Immunoglobulins Structure and Function Immunoglobulins Structure and Function

Immunoglobulins: Structure and Function
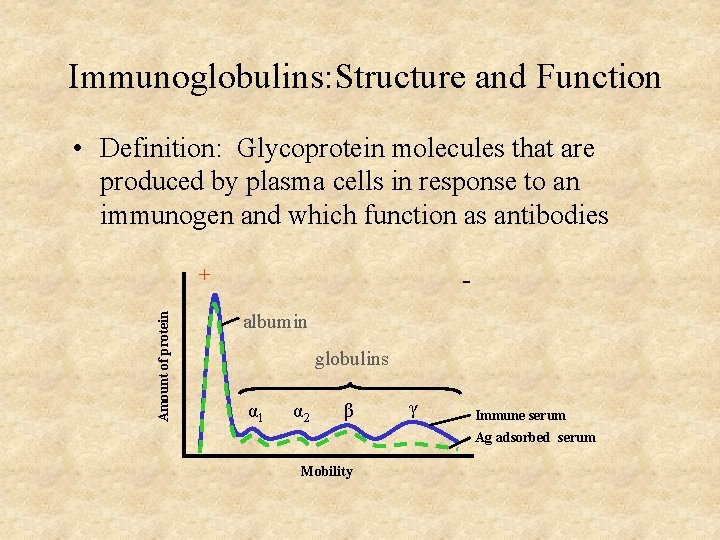
Immunoglobulins: Structure and Function • Definition: Glycoprotein molecules that are produced by plasma cells in response to an immunogen and which function as antibodies Amount of protein + albumin globulins α 1 α 2 β γ Immune serum Ag adsorbed serum Mobility

General Functions of Immunoglobulins • Ag binding – Can result in protection – Valence • Effector functions (Usually require Ag binding) – Fixation of complement – Binding to various cells

Basic Immunoglobulin Structure • Immunoglobulins - heterogeneous • Myeloma proteins - homogeneous immunoglobulins
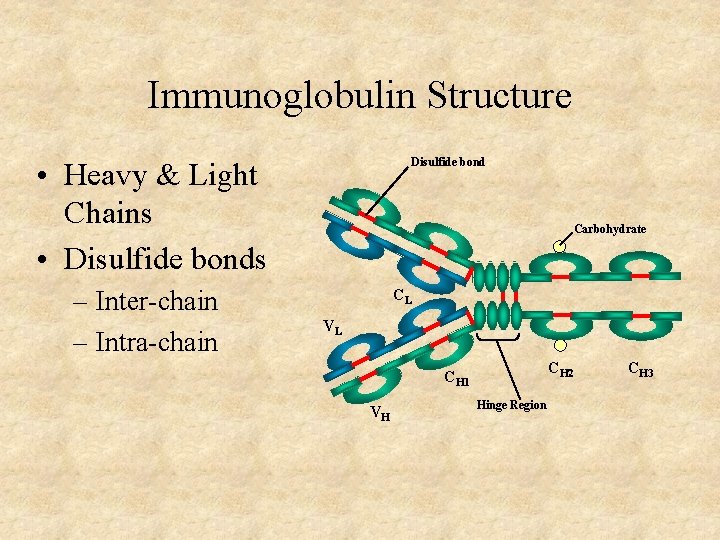
Immunoglobulin Structure Disulfide bond • Heavy & Light Chains • Disulfide bonds – Inter-chain – Intra-chain Carbohydrate CL VL CH 2 CH 1 VH Hinge Region CH 3
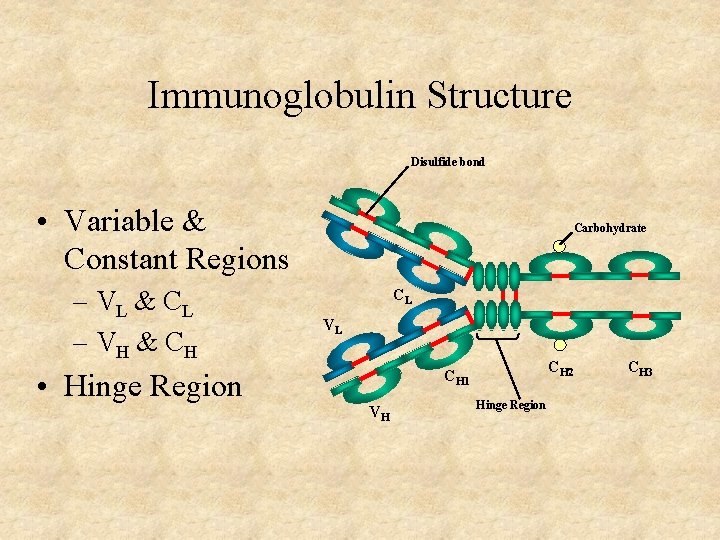
Immunoglobulin Structure Disulfide bond • Variable & Constant Regions – VL & C L – VH & C H • Hinge Region Carbohydrate CL VL CH 2 CH 1 VH Hinge Region CH 3
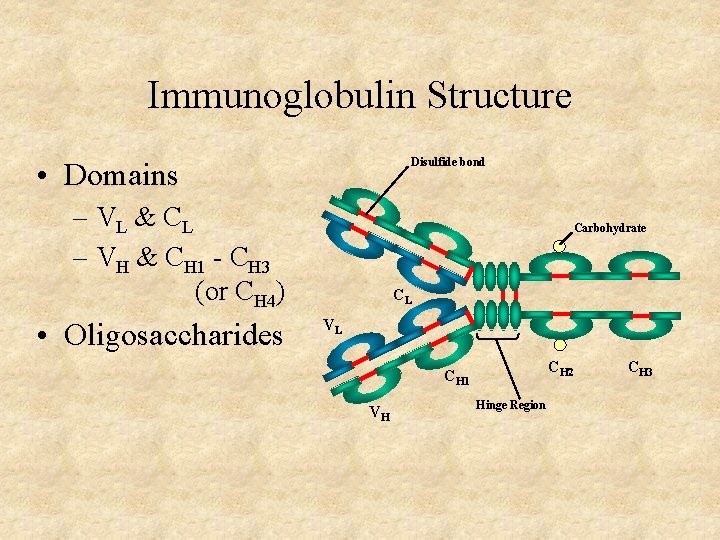
Immunoglobulin Structure Disulfide bond • Domains – VL & C L – VH & CH 1 - CH 3 (or CH 4) • Oligosaccharides Carbohydrate CL VL CH 2 CH 1 VH Hinge Region CH 3

Ig. G molecule Used with permission from: Dr. Mike Clark, Immunology Division, Department of Pathology Cambridge University, Cambridge, England
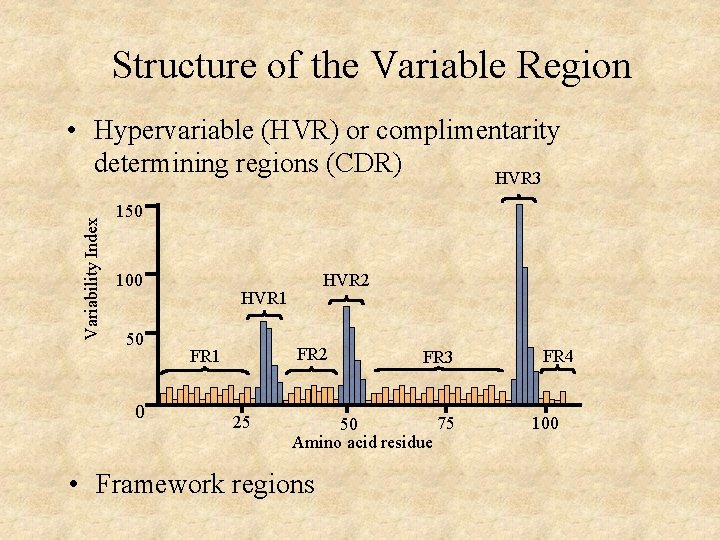
Structure of the Variable Region Variability Index • Hypervariable (HVR) or complimentarity determining regions (CDR) HVR 3 150 100 50 0 HVR 2 HVR 1 FR 2 FR 1 25 FR 3 75 50 Amino acid residue • Framework regions FR 4 100
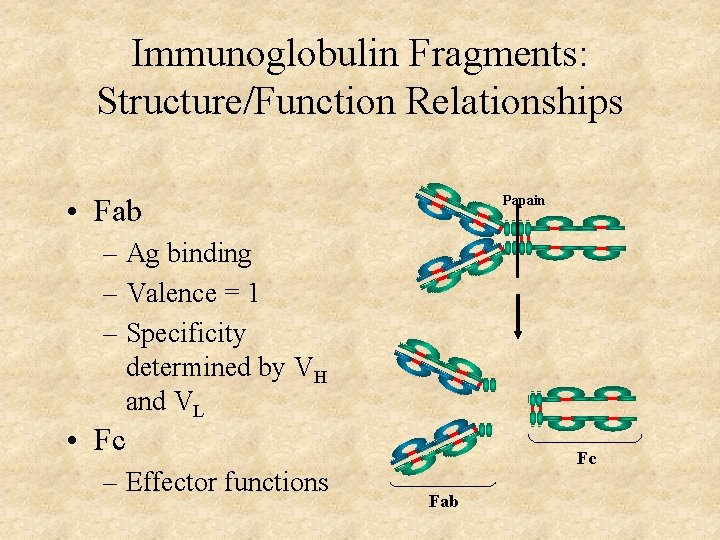
Immunoglobulin Fragments: Structure/Function Relationships • Fab Papain – Ag binding – Valence = 1 – Specificity determined by VH and VL • Fc – Effector functions Fc Fab
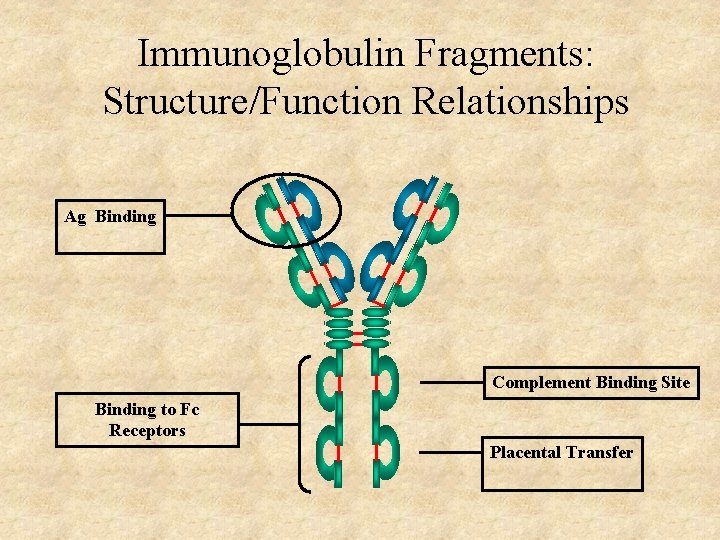
Immunoglobulin Fragments: Structure/Function Relationships Ag Binding Complement Binding Site Binding to Fc Receptors Placental Transfer
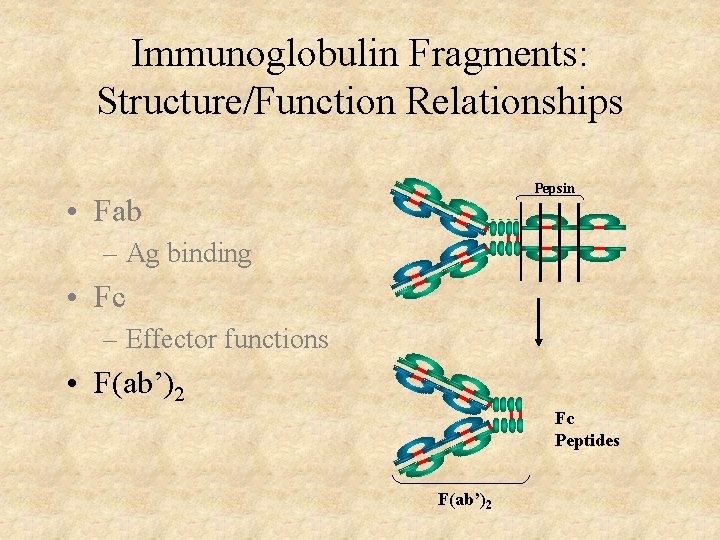
Immunoglobulin Fragments: Structure/Function Relationships Pepsin • Fab – Ag binding • Fc – Effector functions • F(ab’)2 Fc Peptides F(ab’)2

Human Immunoglobulin Classes • • • Ig. G - Gamma (γ) heavy chains Ig. M - Mu (µ) heavy chains Ig. A - Alpha (α) heavy chains Ig. D - Delta (δ) heavy chains Ig. E - Epsilon (ε) heavy chains

Human Immunoglobulin Subclasses • Ig. G Subclasses – Ig. G 1 - Gamma 1 (γ 1) heavy chains – Ig. G 2 - Gamma 2 (γ 2) heavy chains – Ig. G 3 - Gamma 3 (γ 3) heavy chains – Ig. G 4 - Gamma 4 (γ 4) heavy chains • Ig. A subclasses – Ig. A 1 - Alpha 1 (α 1) heavy chains – Ig. A 2 - Alpha 2 (α 2) heavy chains

Human Immunoglobulin Light Chain Types • Kappa (κ) • Lambda (λ)

Human Immunoglobulin Light Chain Subtypes • Lambda light chains – Lambda 1 (λ 1) – Lambda 2 (λ 2) – Lambda 3 (λ 3) – Lambda 4 (λ 4)

Immunoglobulins • Nomenclature – Ig. M (kappa) – Ig. A 1(lambda 2) – Ig. G • Heterogeneity
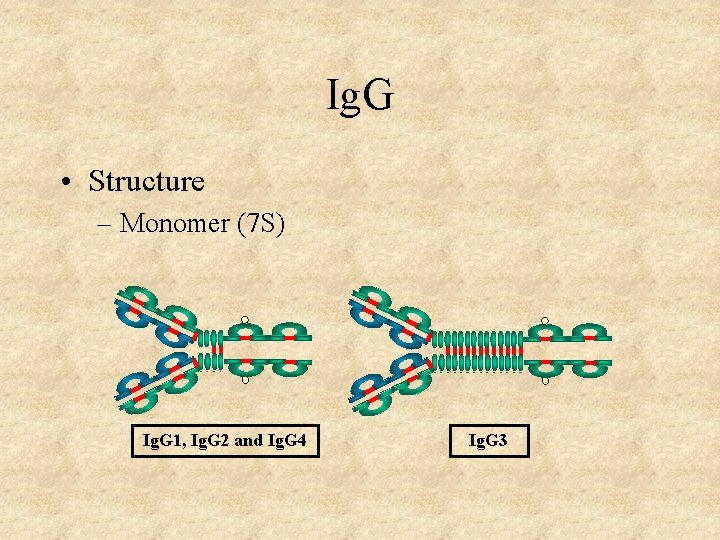
Ig. G • Structure – Monomer (7 S) Ig. G 1, Ig. G 2 and Ig. G 4 Ig. G 3

Ig. G • Structure • Properties – Major serum Ig (systemic immunity) – Major Ig in extravascular spaces – Placental transfer – Does not require Ag binding (± Ig. G 2) – Fixes complement (± Ig. G 4) – Binds to Fc receptors (± Ig. G 2, Ig. G 4) • Phagocytes - opsonization • K cells - ADCC
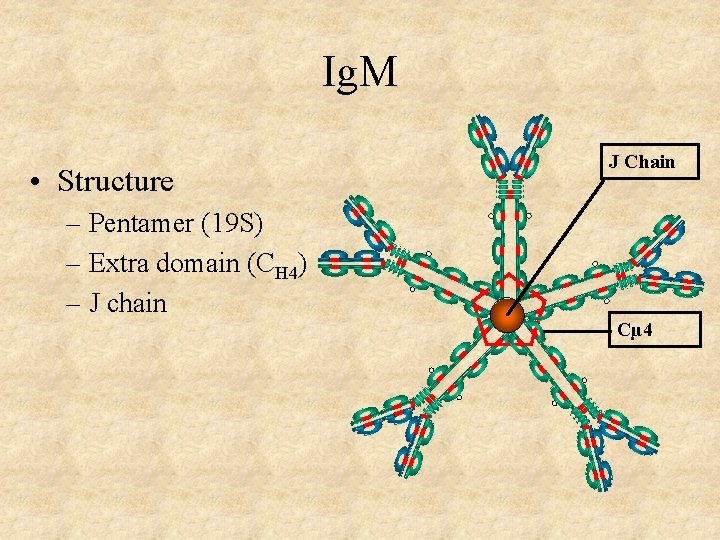
Ig. M • Structure – Pentamer (19 S) – Extra domain (CH 4) – J chain J Chain Cµ 4

Ig. M • Structure • Properties – 3 rd highest serum Ig – First Ig made by fetus and B cells – Fixes complement
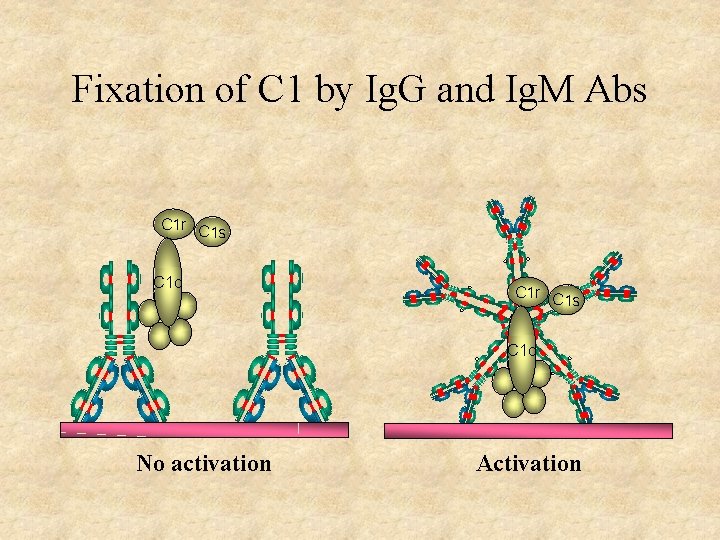
Fixation of C 1 by Ig. G and Ig. M Abs C 1 r C 1 s C 1 q No activation Activation
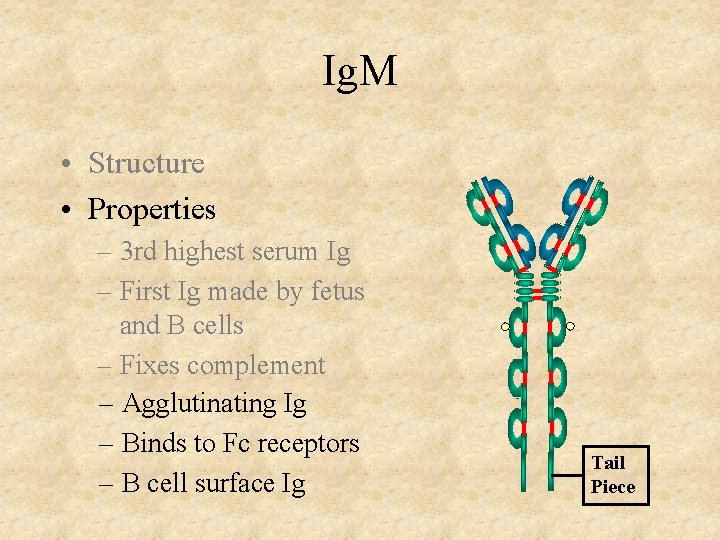
Ig. M • Structure • Properties – 3 rd highest serum Ig – First Ig made by fetus and B cells – Fixes complement – Agglutinating Ig – Binds to Fc receptors – B cell surface Ig Tail Piece
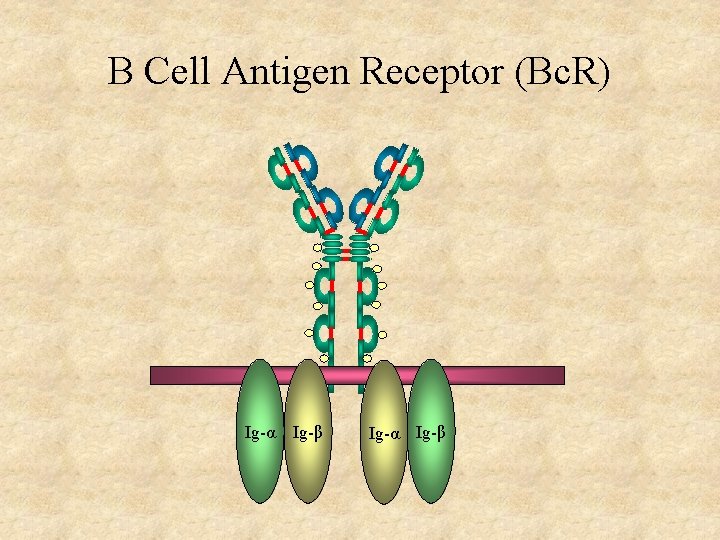
B Cell Antigen Receptor (Bc. R) Ig-α Ig-β
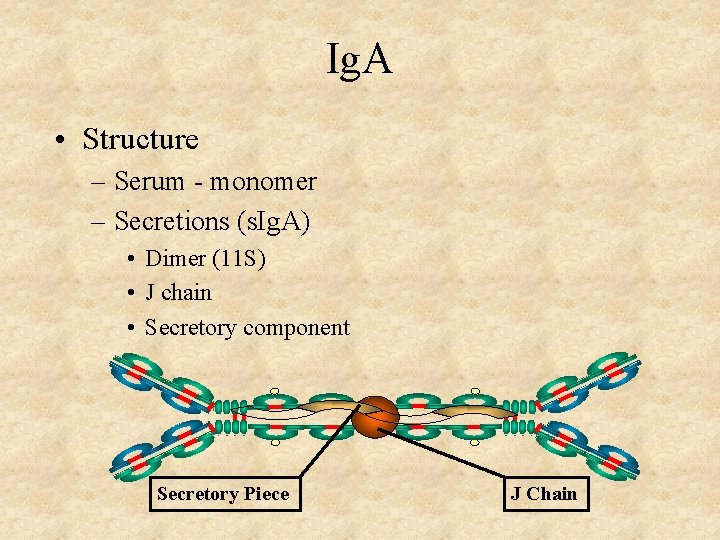
Ig. A • Structure – Serum - monomer – Secretions (s. Ig. A) • Dimer (11 S) • J chain • Secretory component Secretory Piece J Chain
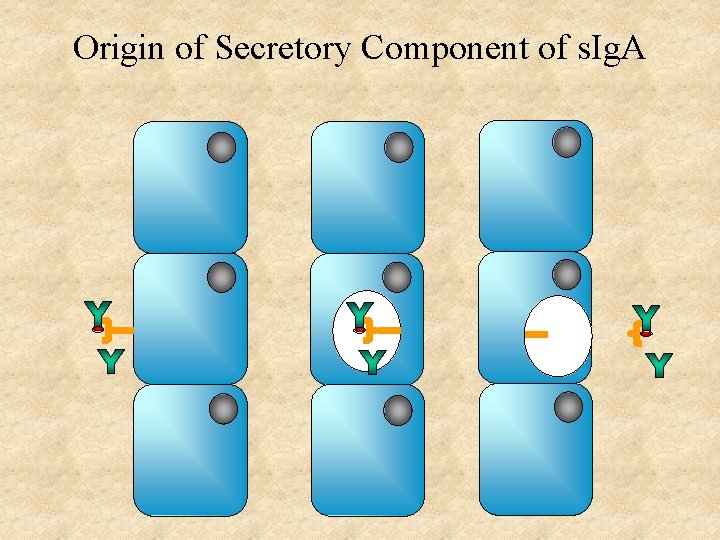
Origin of Secretory Component of s. Ig. A

Ig. A • Structure • Properties – 2 nd highest serum Ig – Major secretory Ig (Mucosal or Local Immunity) • Tears, saliva, gastric and pulmonary secretions – Does not fix complement (unless aggregated) – Binds to Fc receptors on some cells
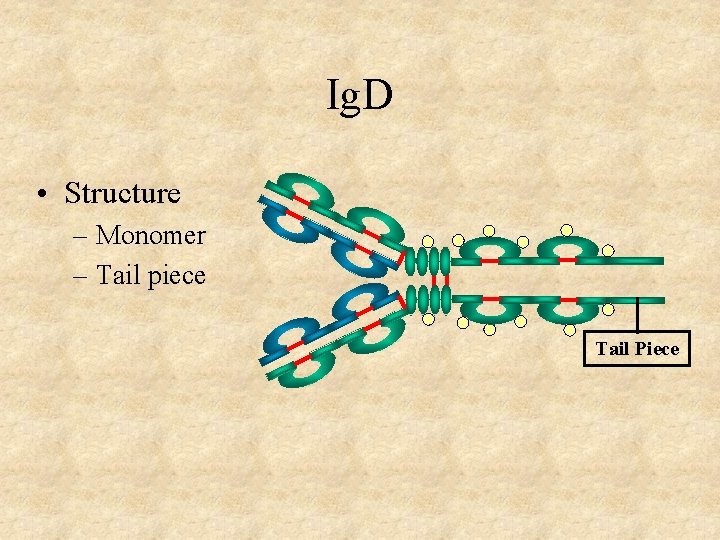
Ig. D • Structure – Monomer – Tail piece Tail Piece

Ig. D • Structure • Properties – 4 th highest serum Ig – B cell surface Ig – Does not bind complement
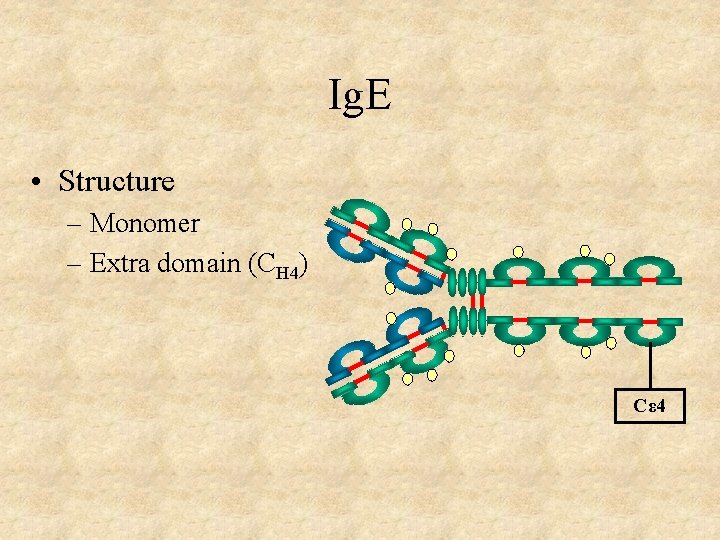
Ig. E • Structure – Monomer – Extra domain (CH 4) Cε 4

Ig. E • Structure • Properties – Least common serum Ig • Binds to basophils and mast cells (Does not require Ag binding) – Allergic reactions – Parasitic infections (Helminths) • Binds to Fc receptor on eosinophils – Does not fix complement
- Slides: 31