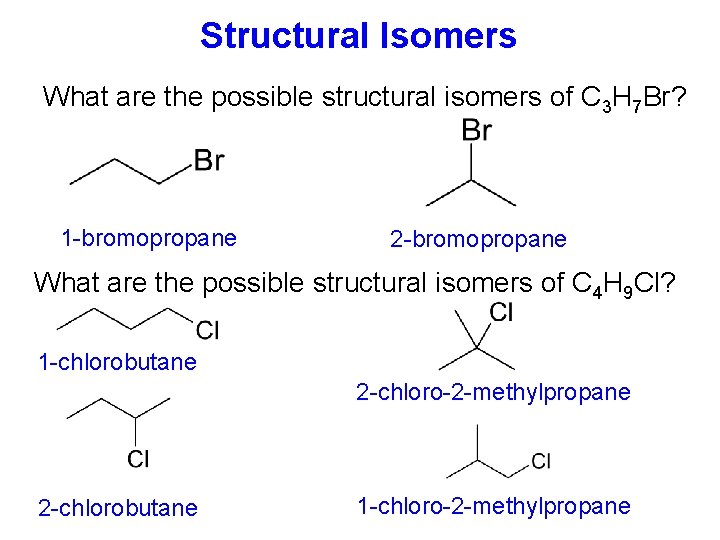
Structural Isomers What are the possible structural isomers of C 3 H 7 Br? 1 -bromopropane 2 -bromopropane What are the possible structural isomers of C 4 H 9 Cl? 1 -chlorobutane 2 -chloro-2 -methylpropane 2 -chlorobutane 1 -chloro-2 -methylpropane
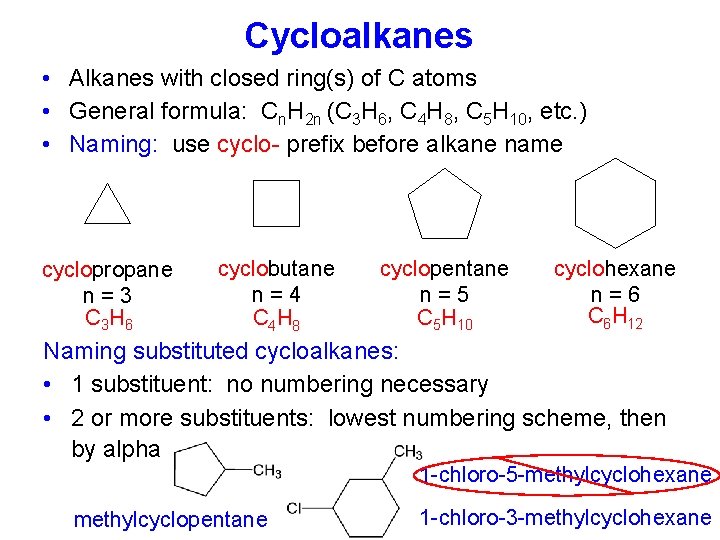
Cycloalkanes • Alkanes with closed ring(s) of C atoms • General formula: Cn. H 2 n (C 3 H 6, C 4 H 8, C 5 H 10, etc. ) • Naming: use cyclo- prefix before alkane name cyclopropane n=3 C 3 H 6 cyclobutane n=4 C 4 H 8 cyclopentane n=5 C 5 H 10 cyclohexane n=6 C 6 H 12 Naming substituted cycloalkanes: • 1 substituent: no numbering necessary • 2 or more substituents: lowest numbering scheme, then by alpha 1 -chloro-5 -methylcyclohexane methylcyclopentane 1 -chloro-3 -methylcyclohexane
Fun Common Names windowpane basketane broken windowpane boxane

Teepane
- Slides: 25