Amino Acids Peptides and Proteins Polypeptides and Proteins








- Slides: 19

Amino Acids, Peptides and Proteins

Polypeptides and Proteins • In 1902, Emil Fischer proposed that proteins are long chains of amino acids joined by peptide bonds • Peptide bond: bond the special name given to the amide bond between the -carboxyl group of one amino acid and the -amino group of another

Peptides ▫ peptide: the name given to a short polymer of amino acids joined by peptide bonds; they are classified by the number of amino acids in the chain ▫ dipeptide: a molecule containing two amino acids joined by a peptide bond ▫ tripeptide: tripeptide a molecule containing three amino acids joined by peptide bonds ▫ polypeptide: polypeptide a macromolecule containing many amino acids joined by peptide bonds ▫ protein: protein a biological macromolecule of molecular weight 5000 g/mol or greater, consisting of one or more polypeptide chains

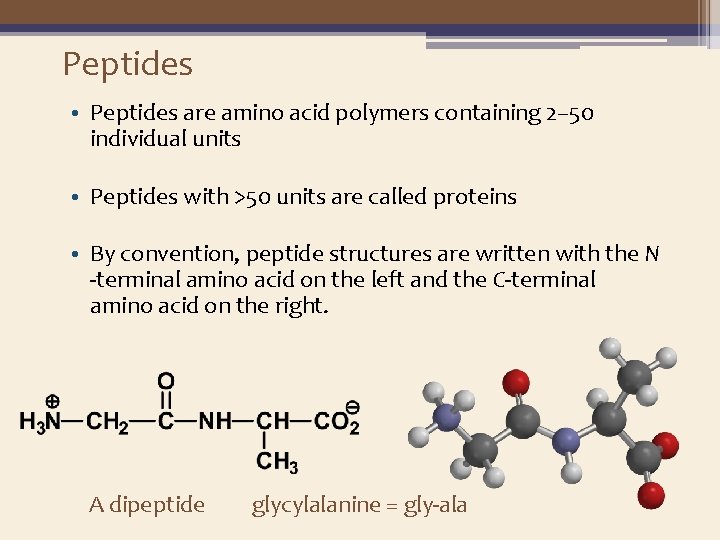
Peptides • Peptides are amino acid polymers containing 2– 50 individual units • Peptides with >50 units are called proteins • By convention, peptide structures are written with the N -terminal amino acid on the left and the C-terminal amino acid on the right. A dipeptide glycylalanine = gly-ala

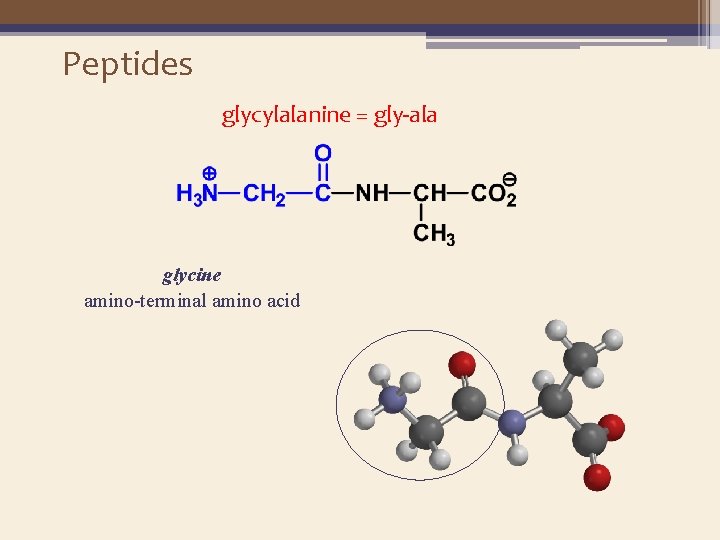
Peptides glycylalanine = gly-ala glycine amino-terminal amino acid

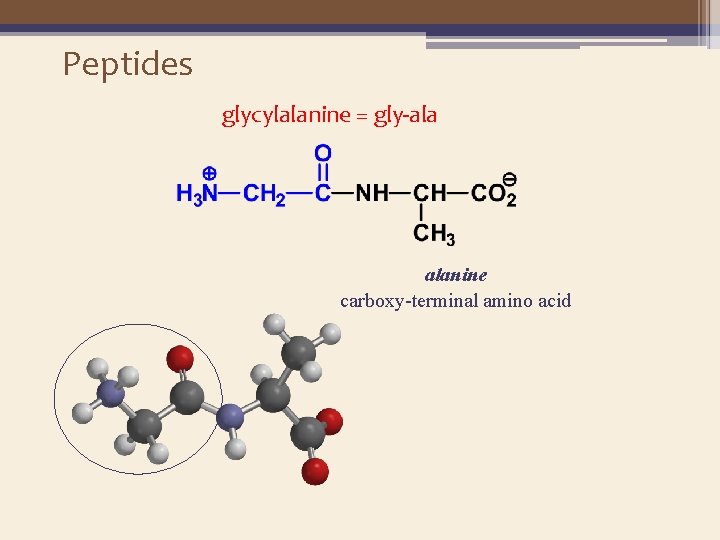
Peptides glycylalanine = gly-ala alanine carboxy-terminal amino acid

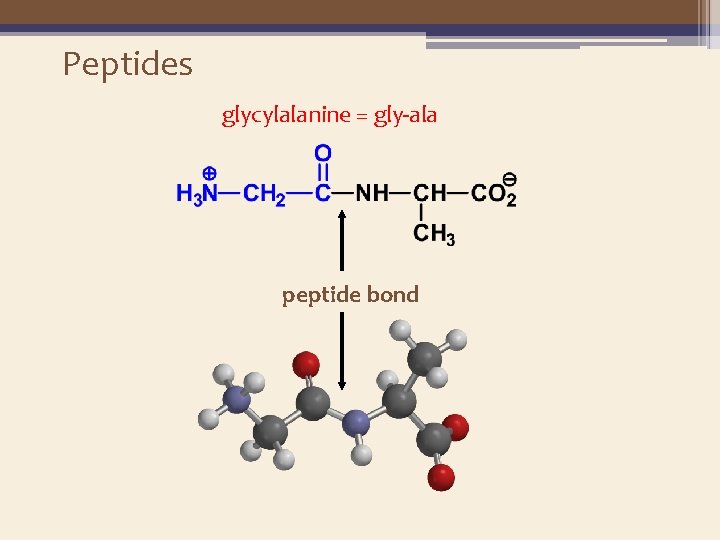
Peptides glycylalanine = gly-ala peptide bond

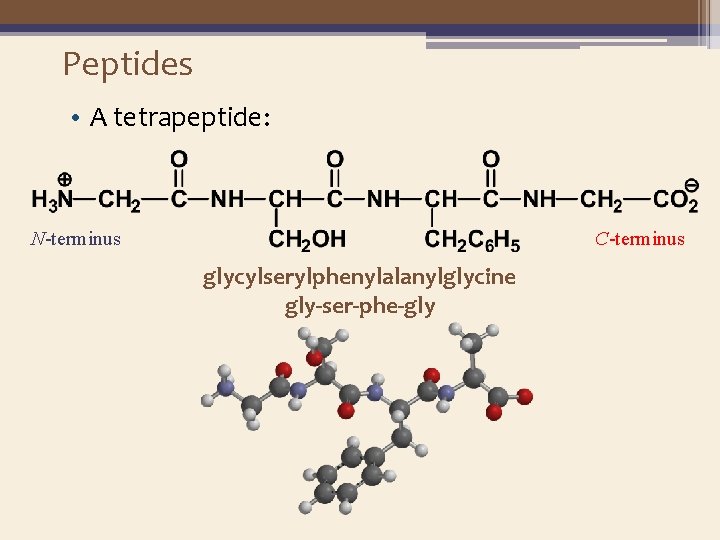
Peptides • A tetrapeptide: N-terminus C-terminus glycylserylphenylalanylglycine gly-ser-phe-gly

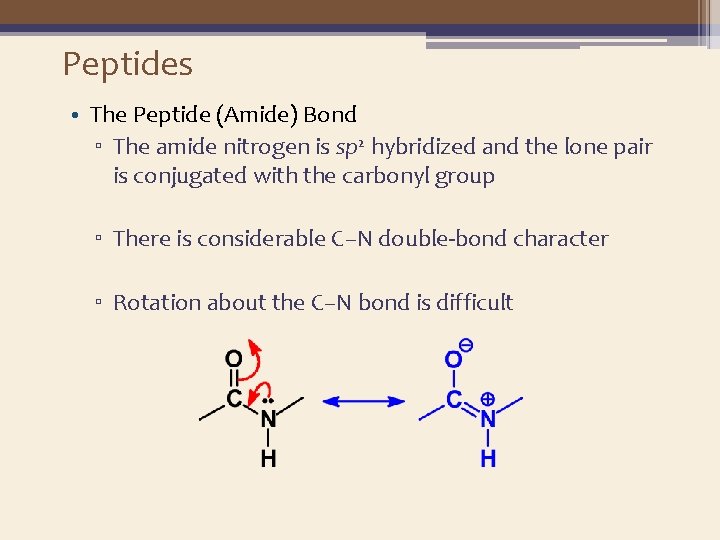
Peptides • The Peptide (Amide) Bond ▫ The amide nitrogen is sp 2 hybridized and the lone pair is conjugated with the carbonyl group ▫ There is considerable C–N double-bond character ▫ Rotation about the C–N bond is difficult

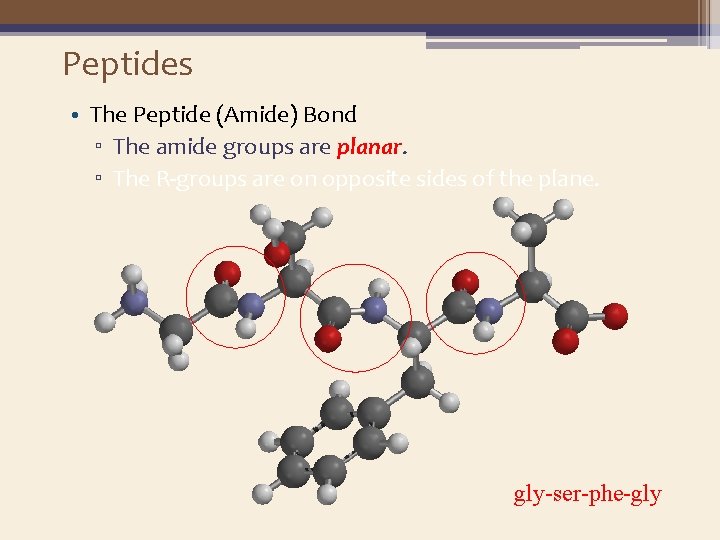
Peptides • The Peptide (Amide) Bond ▫ The amide groups are planar. ▫ The R-groups are on opposite sides of the plane. gly-ser-phe-gly

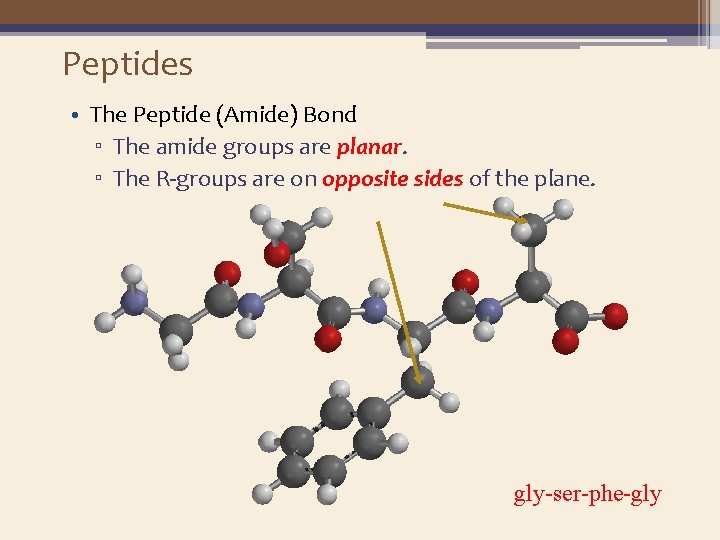
Peptides • The Peptide (Amide) Bond ▫ The amide groups are planar. ▫ The R-groups are on opposite sides of the plane. gly-ser-phe-gly

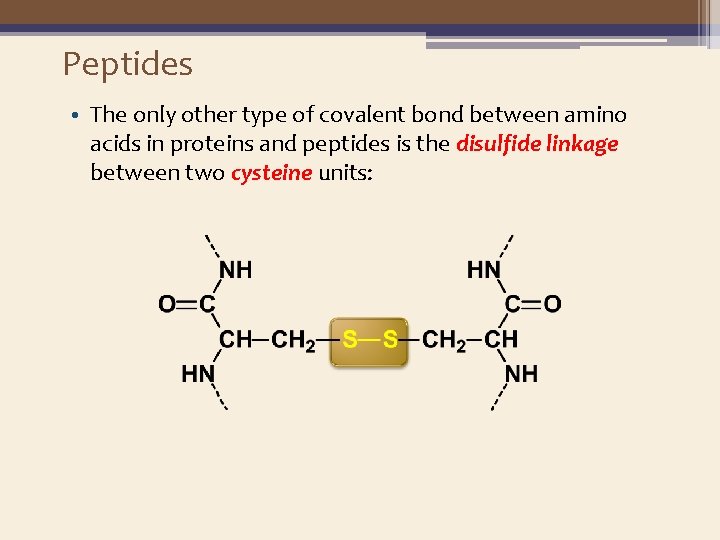
Peptides • The only other type of covalent bond between amino acids in proteins and peptides is the disulfide linkage between two cysteine units:

Peptides • Note: ▫ Thiols are readily oxidized to disulfides. ▫ Disulfides are readily reduced to thiols.

Peptides • Disulfide links serve to connect polypeptide chains:

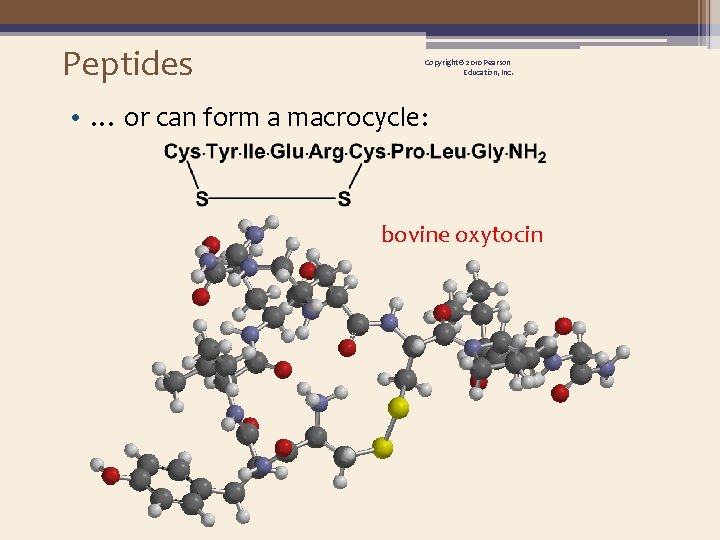
Peptides Copyright © 2010 Pearson Education, Inc. • … or can form a macrocycle: bovine oxytocin

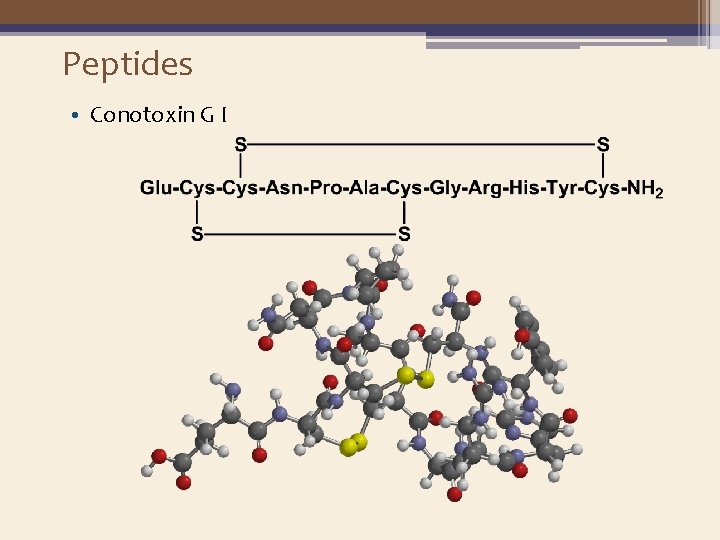
Peptides • Conotoxin G I ▫ A peptide neurotoxin isolated from the venom of the fishhunting cone snail Conus geographicus. ▫ It is a paralytic toxin that blocks nicotinic cholinoreceptors.

Peptides • Conotoxin G I

Structure Determination of Peptides • Amino Acid Analysis. Find out which amino acids and how many make up the peptide • Terminal Residue Analysis. Find out what’s on the ends N-Terminal Analysis Edman degradation C-Terminal Analysis Carboxypeptidase

Structure Determination of Peptides • Partial Hydrolysis (enzymatic) ▫ Hydrolyze the peptide into smaller fragments. Trypsin - Cleaves at lys and arg Chymotrypsin - Cleaves at phe, tyr, and trp Pepsin - Cleaves at phe, tyr, trp, leu, asp, glu Cyanogen bromide (not enzymatic) - Cleaves at met ▫ Determine the sequence of the fragments. Successive Edman degradations.