Carboxylic acid derivatives Ketoacids glyoxylic acid phenylglyoxylic acid
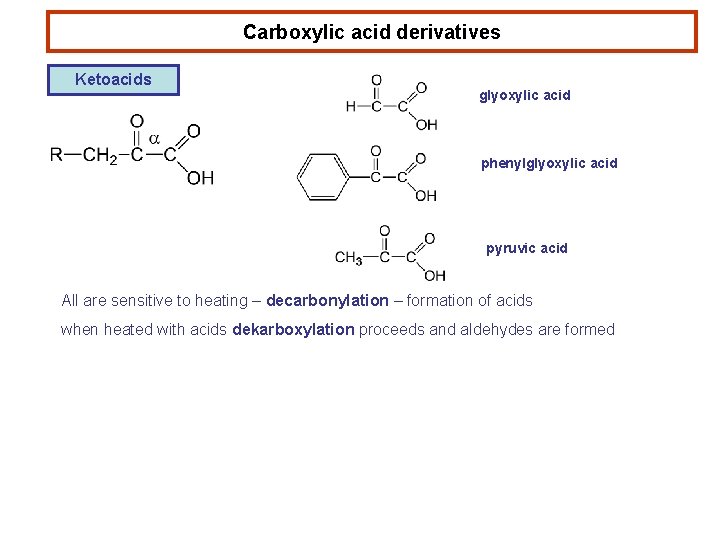
Carboxylic acid derivatives Ketoacids glyoxylic acid phenylglyoxylic acid pyruvic acid All are sensitive to heating – decarbonylation – formation of acids when heated with acids dekarboxylation proceeds and aldehydes are formed
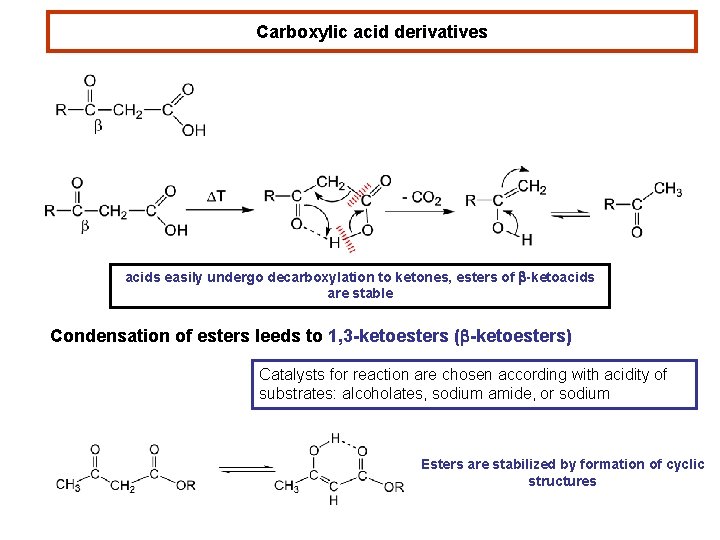
Carboxylic acid derivatives acids easily undergo decarboxylation to ketones, esters of b-ketoacids are stable Condensation of esters leeds to 1, 3 -ketoesters (b-ketoesters) Catalysts for reaction are chosen according with acidity of substrates: alcoholates, sodium amide, or sodium Esters are stabilized by formation of cyclic structures
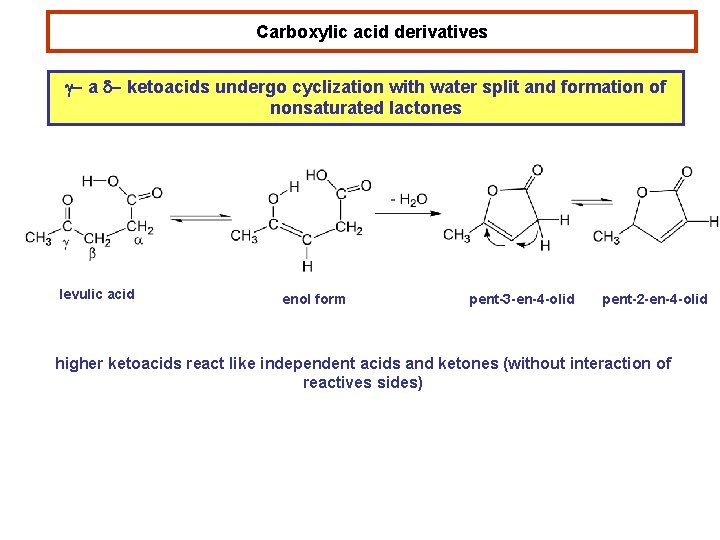
Carboxylic acid derivatives g- a d- ketoacids undergo cyclization with water split and formation of nonsaturated lactones levulic acid enol form pent-3 -en-4 -olid pent-2 -en-4 -olid higher ketoacids react like independent acids and ketones (without interaction of reactives sides)

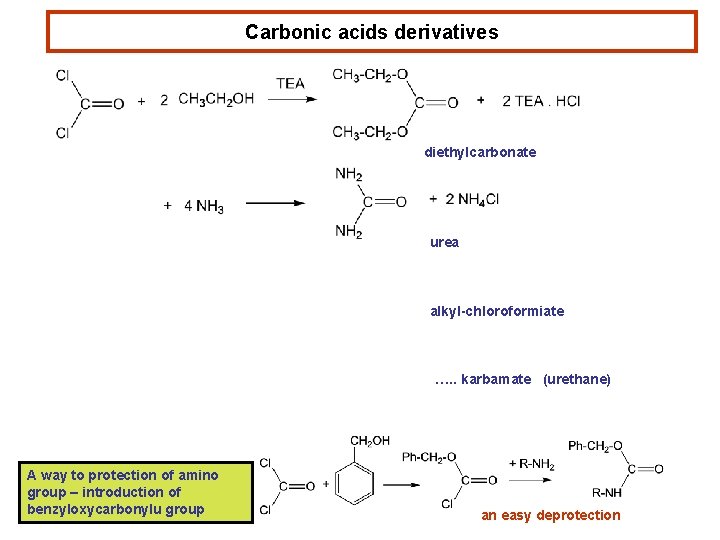
Carbonic acids derivatives diethylcarbonate urea alkyl-chloroformiate …. . karbamate (urethane) A way to protection of amino group – introduction of benzyloxycarbonylu group an easy deprotection
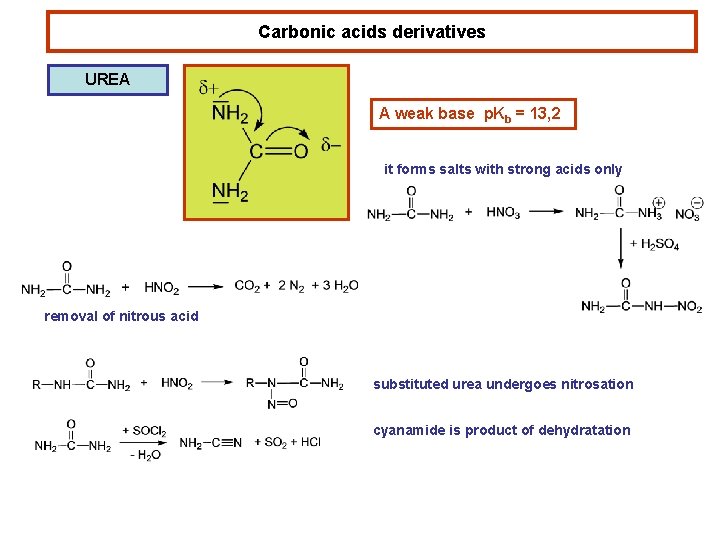
Carbonic acids derivatives UREA A weak base p. Kb = 13, 2 it forms salts with strong acids only removal of nitrous acid substituted urea undergoes nitrosation cyanamide is product of dehydratation
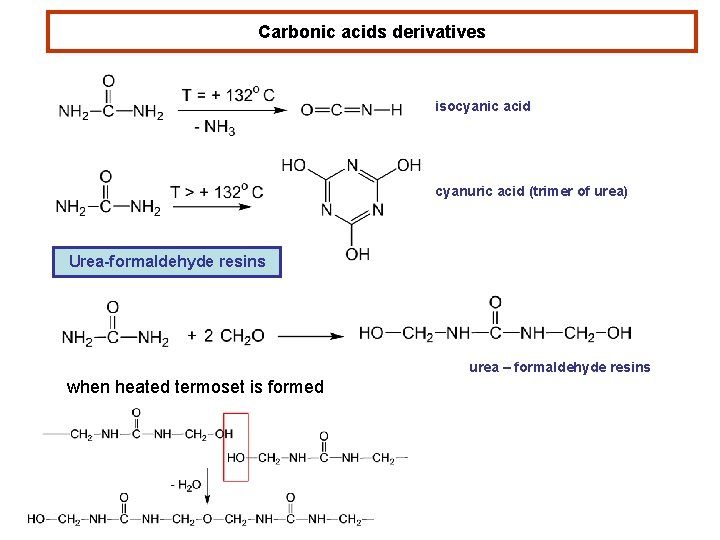
Carbonic acids derivatives isocyanic acid cyanuric acid (trimer of urea) Urea-formaldehyde resins urea – formaldehyde resins when heated termoset is formed

Carbonic acids derivatives
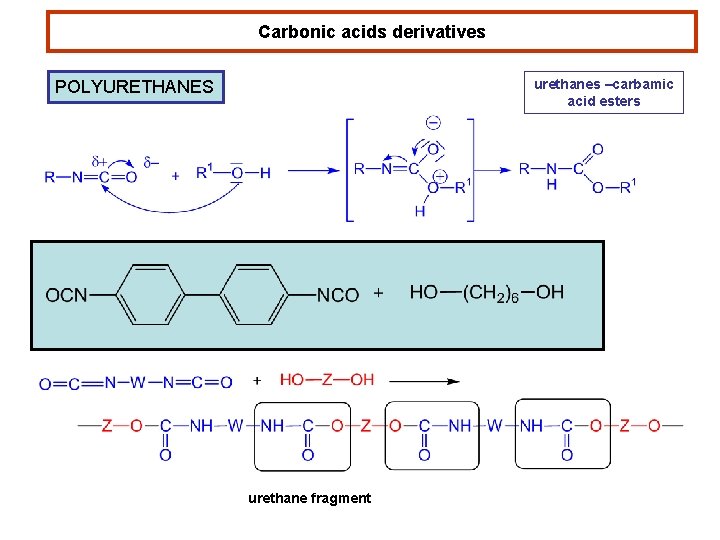
Carbonic acids derivatives urethanes –carbamic acid esters POLYURETHANES urethane fragment
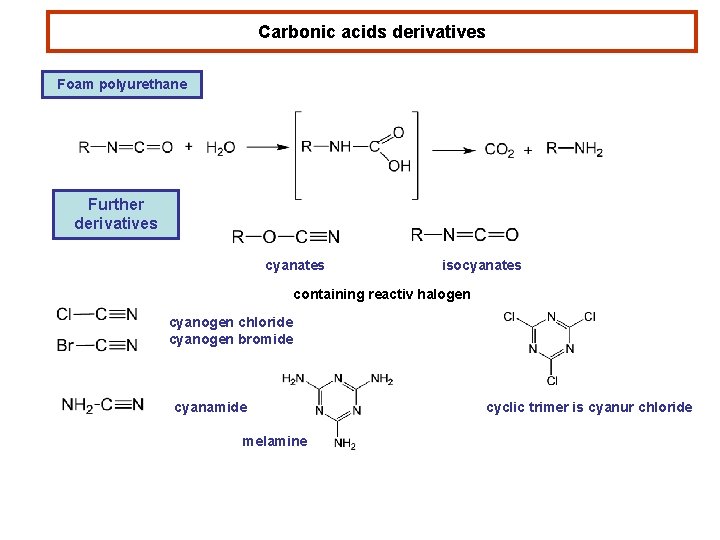
Carbonic acids derivatives Foam polyurethane Further derivatives cyanates isocyanates containing reactiv halogen cyanogen chloride cyanogen bromide cyanamide melamine cyclic trimer is cyanur chloride
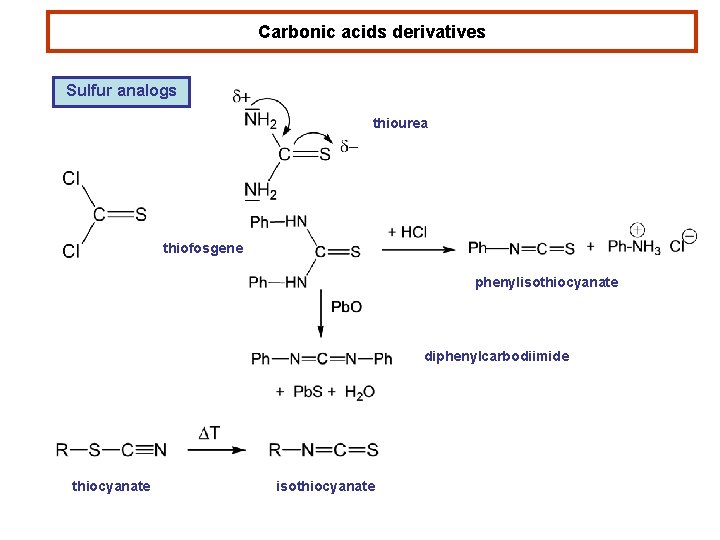
Carbonic acids derivatives Sulfur analogs thiourea thiofosgene phenylisothiocyanate diphenylcarbodiimide thiocyanate isothiocyanate
- Slides: 11