Acetic acid water Acetylsalicylic acid Benzoic acid is
Acetic acid +water Acetylsalicylic acid Benzoic acid is used for foods while benzoin resin is used in cosmetics, lotions and soaps
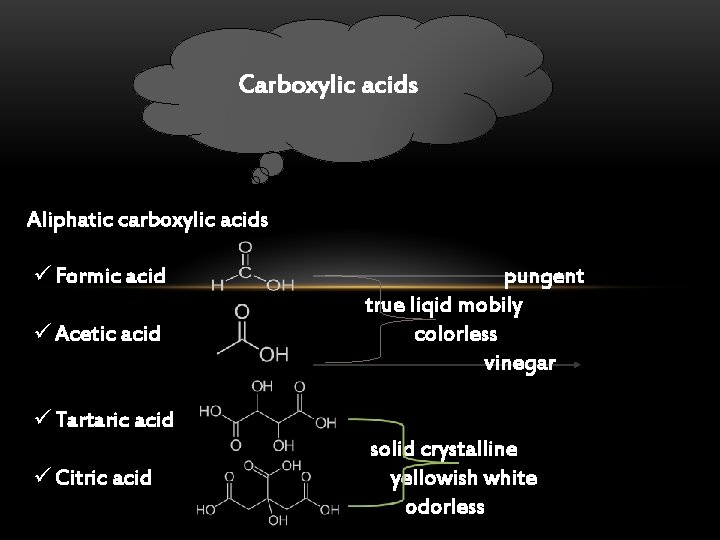
Carboxylic acids Aliphatic carboxylic acids ü Formic acid ü Acetic acid pungent true liqid mobily colorless vinegar ü Tartaric acid ü Citric acid solid crystalline yellowish white odorless
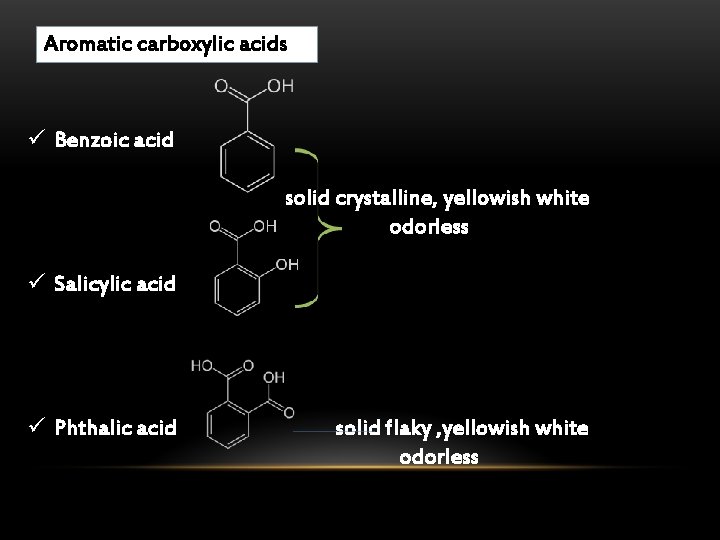
Aromatic carboxylic acids ü Benzoic acid solid crystalline, yellowish white odorless ü Salicylic acid ü Phthalic acid solid flaky , yellowish white odorless

Ignition test Aliphatic C. A: inflammable , non luminous and no smoky Aromatic C. A: inflammable , luminous and smoky Appearance change: formic and acetic acid volatilization tartaric acid swelling up and charring* citric acid melting and formation yellow resin*blackening benzoic, salicylic, phthalic acid melting

Color flame: No change with all Odor: Formic , acetic acid : no change Tartaric, citric acid: burnt sugar odor. Benzoic , phthalic acid: irritant odor (suffocating). Salicylic acid: phenolic odor. Residue: No residue with all except Tartaric acid : black residue. Citric acid: yellow stain.

Acid base test Acidity test: Na 2 CO 3 solution(conc) + acid → warm → effervescent Strong effervescent that means acid. Weak effervescent that means acidic. Conc. Hcl test: when substance soluble in 10% Na. OH that means acid. if add Hc. L in same test tube→ reprecipted again

acids Formic Acetic tartaric citric benzoic phthalic salicylic H 2 O Miscible L. P Change blue to red soluble Change blue to red Insoluble in H 2 O , dil HCL but soluble in 10%Na. OH Not done All strong acid Acidity test Conc. HCL effervescent Reppt.

Preliminary test Soda lime test: Formic and acetic acid: not done for solid only. Tartaric and citric acid: burnt sugar odor on hot. Benzoic and phthalic acid: benzene odor on hot. Salicylic acid: phenolic odor on hot.

30%Na. OH: The same result of soda lime Fe. CL 3: No reaction with all. Conc. H 2 SO 4: Formic and acetic acid: effervescent due to CO 2 evolution on cold. Tartaric acid : effervescent and heavy charring on hot. Citric acid: effervescent and yellow stain after long time it blackening. benzoic, phthalic and salicylic acids: no reaction

General test Neutral Fe. CL 3: 1 ml of acids +2 ml NH 4 OH→ heat until complete disapperance of ammonia odor then wait 2 -3 min. to cooling then add 2 -3 dps of neutral Fe. CL 3→ formic , acetic acids: blood red color give brown if heat it. Tartaric, citric acids: lemon yellow color. Benzoic, phthalic acids: buff ppt. Salicylic acid: no need to use NH 4 OH just salicylic +H 2 O +neutral. Fe. CL 3 to give violet color.
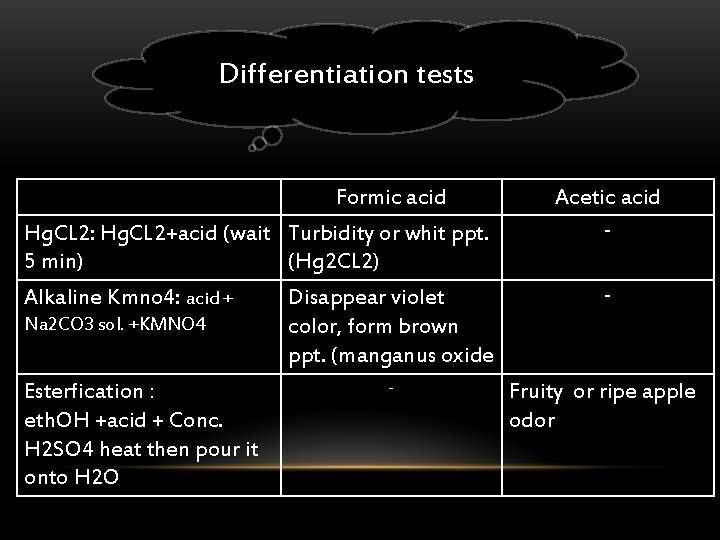
Differentiation tests Formic acid Hg. CL 2: Hg. CL 2+acid (wait Turbidity or whit ppt. 5 min) (Hg 2 CL 2) Alkaline Kmno 4: acid + Na 2 CO 3 sol. +KMNO 4 Esterfication : eth. OH +acid + Conc. H 2 SO 4 heat then pour it onto H 2 O Disappear violet color, form brown ppt. (manganus oxide - Acetic acid - Fruity or ripe apple odor

Conc. H 2 SO 4: (hot) Tartaric acid Citric acid Eff. And charring Eff. And yellow stain Denges: acid+ H 2 O+ denges reagent boiling (without cooling) + KMNO 4 - Benzoic acid Discoloration of violet color anf form white ppt. Phthalic acid Phthaline test: Phenol+ acid +Conc. H 2 SO 4 heat then pour it onto 10% Na. OH - Unstable pink color Flourescence; Acid + resercinol+ Conc. H 2 SO 4 heat then pour it onto 10% Na. OH - Reddish brown color give green flourescence if dilute it with water

Salicylic acid Esterfication: methanol+ saliucylic acid+ Conc H 2 SO 4 heat then pour it onto 10 ml of H 2 O → vicks odor of winter green oil due to methyl salicylate formation. Formaldehyde: Formaldehyde+ salicylic acid + Conc. H 2 SO 4→ crimson red color on the wall. Phthaline test: Salicylic acid + phthalic unhydride +Conc. H 2 SO 4 heat then pour it onto 10 ml of 10% Na. OH→ unstable pink color

IR location COOH stretching band at 1710 cm-1
- Slides: 14