Electron Configuration Notes Ground State The lowest energy


























- Slides: 28

Electron Configuration Notes

• Ground State – The lowest energy state of an atom. • Excited State – State in which an atom has a higher potential energy then it has in its ground state.

• Heisenberg Uncertainty Principle – It is impossible to determine simultaneously both the position and velocity of an electron or any other particle. (Can’t exactly predict where electrons will be, just give a general idea. ) • Orbital – Three dimensional region around the nucleus that indicates the probable location of an electron.

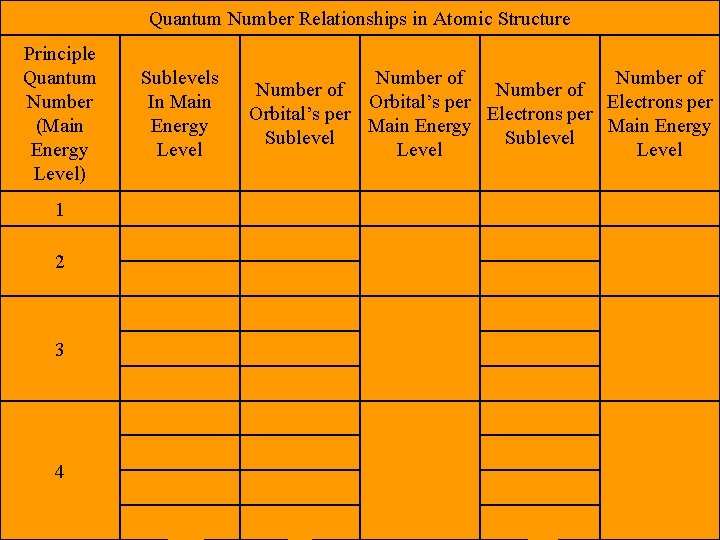
• Quantum Numbers – Numbers that specify the properties of atomic orbital’s and the properties of electrons in orbital’s. • Principle Quantum Number – (n) The main energy level occupied by the electron. – Positive numbers only 1, 2, 3, etc

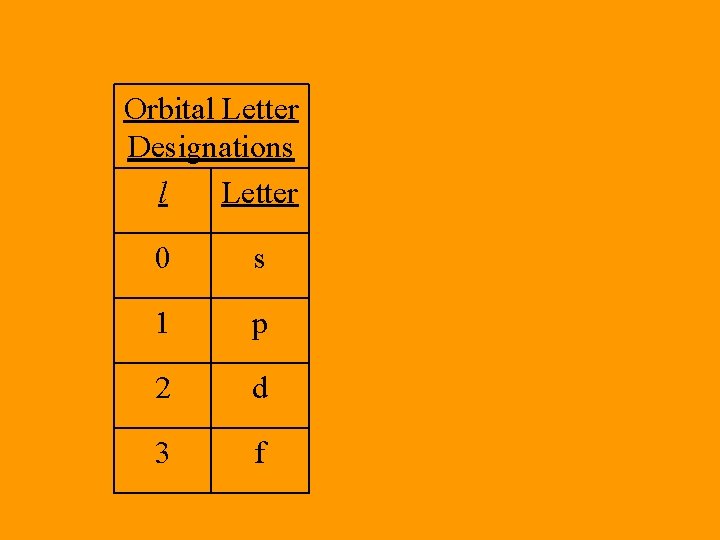
• Angular Momentum Quantum Number – (l) The shape of the orbital. – Starts at 0 and then positive integers till n – 1 is reached • Ex. If n = 2, then l = 0, 1 • Ex. If n = 4, then l = 0, 1, 2, 3

Orbital Letter Designations l Letter 0 s 1 p 2 d 3 f

• Magnetic Quantum Numbers – (m) The orientation of an orbital around the nucleus. – Starts at –l and it each integer to l • Ex. If l = 2, then m = -2, -1, 0, 1, 2 • Ex. If l = 3, then m = -3, -2, -1, 0 , 1, 2, 3 • Ex. If n = 5, then l = 0, 1, 2, 3, 4, then m = -4, -3, -2, -1, 0, 1, 2, 3, 4

• Spin Quantum Number – Indicate the two fundamental spin states of an electron in an orbital. – Can only be + ½ or – ½

Quantum Number Relationships in Atomic Structure Principle Quantum Number (Main Energy Level) Sublevels In Main Energy Level 1 s 1 p 3 d 5 10 s 1 2 p 3 6 d 5 f 7 2 3 4 Number of Orbital’s per Electrons per Main Energy Sublevel Level 1 4 2 2 6 2 8 2 9 16 6 10 14 18 32

• Electron Configuration – The arrangement of electron’s in an atom. • Aufbau Principle – An electron occupies the lowest energy orbital that can receive it. • Pauli Exclusion Principle – No two electrons in the same atom can have the same set of four quantum numbers. • Hunds Rule – Orbital’s of equal energy are each occupied by one electron before any orbital is occupied by a second electron.

1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 6 6 s 2 4 f 14 5 d 10 6 p 6 7 s 2 5 f 14 6 d 10

Orbital Sample 1 • The electron configuration of boron is 1 s 22 p 1. How many electrons are present in an atom of boron? What is the atomic number of boron? Write the orbital notation for boron.

Number of electrons = super scripts added together • Number of electrons = 2 + 1 • Number of electrons = 5 Atomic Number = look on PT • Atomic Number = 5

Orbital Notation = ___ 1 s ___ 2 px ___ 2 py ___ 2 pz • Start with the first orbital… ___ 1 s ___ 2 px ___ 2 py ___ 2 pz • Move onto the next orbital… ___ 1 s ___ 2 px • According to Hunds rule, we have to fill each orbital of the 2 p level with one electron first, then go back and fill with the second electron… ___ 1 s ___ 2 px ___ 2 py ___ 2 pz

Orbital Sample 2 The electron configuration of nitrogen is 1 s 22 p 3. How many electrons are present in a nitrogen atom? What is the atomic number of nitrogen? Write the orbital notation for nitrogen.

Number of electrons = super scripts added together • Number of electrons = 2 + 3 • Number of electrons = 7 Atomic Number = look on PT • Atomic Number = 7

Orbital Notation = ___ 1 s ___ 2 px ___ 2 py ___ 2 pz • Start with the first orbital… ___ 1 s ___ 2 px ___ 2 py ___ 2 pz • Move onto the next orbital… ___ 1 s ___ 2 px • According to Hunds rule, we have to fill each orbital of the 2 p level with one electron first, then go back and fill with the second electron… ___ 1 s ___ 2 px ___ 2 py ___ 2 pz

Orbital Sample 3 The electron configuration of fluorine is 1 s 22 p 5. What is the atomic number of fluorine? How many of its p orbital’s are filled? How many unpaired electrons does a fluorine atom contain?

Atomic Number = look on PT • Atomic Number = 9 How many of its p orbital’ are filled = ___ ___ ___ 1 s 2 s 2 px 2 py 2 pz How many of its p orbital’s are filled = 2 How many unpaired electrons does a fluorine atom contain = 1

• Octet of Electrons – When the s and p orbital’s are filled. Octet = 8 s orbital = 2 + p orbital = 6 2+6=8 • Nobel Gas – The group 18 elements. No unpaired electrons and all orbital’s of the energy level are full.

E- Config. Sample 1 Write both the complete electronconfiguration notation and the noble-gas notation for iron, Fe.

Write the complete electron-configuration notation… • Figure out number of electrons In a stable atom, number of protons = number of electrons Fe = 26 • Start writing electron configuration till number of electrons is reached 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6

Write the noble-gas notation… • Find the highest filled p orbital and its Noble gas 3 p 6 = Ar • Write the noble gas in brackets and then the remainder of the full electron configuration [Ar] 4 s 23 d 6

E- Config. Sample 2 Write both the complete electron configuration notation and the noble-gas notation for iodine, I.

Write the complete electron-configuration notation… • Figure out number of electrons In a stable atom, number of protons = number of electrons I = 53 • Start writing electron configuration till number of electrons is reached 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 5 s 2 4 d 10 5 p 5

Write the noble-gas notation… • Find the highest filled p orbital and its Noble gas 4 p 6 = Kr • Write the noble gas in brackets and then the remainder of the full electron configuration [Kr] 5 s 24 d 105 p 5

E- Config. Sample 3 Without consulting the periodic table or a table in this chapter, write the complete electron configuration for the element with atomic number 25.

Write the complete electron-configuration notation… • Figure out number of electrons Given 25 • Start writing electron configuration till number of electrons is reached 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 5