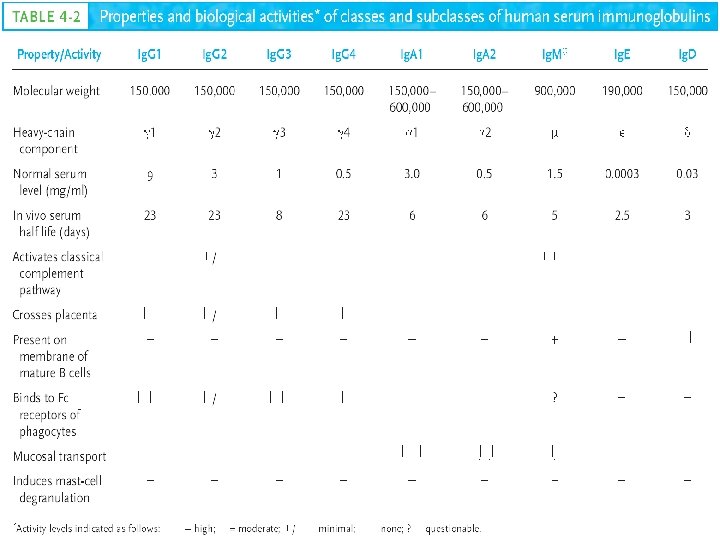
Immunoglobulin Structure and Function Immunoglobulins Structure and Function

Immunoglobulin Structure and Function

Immunoglobulins: Structure and Function • Definition: Glycoprotein molecules that are produced by plasma cells in response to an immunogen and which function as antibodies Amount of protein + albumin globulins α 1 α 2 β γ Immune serum Ag adsorbed serum Mobility

ANTIBODIES • Immunoglobulins : Proteins of animal origin endowed with antibody activity • Immune sera • Gamma globulins • All antibodies are immunoglobulins, but all immunoglobulins may not be antibodies • Immunoglobulins : Structural & Chemical concept • Antibodies : Functional concept • Immunogloblins : 20 – 25 % of total serum proteins • Five classes of Immunoglobulins : Ig. G, Ig. A, IM, Ig. D, Ig. E

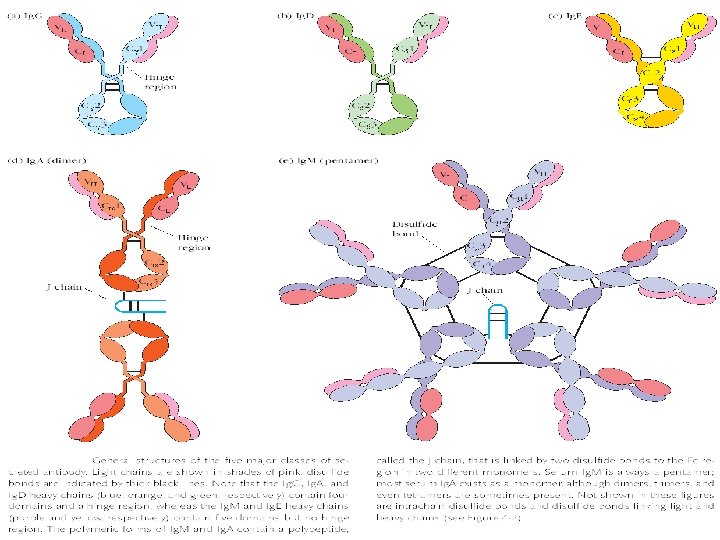
STRUCTURE • • Cleavage study : Fc and Fab fractions Glycoproteins consisting of 2 pairs of polypeptide chains L chains (Light) & H chains (Heavy) L chains : Mol wt 25, 000, Attached to heavy chains by a disulphide bond - Occur in 2 varieties : 1. Kappa (Ќ) 2. Lambda (λ) • H chains : Mol wt 50, 000, Two h chains joined together by 1 -5 SS bonds - 5 classes of H chains : ץ α μ δ ε
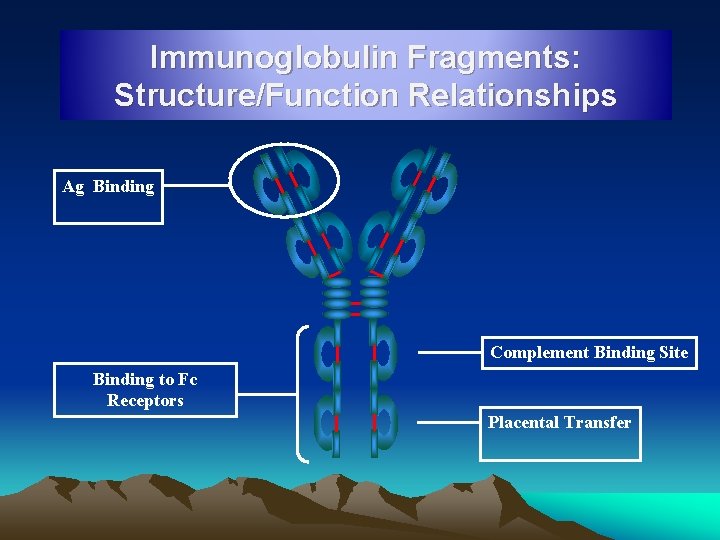
Immunoglobulin Fragments: Structure/Function Relationships Ag Binding Complement Binding Site Binding to Fc Receptors Placental Transfer
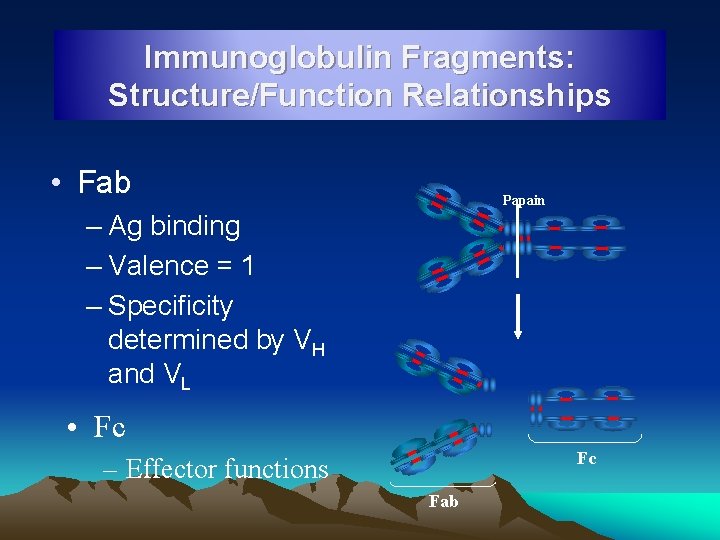
Immunoglobulin Fragments: Structure/Function Relationships • Fab Papain – Ag binding – Valence = 1 – Specificity determined by VH and VL • Fc Fc – Effector functions Fab
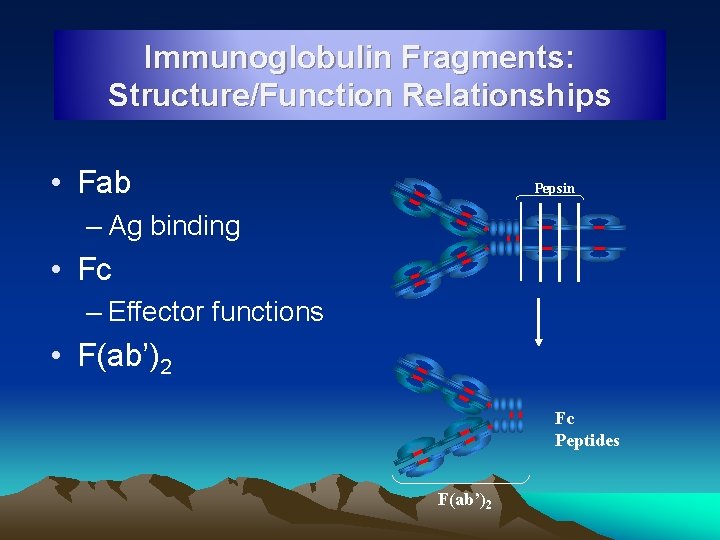
Immunoglobulin Fragments: Structure/Function Relationships • Fab Pepsin – Ag binding • Fc – Effector functions • F(ab’)2 Fc Peptides F(ab’)2
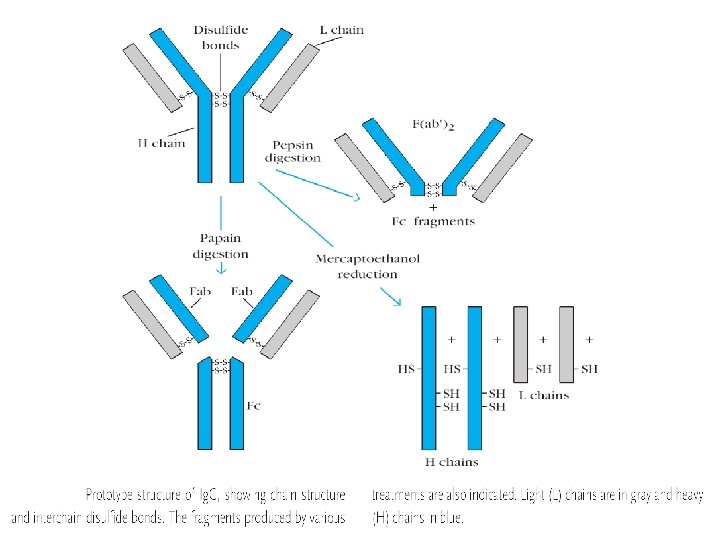
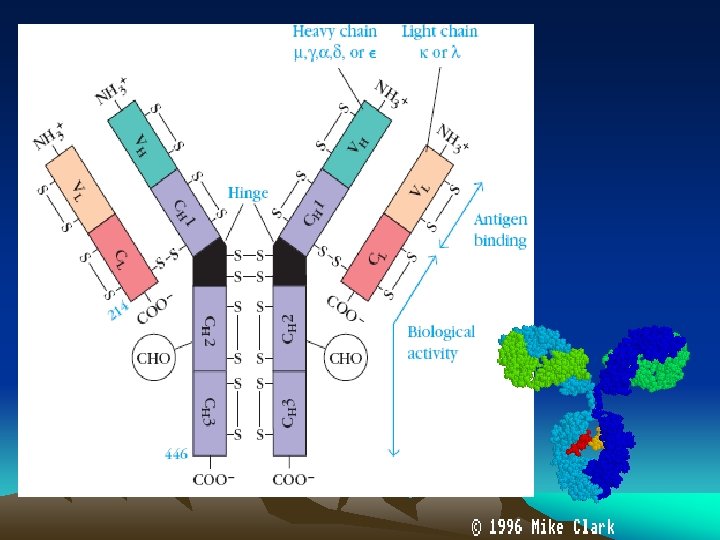
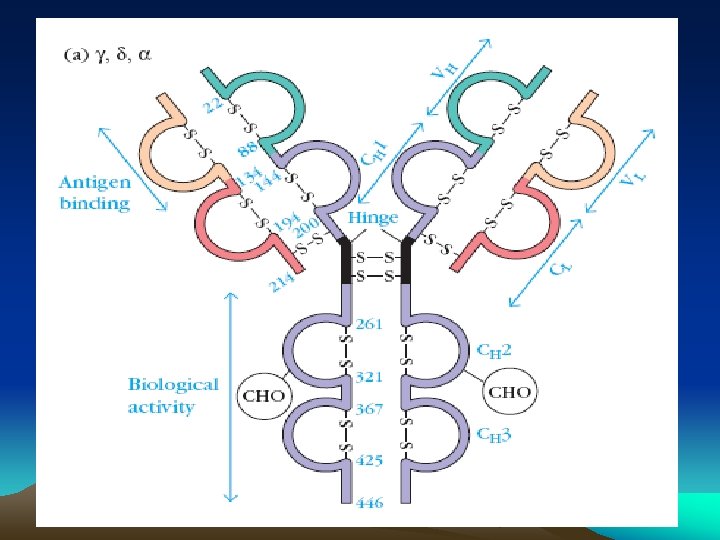

• Antigen combing site : Aminoterminus • Fc fragment site : Carboxy terminal for other biological activities - Complement fixation - Placental transfer - Skin fixation & catabolic rate • Variable and Constant regions • Hypervariable regions (Hot spots) : CDR’s (Complementarity determining regions) • Fd piece : portion of H chain in Fab fragment • Globular domains : Inter chain disulphide bonds form loops in the peptide chain and lops are compactly folded, having specific functions - Vl and VH formation of specific antigen binding site - CH 2 binds C 1 q complement - CH 3 mediates adherence to monocyte surface • Hinge region : Flexible exposed to enzymes an chemicals
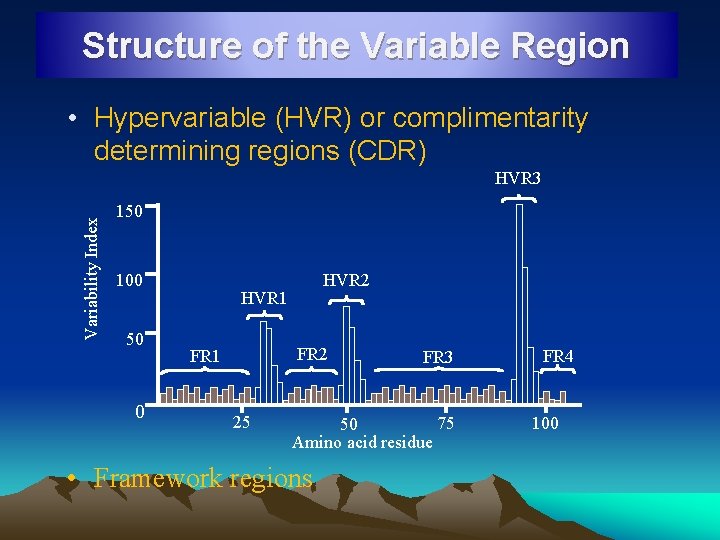
Structure of the Variable Region • Hypervariable (HVR) or complimentarity determining regions (CDR) Variability Index HVR 3 150 100 50 0 HVR 2 HVR 1 FR 2 FR 1 25 FR 3 75 50 Amino acid residue • Framework regions FR 4 100

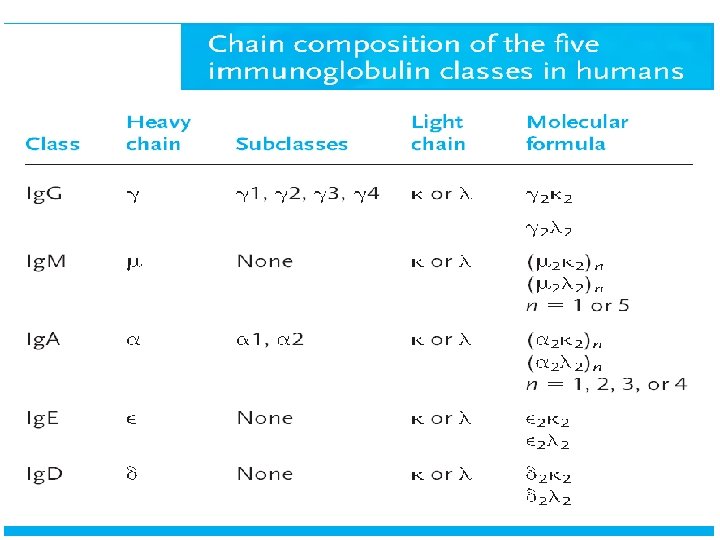
Human Immunoglobulin Classes • • • Ig. G - Gamma (γ) heavy chains Ig. A - Alpha (α) heavy chains Ig. M - Mu (µ) heavy chains Ig. D - Delta (δ) heavy chains Ig. E - Epsilon (ε) heavy chains

Human Immunoglobulin Subclasses • Ig. G Subclasses – Ig. G 1 - Gamma 1 (γ 1) heavy chains – Ig. G 2 - Gamma 2 (γ 2) heavy chains – Ig. G 3 - Gamma 3 (γ 3) heavy chains – Ig. G 4 - Gamma 4 (γ 4) heavy chains • Ig. A subclasses – Ig. A 1 - Alpha 1 (α 1) heavy chains – Ig. A 2 - Alpha 2 (α 2) heavy chains

Ig. G • • • Major serum immunoglobulin – 80% Molecular wt 1, 50, 000 (7 S) , occasionally in polymersed form Distributed equally between intravascular an extra vascular Half life : 23 days Serum concentration : 8 to 16 mg per ml Transported across placenta Binds to microoranisms and enhances phagocytosis General purpose antibody active in blood and tissues Participates in Complement fixation, Precipitation and neutralization of toxins and viruses • Four sub classes : Ig. G 1, Ig. G 2, Ig. G 3, Ig. G 4 - Serum conc : 65%, 23%, 8%, 4% resp
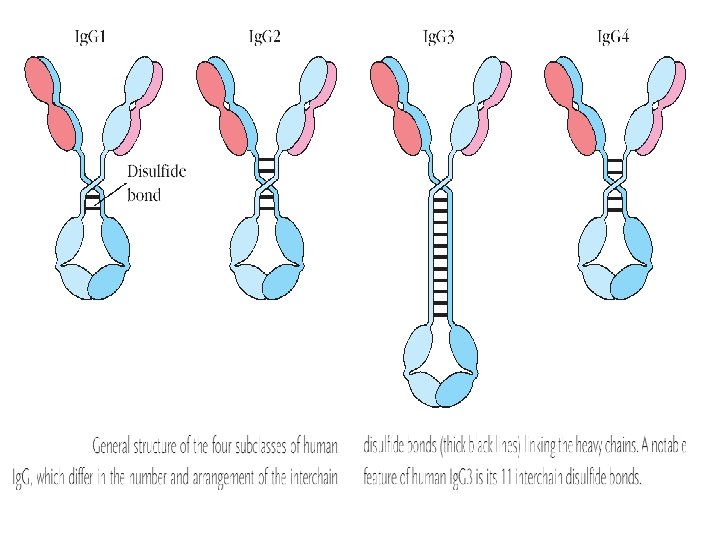
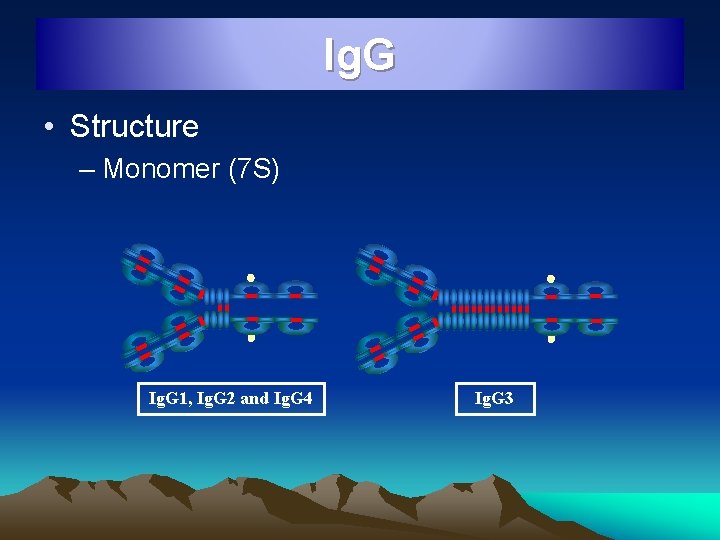
Ig. G • Structure – Monomer (7 S) Ig. G 1, Ig. G 2 and Ig. G 4 Ig. G 3
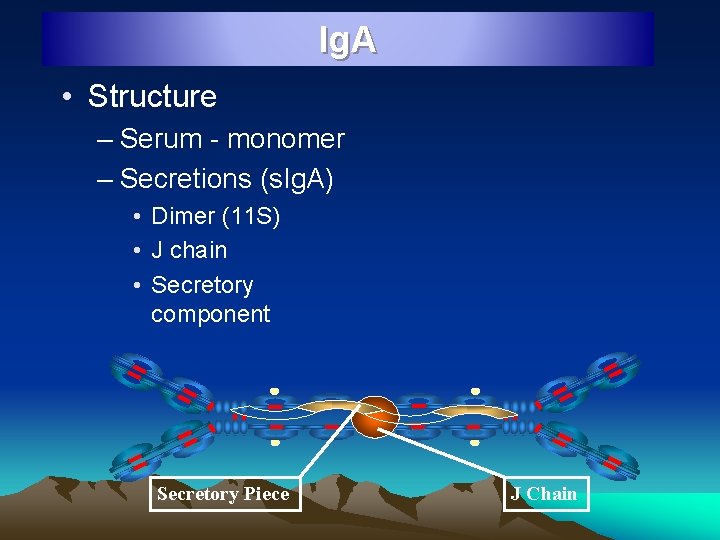
Ig. A • Structure – Serum - monomer – Secretions (s. Ig. A) • Dimer (11 S) • J chain • Secretory component Secretory Piece J Chain

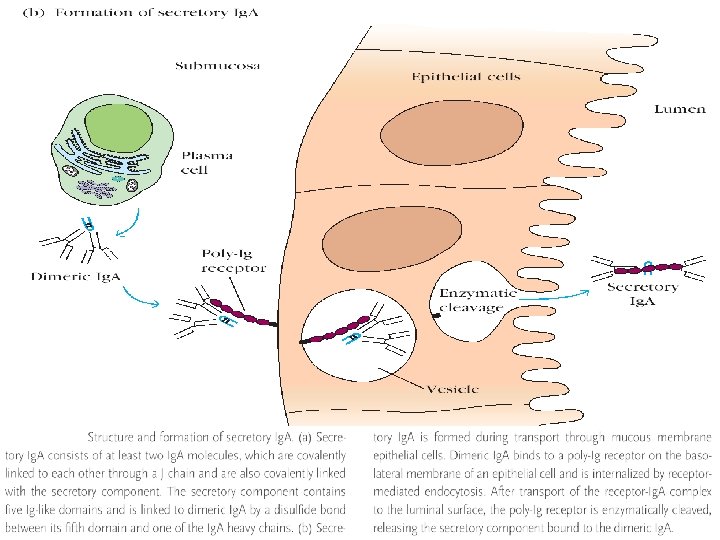
Ig. A • 10 to 13% of serum immunoglobulin, 0. 6 to 4. 2 mg/ml • Major Ig in colostrum, saliva and tears, Half life 6 -8 days • 1. Serum Ig. A – Monomeric 7 S (Mol wt 1, 60, 000) 2. Secretory Ig. A : Dimeric SIg. A , 11 S (4, 00 mol wt) J chain : Ig. A found on mucosal surfaces and in secretions is a dimer joined by 2 monomer units joined together at carboxy terminals by a glycoprotein Secretory component : Glycine rich polypeptide produced by mucosal cells, protect Ig. A from denaturation by bacterial proteases • Antibody paste, inhibit adherence of microorganisms to mucosal surface, promotes phagocytosis, activate alternate Complement pathway • Two sub class : Ig. A 1 & Ig. A 2
Origin of Secretory Component of s. Ig. A
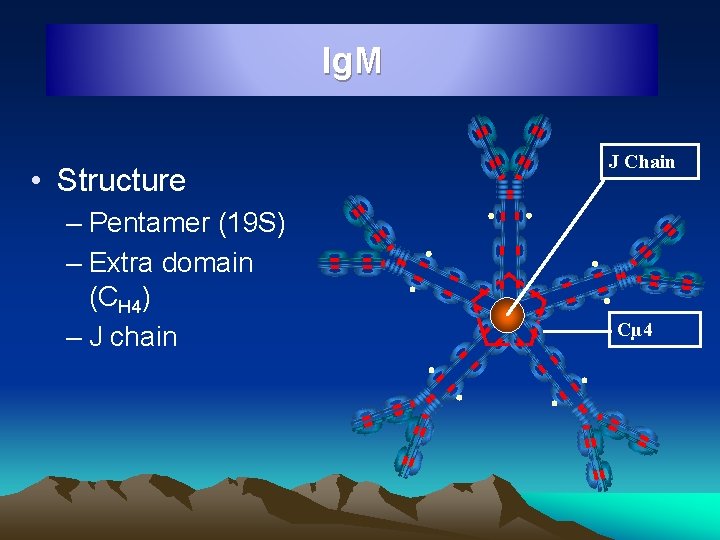
Ig. M • Structure – Pentamer (19 S) – Extra domain (CH 4) – J chain J Chain Cµ 4

Ig. M • • • 5 - 8% of serum Ig, 0. 5 – 2 mg/ml, Half life 5 days Millionaire molecule : 19 S, 9, 000 mol wt Polymers of 5 subunits with J chain Oldest and earliest, short lived Ig, 80% intravascular Produced by foetus at 20 weeks of age Iso haemagglutinins, Natural abs to micro organisms, Abs to Typhoid O Ag, Reagin Abs in syphilis • Monomeric Ig. M is the major antibody receptor on surface of B lymphocytes for antigen recognition • A single Ig. M molecule bring about Immune haemolysis, thousand times more effective in opsonisation, 100 times effective in bactericidal action and 20 times in agglutination
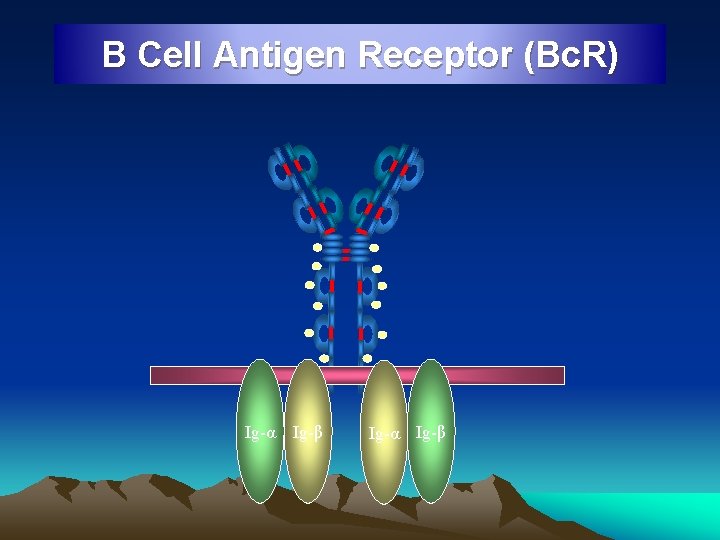
B Cell Antigen Receptor (Bc. R) Ig-α Ig-β
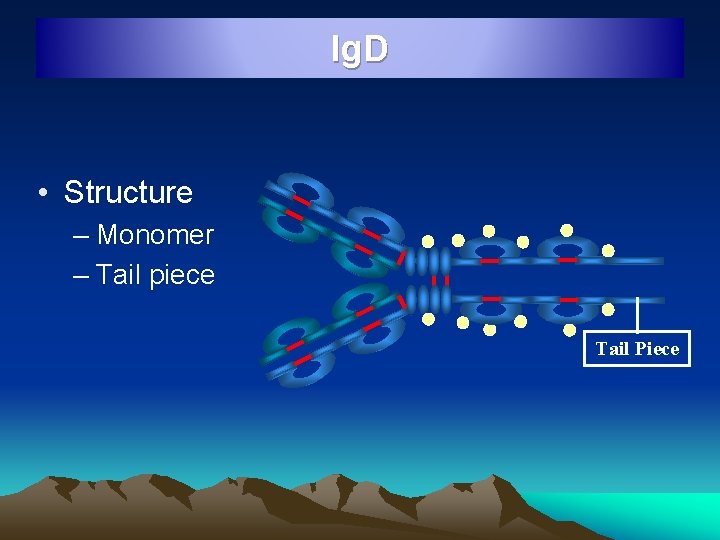
Ig. D • Structure – Monomer – Tail piece Tail Piece

Ig. D • Resembles Ig. G structurally • 3 mg / 100 ml conc in serum • Mostly intravascular • Half life 3 days • Occur on the surface of B lymphocytes and serve as recognition receptors for antigens
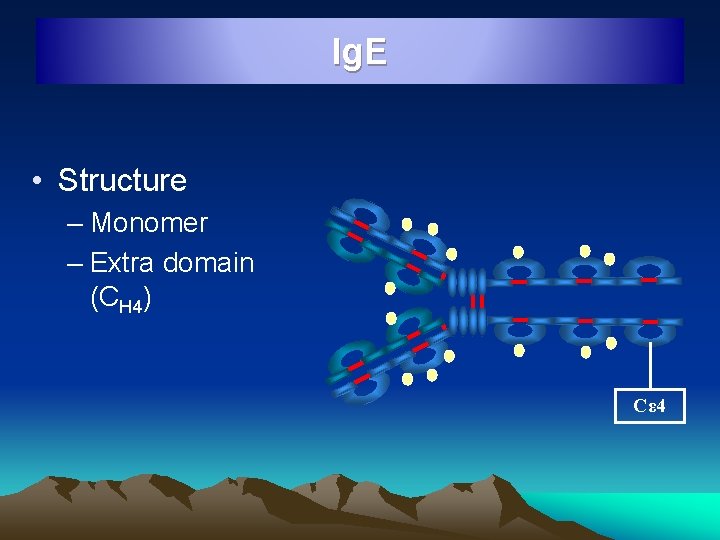
Ig. E • Structure – Monomer – Extra domain (CH 4) Cε 4

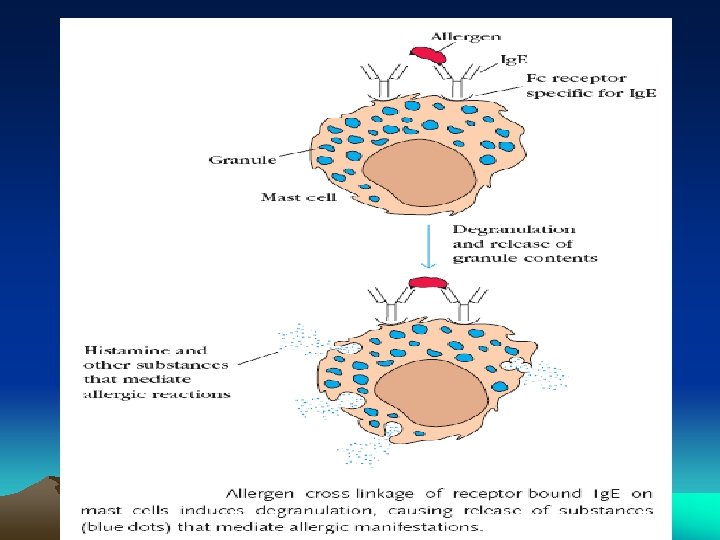
Ig. E • • Atopic reagin antibodies, Ishizaka 1966 8 S molecule, Molecular wt 1, 90, 000, Half life 2 days Extravascular in distribution Elevated levels in atopic conditions like, asthma, hay fever, eczema and parasitic infections • Chiefly produced in linings of respiratory and intestinal tracts • Responsible for Anaphylactic type of hypersensitivity • Protect against pathogens by mast cell de-granulations and release of inflammatory mediators

ABNORMAL IMMUNOGLOBULINS • Structurally similar proteins to antibodies in serum • Seen in many pathological processes • Bence Jones Proteins in Multiple myeloma : Light chains of immunoglobulins, either kappa or lambda - Identified in urine by coagulation when heated to 50 0 C but redissolving at 70 0 C - Multiple myeloma may affect Ig G, A, D, E • Waldenstrom’s macrogluobulinemia : Myeloma of Ig. M producing plasma cells – Excessive production of M proteins • Cryoglobulinemia : Formation of a gel or precipitate on cooling the serum, which redissolves on warming - Associated with myelomas, macroglobulinaemias and autoimmune conditions such as systemic lupus erythematosus

Immunoglobulin specificities • Idiotopes : Specific antigenic determinants on paratope • Idiotype : Sum total of idiotopes on Ig molecule • Antiidiotypic antibodies : Produced by immunization with Fab fragments, resembles epitopes of original antigen • Used as vaccines to protect against pathogen or tumour

Isotypic and Allotypic specificities • Genetically determined specificities based on their antigenic structure • Isotypic specificity : Antigenic specificity which distinguish between the different classes and subclasses of Ig’s present in all normal individuals of a given species • Allotypic specificity : Antigenic specificity which distinguish Ig’s of the same class between different groups of individuals in the same species • Anti-allotype specific abs may develop following blood transfusion or passage of maternal Ig. G into foetus and also seen in sera containing RA factor. • 2 Allotype systems in humans : 1. Gm system 2. In. V system • Gm is associated with Fc portion of Ig. G heavy chain, more than 25 Gm types identified so far • In. V associated with kappa light chain (Km) and 3 Km types • Am : Genetic markers associated with Ig. A


- Slides: 38