Development of a Solid Acid Catalyst Alkylation Process






























- Slides: 50

Development of a Solid Acid Catalyst Alkylation Process Alky. Clean Solid Acid Alkylation October 6, 2006 -1 -

Alky. Clean solid acid alkylation TDC_2006 - 2 Presentation Outline n Introduction n Process Development n Demonstration Unit n Economic Benchmarking n Summary

Introduction TDC_2006 - 3 Alky. Clean process for gasoline alkylate n Mandate: Cleaner fuels and “Greener” refining processes n Answer: Alkylate = Clean Gasoline High RON & MON, virtually no olefins, aromatics or sulfur, low RVP n Problem: Safety, environmental and reliability issues associated with current liquid acid technologies n Challenge: Develop and demonstrate an environmentally friendly and competitive Solid Acid Catalyst (SAC) technology to replace HF and H 2 SO 4 technologies

Introduction Alkylation processes TDC_2006 - 4 H 2 SO 4 (Sulfuric acid) HF (Hydrogen fluoride) Alky. Clean (Solid acid) Liquid Gas Solid 80 kg/ton alkylate 4000 gram/ton alkylate <400 gram/ton alkylate

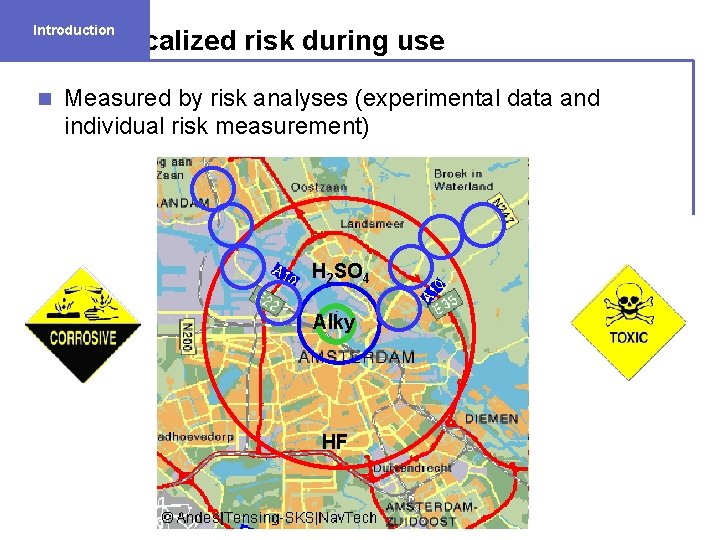
Introduction Localized risk during use n Measured by risk analyses (experimental data and individual risk measurement) H 2 SO 4 Alky HF

Introduction TDC_2006 - 6 Alkylation market drivers n Economic driver – increases quantity of gasoline n Environmental driver – high quality RFG blend stock n No olefins, aromatics, or S n Low volatility (“RVP”) n High octane, RON & MON n MTBE replacement

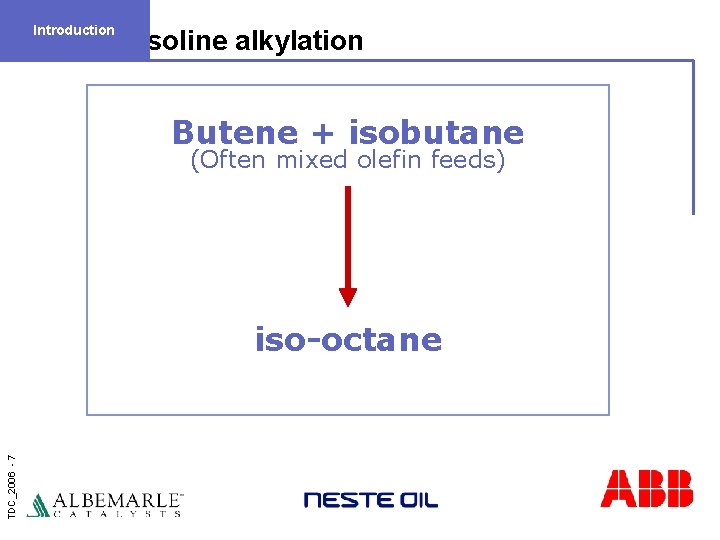
Introduction Gasoline alkylation Butene + isobutane (Often mixed olefin feeds) TDC_2006 - 7 iso-octane

Introduction Gasoline alkylation chemistry n Reaction of C 3 -C 5 olefins with isobutane to produce primarily gasoline boiling range C 7 -C 9 isoparaffins n Primary reaction: IC 4 + C 4= ® n Secondary reactions yield: DMHs Undesirable C 5 - C 7 s. Low Octane C 9 + High RVP or High B. P. TMPs Preferred– High Octane Selectivity to TMPs favored by: n Higher isobutane/olefin (I/O) ratio at catalytic sites n Higher hydrogen transfer rates (catalyst function) n Lower reactor operating temperature TDC_2006 - 8 C 5+ alkylate from C 4 olefins: n n RON: 95 -96 , MON: 92 -94 , RVP: 4 -5 psia

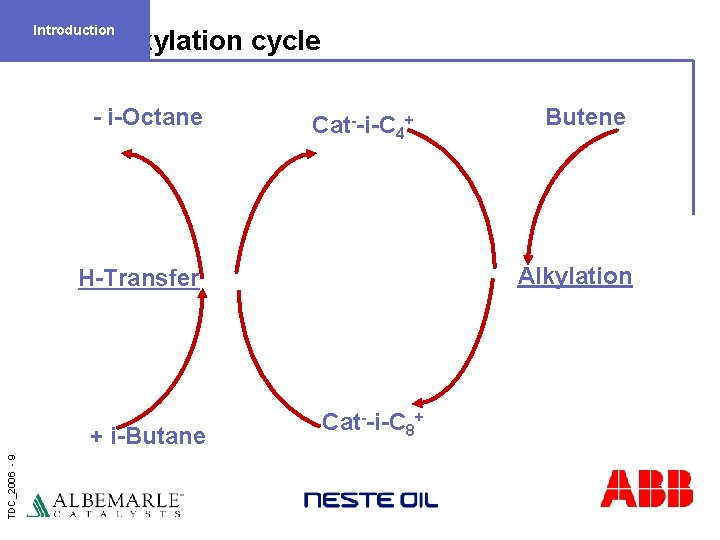
Introduction Alkylation cycle - i-Octane Cat--i-C 4+ Alkylation H-Transfer TDC_2006 - 9 + i-Butane Butene Cat--i-C 8+

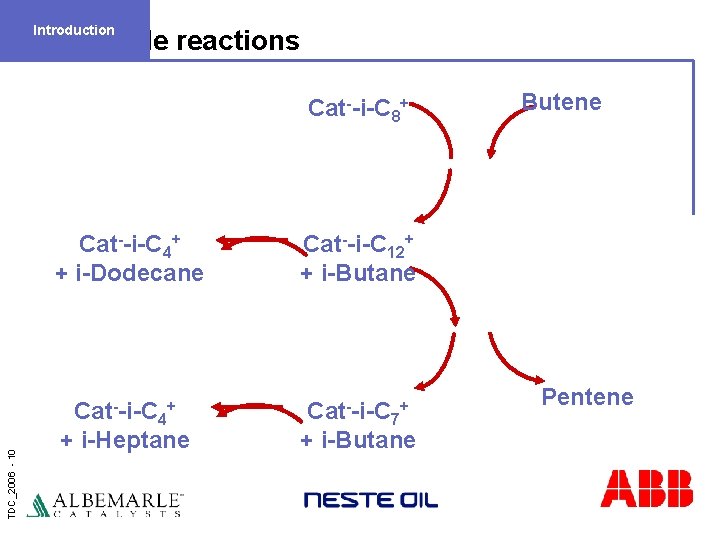
Introduction Side reactions TDC_2006 - 10 Cat--i-C 8+ Cat--i-C 4+ + i-Dodecane Cat--i-C 12+ + i-Butane Cat--i-C 4+ + i-Heptane Cat--i-C 7+ + i-Butane Butene Pentene

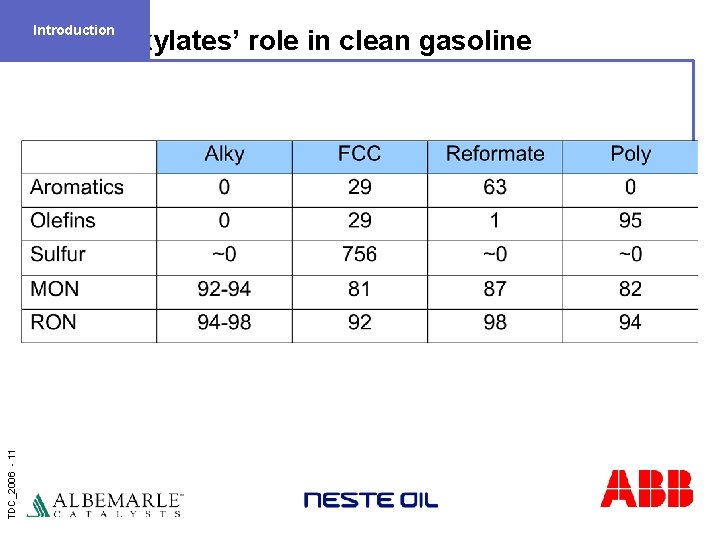
Introduction TDC_2006 - 11 Alkylates’ role in clean gasoline

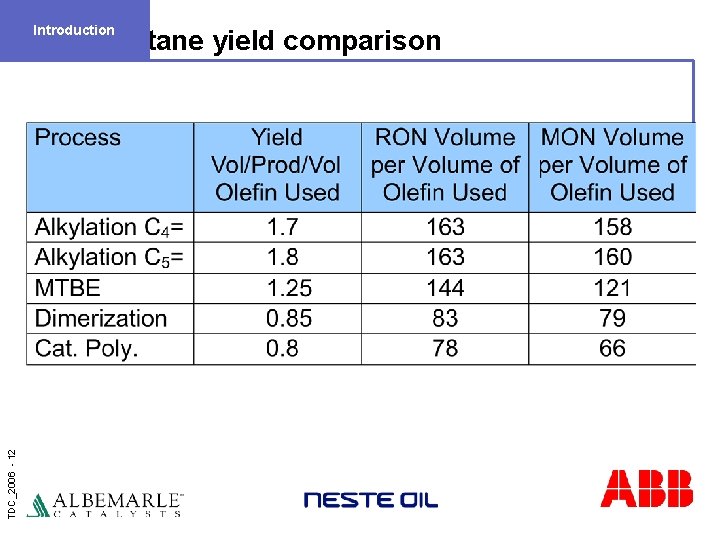
Introduction TDC_2006 - 12 Octane yield comparison

Introduction Alky. Clean catalyst Features n True solid acid: no halogens or volatile components n Properties tailored to yield high quality alkylate, with maximized activity and stability n Robust: low sensitivity towards feedstock composition variation and common impurities Successful commercial scale-up TDC_2006 - 13 n Successful commercial trial production of the original catalyst in 2002 and of a new optimized version in 2004

Introduction Joint venture development progression n ABB Lummus Global and Albemarle Catalysts n n Cooperation since 1996 Neste Oil n TDC_2006 - 14 Initiated R&D effort 1994 Joined the team in 2001 for technology demonstration

Introduction TDC_2006 - 15 Development and demonstration status n Bench scale development work completed n Alky. Clean catalyst manufactured at commercial scale n Demonstration unit constructed and initially operated during 2002 -2003, proving key technology aspects and process operability n Further bench scale effort focused on improvement of catalyst/ process performance and resulting economics n Successfully completed demonstration of these catalyst and processing improvements in 2004 n Technology offered for license beginning 2005 n Bench-scale work continues to expand data base and support next generation catalyst

Alky. Clean solid acid alkylation TDC_2006 - 16 Presentation Outline n Introduction n Process Development n Demonstration Unit n Economic Benchmarking n Summary

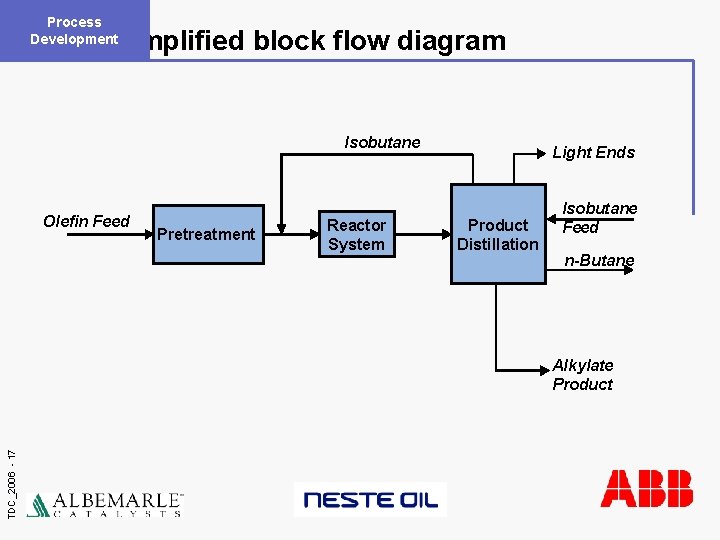
Process Development Simplified block flow diagram Isobutane Olefin Feed Pretreatment Reactor System Light Ends Product Distillation Isobutane Feed n-Butane TDC_2006 - 17 Alkylate Product

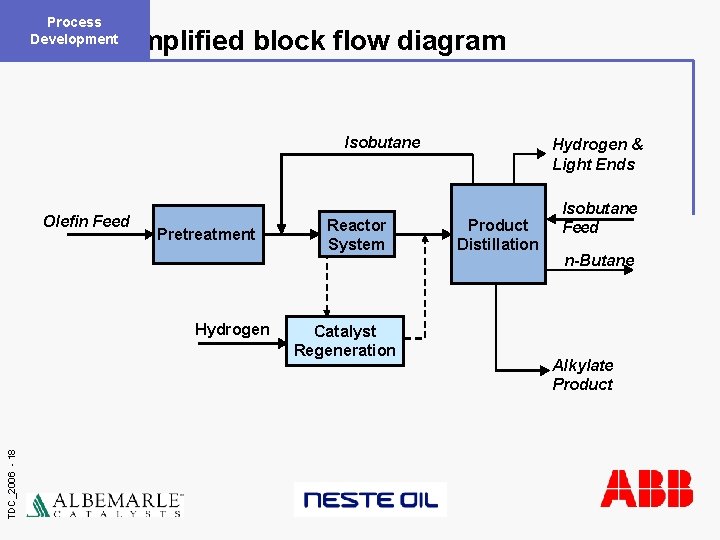
Process Development Simplified block flow diagram Isobutane Olefin Feed Pretreatment TDC_2006 - 18 Hydrogen Reactor System Catalyst Regeneration Hydrogen & Light Ends Product Distillation Isobutane Feed n-Butane Alkylate Product

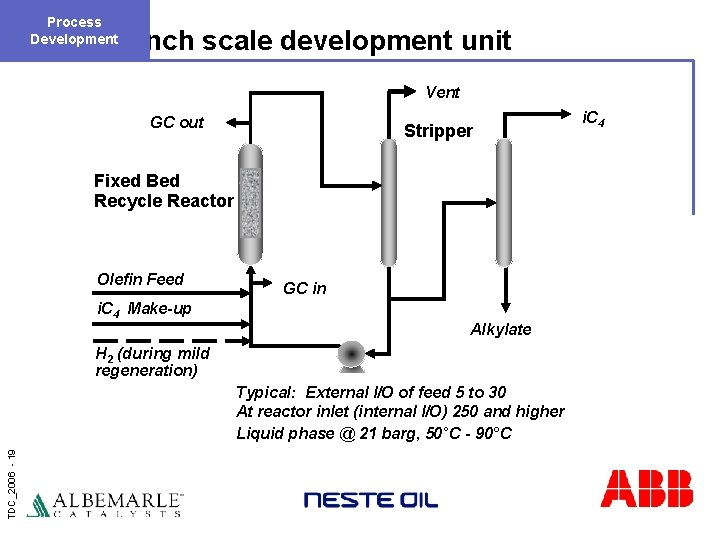
Process Development Bench scale development unit Vent GC out Stripper Fixed Bed Recycle Reactor Olefin Feed GC in i. C 4 Make-up Alkylate H 2 (during mild regeneration) TDC_2006 - 19 Typical: External I/O of feed 5 to 30 At reactor inlet (internal I/O) 250 and higher Liquid phase @ 21 barg, 50°C - 90°C i. C 4

Process Development TDC_2006 - 20 Cyclic pilot unit in Amsterdam

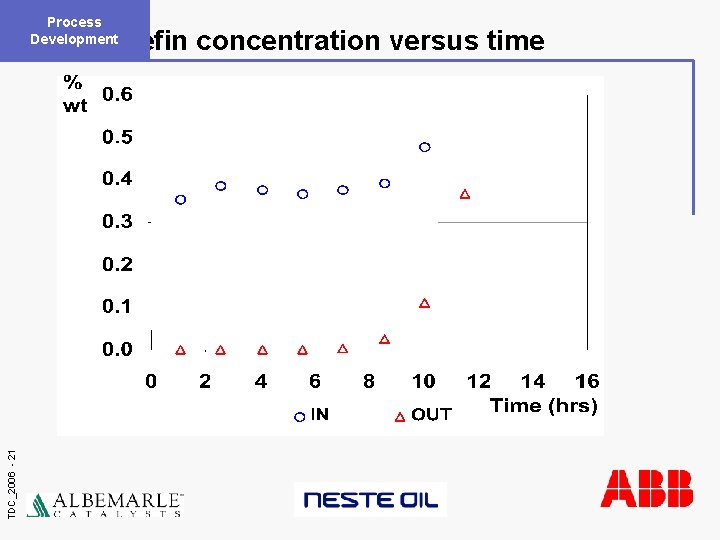
Process Development TDC_2006 - 21 Olefin concentration versus time

Process Development TDC_2006 - 22 Effect of regeneration procedure Exp. Regenerant (°C) T (bar) P (hr) Time (hr) Cat. Life 0 Fresh catalyst 1 H 2 gas 250 21 1 10 1 a H 2 gas 250 21 1 10 1 b H 2 gas 250 21 1 10 2 6. 5 i-C 4 liquid with dissolved H 2 90 21 66 3 4 i. C 4 liquid with dissolved H 2 115 30 18 10

Process Development NEW TITLE Conclusions – Regeneration after olefin breakthrough n Regeneration at 250°C in H 2 (gas phase) completely recovers activity and selectivity n Regeneration with i-C 4 and dissolved H 2 (liquid phase) not successful TDC_2006 - 23 Next – Investigated short cycle mild regeneration n Alternating periods of alkylation and liquid phase regeneration with i-C 4 and dissolved H 2 n Regeneration occurs prior to significant olefin breakthrough

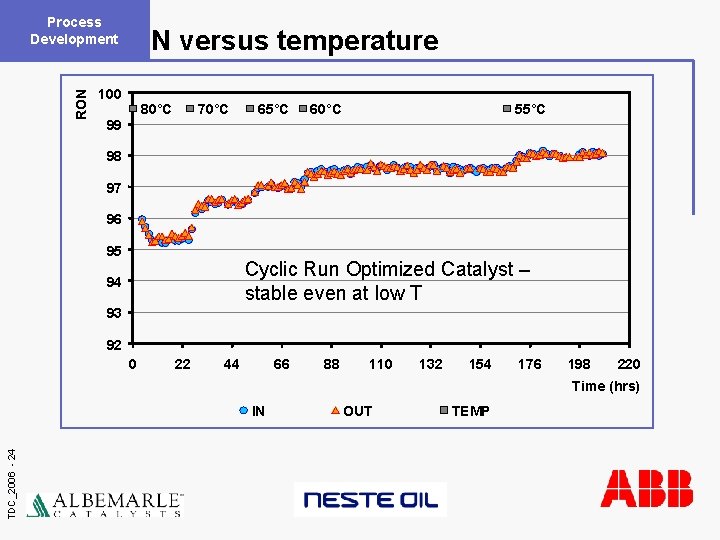
Process Development RON versus temperature 100 80°C 70°C 65°C 60°C 55°C 99 98 97 96 95 Cyclic Run Optimized Catalyst – stable even at low T 94 93 92 0 22 44 66 88 110 132 154 176 198 220 Time (hrs) TDC_2006 - 24 IN OUT TEMP

Process Development Process key – cyclic reactor operation Short cycle alkylation / mild regeneration TDC_2006 - 25 n Alternating periods of alkylation and liquid phase mild regeneration with i-C 4 and dissolved H 2 n Seamless – no change in operating conditions; hydrogen injection substituted for olefin feed n Mild regeneration is pre-emptive – occurs prior to excessive deactivation and formation of ‘hard’ coke n Allows for continuous operation and maintenance of product quality n First patent granted in 1999 – US 5, 986, 158

Process Development High temperature regeneration (HTR) n Gradual catalyst deactivation, over time under cyclic operation, necessitates off-line HTR n HTR: hot hydrogen strip at 250°C – completely recovers activity and selectivity TDC_2006 - 26 n HTR undertaken before formation of hardest coke species (e. g. high MW condensed cyclics), which would require oxidative ‘burn-off’ n Required HTR frequency 4 -30 days depending on operating severity n Effectiveness of HTR to fully restore activity proven over > 6 months of operation

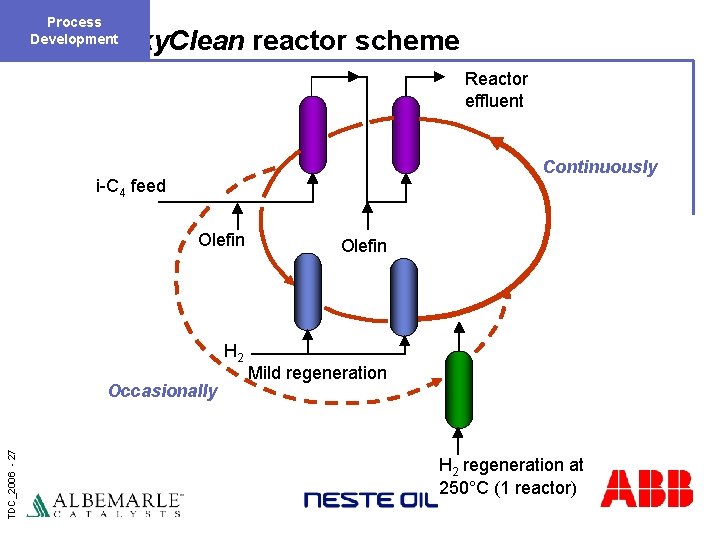
Process Development Alky. Clean reactor scheme Reactor effluent Continuously i-C 4 feed Olefin H 2 TDC_2006 - 27 Occasionally Olefin Mild regeneration H 2 regeneration at 250°C (1 reactor)

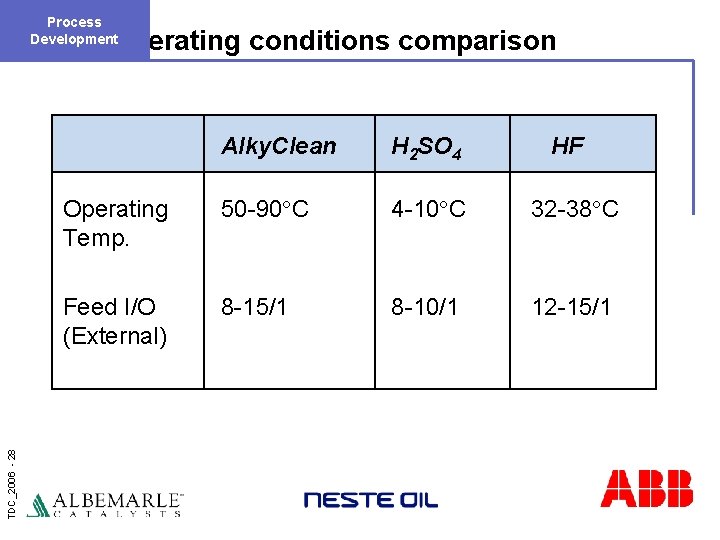
Process Development TDC_2006 - 28 Operating conditions comparison Alky. Clean H 2 SO 4 HF Operating Temp. 50 -90°C 4 -10°C 32 -38°C Feed I/O (External) 8 -15/1 8 -10/1 12 -15/1

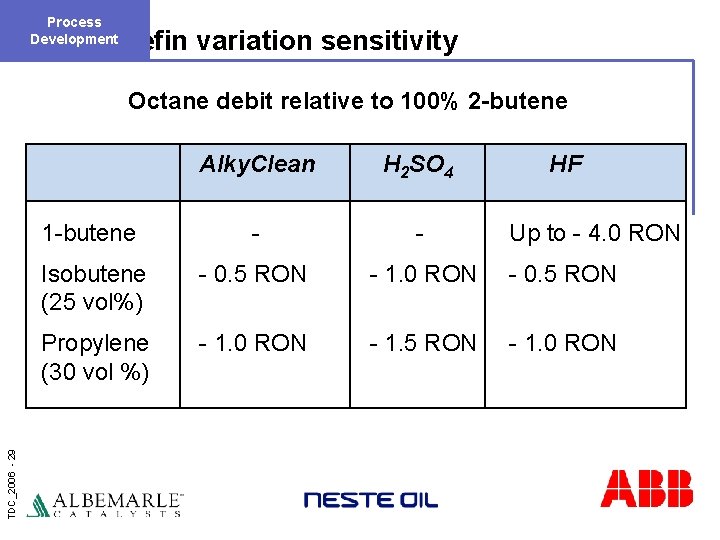
Process Development Olefin variation sensitivity TDC_2006 - 29 Octane debit relative to 100% 2 -butene Alky. Clean H 2 SO 4 HF 1 -butene - - Isobutene (25 vol%) - 0. 5 RON - 1. 0 RON - 0. 5 RON Propylene (30 vol %) - 1. 0 RON - 1. 5 RON - 1. 0 RON Up to - 4. 0 RON

Process Development TDC_2006 - 30 Results of feedstock impurity testing n Water saturated feed gave the same results as dry feed n After spiking total reactor feed with: 600 ppmw DME, 200 ppmw CH 3 SH, 1200 ppmw H 2 S 1800 ppmw butadiene (each separately) n Any activity loss could be recovered by high temperature regeneration with H 2 at 250°C

Process Development TDC_2006 - 31 Catalyst testing results n Sensitivity to olefin composition (C 3=, n-C 4=, i-C 4=) variation is relatively low compared to HF/H 2 SO 4 n Exposure to high levels of typical feed impurities ( H 2 O, oxygenates, sulfur compounds, butadiene) does not cause irreversible deactivation n Commercial feeds can be converted with good activity, selectivity and stability, yielding high quality product with no co-production of an ASO (heavy hydrocarbon bleed stream) without “clean up” facilities

Alky. Clean solid acid alkylation TDC_2006 - 32 Presentation Outline n Introduction n Process Development n Demonstration Unit n Economic Benchmarking n Summary

TDC_2006 - 33 Demonstration Unit Alky. Clean demonstration unit n Neste Oil joined team in early 2001 for technology demonstration n ABB Lummus Global’s basic engineering completed 2001 n Demonstration unit construction completed in 2002; operates at Neste facilities in Porvoo, Finland with actual refinery feed streams; 10 BPD production capacity n Contains all key elements and is analogous to commercial design n Allows for proving operability, confirmation of design parameters and reliable scale-up

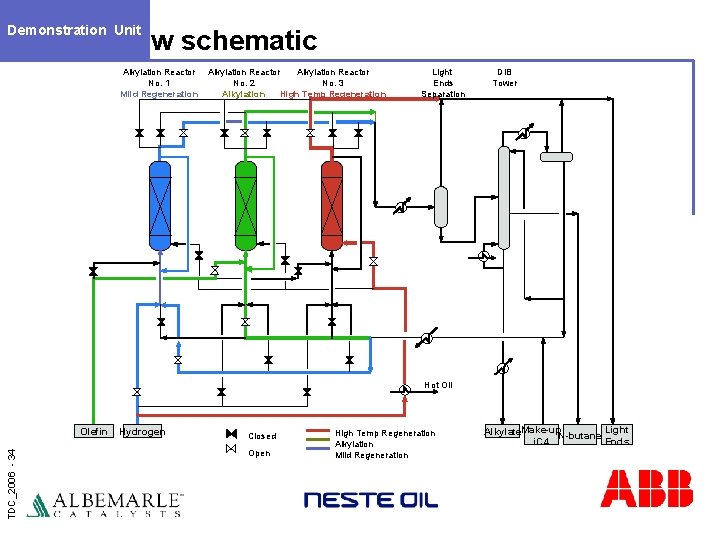
Demonstration Unit Flow schematic Alkylation Reactor No. 1 Mild Regeneration Alkylation Reactor No. 2 No. 3 Alkylation High Temp Regeneration Light Ends Separation DIB Tower Hot Oil TDC_2006 - 34 Olefin Hydrogen Closed Open High Temp Regeneration Alkylation Mild Regeneration Alkylate. Make-up. N-butane Light i. C 4 Ends

Demonstration Unit TDC_2006 - 35 Outside view

Demonstration Unit TDC_2006 - 36 Reactor section

Demonstration Unit TDC_2006 - 37 Reactor lower section

Demonstration Unit TDC_2006 - 38 Alky. Clean demonstration unit n Demonstration unit construction completed in 2002; operates at Neste’s facilities in Porvoo, Finland with actual refinery feed streams n Contains all key elements and is analogous to commercial design n Allowed for proving operability, confirmation of design parameters and reliable scale-up

Demonstration Unit Operation summary n Unit reliably operated for over two years utilizing refinery slipstreams, both C 4 and C 3/C 4 mixed olefins n Alkylate quality comparable to Porvoo HF unit n Key technology aspects proven n TDC_2006 - 39 n n Operated continuously with multiple high temperature regenerations n Catalyst activity recovered consistently Performance data obtained over a wide range of conditions n Support correlations/modeling effort and economic benchmarking n Some surprises, leading to insights and opportunities for catalyst/process optimization Absolutely no fouling, plugging, corrosion, erosion or degradation to the plant over the years of operation

Demonstration Unit Recent operations n In April 2004 second generation of catalyst tested: Successful bench scale catalyst / processing optimization effort n As with the first generation, commercial trial manufacture of the new improved catalyst n Demo unit modifications incorporated operational improvements n n Demonstration operated successfully for another six months Benefits of operational improvements confirmed n Improved catalyst activity and stability confirmed n Established excellent correlation between this unit and the bench scale unit n Demonstration unit available for client feedstock testing n Bench scale unit continues to operate for parametric optimization TDC_2006 - 40 n

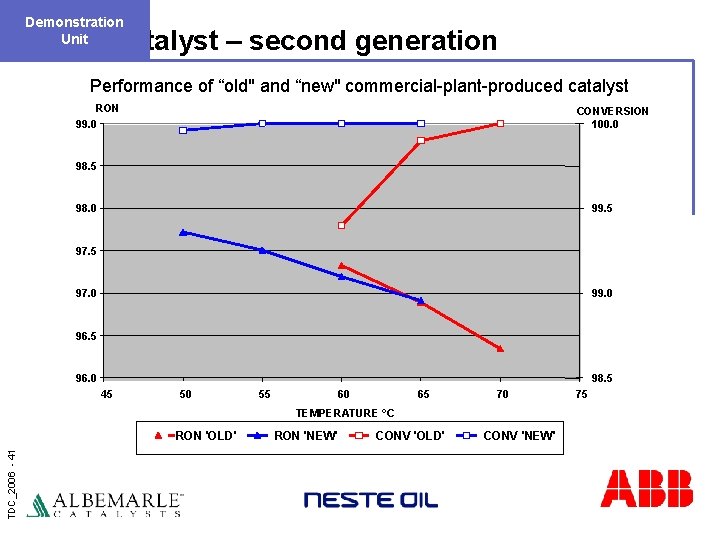
Demonstration Unit Catalyst – second generation Performance of “old" and “new" commercial-plant-produced catalyst RON 99. 0 CONVERSION 100. 0 98. 5 98. 0 99. 5 97. 0 99. 0 96. 5 96. 0 98. 5 45 50 55 60 65 70 TEMPERATURE °C TDC_2006 - 41 RON 'OLD' RON 'NEW' CONV 'OLD' CONV 'NEW' 75

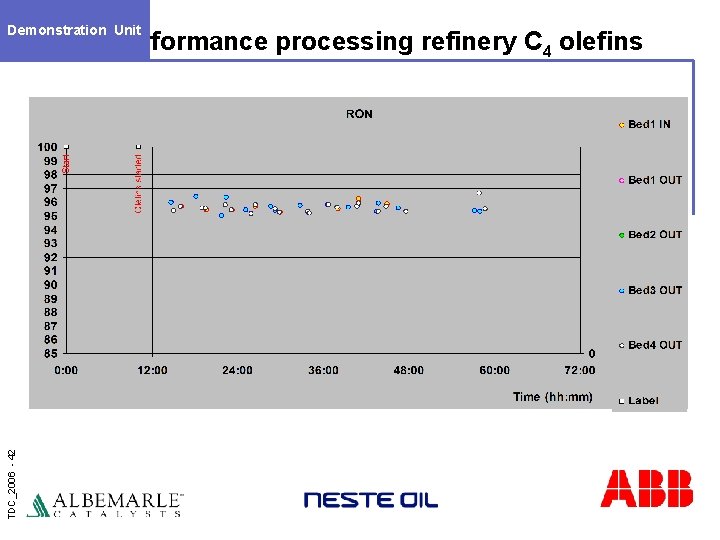
Demonstration Unit TDC_2006 - 42 Performance processing refinery C 4 olefins

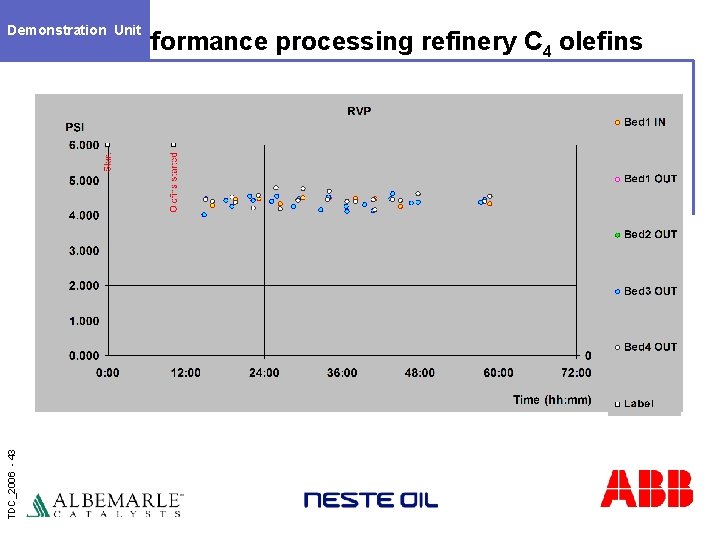
Demonstration Unit TDC_2006 - 43 Performance processing refinery C 4 olefins

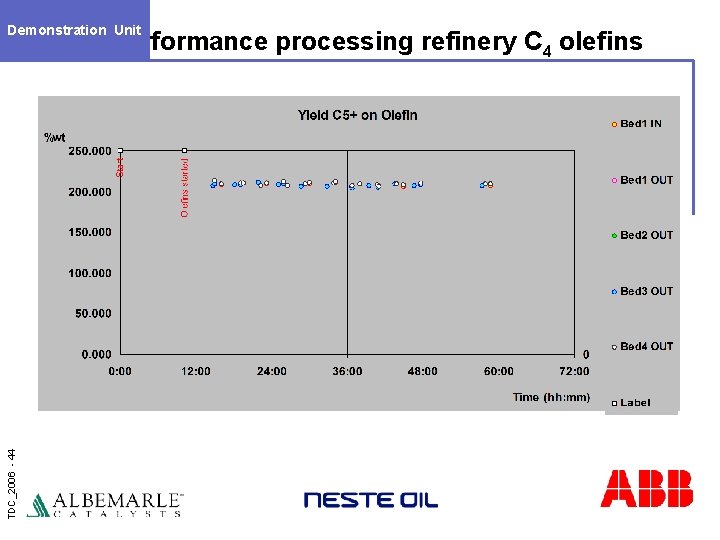
Demonstration Unit TDC_2006 - 44 Performance processing refinery C 4 olefins

Alky. Clean solid acid alkylation TDC_2006 - 45 Presentation Outline n Introduction n Process Development n Demonstration Unit n Economic Benchmarking n Summary

Economic Benchmarking Design feed composition TDC_2006 - 46 FCC C 4 s

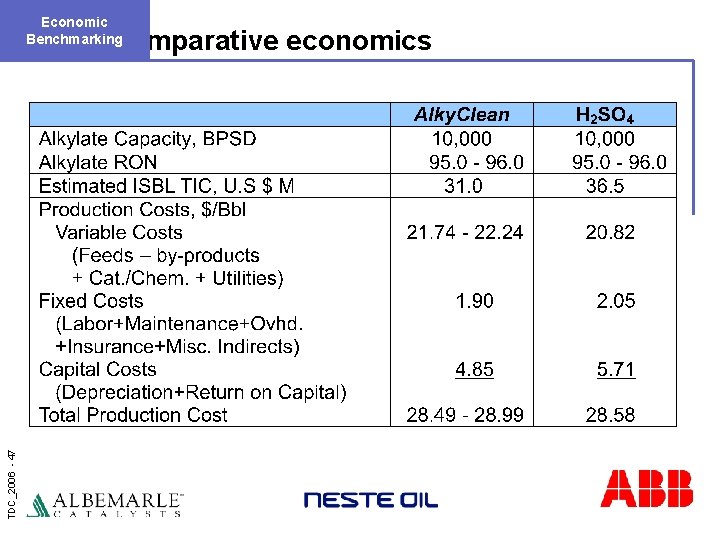
Economic Benchmarking TDC_2006 - 47 Comparative economics

Alky. Clean solid acid alkylation TDC_2006 - 48 Presentation Outline n Introduction n Process Development n Demonstration Unit n Economic Benchmarking n Summary

TDC_2006 - 49 Summary Benefits of the Alky. Clean process n True solid acid catalyst eliminates the hazards associated with liquid acids n Low emissions / environmental impact n No production of acid soluble oil (ASO) n No product post treatment needed n No refrigeration or alloy construction; common refinery equipment, non-corrosive/erosive n Reduced maintenance and manpower n Lower sensitivity towards olefin feed composition n Robust with respect to key impurities n Competitive economics with comparable alkylate quality

Summary In conclusion TDC_2006 - 50 The Alky. Clean process n Offers significant environmental and operational benefits relative to existing liquid acid technologies at a competitive cost n FYI… Wall Street Journal Europe Innovation Award 2002