The Structure and Function of Macromolecules Carbohydrates and
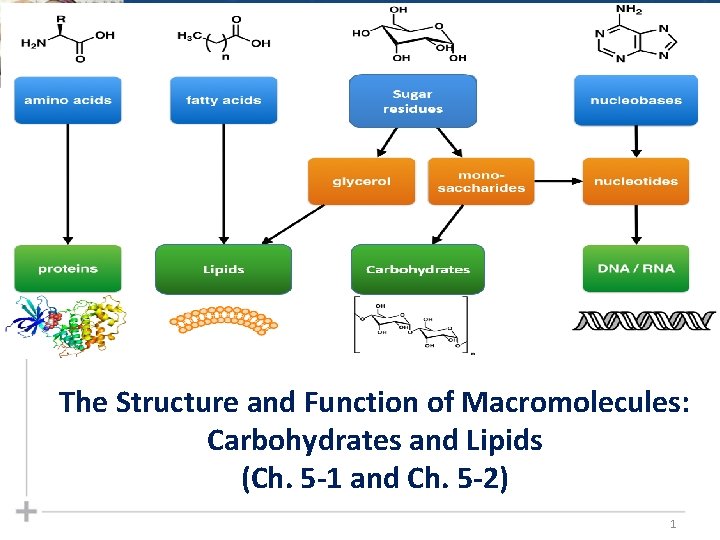
The Structure and Function of Macromolecules: Carbohydrates and Lipids (Ch. 5 -1 and Ch. 5 -2) 1

Learning Objectives Describe the properties of the monomers and the type of bonds that connect the monomers in biological macromolecules. Explain how hydrolysis and dehydration synthesis are used to cleave and form covalent bonds between monomers. Explain how complex carbohydrates are comprised of sugar monomers whose structures determine the properties and functions of the molecules.

Learning Objectives Explain why lipids are nonpolar. Explain how the differences in saturation determine the structure and function of lipids. Describe how phospholipids contain polar regions that interact with other polar molecules, such as water, and with nonpolar regions that are often hydrophobic.

The FOUR Classes of Large Biomolecules • All living things are made up of four classes of large biological molecules: • • Carbohydrates Lipids Protein Nucleic Acids • Macromolecules are large molecules composed of thousands of covalently bonded atoms • Molecular structure determines function 4

The FOUR Classes of Large Biomolecules • Macromolecules are polymers, built from monomers covalently bonded together • A polymer is a long molecule consisting of many similar building blocks • These small building-block molecules are called monomers 5
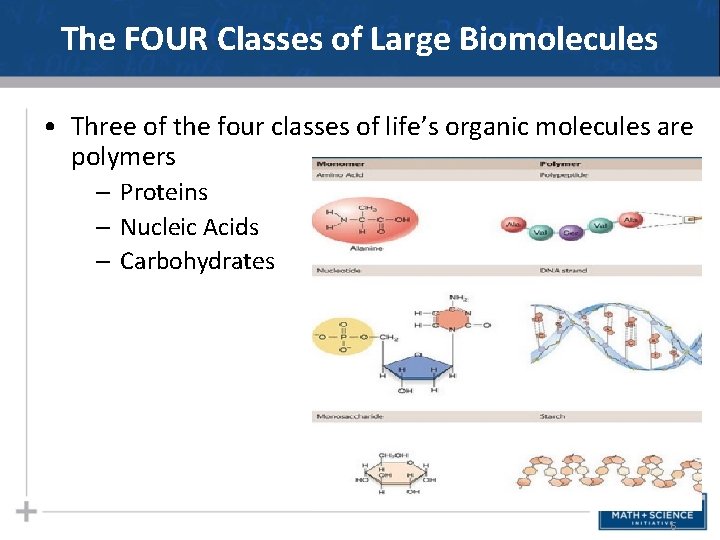
The FOUR Classes of Large Biomolecules • Three of the four classes of life’s organic molecules are polymers – Proteins – Nucleic Acids – Carbohydrates 6

Dehydration Synthesis and Hydrolysis • Dehydration synthesis occurs when two monomers bond together through the loss of a water molecule (AKA: condensation reaction) • In hydrolysis - polymers are disassembled to monomers by a reaction that is essentially the reverse of the dehydration reaction 7
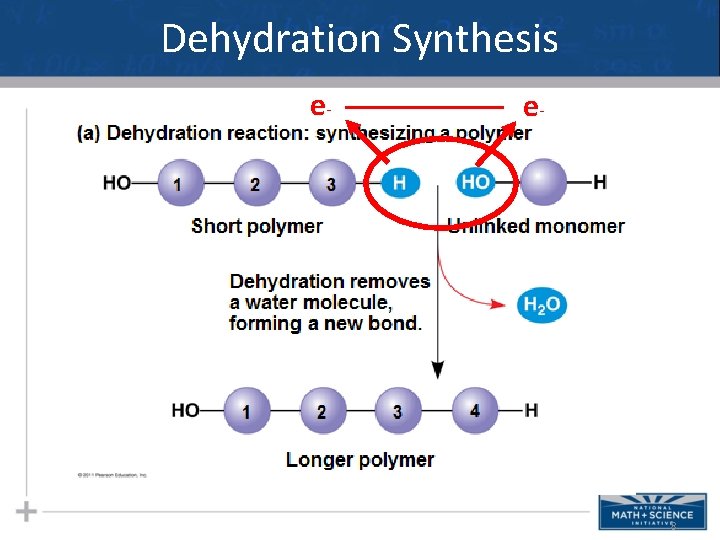
Dehydration Synthesis e- _____ e- 8
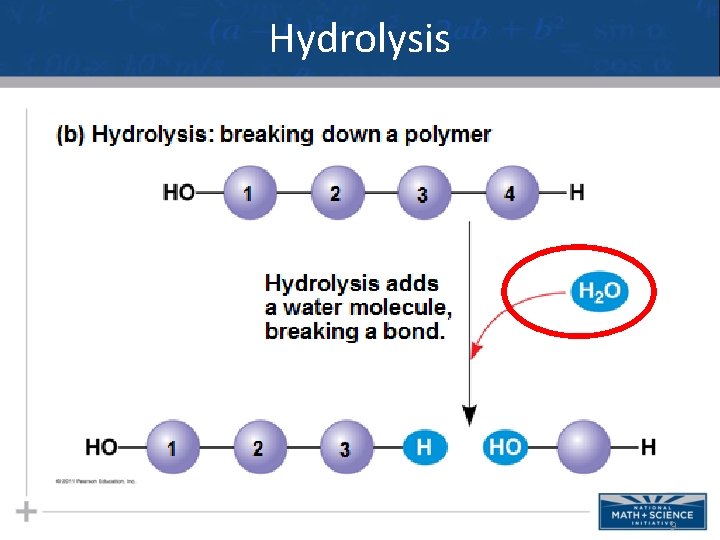
Hydrolysis 9

Carbohydrates • Carbohydrates - Serve as readily available energy for living things. • Usually end in –ose. • Carbohydrates include sugars and the polymers of sugars • Monosaccharides – are the simplest carbohydrates, or monomers of a single sugar • Polysaccharides – are carbohydrate macromolecules, or polymers composed of many sugar building blocks 10
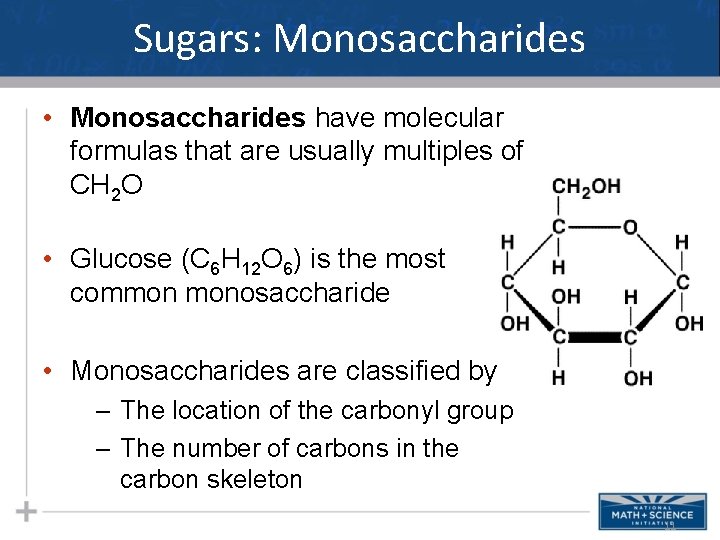
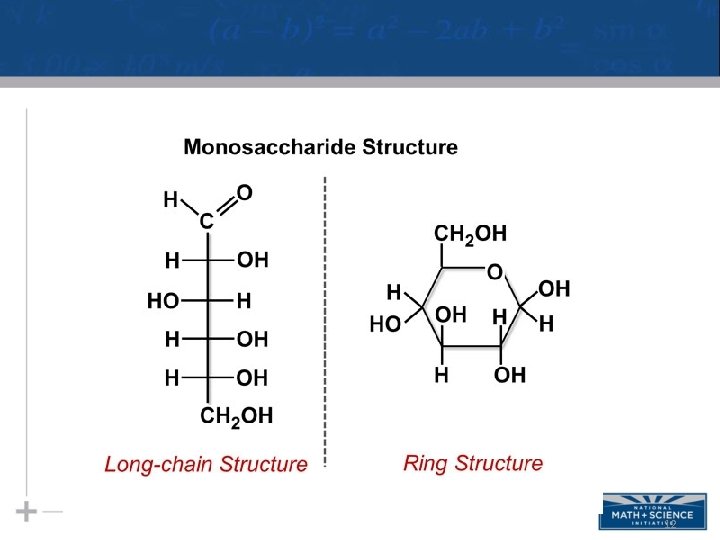
Sugars: Monosaccharides • Monosaccharides have molecular formulas that are usually multiples of CH 2 O • Glucose (C 6 H 12 O 6) is the most common monosaccharide • Monosaccharides are classified by – The location of the carbonyl group – The number of carbons in the carbon skeleton 11
12

13
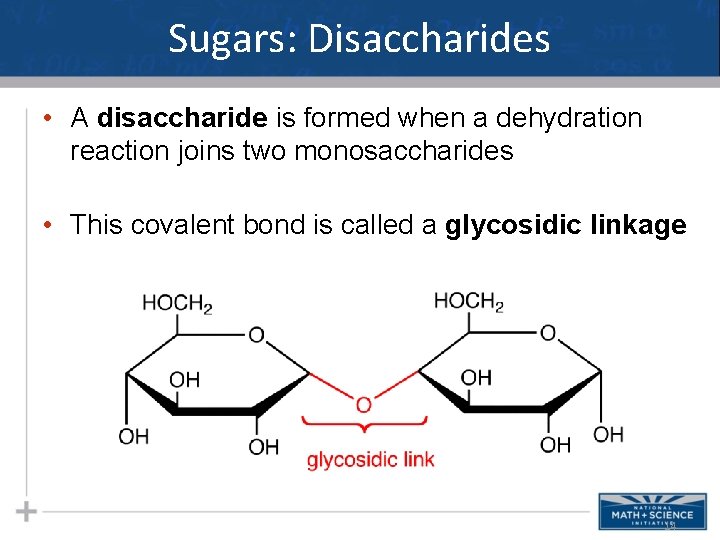
Sugars: Disaccharides • A disaccharide is formed when a dehydration reaction joins two monosaccharides • This covalent bond is called a glycosidic linkage 14
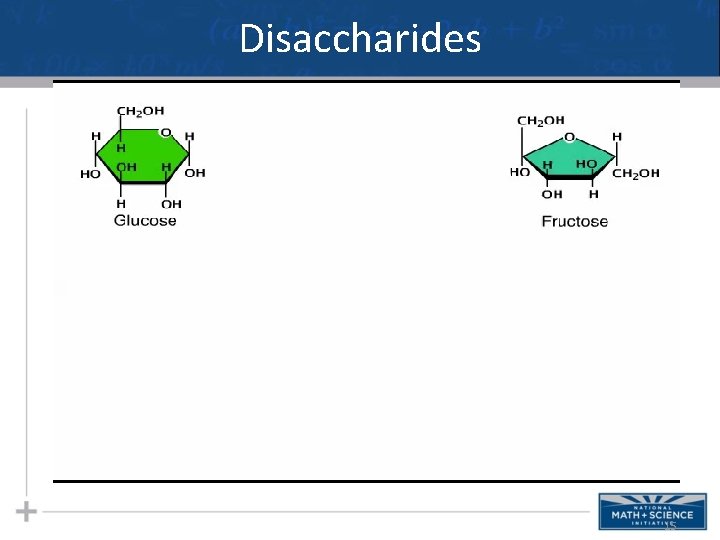
Disaccharides 15
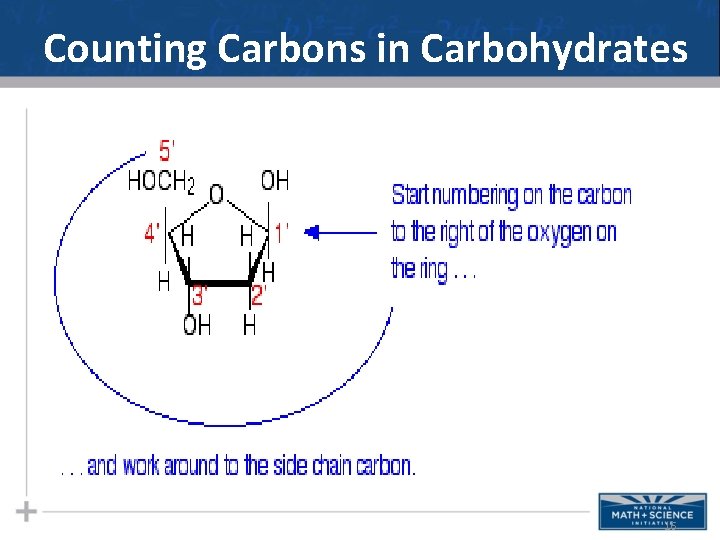
Counting Carbons in Carbohydrates 16
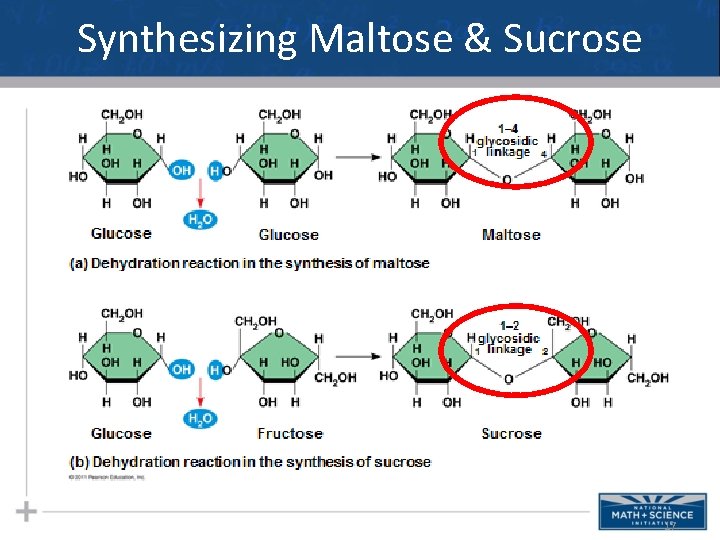
Synthesizing Maltose & Sucrose 17

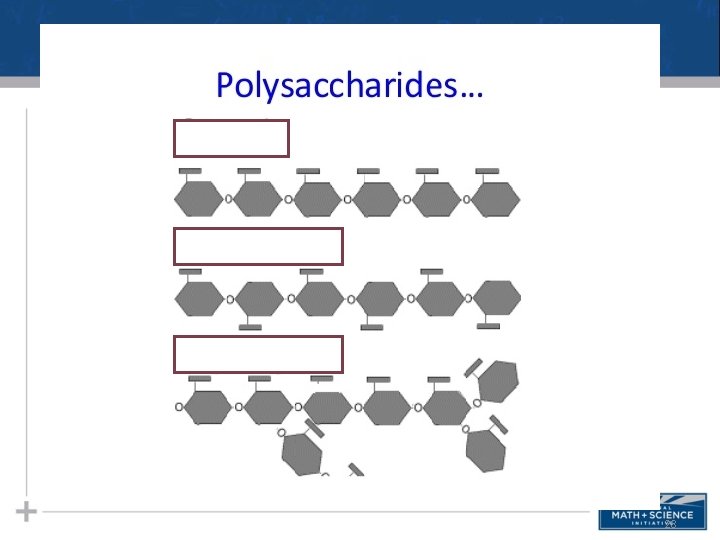
Polysaccharides • Polysaccharides, the polymers of sugars, have storage and structural roles • The structure and function of a polysaccharide are determined by its sugar monomers and the positions of glycosidic linkages 18
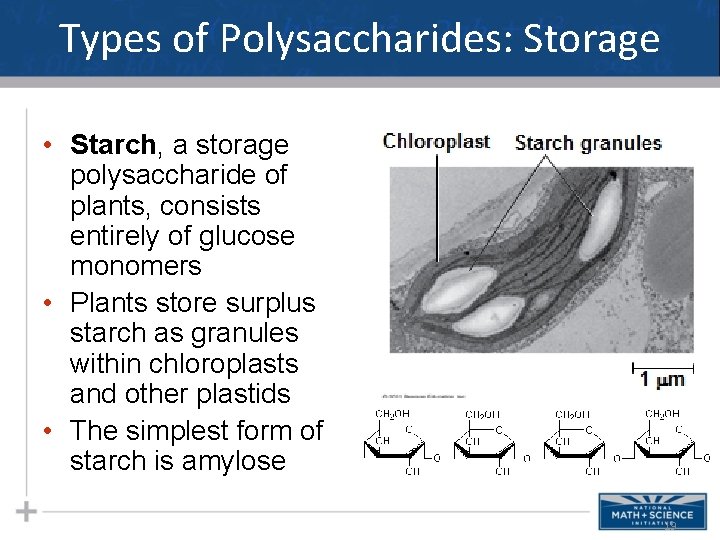
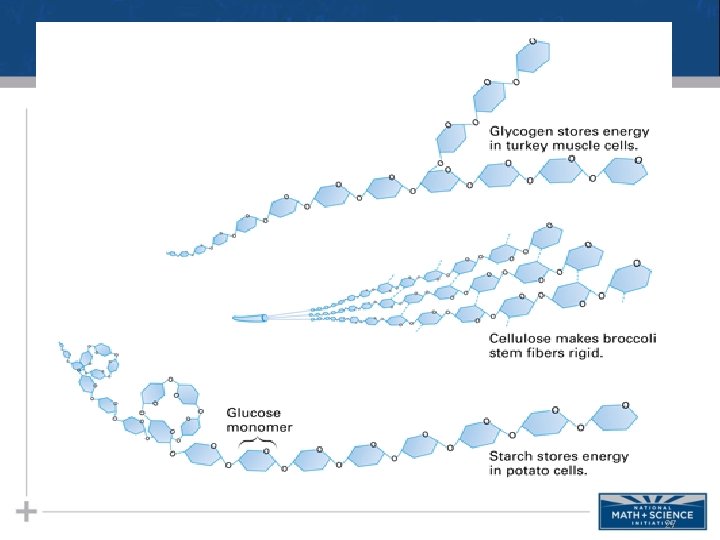
Types of Polysaccharides: Storage • Starch, a storage polysaccharide of plants, consists entirely of glucose monomers • Plants store surplus starch as granules within chloroplasts and other plastids • The simplest form of starch is amylose 19
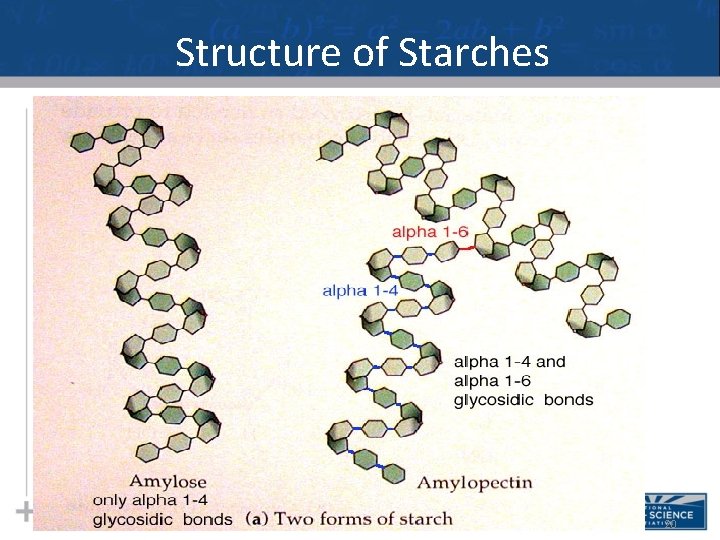
Structure of Starches 20

Types of Polysaccharides: Storage • Glycogen is a storage polysaccharide in animals • Humans and other vertebrates store glycogen mainly in liver and muscle cells 21
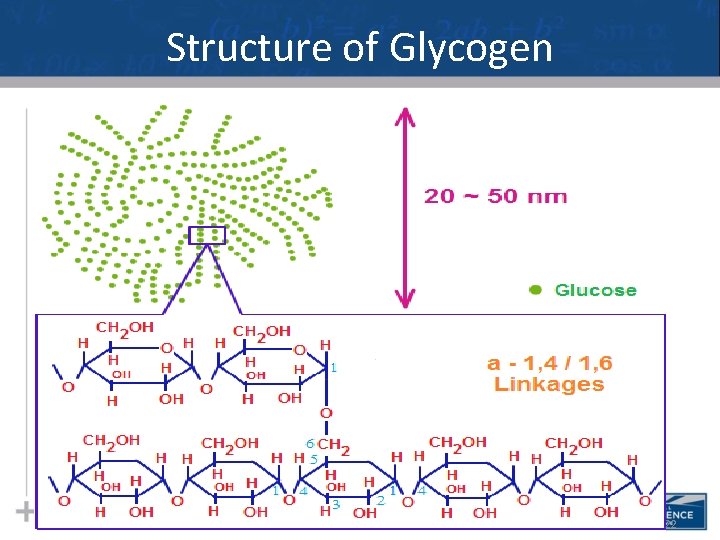
Structure of Glycogen 22

Types of Polysaccharides: Structural • The polysaccharide cellulose is a major component of the tough wall of plant cells • To digest cellulose, organisms must produce the enzyme cellulase (humans don’t produce this enzyme) • Like starch, cellulose is a polymer of glucose, but the glycosidic linkages differ • Remember…change the structure, change the function… 23
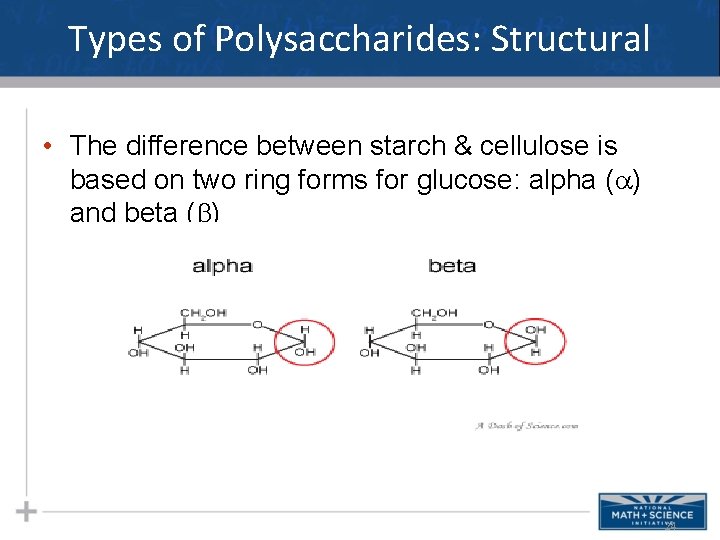
Types of Polysaccharides: Structural • The difference between starch & cellulose is based on two ring forms for glucose: alpha ( ) and beta ( ) 24
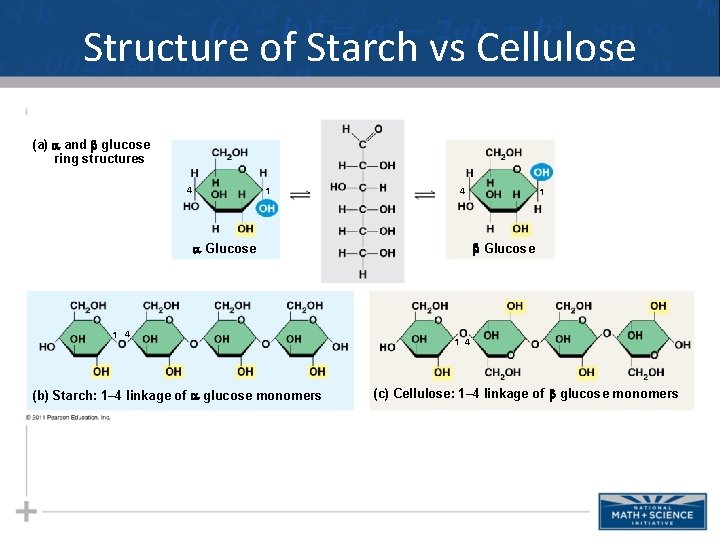
Structure of Starch vs Cellulose (a) and glucose ring structures 4 1 4 Glucose 1 4 (b) Starch: 1– 4 linkage of glucose monomers 1 1 4 (c) Cellulose: 1– 4 linkage of glucose monomers
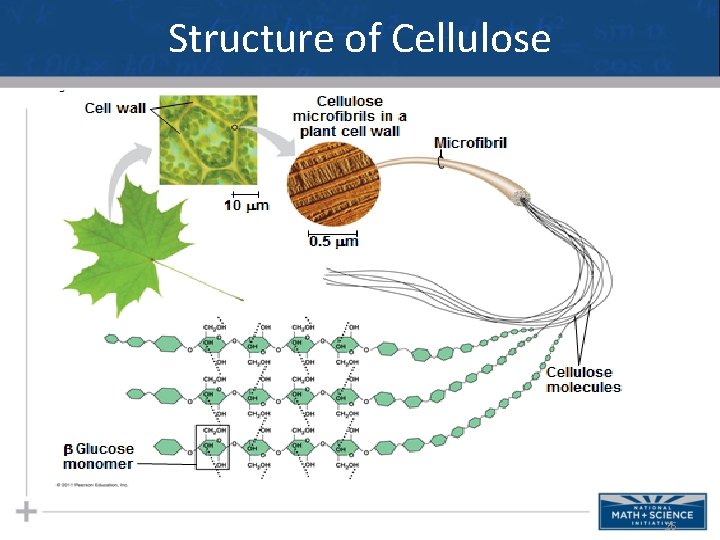
Structure of Cellulose 26
27
28

Polysaccharide Random Acts of Biology • Cellulose in human food passes through the digestive tract as insoluble fiber • Some microbes use enzymes to digest cellulose • Many herbivores, from cows to termites, have symbiotic relationships with these microbes • Chitin, another structural polysaccharide, is found in the exoskeleton of arthropods (crunch!) • Chitin also provides structural support for the cell walls of many fungi 29
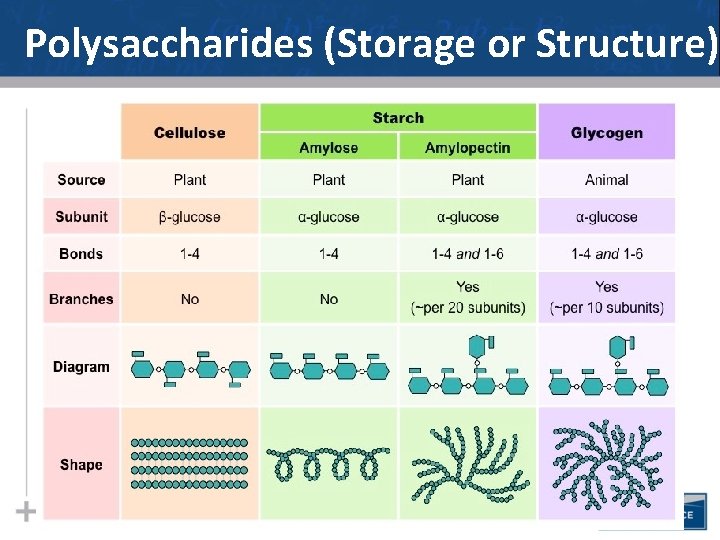
Polysaccharides (Storage or Structure) 30
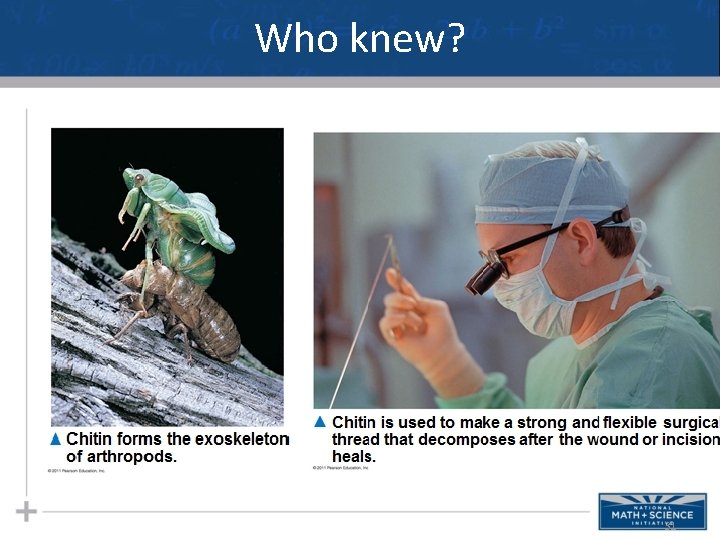
Who knew? 31

Learning Objectives Explain why lipids are nonpolar. Explain how the differences in saturation determine the structure and function of lipids. Describe how phospholipids contain polar regions that interact with other polar molecules, such as water, and with nonpolar regions that are often hydrophobic.

Lipids • Lipids – vary in form and function, provide stored energy & insulation and cell membrane structure for living things. • The most biologically important lipids are fats, phospholipids, and steroids • The unifying feature of lipids is having little or no affinity for water (water fearing) • Lipids are hydrophobic because they consist mostly of hydrocarbons, which form nonpolar covalent bonds 33

Structure of Lipids • Lipids are the one class of large biological molecules that do not form polymers • Fats (or triglycerides) are constructed from two types of smaller molecules: glycerol and fatty acids • Glycerol is a three-carbon alcohol with a hydroxyl group attached to each carbon 34

• A fatty acid consists of a carboxyl group attached to a long carbon skeleton 35
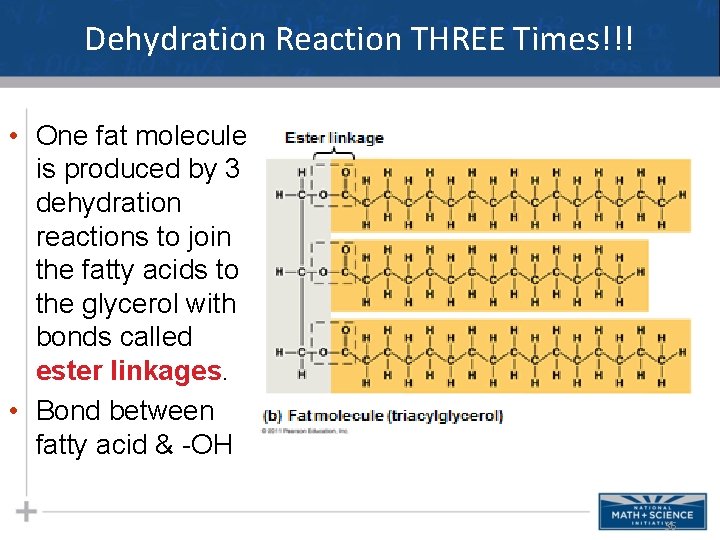
Dehydration Reaction THREE Times!!! • One fat molecule is produced by 3 dehydration reactions to join the fatty acids to the glycerol with bonds called ester linkages. • Bond between fatty acid & -OH 36

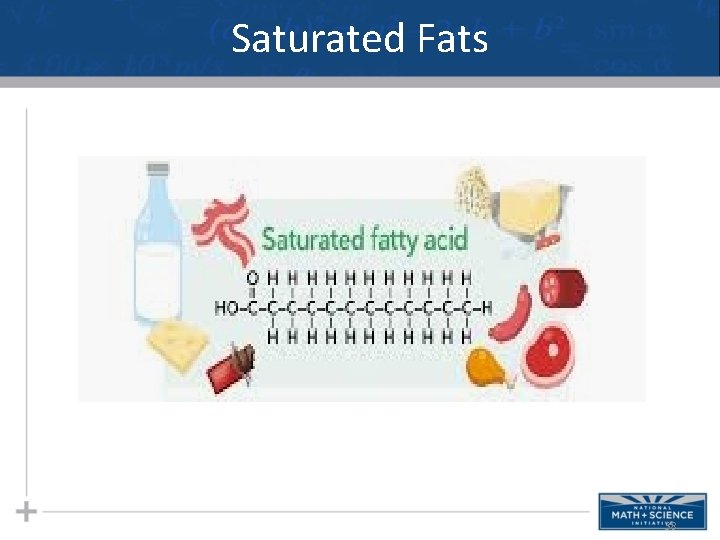
Saturated Fats • Fats made from saturated fatty acids are called saturated fats, and are solid at room temperature • Most animal fats are saturated (lard) • Saturated fatty acids have the maximum number of hydrogen atoms possible and no double bonds 37
Saturated Fats 38

Saturated Fats • A diet rich in saturated fats may contribute to cardiovascular disease through plaque deposits • Hydrogenation is the process of converting unsaturated fats to saturated fats by adding hydrogen • A pair of hydrogen atoms must be “added” across the double bond to form 2 covalent bonds between 2 different carbons and the 2 different hydrogen atoms added. • Ex. Peanut butter & margarine – hydrogenated to prevent lipids from separating out in liquid 39

Unsaturated Fats • Fats made from unsaturated fatty acids are called unsaturated fats or oils, and are liquid at room temperature • Plant fats and fish fats are usually unsaturated • Unsaturated fatty acids have one or more double bonds 40

Unsaturated Fats • Unsaturated means a PAIR of H’s is now “missing” thus a double bond forms between neighboring C’s in the carbon chain of the fatty acid. • This causes a “bend” or “kink” in the chain which alters the shape or conformation of the fat. 41
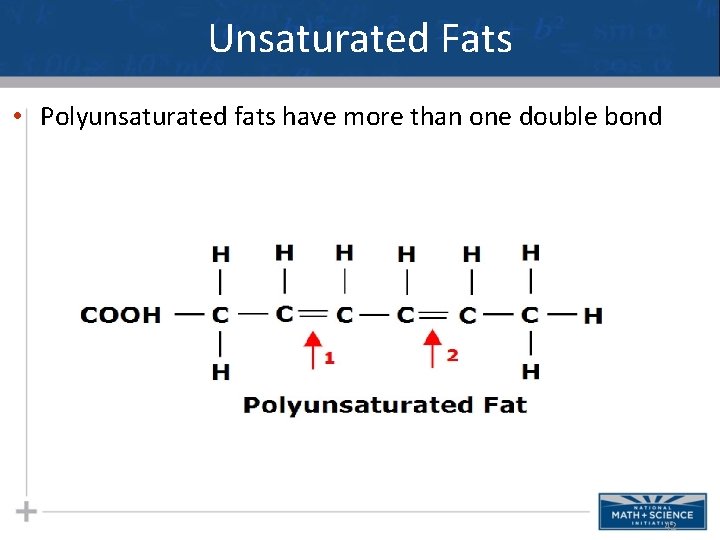
Unsaturated Fats • Polyunsaturated fats have more than one double bond 42
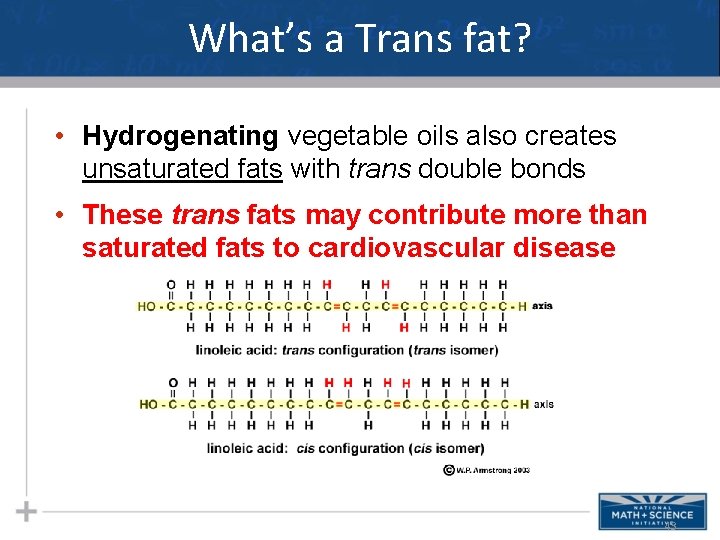
What’s a Trans fat? • Hydrogenating vegetable oils also creates unsaturated fats with trans double bonds • These trans fats may contribute more than saturated fats to cardiovascular disease 43
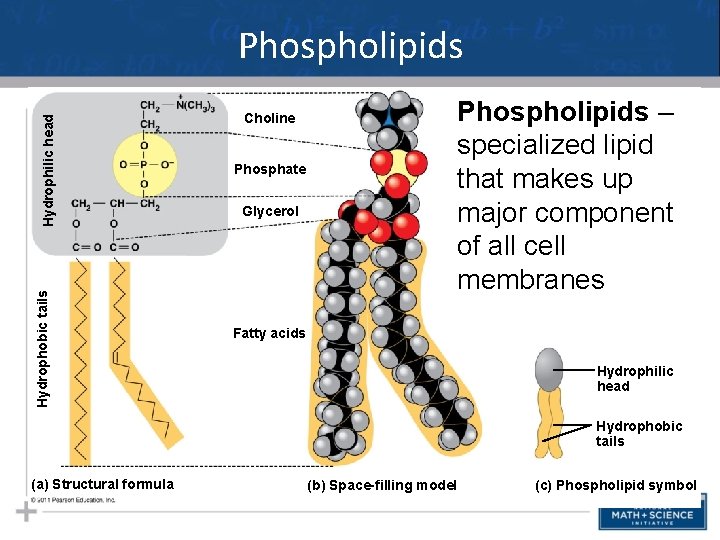
Hydrophobic tails Hydrophilic head Phospholipids Choline Phosphate Glycerol Phospholipids – specialized lipid that makes up major component of all cell membranes Fatty acids Hydrophilic head Hydrophobic tails (a) Structural formula (b) Space-filling model (c) Phospholipid symbol
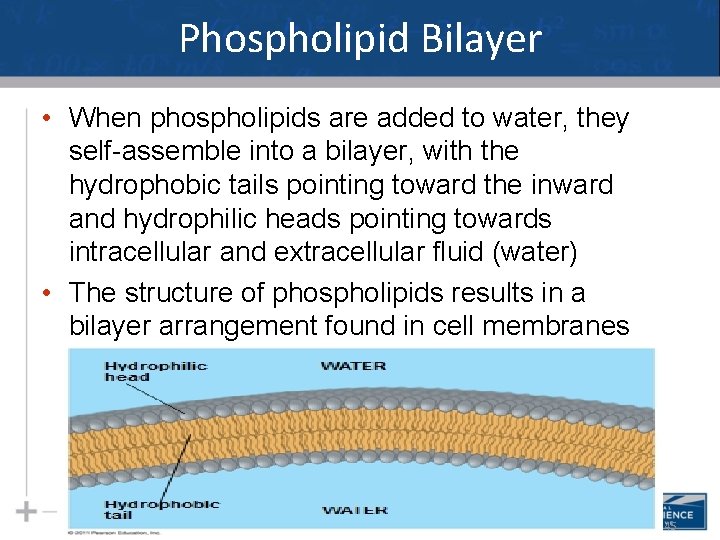
Phospholipid Bilayer • When phospholipids are added to water, they self-assemble into a bilayer, with the hydrophobic tails pointing toward the inward and hydrophilic heads pointing towards intracellular and extracellular fluid (water) • The structure of phospholipids results in a bilayer arrangement found in cell membranes 45

Steroids • Steroids are lipids characterized by a carbon skeleton consisting of four fused rings • Cholesterol, an important steroid, helps stabilize animal cell membranes • Although cholesterol is essential in animals, high levels in the blood may contribute to cardiovascular disease 46

Learning Objectives Summaries
- Slides: 47