Macromolecules Carbohydrates Lipids Proteins Nucleic Acids Building blocks

Macromolecules Ø Carbohydrates Ø Lipids Ø Proteins Ø Nucleic Acids Building blocks Of life Science Is Real© by Ana Ulrich
What is organic chemistry? Organic Chemistry is the study of compounds that contain bonds with carbon atoms. All living things contain the element carbon.

What’s The big Deal About Carbon? No other element comes close to matching carbon’s versatility! These Mighty Atoms: • Can form millions of different large and complex structures. • Easily Bond with elements and each other • Can form chains and rings. • Can form single, double, or triple bonds Benzene

Macromolecules AKA Organic molecules or biomolecules Macromolecules are large organic compounds formed from hundreds or thousands of smaller molecules. • Small molecules that join together are monomers. (mono means “one”). Monomers are like links in a chain. • When monomers join together they form long polymers. (poly means “many). Polymers are chains of monomers.
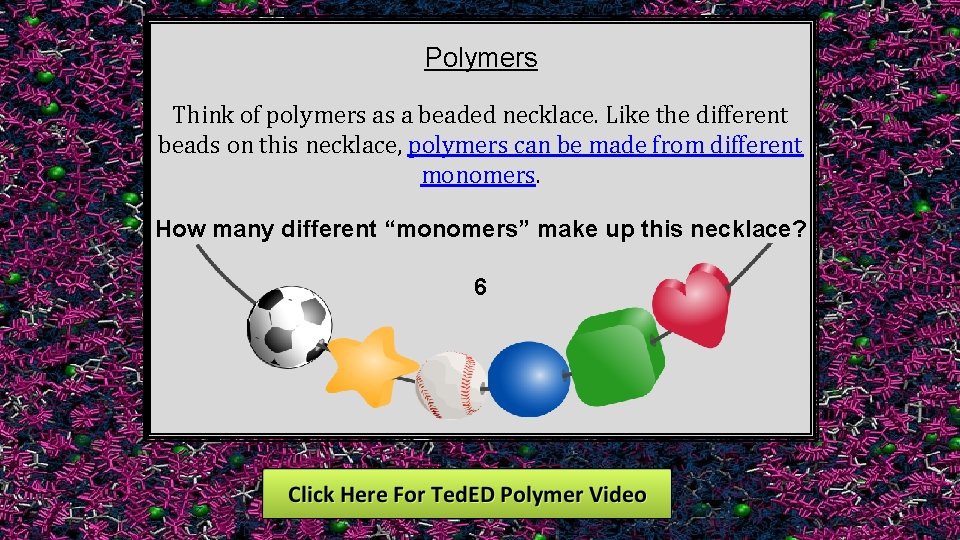
Polymers Think of polymers as a beaded necklace. Like the different beads on this necklace, polymers can be made from different monomers. How many different “monomers” make up this necklace? 6

What are the groups of macromolecules? Macromolecules in living things are grouped based on their chemical compositions 4 Main Macromolecules are: • • Carbohydrates Lipids Proteins Nucleic Acids

CARBOHYDRATES! AKA: Sugars and Starches Usually colorless, water soluble, and crystalized. Composed of: Carbon, Hydrogen, and Oxygen Atoms (CHO) Monomer: Monosaccharaides Polymer: Disaccharides and Polysaccharides

CARBOHYDRATES! Carbohydrates are classified in groups based on number of different molecules. 1 Molecule - Monosaccharaide 2 Molecules - Disaccharides 3 or More Molecules – Polysaccharides

Functions of CARBOHYDRATES 1. Readily Available Source of Short Term Energy. QUICK! 2. Energy Storage • Glycogen stores energy in animals -liver and muscle cells • Starch stores energy in plants 3. Structural Cell Walls in Bacteria and Plants Exoskeletons of Insects, Arthropods, and Crustaceans.

Monosaccharide Carbohydrates AKA: Simple Sugars Glucose Examples: • Glucose - “Blood Sugar” immediately used • Fructose - In Fruits *Hint* “Mono” Means One! Fructose

Polysaccharide Carbohydrates AKA: Complex Carbohydrates What does “poly” mean? Examples: • Glycogen: Energy storage in animals. • Starch: Energy storage in plants • Cellulose: Builds cell walls for plants

Lipids! AKA: Fats, Oils, and Waxes Composed of: Carbon, Hydrogen, and Oxygen atoms (CHO) Monomer: Fatty Acids & Glycerol Polymer: Fats, Oils, Waxes Functions: • Stores Energy in Long Term • Make Up Cell Membranes • Steroids Examples: • Fats & Oils: Energy Storage • Waxes: Repel Water • Steroids: Cholesterol, Sex Hormones, and Cortisol • Phospholipid Cell Membranes: Protect and Regulate Cell Homeostasis **Lipids are generally NOT soluble in water. **
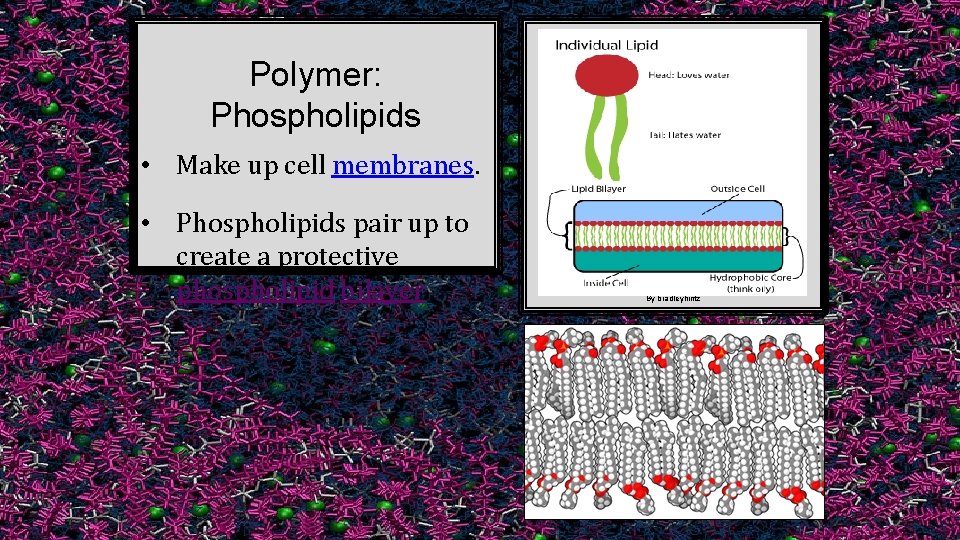
Polymer: Phospholipids • Make up cell membranes. • Phospholipids pair up to create a protective phospholipid bilayer. By bradleyhintz
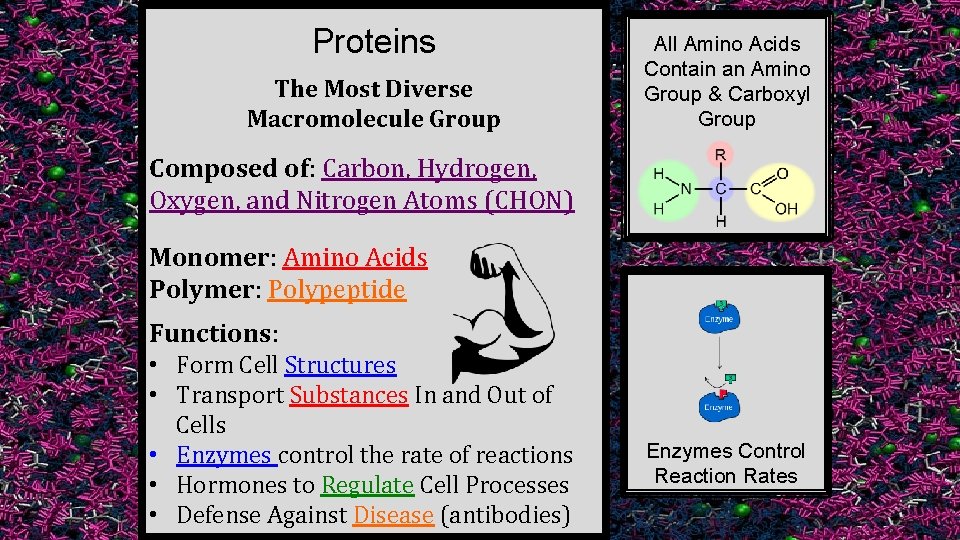
Proteins The Most Diverse Macromolecule Group All Amino Acids Contain an Amino Group & Carboxyl Group Composed of: Carbon, Hydrogen, Oxygen, and Nitrogen Atoms (CHON) Monomer: Amino Acids Polymer: Polypeptide Functions: • Form Cell Structures • Transport Substances In and Out of Cells • Enzymes control the rate of reactions • Hormones to Regulate Cell Processes • Defense Against Disease (antibodies) Enzymes Control Reaction Rates

Protein structure and function Amino acids link together to form a polypeptide. About 20 Amino Acids are found in nature. A protein is built from one or more polypeptides. Formed by RNA
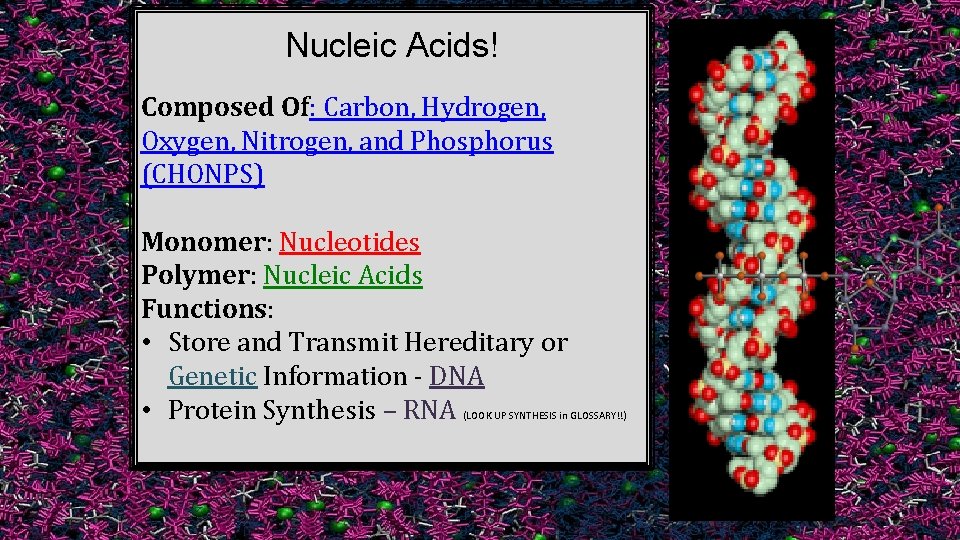
Nucleic Acids! Composed Of: Carbon, Hydrogen, Oxygen, Nitrogen, and Phosphorus (CHONPS) Monomer: Nucleotides Polymer: Nucleic Acids Functions: • Store and Transmit Hereditary or Genetic Information - DNA • Protein Synthesis – RNA (LOOK UP SYNTHESIS in GLOSSARY!!)
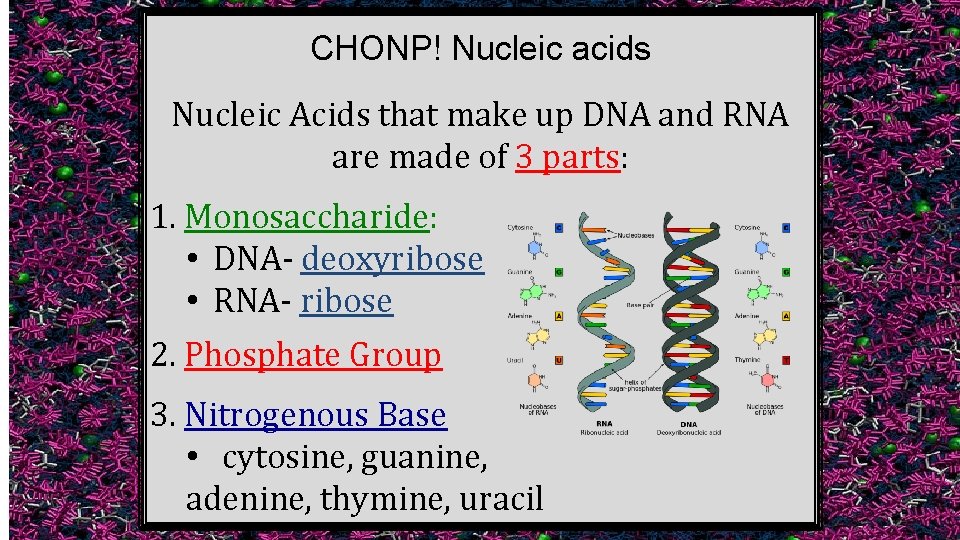
CHONP! Nucleic acids Nucleic Acids that make up DNA and RNA are made of 3 parts: 1. Monosaccharide: • DNA- deoxyribose • RNA- ribose 2. Phosphate Group 3. Nitrogenous Base • cytosine, guanine, adenine, thymine, uracil
Let’s Sum It up! Use This video to answer questions on your student handout!

Students – Go to Kahoot. it Get Your Electroni c Devices Ready for FUN! Let’s See What You’ve Learned! Teachers DO NOT need an account to facilitate! It’s Easy and Effective! Students do not need accounts! Students LOVE Kahoot!!! Science Is Real© by Ana Ulrich
- Slides: 19