Bohr Diagrams Bohr diagrams show many electrons appear

Bohr Diagrams: § Bohr diagrams show many electrons appear in each electron shell around an atom. § Each shell holds a maximum number of electrons (2, 8, 8, 18) § Electrons in the outermost shell are called valence electrons § If the valence shell is full = the atom is stable § If the valence shell is not full = the atom is not stable Note: Think of the shells as being 3 D like spheres, not 2 D like circles!
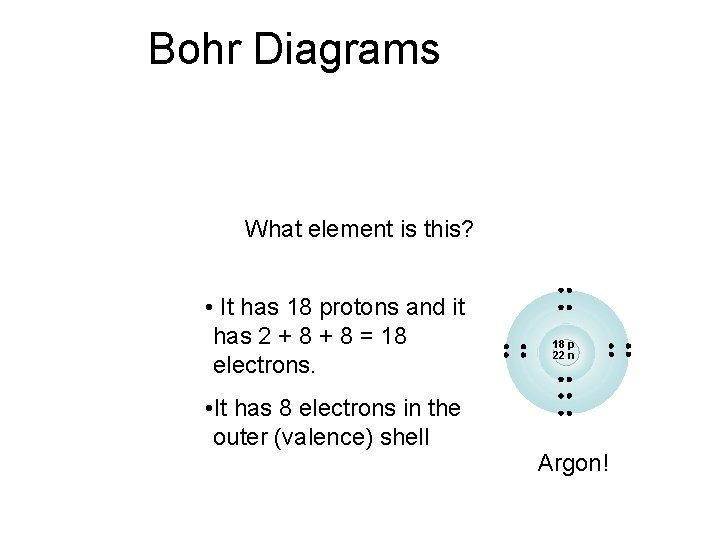
Bohr Diagrams What element is this? • It has 18 protons and it has 2 + 8 = 18 electrons. • It has 8 electrons in the outer (valence) shell 18 p 22 n Argon!
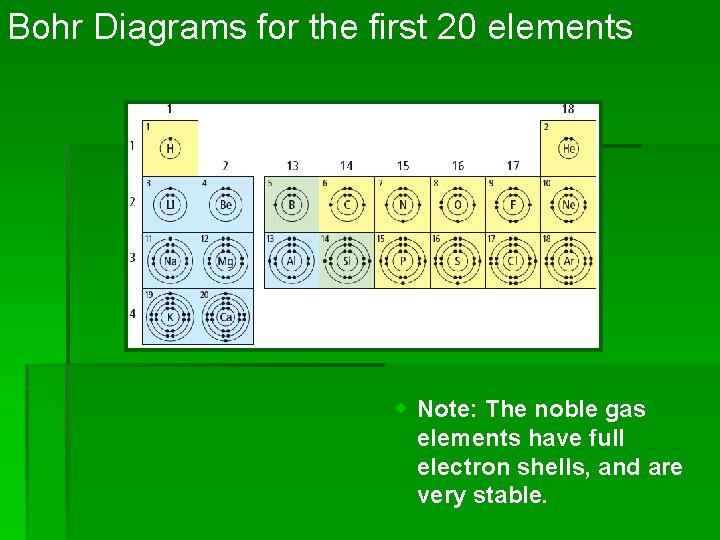
Bohr Diagrams for the first 20 elements w Note: The noble gas elements have full electron shells, and are very stable.

Bonding Types: § When two atoms get close together, their valence electrons interact.

Ionic Bonds: § Metals give electrons to non-metals (transfer of valence electrons) § cations (+ ions) and anions (- ions) form § For example, lithium and oxygen form an ionic bond in the compound Li 2 O + Lithium Oxygen Electrons are transferred from the cations to the anion Li+ O 2 Li+ Lithium oxide, Li 2 O

§ Bohr Diagram for Li. F - Lithium fluoride On board

Covalent Bonds: § Formed between two or more non-metals § Valence electrons are shared between atoms § A group of covalently bonded atoms are called a molecule + Hydrogen fluoride Fluorine Electrons are shared

§ Bohr Diagram for CH 4 – Methane (or carbon tetrahydride) On Board

Lewis Diagrams: § Like simplified Bohr diagrams § Only valence electrons are shown § Dots representing electrons are placed around the element symbols (on 4 sides, imagine a box around the symbol) § Electron dots are placed singularly, until the fifth electron is reached, then they are paired. Example: Nitrogen atom
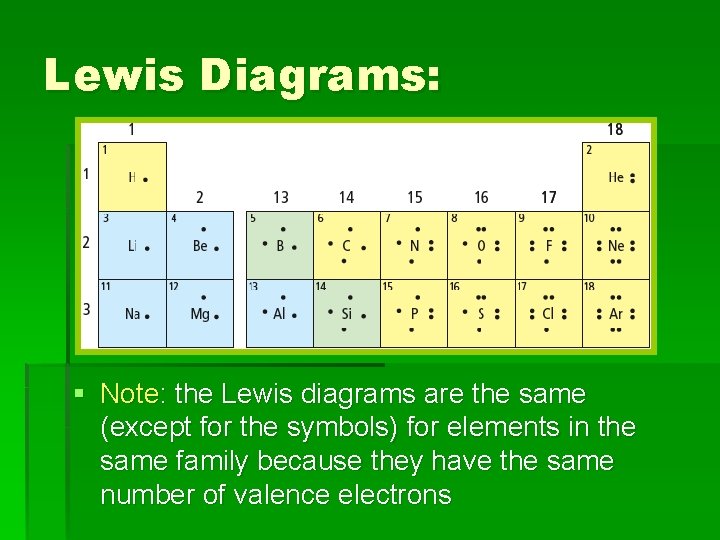
Lewis Diagrams: § Note: the Lewis diagrams are the same (except for the symbols) for elements in the same family because they have the same number of valence electrons

Lewis Diagrams for Ions: § For positive ions: one electron dot is removed from the valence shell for each positive charge of the ion. § For negative ions: one electron dot is added to each valence shell for each negative charge of the ion. § Square brackets and the charge are placed around each ion Example: Nitrogen ion
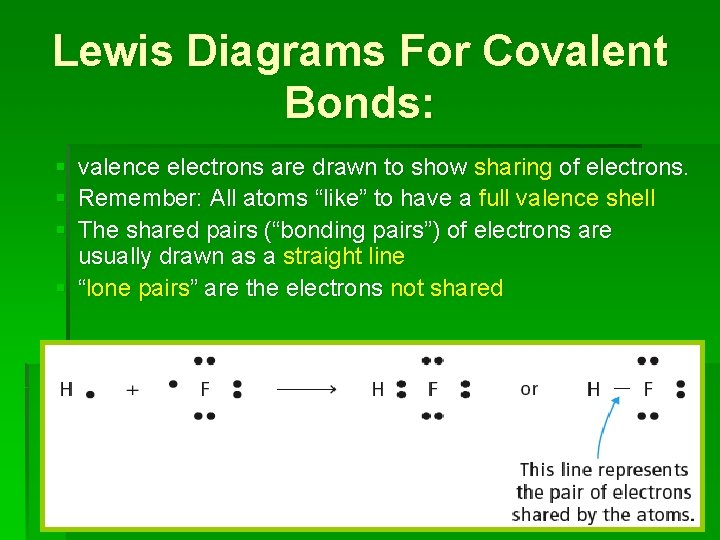
Lewis Diagrams For Covalent Bonds: § valence electrons are drawn to show sharing of electrons. § Remember: All atoms “like” to have a full valence shell § The shared pairs (“bonding pairs”) of electrons are usually drawn as a straight line § “lone pairs” are the electrons not shared

Lewis Diagrams For Ionic Bonds: – Be • • • Cl • • Be • • • • • • Cl • • Each beryllium has two electrons to transfer away, and each chlorine wants one more electron • • • • • Cl • • • Since Be 2+ wants to donate 2 electrons and each Cl– wants to accept only one, two Cl– ions are necessary • • Cl • • • Be • • • – 2+ • • • Cl • • • The ionic compound Beryllium chloride is formed
- Slides: 13