Amino Acids Peptides and Proteins Classification of Amino












- Slides: 41

Amino Acids, Peptides, and Proteins.

Classification of Amino Acids

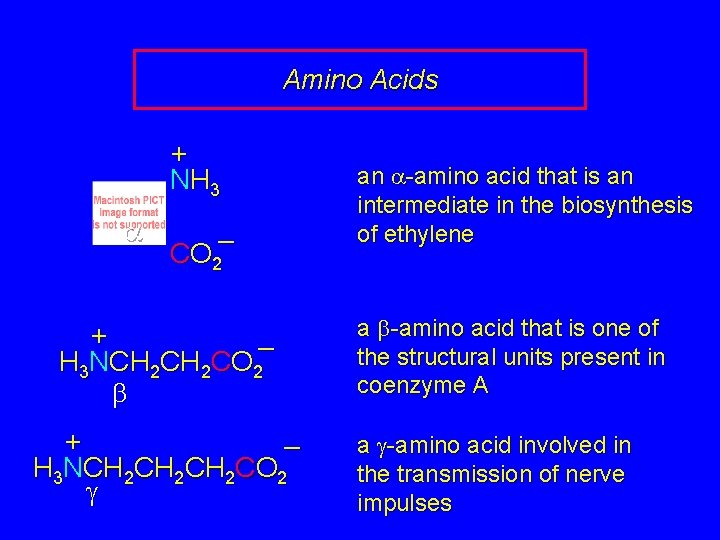
Fundamentals While their name implies that amino acids are compounds that contain an —NH 2 group and a —CO 2 H group, these groups are actually present as —NH 3+ and —CO 2– respectively. They are classified as a, b, g, etc. amino acids according the carbon that bears the nitrogen.

Amino Acids + NH 3 a CO 2– + – H 3 NCH 2 CO 2 b + – H 3 NCH 2 CH 2 CO 2 g an a-amino acid that is an intermediate in the biosynthesis of ethylene a b-amino acid that is one of the structural units present in coenzyme A a g-amino acid involved in the transmission of nerve impulses

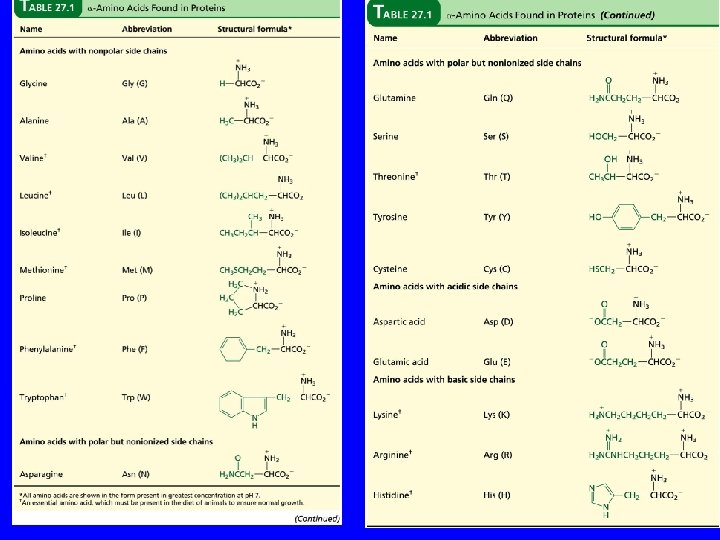
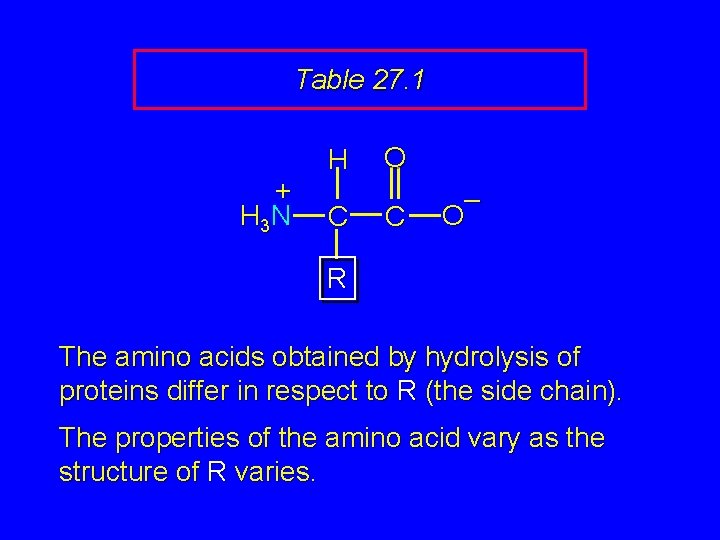
The 20 Key Amino Acids More than 700 amino acids occur naturally, but 20 of them are especially important. These 20 amino acids are the building blocks of proteins. All are a-amino acids. They differ in respect to the group attached to the a carbon. These 20 are listed in Table 27. 1 (p 1054 -1055).



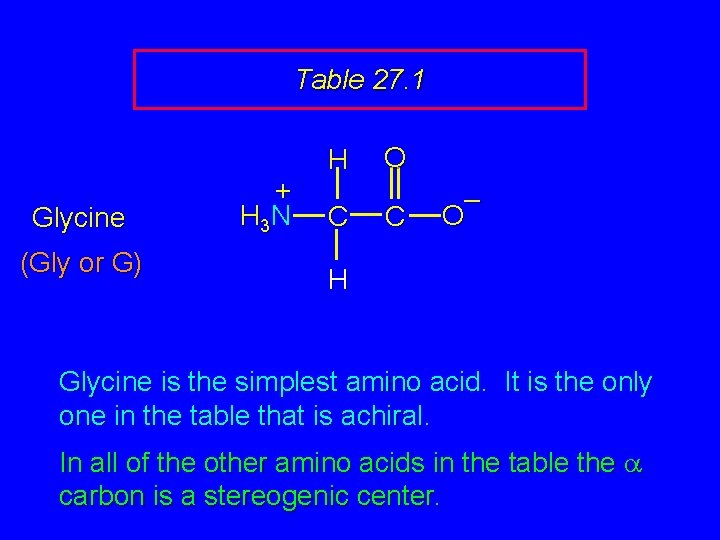
Table 27. 1 + H 3 N H C O C – O R The amino acids obtained by hydrolysis of proteins differ in respect to R (the side chain). The properties of the amino acid vary as the structure of R varies.

Table 27. 1 Glycine (Gly or G) + H 3 N H C O C – O H Glycine is the simplest amino acid. It is the only one in the table that is achiral. In all of the other amino acids in the table the a carbon is a stereogenic center.

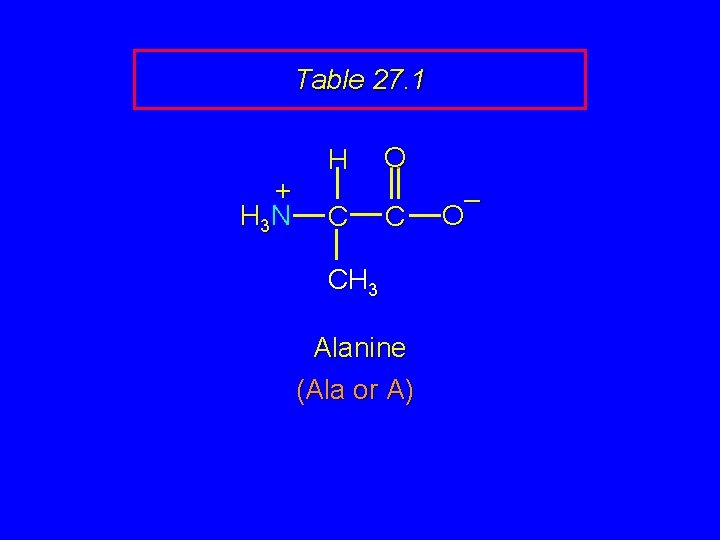
Table 27. 1 + H 3 N H C O C CH 3 Alanine (Ala or A) – O

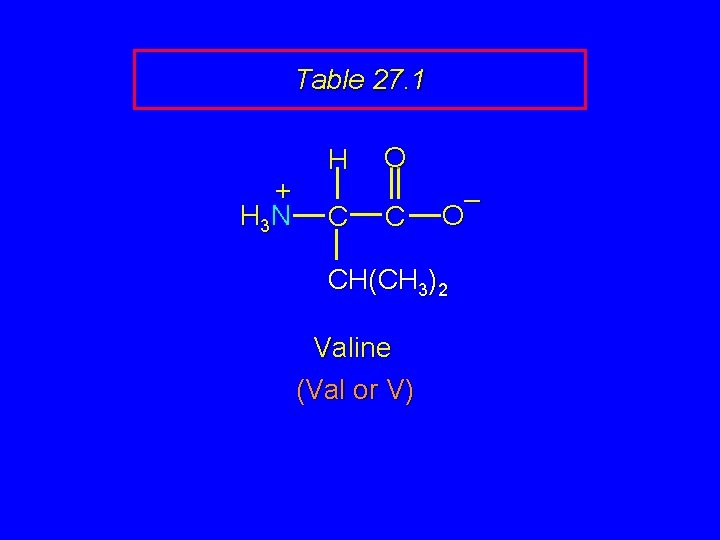
Table 27. 1 + H 3 N H C O C – O CH(CH 3)2 Valine (Val or V)

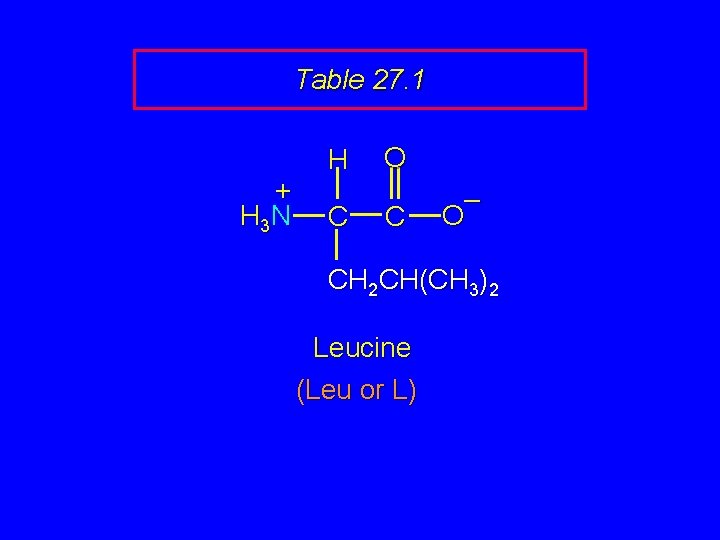
Table 27. 1 + H 3 N H C O C – O CH 2 CH(CH 3)2 Leucine (Leu or L)

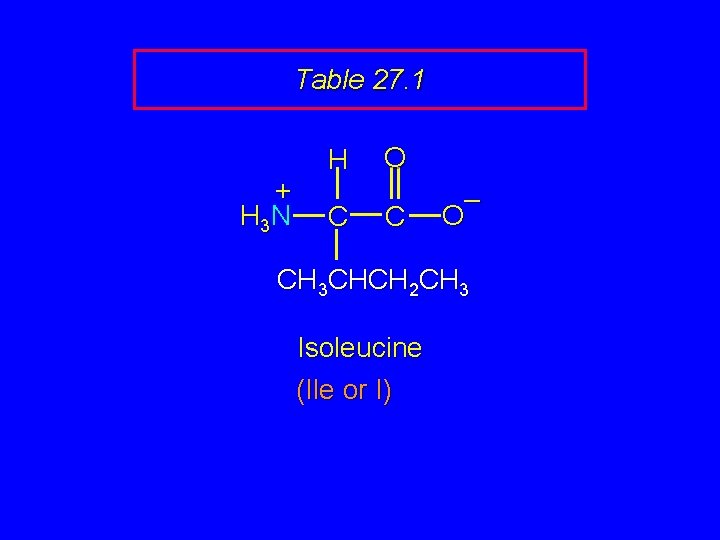
Table 27. 1 + H 3 N H C O C – O CH 3 CHCH 2 CH 3 Isoleucine (Ile or I)

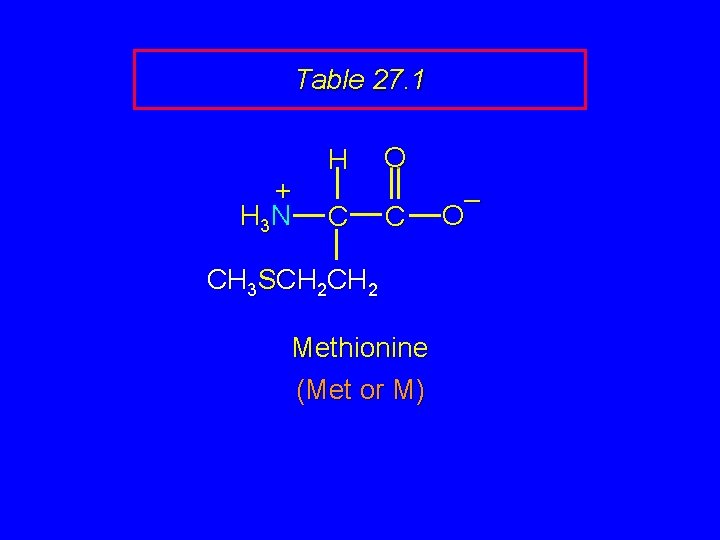
Table 27. 1 + H 3 N H C O C CH 3 SCH 2 Methionine (Met or M) – O

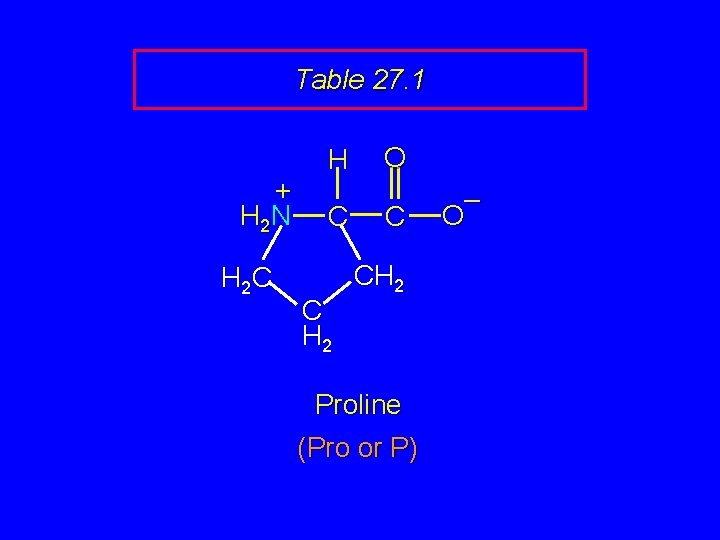
Table 27. 1 + H 2 N H 2 C H C C H 2 O C CH 2 Proline (Pro or P) – O

Table 27. 1 + H 3 N H C O C – O CH 2 Phenylalanine (Phe or F)

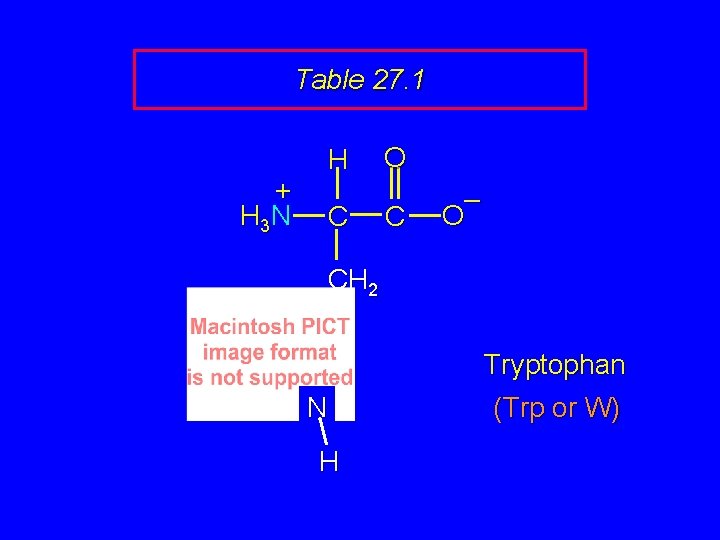
Table 27. 1 + H 3 N H C O C – O CH 2 Tryptophan N H (Trp or W)

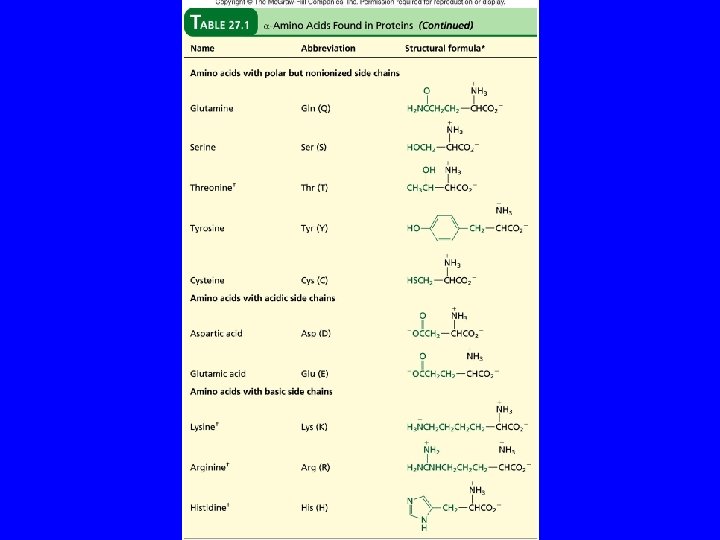
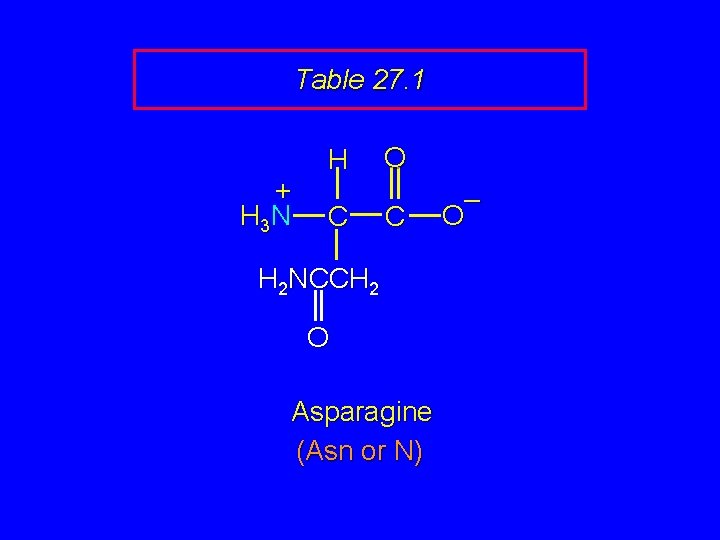
Table 27. 1 + H 3 N H C O C H 2 NCCH 2 O Asparagine (Asn or N) – O

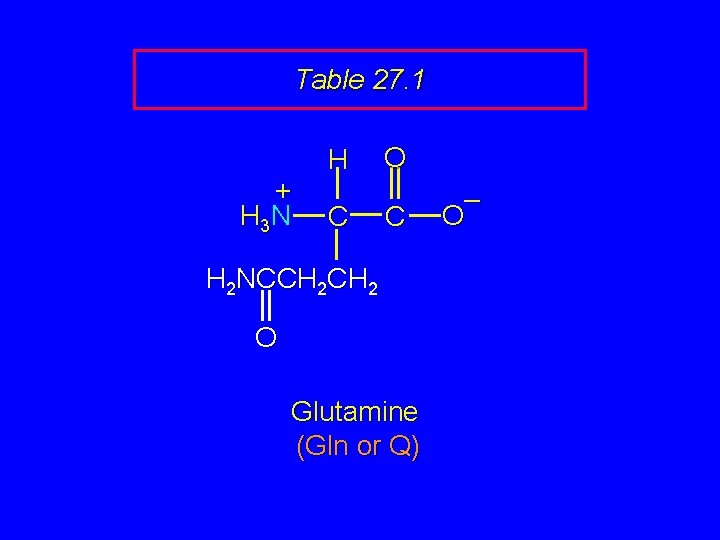
Table 27. 1 + H 3 N H C O C H 2 NCCH 2 O Glutamine (Gln or Q) – O

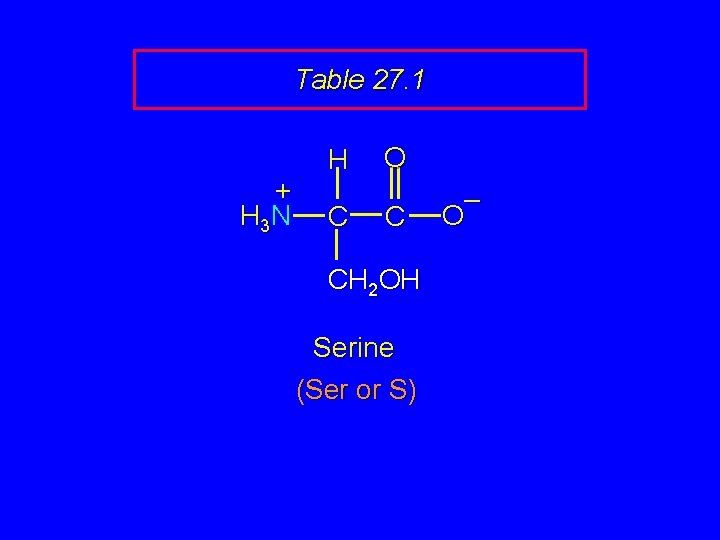
Table 27. 1 + H 3 N H C O C CH 2 OH Serine (Ser or S) – O

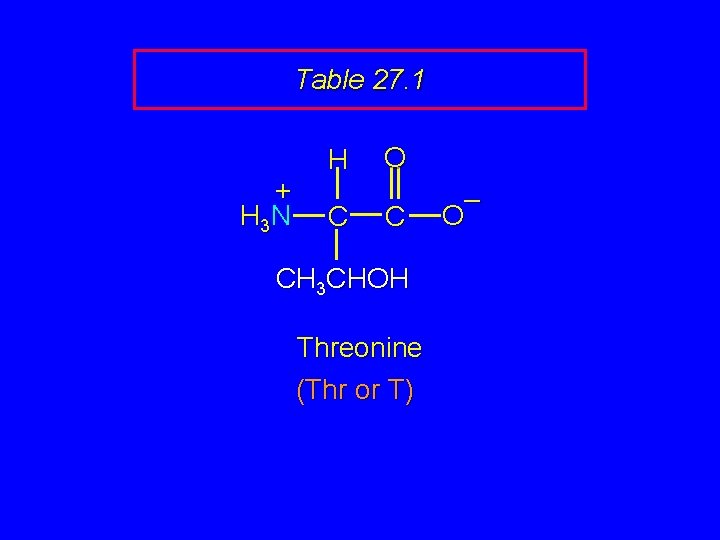
Table 27. 1 + H 3 N H C O C CH 3 CHOH Threonine (Thr or T) – O

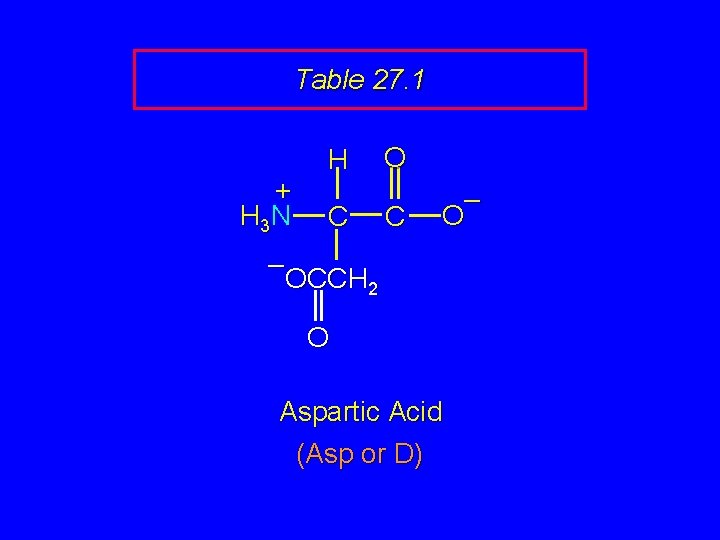
Table 27. 1 + H 3 N H C O C – OCCH 2 O Aspartic Acid (Asp or D)

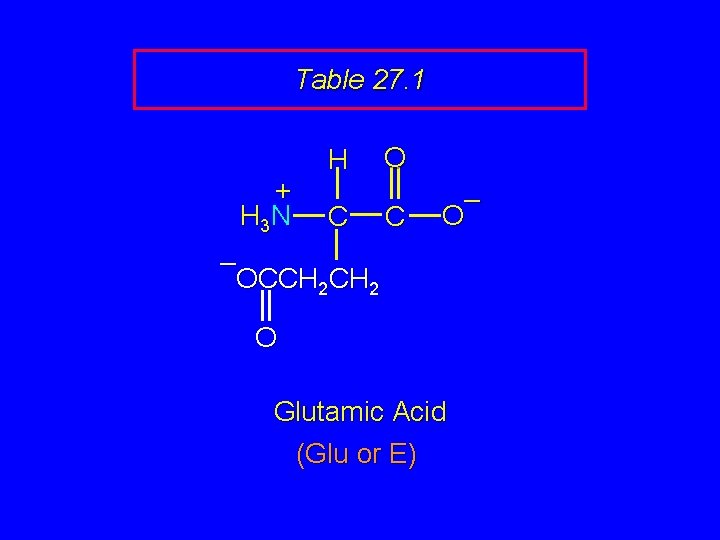
Table 27. 1 + H 3 N H C O C – OCCH 2 O Glutamic Acid (Glu or E)

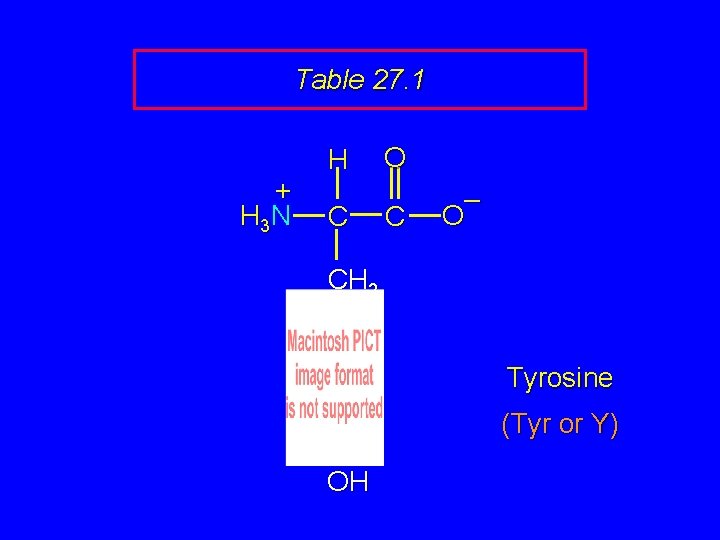
Table 27. 1 + H 3 N H C O C – O CH 2 Tyrosine (Tyr or Y) OH

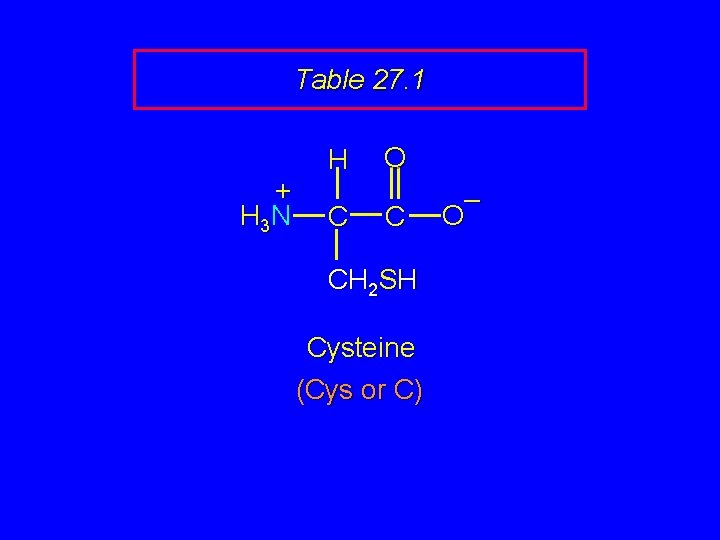
Table 27. 1 + H 3 N H C O C CH 2 SH Cysteine (Cys or C) – O

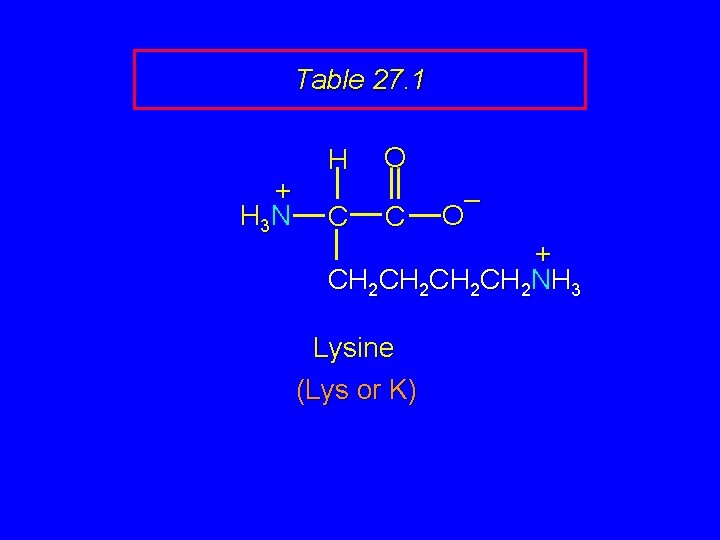
Table 27. 1 + H 3 N H C O C – O + CH 2 CH 2 NH 3 Lysine (Lys or K)

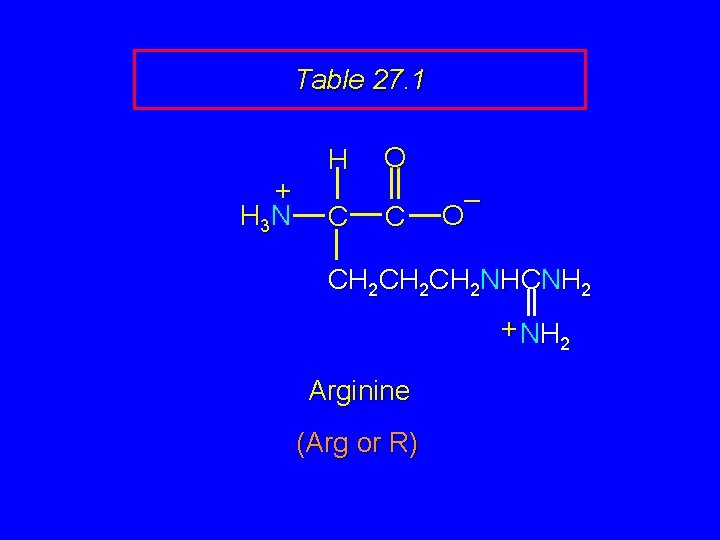
Table 27. 1 + H 3 N H C O C – O CH 2 CH 2 NHCNH 2 + NH 2 Arginine (Arg or R)

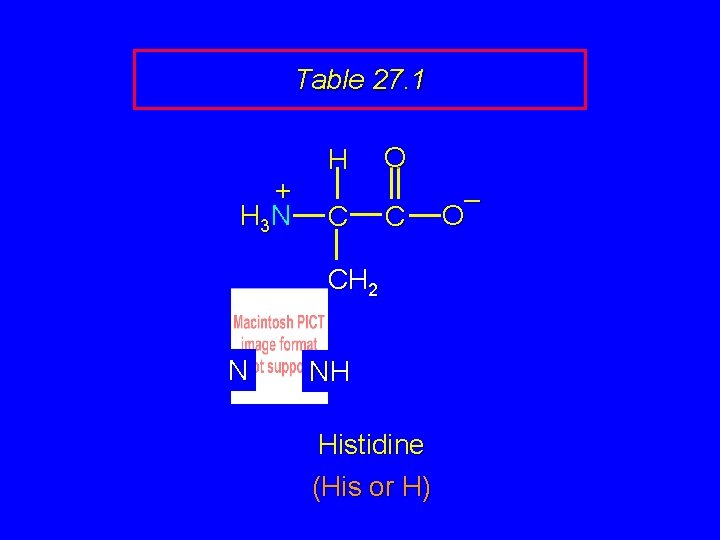
Table 27. 1 + H 3 N H C O C CH 2 N NH Histidine (His or H) – O

27. 2 Stereochemistry of Amino Acids

Configuration of a-Amino Acids Glycine is achiral. All of the other amino acids in proteins have the L-configuration at their a carbon. – CO 2 + H 3 N H R

27. 7 Peptides

Peptides are compounds in which an amide bond links the amino group of one a-amino acid and the carboxyl group of another. An amide bond of this type is often referred to as a peptide bond.

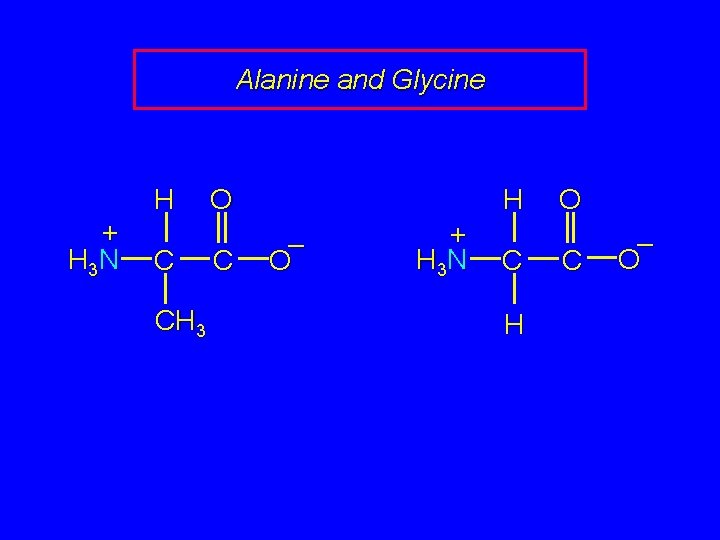
Alanine and Glycine H + H 3 N C CH 3 H O C – O + H 3 N C H O C – O

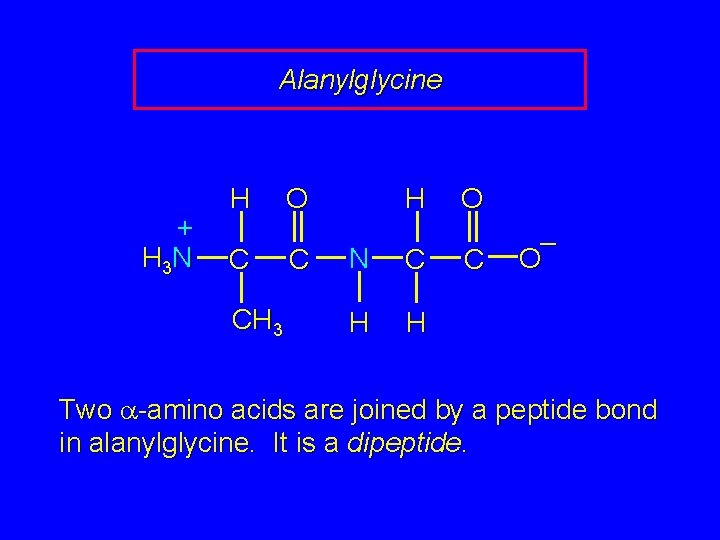
Alanylglycine + H 3 N H C CH 3 H O C N C H H O C – O Two a-amino acids are joined by a peptide bond in alanylglycine. It is a dipeptide.

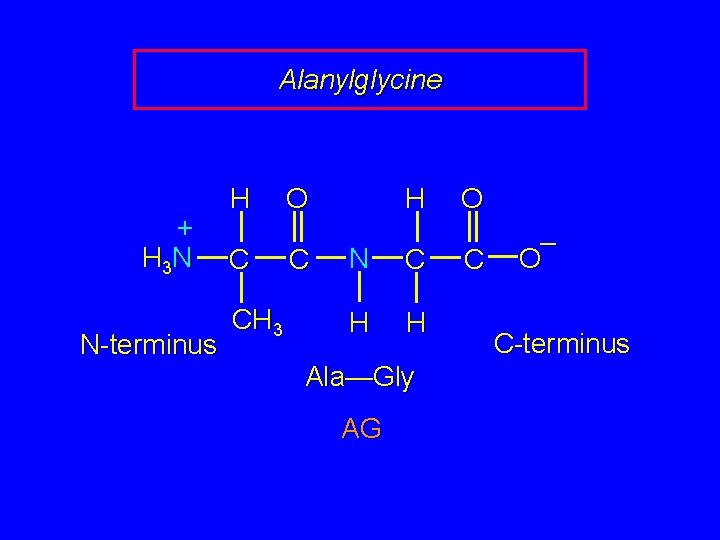
Alanylglycine + H 3 N N-terminus H C CH 3 H O C N C H H Ala—Gly AG O C – O C-terminus

Alanylglycine and glycylalanine are constitutional isomers + H 3 N H C C CH 3 + H 3 N H C H H O N C H H H O C N C H CH 3 O C – O Alanylglycine Ala—Gly AG – O Glycylalanine Gly—Ala GA O C

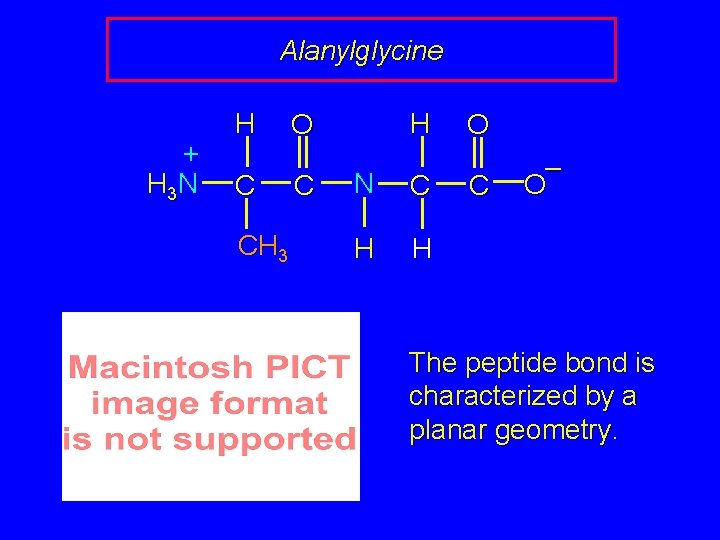
Alanylglycine + H 3 N H C CH 3 H O C N C H H O C – O The peptide bond is characterized by a planar geometry.

Higher Peptides are classified according to the number of amino acids linked together. dipeptides, tripeptides, tetrapeptides, etc. Leucine enkephalin is an example of a pentapeptide.

Leucine Enkephalin Tyr—Gly—Phe—Leu YGGFL

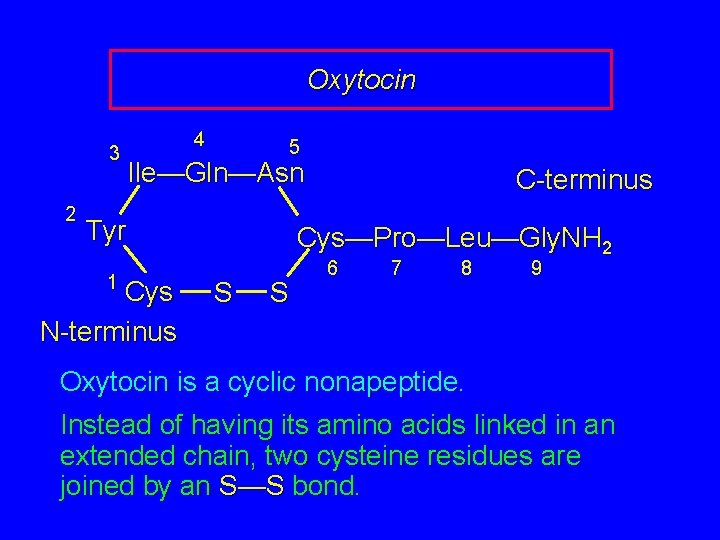
Oxytocin 3 2 4 5 Ile—Gln—Asn Tyr 1 Cys N-terminus Cys—Pro—Leu—Gly. NH 2 S S 6 7 8 9 Oxytocin is a cyclic nonapeptide. Instead of having its amino acids linked in an extended chain, two cysteine residues are joined by an S—S bond.

Oxytocin S—S bond An S—S bond between two cysteines is often referred to as a disulfide bridge.