Introduction to macromolecule structure function Macromolecule types 1


















- Slides: 43

Introduction to macromolecule structure / function

Macromolecule types 1) carbohydrates 2) fats 3) nucleic acids 4) proteins

Big and small … Simplest Many unit = monomers together = polymer can polymerize indefinitely (exception: fats)

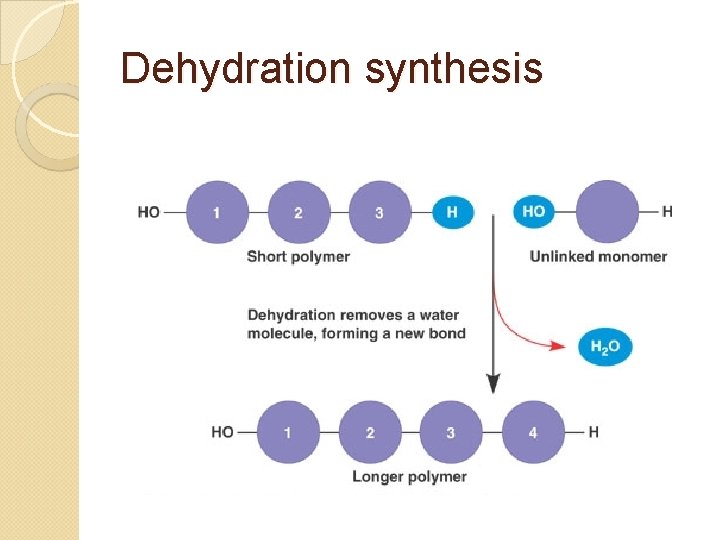
Putting together = dehydration synthesis Uses: storage, construction Reaction also produces H 2 O molecule (H and OH ripped off, combine)

Dehydration synthesis

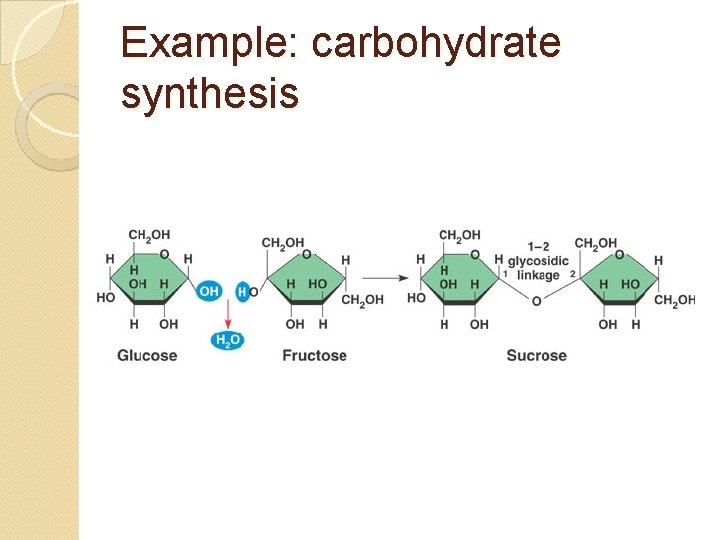
Example: carbohydrate synthesis

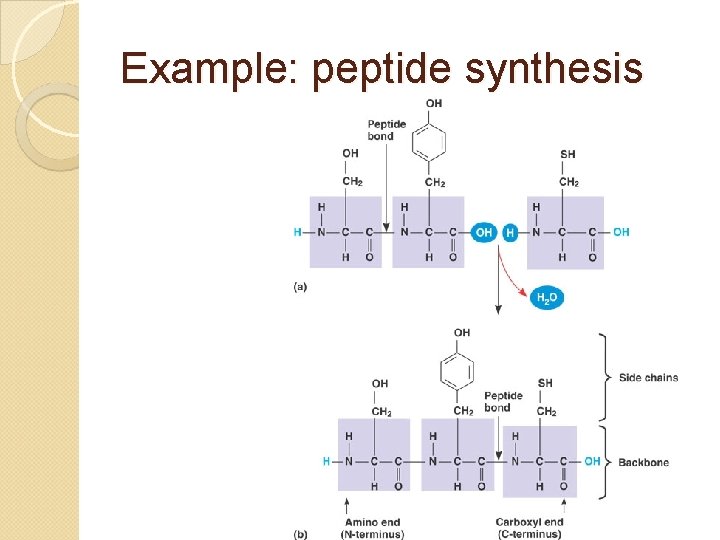
Example: peptide synthesis

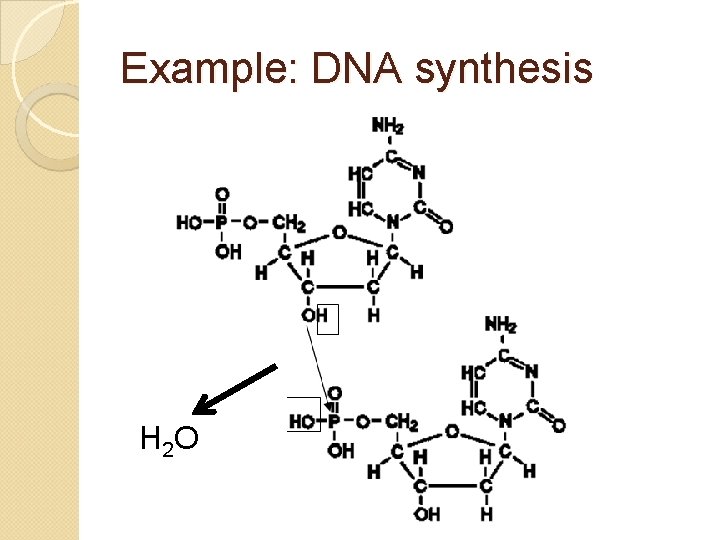
Example: DNA synthesis H 2 O

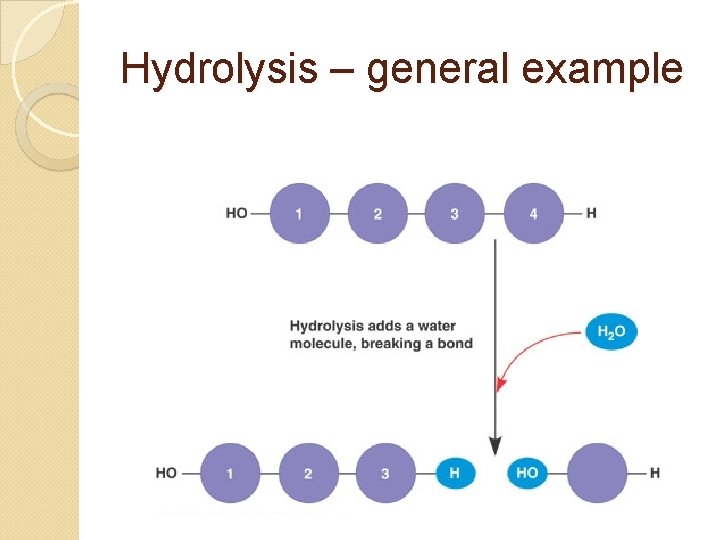
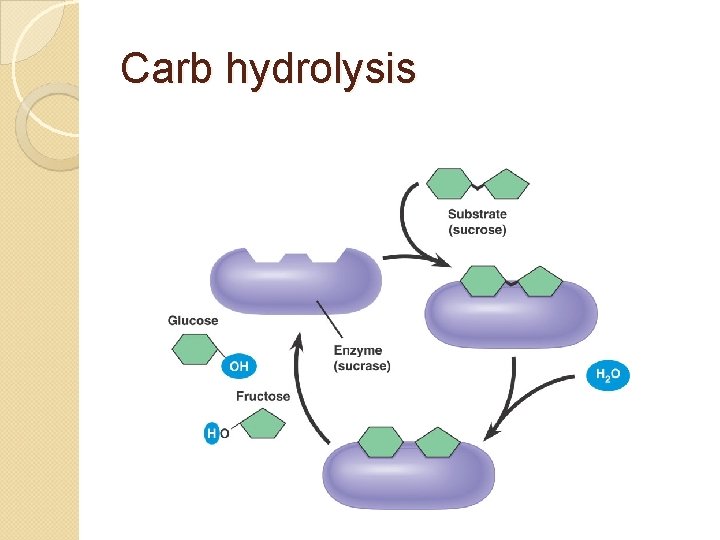
Breaking up polymers = hydrolysis – using water to split a polymer (also splits water itself as H is added to one side, OH to other) Uses: digestion, defense, recycling

Hydrolysis – general example

Carb hydrolysis

Macromolecule overview 1) carbohydrates Roots: –ose (not all the time) Components: C: H: O ~ 1: 2: 1 ratio monosaccharide is monomer

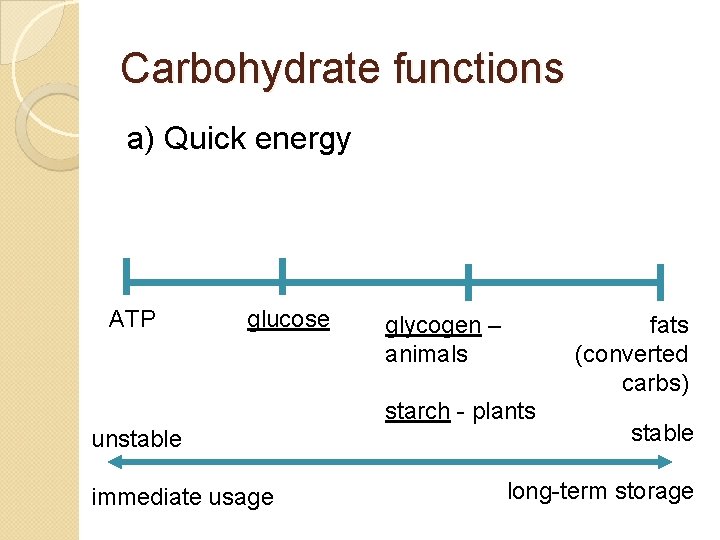
Carbohydrate functions a) Quick energy ATP glucose glycogen – animals fats (converted carbs) starch - plants unstable immediate usage stable long-term storage

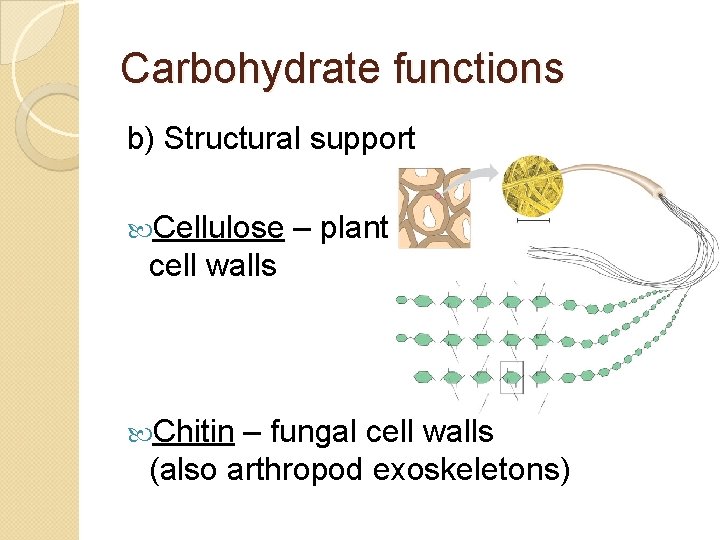
Carbohydrate functions b) Structural support Cellulose – plant cell walls Chitin – fungal cell walls (also arthropod exoskeletons)

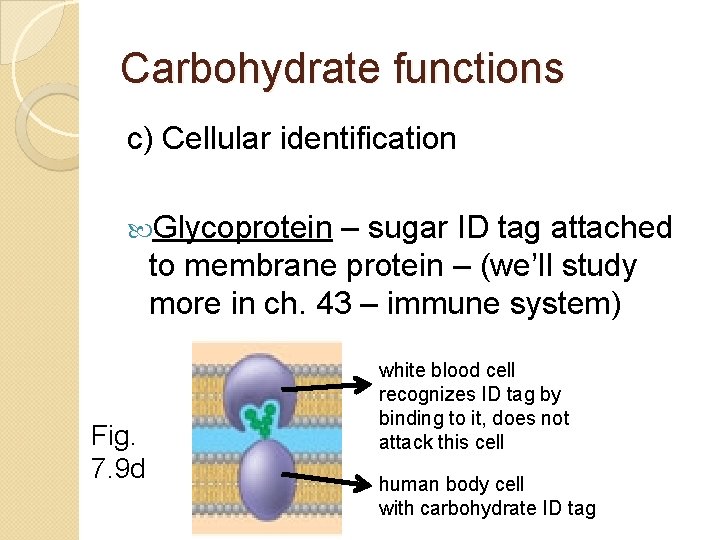
Carbohydrate functions c) Cellular identification Glycoprotein – sugar ID tag attached to membrane protein – (we’ll study more in ch. 43 – immune system) Fig. 7. 9 d white blood cell recognizes ID tag by binding to it, does not attack this cell human body cell with carbohydrate ID tag

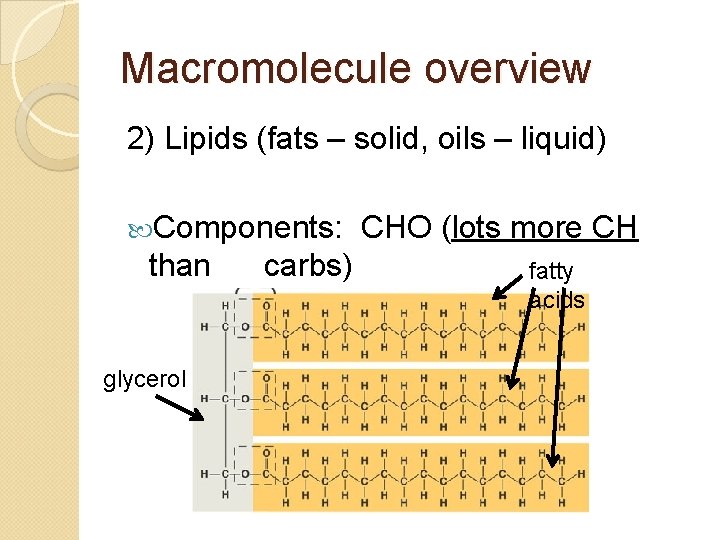
Macromolecule overview 2) Lipids (fats – solid, oils – liquid) Components: than glycerol carbs) CHO (lots more CH fatty acids

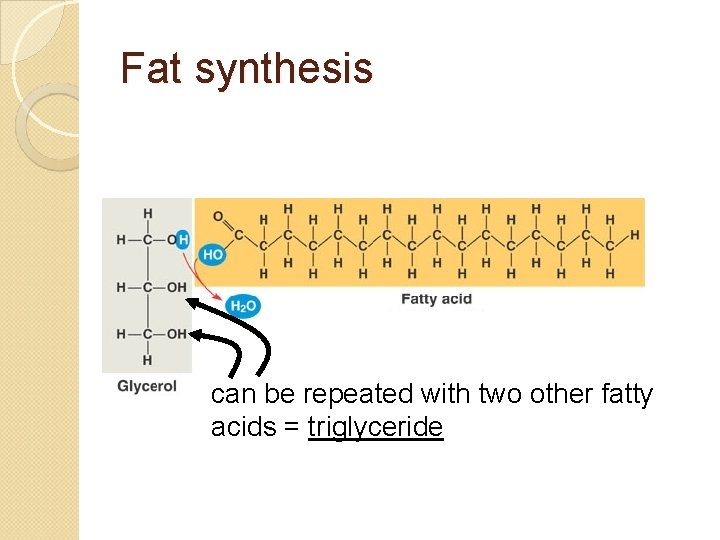
Fat synthesis can be repeated with two other fatty acids = triglyceride

Lipid functions a) Energy storage b) Insulation c) Cell membrane component (phospholipids)

Macromolecule overview 4) Proteins Roots: -ase (for many enzymes) Components: CHONS (sulfur present in certain amino acids) Monomer: amino acids

Protein functions - EVERYTHING a) enzymes – speed up chemical reactions transport proteins – let certain materials in / out of cell c) motor proteins – move things around in cell b) d) receptor proteins – receives signals sent to cell e) transcription factors – binds to DNA to regulate protein production) (and more … this is just a sample)

Protein structure / function How is diversity of function possible? How is specificity possible (each protein only interacts with one particular component)? Answer: each protein has unique conformation (= 3 -D shape) to interact with particular component

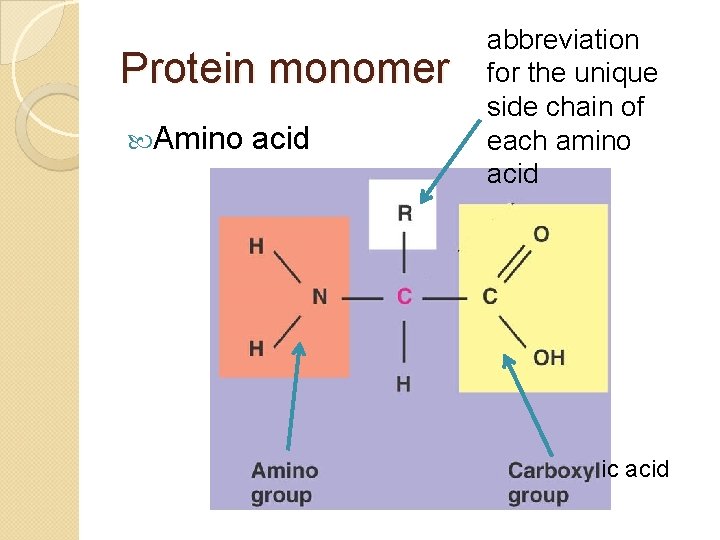
Protein monomer Amino acid abbreviation for the unique side chain of each amino acid ic acid

How get a unique 3 -D conformation? Must have unique combination of bonds that hold it in its precise shape We will talk about different types of bonds that form as a protein folds up Primary structure 2) Secondary structure 3) Tertiary structure 4) Quaternary structure 1) * Really important, please study carefully

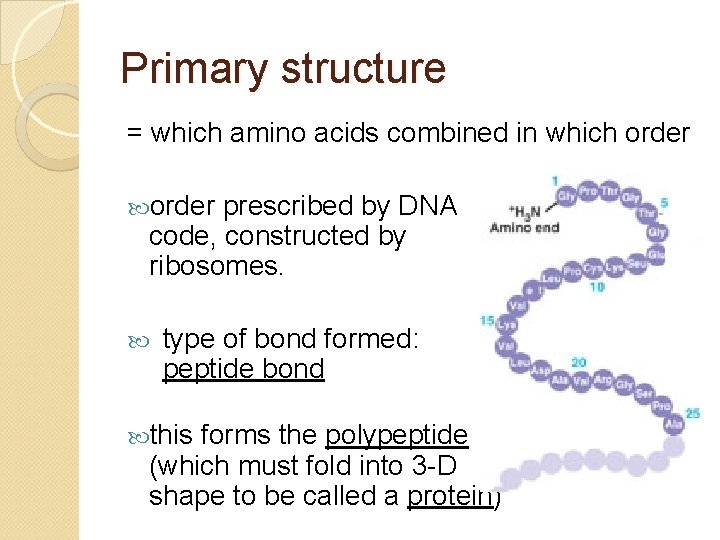
Primary structure = which amino acids combined in which order prescribed by DNA code, constructed by ribosomes. type of bond formed: peptide bond this forms the polypeptide (which must fold into 3 -D shape to be called a protein)

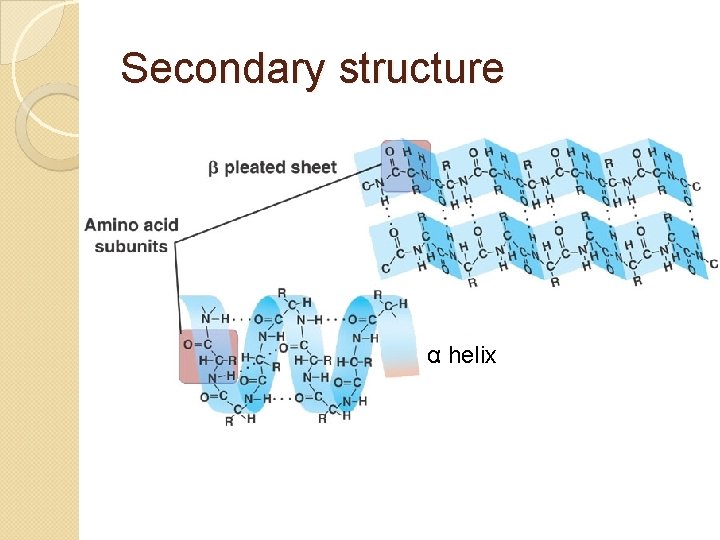
Secondary structure Interactions between “core” parts of amino acids (amino and carboxyl groups) that used to be far away Type of bond: hydrogen bonding (O—H N—H attractions) Because all amino acids have these parts, repeated hydrogen bonding forms two possible shapes: α-helix or β-pleated sheet

Secondary structure α helix

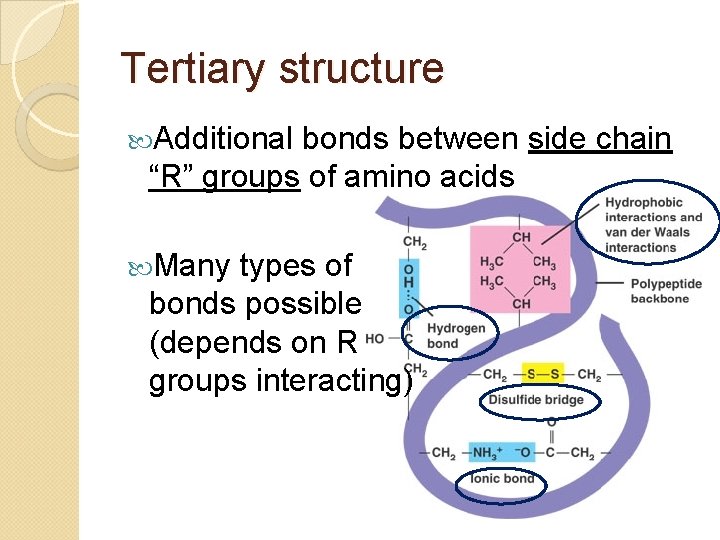
Tertiary structure Additional bonds between side chain “R” groups of amino acids Many types of bonds possible (depends on R groups interacting)

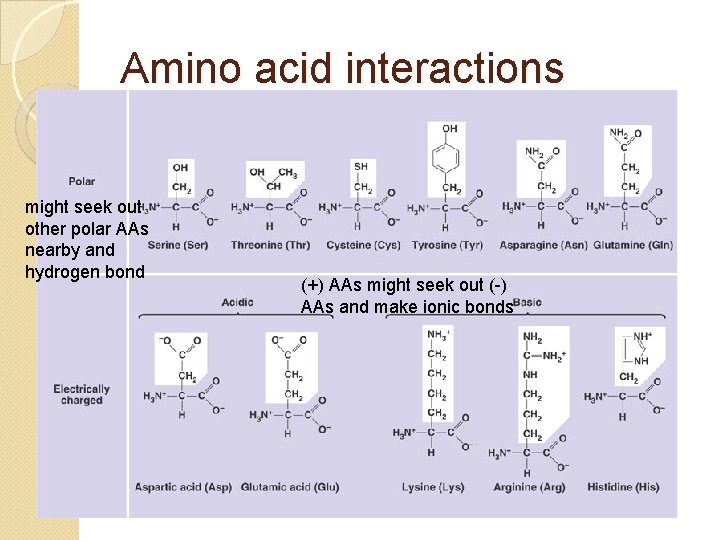
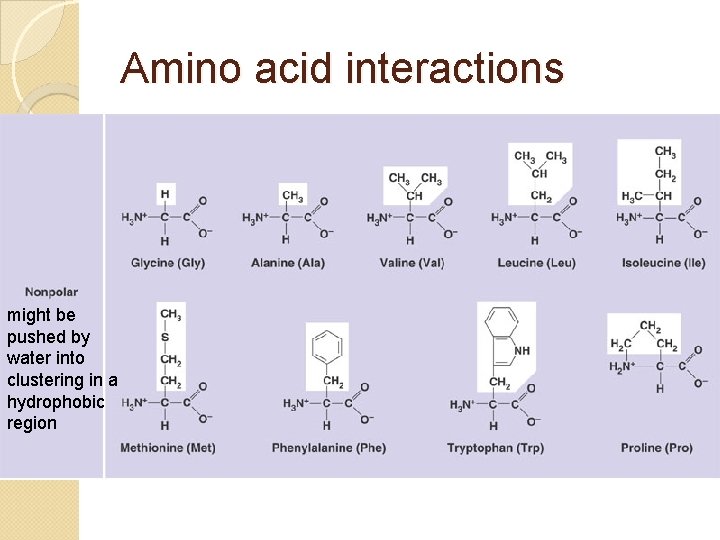
Amino acid interactions might seek out other polar AAs nearby and hydrogen bond (+) AAs might seek out (-) AAs and make ionic bonds

Amino acid interactions might be pushed by water into clustering in a hydrophobic region

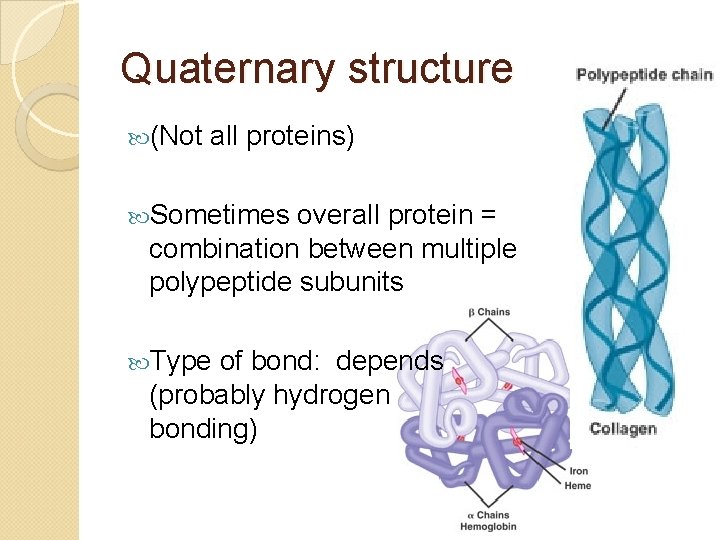
Quaternary structure (Not all proteins) Sometimes overall protein = combination between multiple polypeptide subunits Type of bond: depends (probably hydrogen bonding)

Result of all that bonding Uniquely We shaped protein will focus first on one subgroup: enzymes – bind to a specific substrate to speed up a chemical reaction

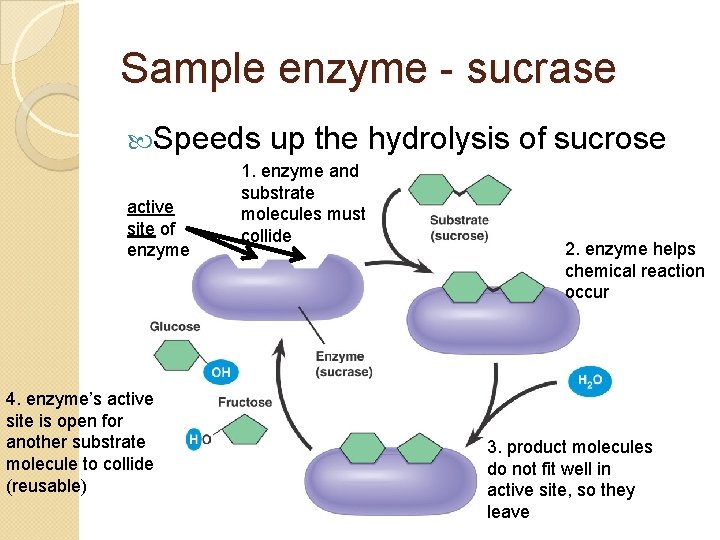
Sample enzyme - sucrase Speeds active site of enzyme 4. enzyme’s active site is open for another substrate molecule to collide (reusable) up the hydrolysis of sucrose 1. enzyme and substrate molecules must collide 2. enzyme helps chemical reaction occur 3. product molecules do not fit well in active site, so they leave

Environmental factors affect enzymes Also discussed in lab 2 introduction 1) temperature changes 2) p. H changes 3) salt concentration changes

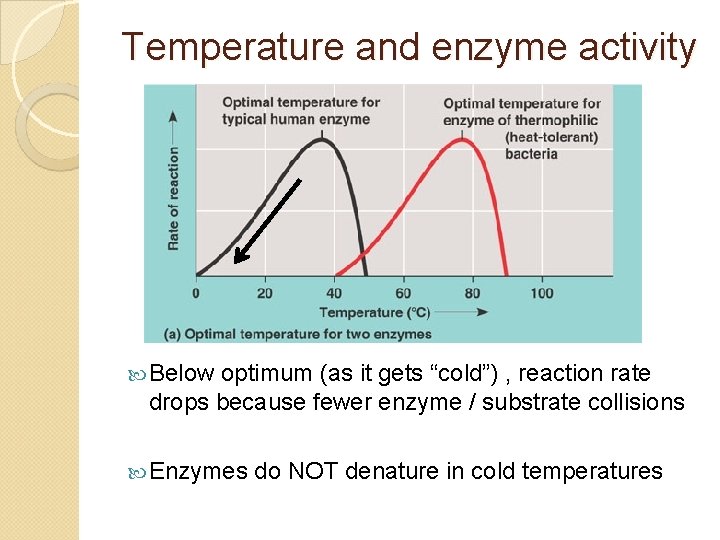
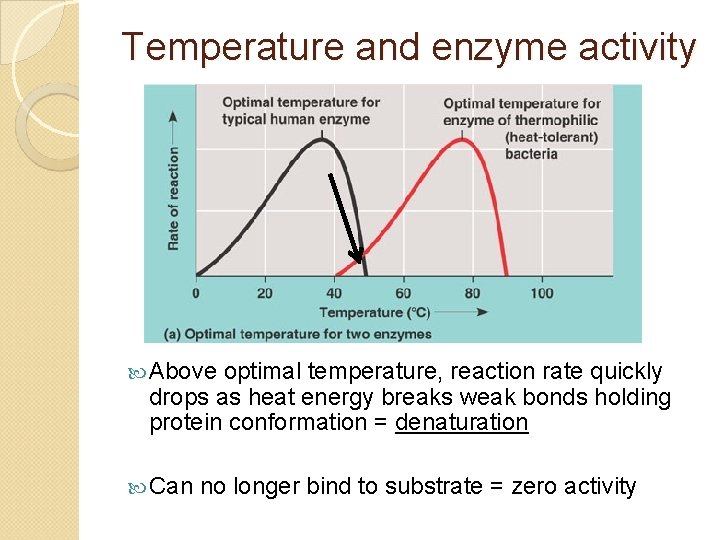
Temperature and enzyme activity Below optimum (as it gets “cold”) , reaction rate drops because fewer enzyme / substrate collisions Enzymes do NOT denature in cold temperatures

Temperature and enzyme activity Above optimal temperature, reaction rate quickly drops as heat energy breaks weak bonds holding protein conformation = denaturation Can no longer bind to substrate = zero activity

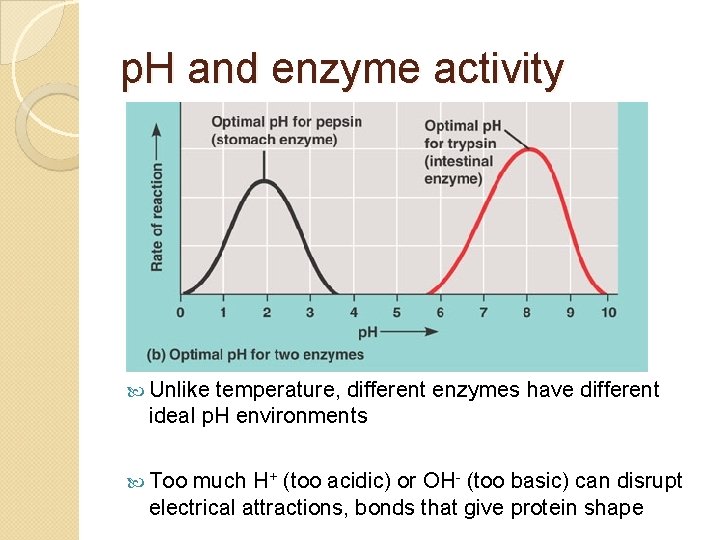
p. H and enzyme activity Unlike temperature, different enzymes have different ideal p. H environments Too much H+ (too acidic) or OH- (too basic) can disrupt electrical attractions, bonds that give protein shape

Salt concentration and enzymes Ideal salt concentration may differ for different enzymes also Salts may interfere with electrical attractions within protein structure (hydrogen / ionic bonding) Salt levels also affect water balance in cells

Maintaining environments A balanced temperature, p. H, and salt concentration must be maintained for protein activity (and survival) Using energy to maintain a balance = homeostasis Cell level homeostasis Overall body homeostasis = temp, p. H and salt levels

Cell regulation of enzyme activity Cells want to control overall enzyme activity a) regulate enzyme production levels (regulate DNA transcription) b) regulate how active these existing enzymes are

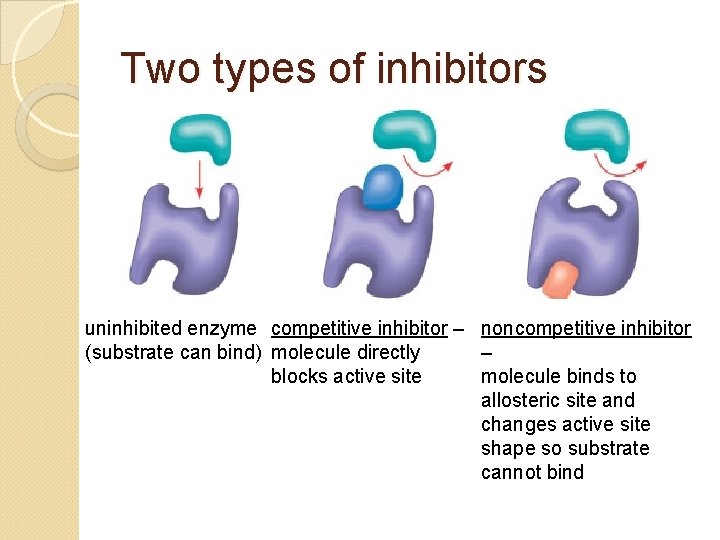
Two types of inhibitors uninhibited enzyme competitive inhibitor – noncompetitive inhibitor (substrate can bind) molecule directly – blocks active site molecule binds to allosteric site and changes active site shape so substrate cannot bind

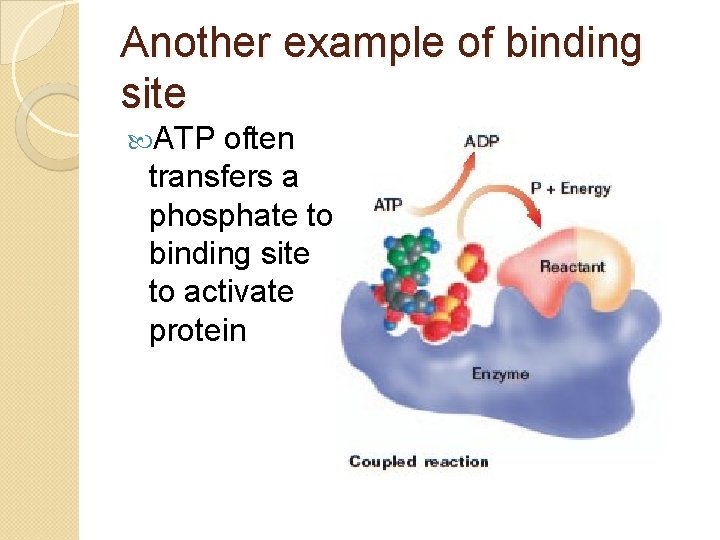
Another example of binding site ATP often transfers a phosphate to binding site to activate protein

Inhibitor / enzyme balance Most inhibitors bind weakly and reversibly to proteins Enzyme regulation = balance of inhibitors moving randomly, binding and falling off enzymes Many of the strongest poisons are irreversible protein inhibitors (ex: cyanide)

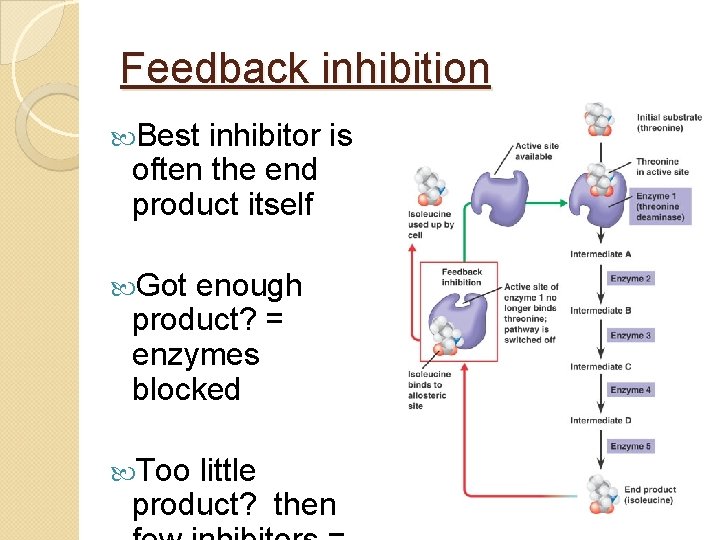
Feedback inhibition Best inhibitor is often the end product itself Got enough product? = enzymes blocked Too little product? then