Protein metabolism BY Dr NAGLAA IBRAHIM AZAB Lecturer

Protein metabolism BY Dr. NAGLAA IBRAHIM AZAB Lecturer Of Medical Biochemistry &Molecular Biology BENHA FACULTY OF MEDICINE

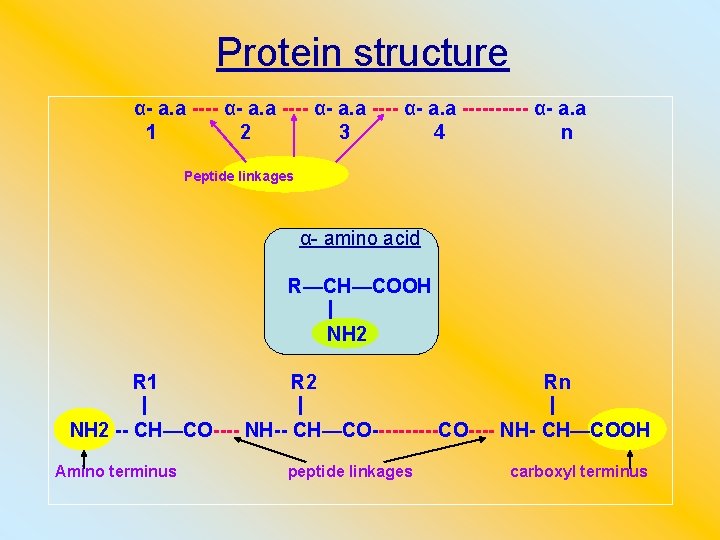
Protein structure α- a. a ---------- α- a. a 1 2 3 4 n Peptide linkages α- amino acid R—CH—COOH NH 2 R 1 R 2 Rn NH 2 -- CH—CO---- NH-- CH—CO-----CO---- NH- CH—COOH Amino terminus peptide linkages carboxyl terminus

Classification of amino acids Structural classification : according to the chemical structure of the side chain ssification • • (essensial & non essential a. as) Metabolic classification : according to the fate of amino acids inside the body (glucogenic, ketogenic and mixed a. as) •
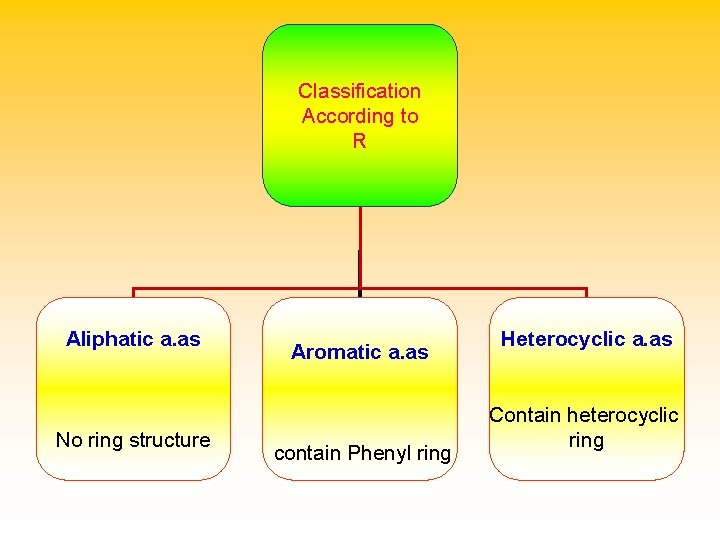
Classification According to R Aliphatic a. as No ring structure Aromatic a. as contain Phenyl ring Heterocyclic a. as Contain heterocyclic ring
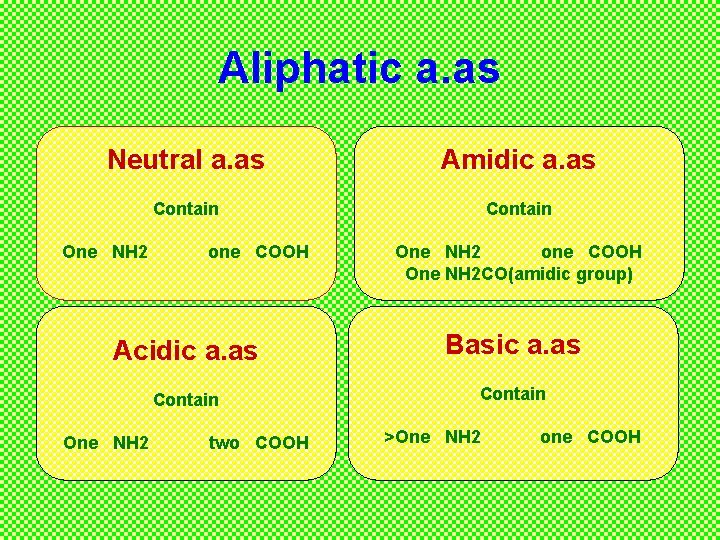
Aliphatic a. as Neutral a. as Amidic a. as Contain One NH 2 one COOH One NH 2 CO(amidic group) Acidic a. as Basic a. as Contain One NH 2 two COOH >One NH 2 one COOH
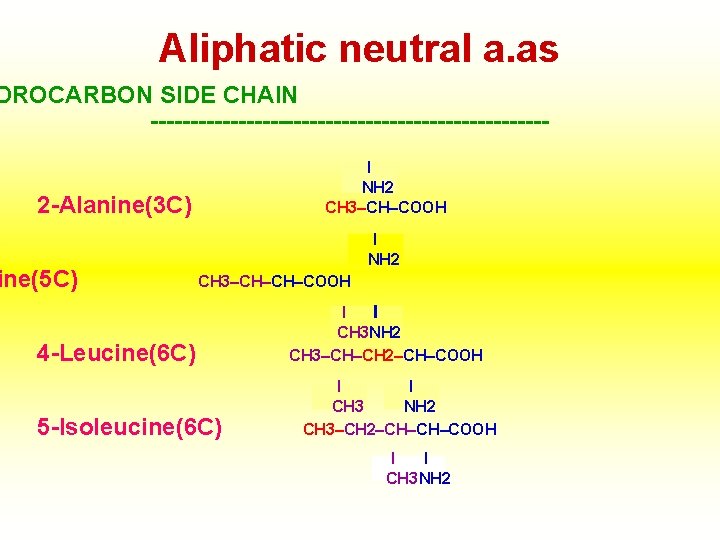
Aliphatic neutral a. as DROCARBON SIDE CHAIN -------------------------I NH 2 CH 3–CH–COOH 2 -Alanine(3 C) ine(5 C) I NH 2 CH 3–CH–CH–COOH 4 -Leucine(6 C) 5 -Isoleucine(6 C) I I CH 3 NH 2 CH 3–CH–CH 2–CH–COOH I I CH 3 NH 2 CH 3–CH 2–CH–CH–COOH I I CH 3 NH 2
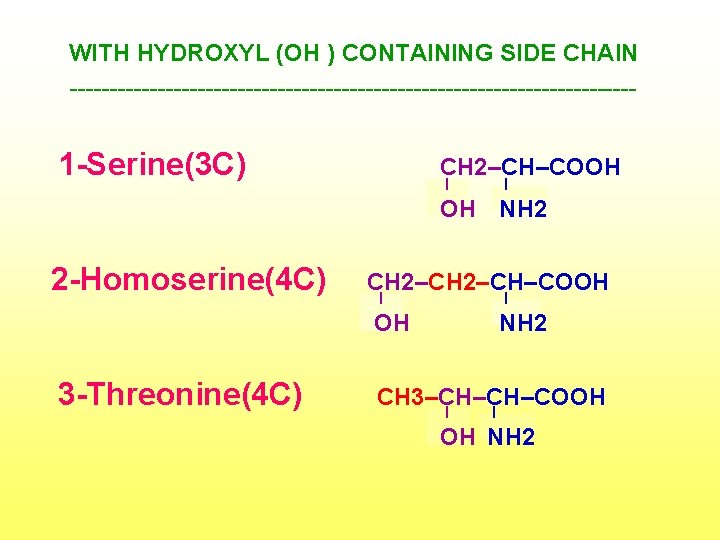
WITH HYDROXYL (OH ) CONTAINING SIDE CHAIN ------------------------------------ 1 -Serine(3 C) CH 2–CH–COOH I I OH NH 2 2 -Homoserine(4 C) 3 -Threonine(4 C) CH 2–CH–COOH I I OH NH 2 CH 3–CH–CH–COOH I I OH NH 2
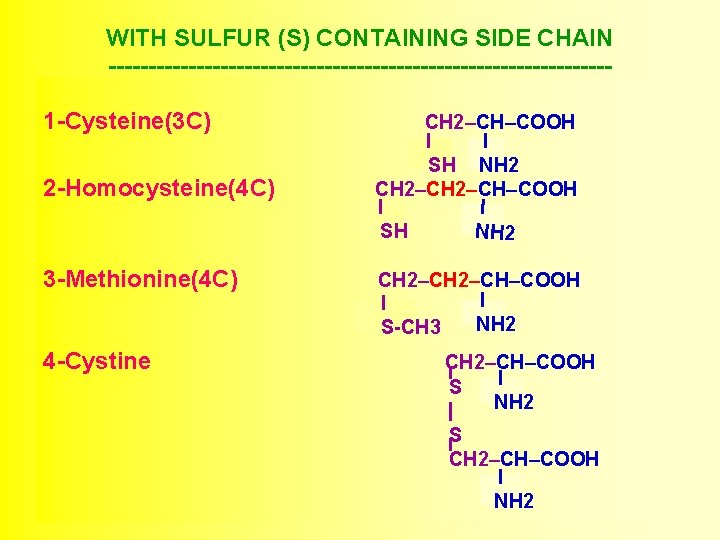
WITH SULFUR (S) CONTAINING SIDE CHAIN -------------------------------1 -Cysteine(3 C) 2 -Homocysteine(4 C) 3 -Methionine(4 C) 4 -Cystine CH 2–CH–COOH I I SH NH 2 CH 2–CH–COOH I I NH 2 SH CH 2–CH–COOH I I NH 2 S-CH 3 CH 2–CH–COOH I S NH 2 S CH 2–CH–COOH I NH 2
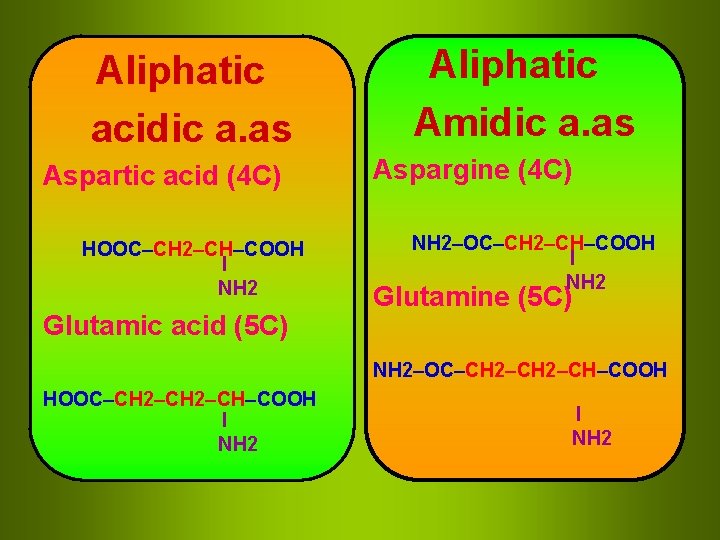
Aliphatic acidic a. as Aspartic acid (4 C) HOOC–CH 2–CH–COOH I NH 2 Glutamic acid (5 C) Aliphatic Amidic a. as Aspargine (4 C) NH 2–OC–CH 2–CH–COOH I NH 2 Glutamine (5 C) NH 2–OC–CH 2–CH–COOH HOOC–CH 2–CH–COOH I NH 2
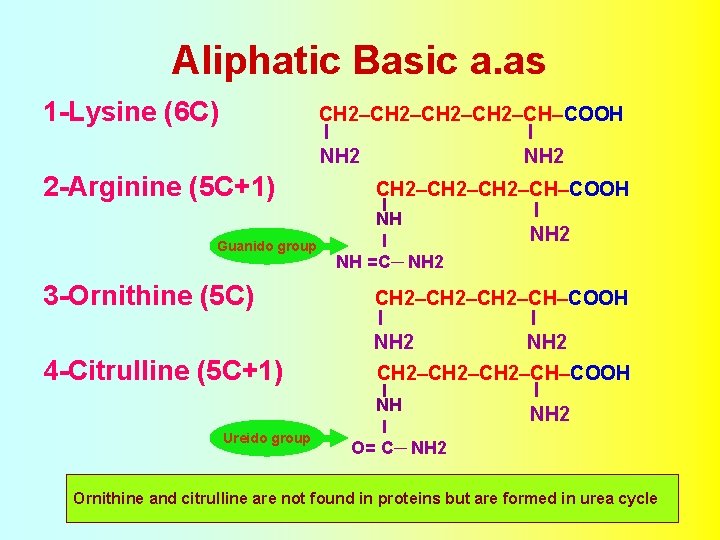
Aliphatic Basic a. as 1 -Lysine (6 C) CH 2–CH–COOH I I NH 2 2 -Arginine (5 C+1) Guanido group CH 2–CH–COOH I NH NH 2 I NH =C─ NH 2 3 -Ornithine (5 C) CH 2–CH–COOH I I NH 2 4 -Citrulline (5 C+1) CH 2–CH–COOH I NH NH 2 Ureido group I O= C─ NH 2 Ornithine and citrulline are not found in proteins but are formed in urea cycle
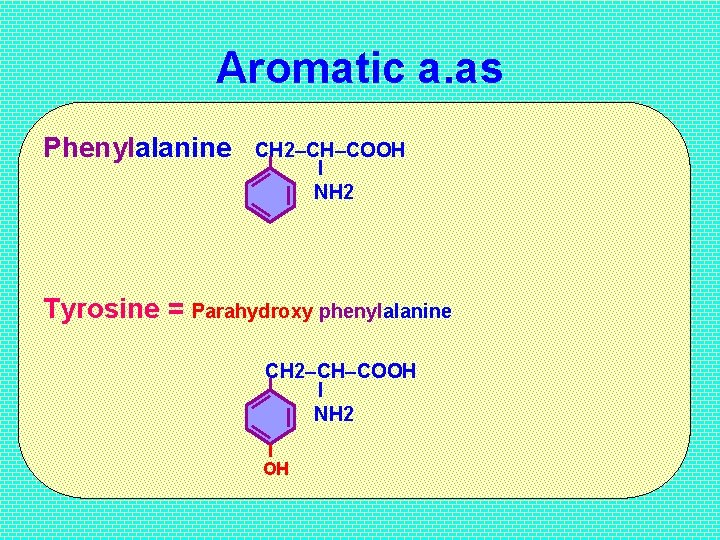
Aromatic a. as Phenylalanine CH 2–CH–COOH I NH 2 Tyrosine = Parahydroxy phenylalanine CH 2–CH–COOH I NH 2 OH
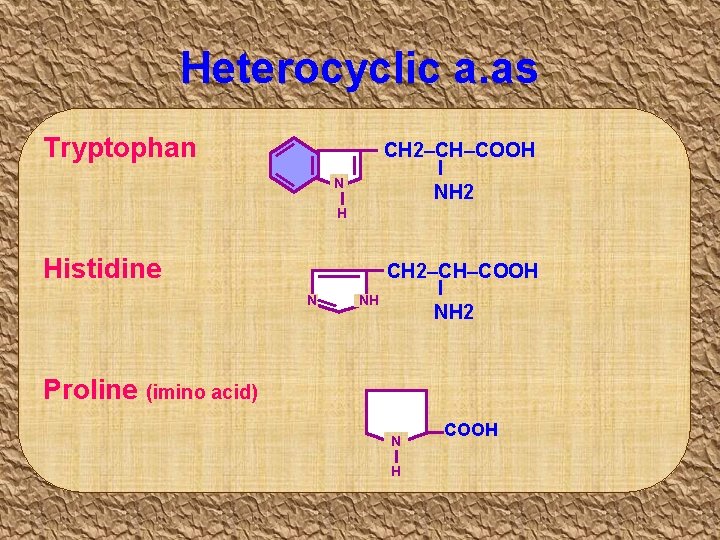
Heterocyclic a. as Tryptophan CH 2–CH–COOH I NH 2 N H Histidine N NH CH 2–CH–COOH I NH 2 Proline (imino acid) N H COOH

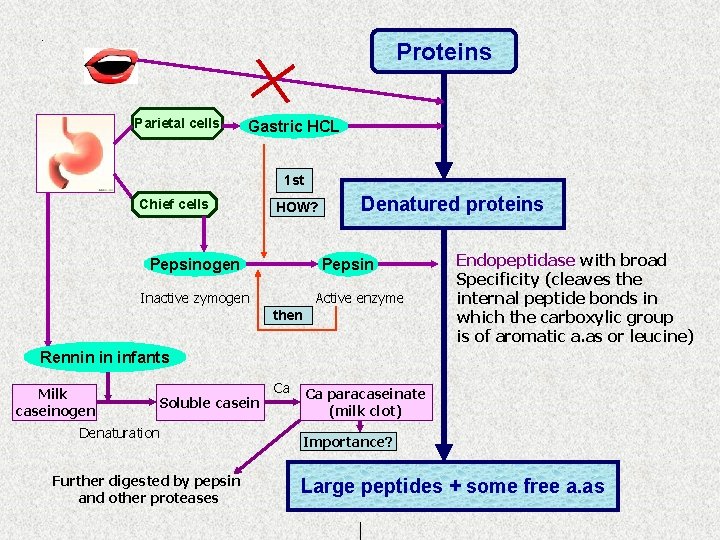
. Proteins Parietal cells Gastric HCL 1 st Chief cells HOW? Pepsinogen Denatured proteins Pepsin Inactive zymogen Active enzyme then Endopeptidase with broad Specificity (cleaves the internal peptide bonds in which the carboxylic group is of aromatic a. as or leucine) Rennin in infants Milk caseinogen Soluble casein Denaturation Further digested by pepsin and other proteases Ca Ca paracaseinate (milk clot) Importance? Large peptides + some free a. as
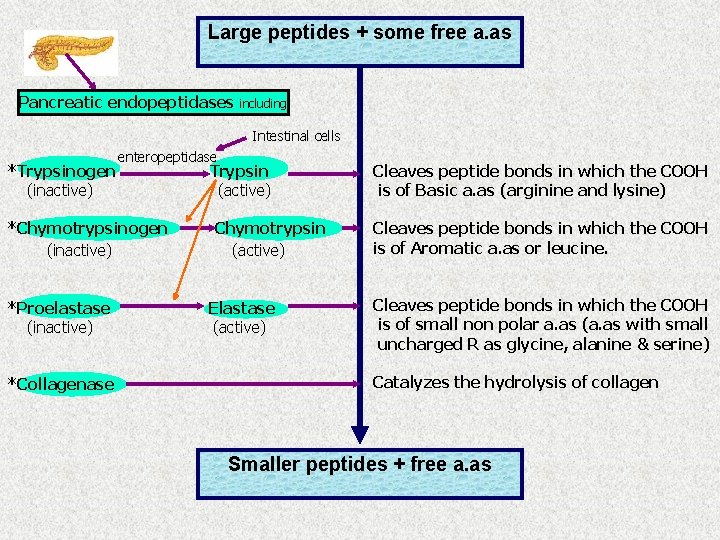
Large peptides + some free a. as Pancreatic endopeptidases including Intestinal cells *Trypsinogen (inactive) enteropeptidase *Chymotrypsinogen (inactive) *Proelastase (inactive) *Collagenase Trypsin (active) Chymotrypsin (active) Elastase (active) Cleaves peptide bonds in which the COOH is of Basic a. as (arginine and lysine) Cleaves peptide bonds in which the COOH is of Aromatic a. as or leucine. Cleaves peptide bonds in which the COOH is of small non polar a. as (a. as with small uncharged R as glycine, alanine & serine) Catalyzes the hydrolysis of collagen Smaller peptides + free a. as
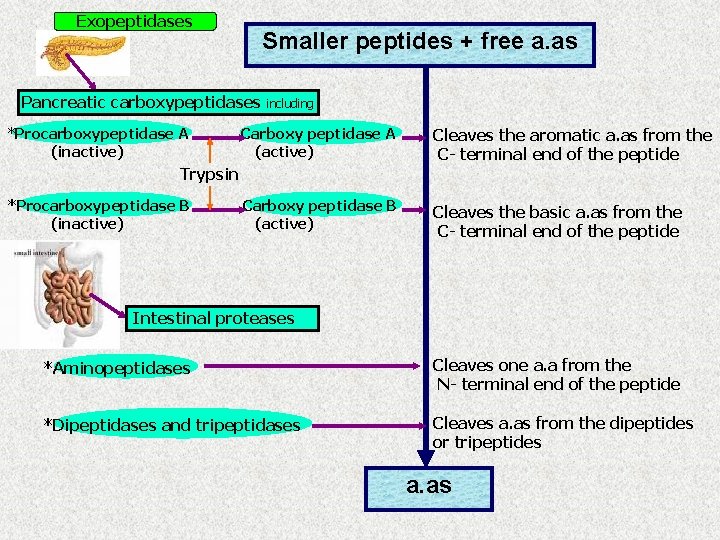
Exopeptidases Smaller peptides + free a. as Pancreatic carboxypeptidases *Procarboxypeptidase A (inactive) including Carboxy peptidase A (active) Cleaves the aromatic a. as from the C- terminal end of the peptide Carboxy peptidase B (active) Cleaves the basic a. as from the C- terminal end of the peptide Trypsin *Procarboxypeptidase B (inactive) Intestinal proteases *Aminopeptidases Cleaves one a. a from the N- terminal end of the peptide *Dipeptidases and tripeptidases Cleaves a. as from the dipeptides or tripeptides a. as

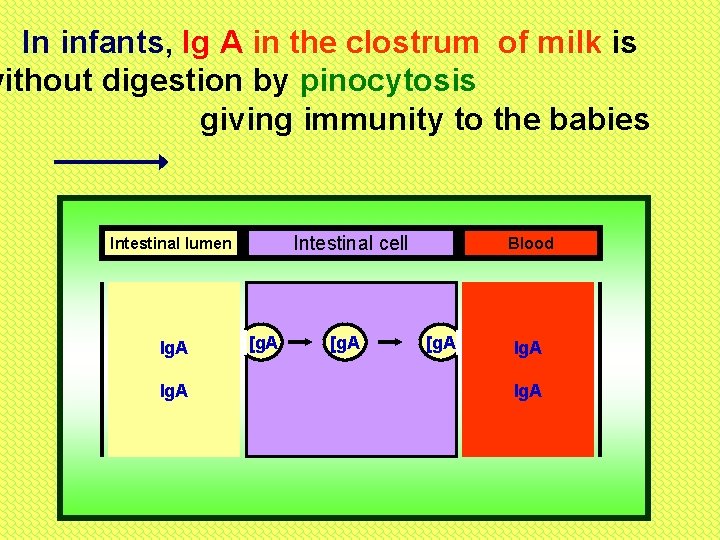
In infants, Ig A in the clostrum of milk is without digestion by pinocytosis giving immunity to the babies Intestinal cell Intestinal lumen Ig. A [g. A Blood [g. A Ig. A

Aa. s resulting from protein digestion are absorbed from the small intestine by: ■ passive transport mechanism (For D-aas). ■ Active transport mechanism (For L-aas and dipeptides): ► Carrier protein transport system ( sodium – amino acid carrier system ). ► Glutathione transport system (γ-glutamyl cycle)
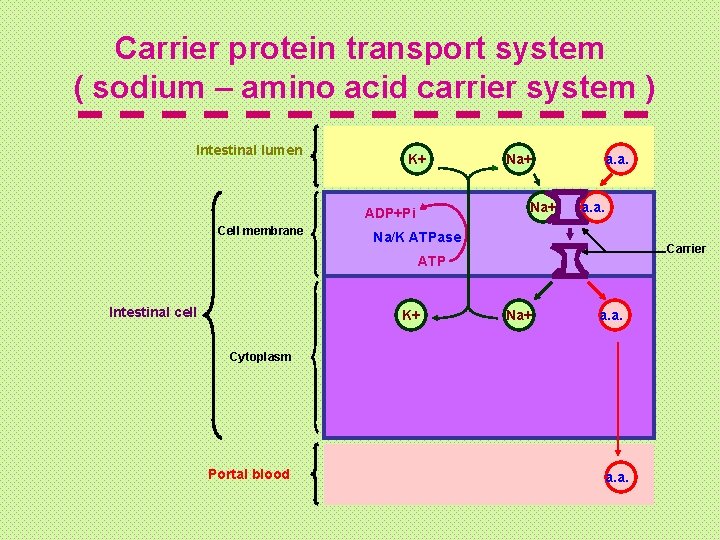
Carrier protein transport system ( sodium – amino acid carrier system ) Intestinal lumen K+ Na+ ADP+Pi Cell membrane Na+ a. a. Na/K ATPase Carrier ATP Intestinal cell K+ Na+ a. a. Cytoplasm Portal blood a. a.

*This system transport the a. a. against its conc. gradiant using energy derived from Na/K+ pump. *Here a. as are absorbed by specific carrier protein in the cell membrane of the small intestinal cells. This carrier protein has one site for the a. a. and another site for the Na+. It transports them from the intestinal lumen across the cell membrane to the cytoplasm. Then the a. a. passes to the blood down its conc. Gradient, while the Na+ is pumped out from the cell to the intestinal lumen by Na/K+ pump utilizing ATP as a source of energy derived from Na/K+ pump.
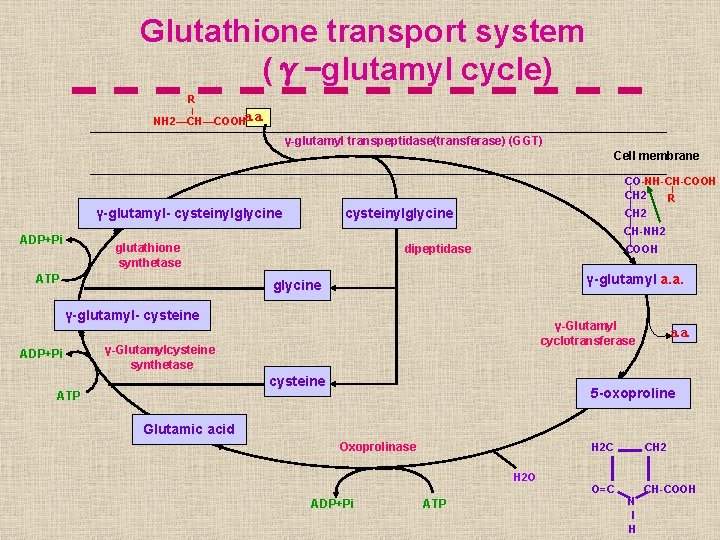
Glutathione transport system (γ-glutamyl cycle) R NH 2—CH—COOHa. a. γ-glutamyl transpeptidase(transferase) (GGT) Cell membrane γ-glutamyl- cysteinylglycine CO-NH-CH-COOH CH 2 R CH 2 cysteinylglycine CH-NH 2 ADP+Pi glutathione synthetase ATP dipeptidase COOH γ-glutamyl a. a. glycine γ-glutamyl- cysteine ADP+Pi γ-Glutamyl cyclotransferase γ-Glutamylcysteine synthetase cysteine a. a. 5 -oxoproline ATP Glutamic acid Oxoprolinase H 2 C H 2 O ADP+Pi ATP O=C CH 2 N I H CH-COOH

►This transport system is for: the transport of a. a. s from the extracellular space to the cytoplasm in the intestine, kidney, brain & liver(bile ductule cells) So it is not important only for the uptake of a. as from the intestinal lumen (a. a. absorption). ► 3 ATP molecules are utilized for the transfer of one a. a. ►Clinical notes: ■The blood conc. of GGT enzyme is increased in cholestasis & chronic alcholism ( so used as a liver function test). ■Oxoprolinuria: Inherited deficiency of glutathione synthetase enzyme , leading to increase levels of 5 oxoproline in blood &urine acidosis & neurological damage

gic reactions to food Quiz
Fate of absorbed a. as Enter in the formation of a. a. pool

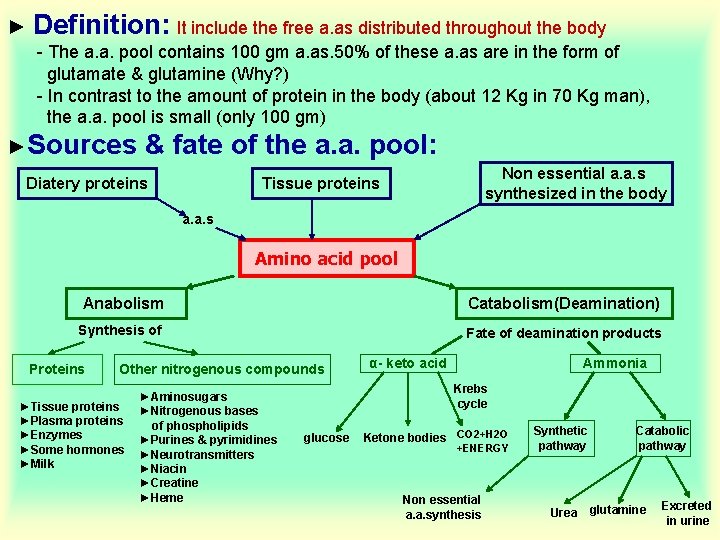
► Definition: It include the free a. as distributed throughout the body - The a. a. pool contains 100 gm a. as. 50% of these a. as are in the form of glutamate & glutamine (Why? ) - In contrast to the amount of protein in the body (about 12 Kg in 70 Kg man), the a. a. pool is small (only 100 gm) ►Sources & fate of the a. a. pool: Diatery proteins Non essential a. a. s synthesized in the body Tissue proteins a. a. s Amino acid pool Anabolism Catabolism(Deamination) Synthesis of Fate of deamination products Proteins Other nitrogenous compounds ►Tissue proteins ►Plasma proteins ►Enzymes ►Some hormones ►Milk ►Aminosugars ►Nitrogenous bases of phospholipids ►Purines & pyrimidines ►Neurotransmitters ►Niacin ►Creatine ►Heme α- keto acid Ammonia Krebs cycle glucose Ketone bodies CO 2+H 2 O +ENERGY Non essential a. a. synthesis Synthetic pathway Urea Catabolic pathway glutamine Excreted in urine


Catabolism of the a. a. s occurs by Deamination (removal of the amino group) either by General methods of deamination: ►Oxidative deamination ►Transdeamination Or Specific methods of deamination: For certain a. a. s

Oxidative deamination ► Definition: It is the oxidation (removal of hydrogen) deamination (removal of the amino group which is liberated as free ammonia) giving α- ketoacid and ammonia (reversible reactions) R— CH — COOH l NH 2
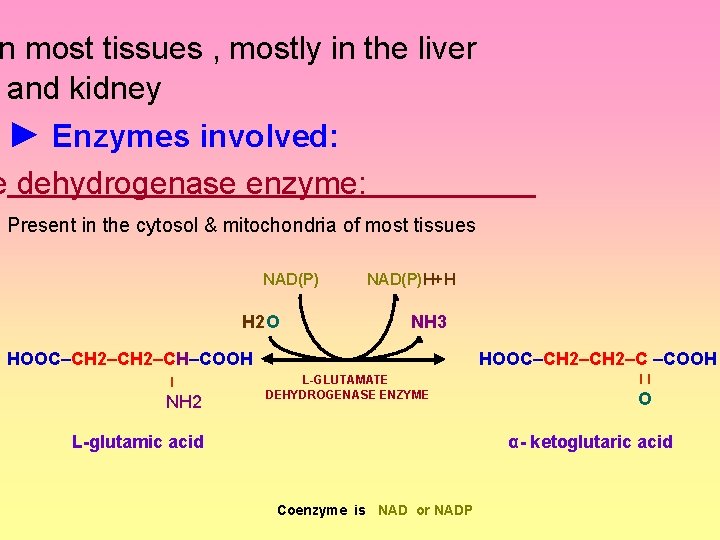
n most tissues , mostly in the liver and kidney ► Enzymes involved: e dehydrogenase enzyme: Present in the cytosol & mitochondria of most tissues NAD(P)H+H H 2 O NH 3 HOOC–CH 2–CH–COOH ◄ ► l NH 2 L-GLUTAMATE DEHYDROGENASE ENZYME L-glutamic acid HOOC–CH 2–C –COOH ll O α- ketoglutaric acid Coenzyme is NAD or NADP

Regulation: The direction of the reaction depends on: 1 - Availability of the substrates: --Relative conc. Of (α-ketoglutarate &NH 3) and (glutamate). --Ratio of NADP : NADPH+H 2 - Allosteric regulation: --Activators : ADP or GDP. -- Inhibitors : ATP , GTP & NADH

QUIZ ---After a protein meal , in which direction the reaction proceeds? Why? ---After a CHO meal , in which direction the reaction proceeds? Why?
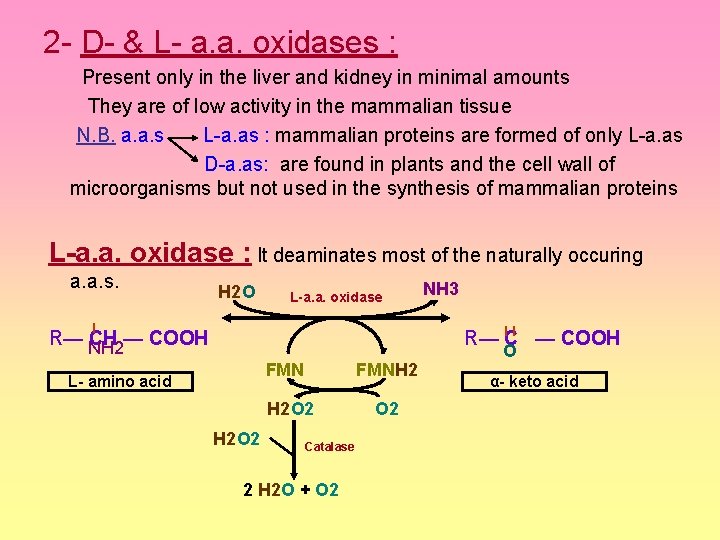
2 - D- & L- a. a. oxidases : Present only in the liver and kidney in minimal amounts They are of low activity in the mammalian tissue N. B. a. a. s L-a. as : mammalian proteins are formed of only L-a. as D-a. as: are found in plants and the cell wall of microorganisms but not used in the synthesis of mammalian proteins L-a. a. oxidase : It deaminates most of the naturally occuring a. a. s. H 2 O L-a. a. oxidase l NH 3 R— l. Cl — COOH R— NH 2 CH — COOH FMN L- amino acid FMNH 2 H 2 O 2 Catalase 2 H 2 O + O 2 O α- keto acid
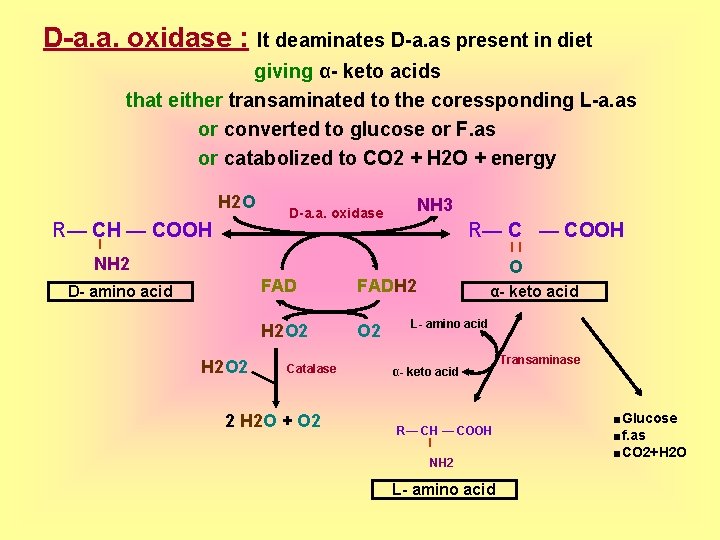
D-a. a. oxidase : It deaminates D-a. as present in diet giving α- keto acids that either transaminated to the coressponding L-a. as or converted to glucose or F. as or catabolized to CO 2 + H 2 O + energy H 2 O R— CH — COOH D-a. a. oxidase NH 3 R— C — COOH l ll NH 2 D- amino acid H 2 O 2 FADH 2 H 2 O 2 Catalase 2 H 2 O + O 2 O α- keto acid L- amino acid α- keto acid R— CH — COOH l NH 2 L- amino acid Transaminase ■Glucose ■f. as ■CO 2+H 2 O

►Importance of oxidative deamination L-glutamate dehydrogenase enzyme is the only a. a. that undergoes oxidative deamination in the mammalian tissue. Oxidative deamination by L- glutamate dehydrogenase is an essential component of transdeamination. So it is important in deamination of most a. as.
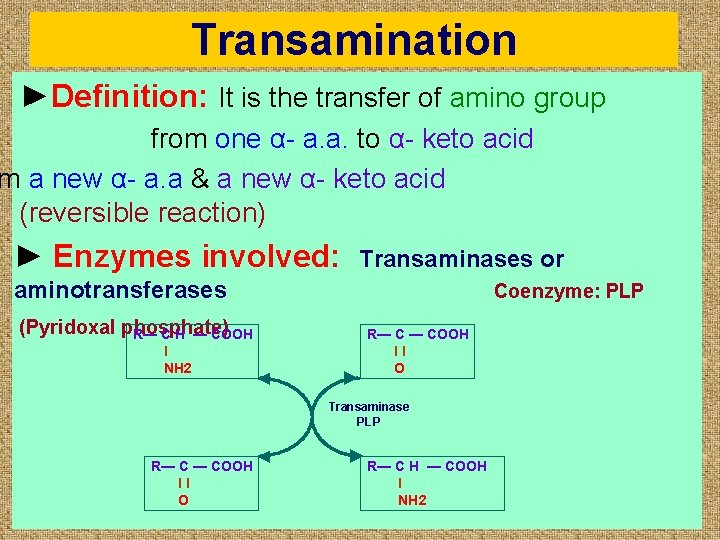
Transamination ►Definition: It is the transfer of amino group from one α- a. a. to α- keto acid m a new α- a. a & a new α- keto acid (reversible reaction) ► Enzymes involved: Transaminases or aminotransferases Coenzyme: PLP (Pyridoxal phosphate) R— C H — COOH l NH 2 ◄ ► R— C — COOH ll O Transaminase PLP R— C — COOH ll O ◄ ►R— C H l NH 2 — COOH

►Site: In the cytosol or both the cytosol & the mitochondria of most cells especially the liver ► All a. as except threonine, lysine, proline&hydroxy proline may undergo transamination
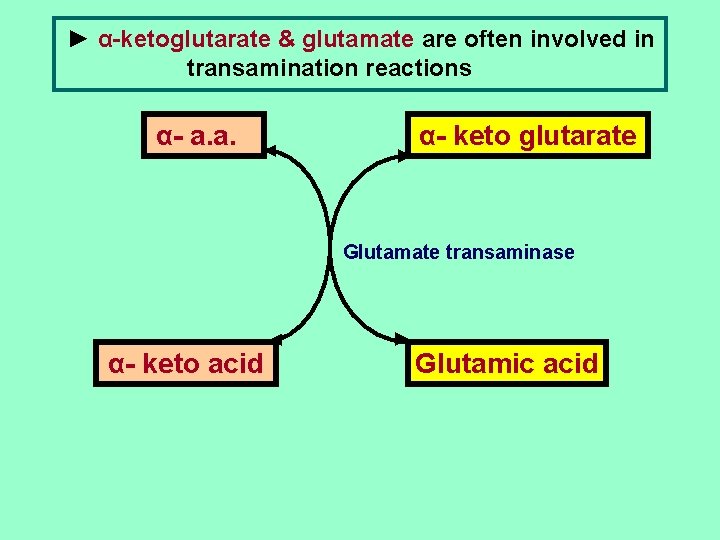
► α-ketoglutarate & glutamate are often involved in transamination reactions α- a. a. ◄ ► α- keto glutarate Glutamate transaminase α- keto acid ◄ ► Glutamic acid
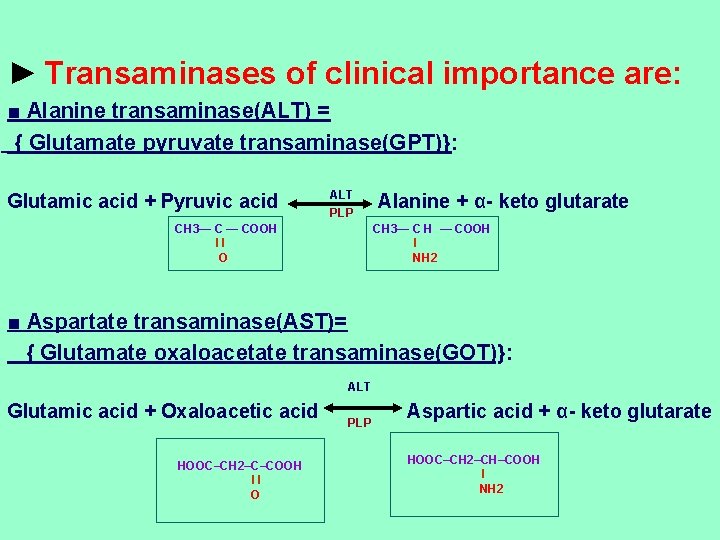
► Transaminases of clinical importance are: ■ Alanine transaminase(ALT) = { Glutamate pyruvate transaminase(GPT)}: Glutamic acid + Pyruvic acid ALT PLP CH 3— C — COOH ll O Alanine + α- keto glutarate CH 3— C H — COOH l NH 2 ■ Aspartate transaminase(AST)= { Glutamate oxaloacetate transaminase(GOT)}: ALT Glutamic acid + Oxaloacetic acid HOOC–CH 2–C–COOH ll O PLP Aspartic acid + α- keto glutarate HOOC–CH 2–CH–COOH l NH 2

►Value of transamination: ■Function: 1 - Degradation of a. as to form α- keto acids. 2 - Synthesis of non essential a. as from CHO. ■Diagnostic value: Transaminases are normally intracellular enzymes. hey are elevated in the blood when damage to the cells producing these enzymes occurs. rease level of both ALT & AST indicates possible damage to the liver cells. * Increase level of AST alone suggest damage to heart muscle , skeletal muscle or kidney.
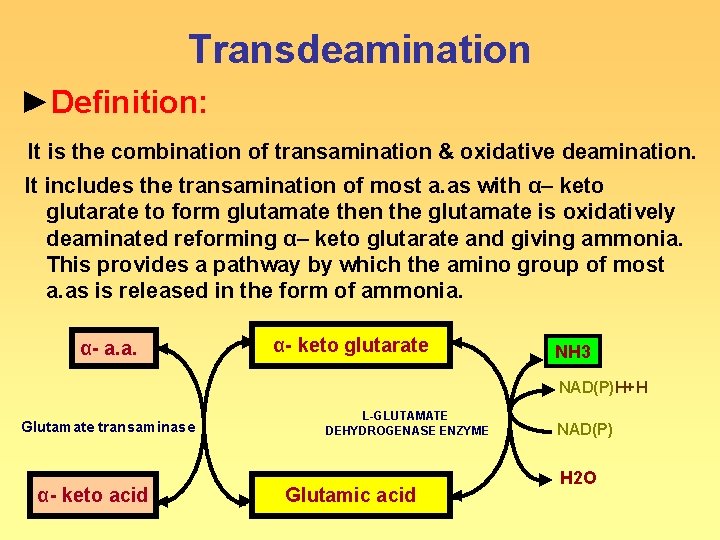
Transdeamination ►Definition: It is the combination of transamination & oxidative deamination. It includes the transamination of most a. as with α– keto glutarate to form glutamate then the glutamate is oxidatively deaminated reforming α– keto glutarate and giving ammonia. This provides a pathway by which the amino group of most a. as is released in the form of ammonia. α- a. a. ◄ ► α- keto glutarate ◄ L-GLUTAMATE DEHYDROGENASE ENZYME Glutamate transaminase α- keto acid ◄ ► Glutamic acid ◄ ► NH 3 ►NAD(P)H+H NAD(P) ► H 2 O

On biochemical basis explain ■Removal of ammonia from a. as can not be explained alone by transamination nor by oxidative deamination alone It can not be explained by transamination alone as no free ammonia is liberated nor by oxidative deamination alone as oxid. Deamination works efficiently on glutamic acid as L- glutamate dehydrogenase is of high activity in the mammalian tissue, while the L- amino acid oxidase which works on most a. as is of low activity. ■The formation of NH 3 from α– a. as occurs mainly via the α– amino nitrogen of glutamate As this occurs by transdeamination, and L- glutamate is the only a. a that undergoes oxidative deamination at an appreciable rate in the mammalian tissue.
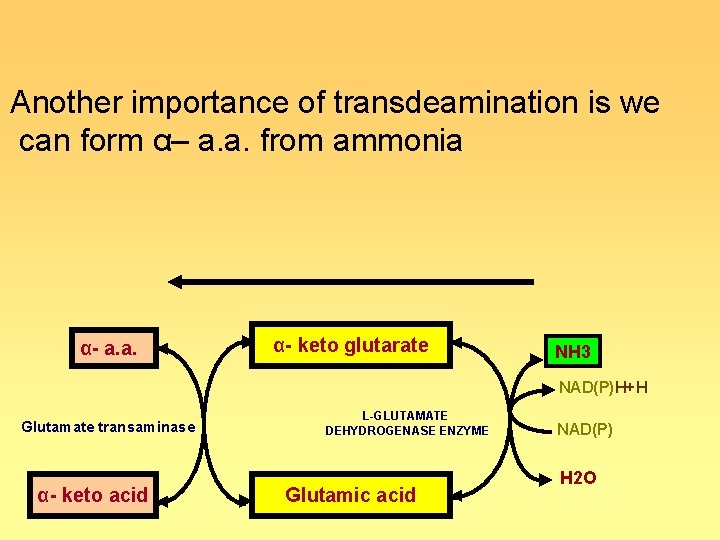
Another importance of transdeamination is we can form α– a. a. from ammonia α- a. a. ◄ ► α- keto glutarate ◄ L-GLUTAMATE DEHYDROGENASE ENZYME Glutamate transaminase α- keto acid ◄ ► Glutamic acid ◄ ► NH 3 ►NAD(P)H+H NAD(P) ► H 2 O

Specific methods of deamination 1 - L- glutamate dehydrogenase: Said before
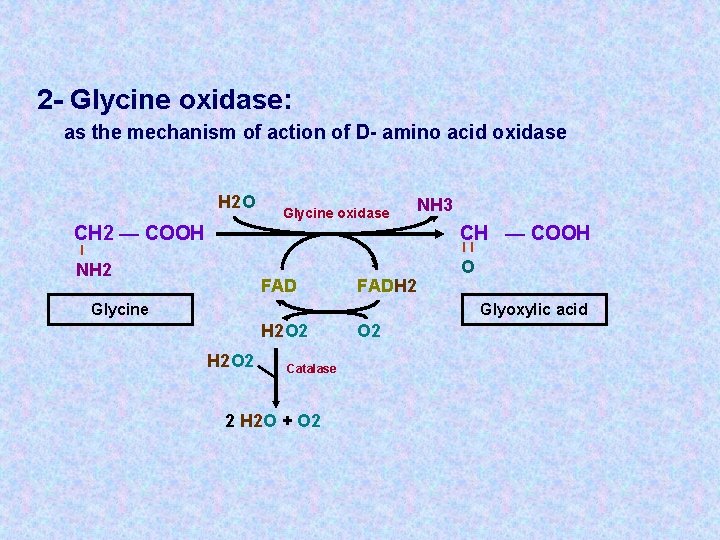
2 - Glycine oxidase: as the mechanism of action of D- amino acid oxidase H 2 O Glycine oxidase NH 3 CH 2 — COOH CH — COOH NH 2 O ll l FADH 2 Glycine Glyoxylic acid H 2 O 2 Catalase 2 H 2 O + O 2
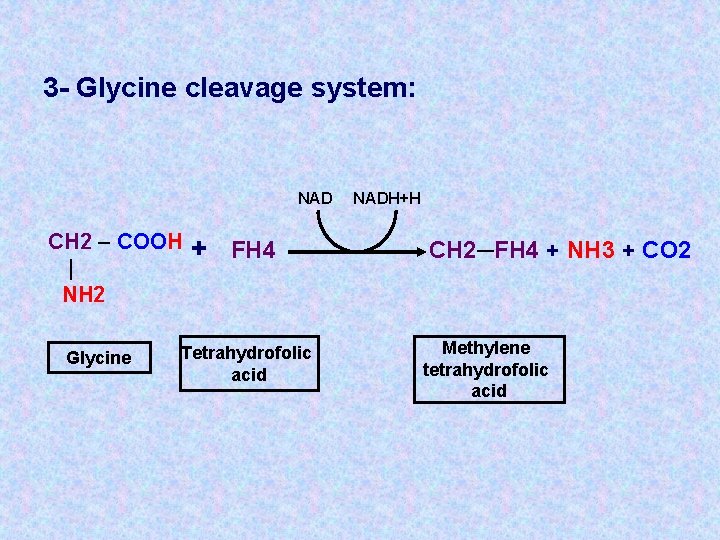
3 - Glycine cleavage system: NAD CH 2 – COOH | NH 2 Glycine + FH 4 Tetrahydrofolic acid NADH+H CH 2─FH 4 + NH 3 + CO 2 Methylene tetrahydrofolic acid
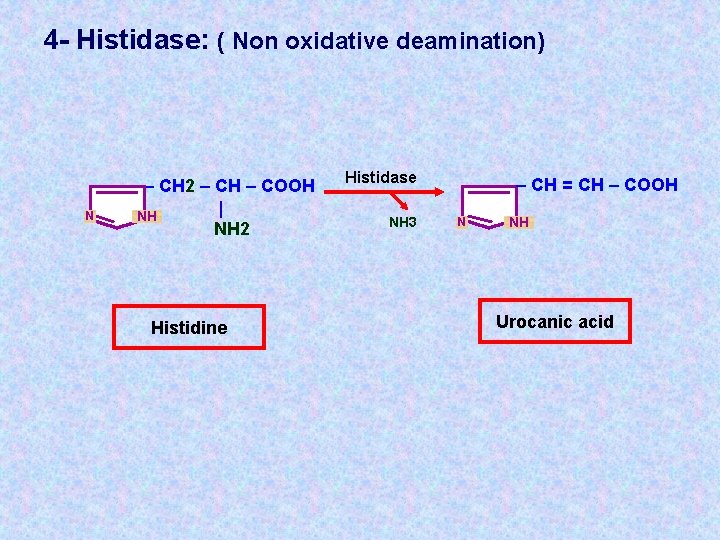
4 - Histidase: ( Non oxidative deamination) N – CH 2 – CH – COOH | NH NH 2 Histidine Histidase NH 3 – CH = CH – COOH N NH Urocanic acid
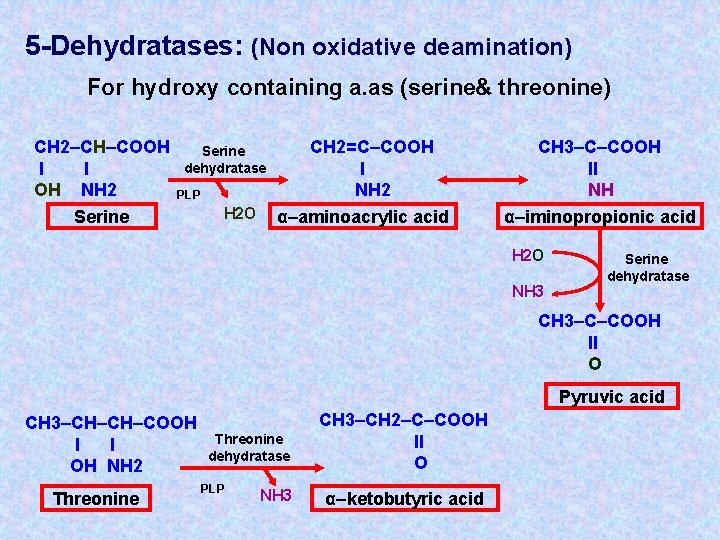
5 -Dehydratases: (Non oxidative deamination) For hydroxy containing a. as (serine& threonine) CH 2–CH–COOH l l OH NH 2 Serine dehydratase PLP H 2 O CH 2=C–COOH l NH 2 α–aminoacrylic acid CH 3–C–COOH ll NH α–iminopropionic acid H 2 O NH 3 Serine dehydratase CH 3–C–COOH ll O Pyruvic acid CH 3–CH–CH–COOH l l OH NH 2 Threonine dehydratase PLP NH 3 CH 3–CH 2–C–COOH ll O α–ketobutyric acid
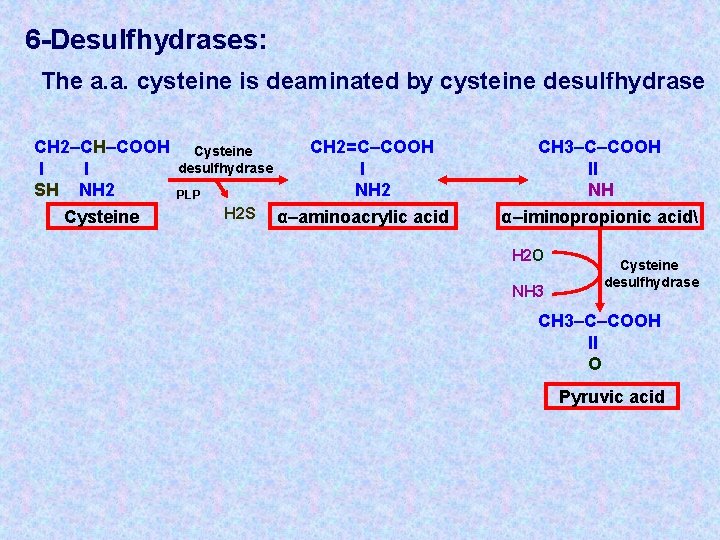
6 -Desulfhydrases: The a. a. cysteine is deaminated by cysteine desulfhydrase CH 2–CH–COOH l l SH NH 2 Cysteine desulfhydrase PLP H 2 S CH 2=C–COOH l NH 2 α–aminoacrylic acid CH 3–C–COOH ll NH α–iminopropionic acid H 2 O NH 3 Cysteine desulfhydrase CH 3–C–COOH ll O Pyruvic acid
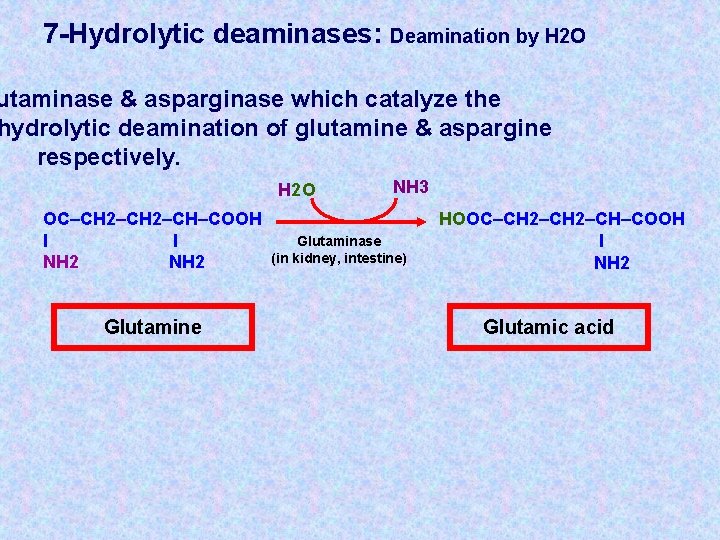
7 -Hydrolytic deaminases: Deamination by H 2 O utaminase & asparginase which catalyze the hydrolytic deamination of glutamine & aspargine respectively. H 2 O OC–CH 2–CH–COOH l l NH 2 Glutamine NH 3 Glutaminase (in kidney, intestine) HOOC–CH 2–CH–COOH l NH 2 Glutamic acid
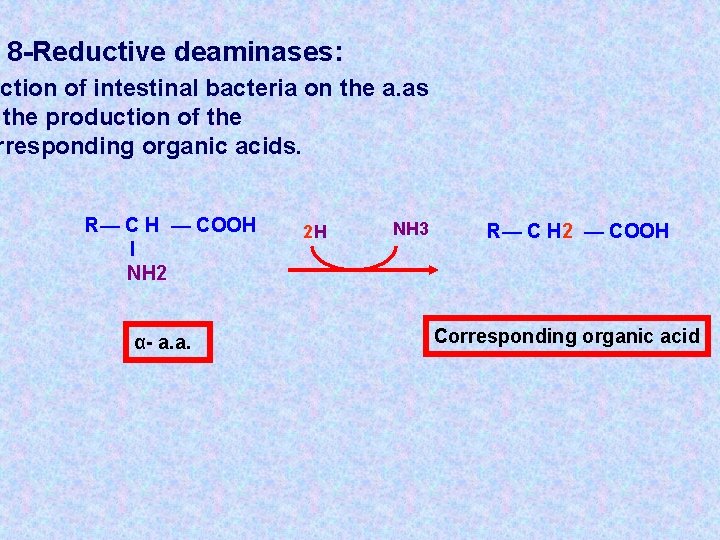
8 -Reductive deaminases: action of intestinal bacteria on the a. as the production of the rresponding organic acids. R— C H — COOH l NH 2 α- a. a. 2 H NH 3 R— C H 2 — COOH Corresponding organic acid
Deamination products NH 3 α- keto acid

Blood level: Normally < 0. 1 mg / dl Urine level: 0. 7 gm / day Sources & fate:

Various nitrogenous compounds as Amino acids Purines& pyrimidines Deamination: --Oxidative deamination --Transdeamination --Specific deamination Methods ( MAIN SOURCE) (amino groups attached to the rings) Some neurotransmitters as Urea secreted into the intestine Monoamines: --serotonin Histamine Catabolism –epinephrine, norepinephrine, dopamine & their metabolites metanephrine, normetanephrine Histaminase &3 methoxy tyramine Monoamine oxidase (MAO) Intestinal bacterial urease NH 3 Synthetic pathway Transdeamination Non essential a. a. synthesis Liver Catabolic pathway (MAIN FATE) Urea Urine glutamine Excreted in urine

►Urea formation: is the main pathway by which the body gets rid of NH 3 ►Glutamine formation: --By the glutamine synthetase enzyme which is a mitochondrial enzyme Glutamic acid + ammonia glutamine synthetase glutamine -- Glutamine is produced in many extra renal tissues esp. important: • In the muscle • In the liver: The formation of glutamine can be considered as a mechanism for scavenging NH 3 that has not been incorporated into urea. • In the brain: It removes the toxic effect of NH 3 in the brain. Then the glutamine goes via the blood to the kidneys where it become hydrolyzed by glutaminase into glutamic acid and NH 3 which is excreted in urine, (This accounts for 60% of the NH 3 excreted in urine) Glutamine glutaminase H 2 O NH 3 Glutamic acid

►NH 3 produced from a. a. deamination in the kidney is directly excreted in urine ( This acconts for 40%of NH 3 excreted in urine) ■N. B. NH 3 produced from a. a deamination in the kidney esp. glutamine regulates acid base balance &preserve cations

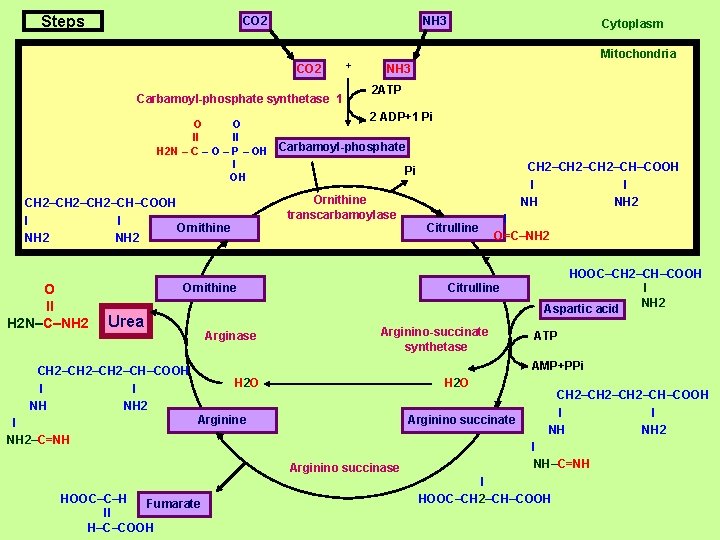
Steps CO 2 NH 3 CO 2 Carbamoyl-phosphate synthetase 1 + Cytoplasm Mitochondria NH 3 2 ATP 2 ADP+1 Pi O O ll ll H 2 N – C – O – P – OH Carbamoyl-phosphate l Pi OH Ornithine transcarbamoylase CH 2–CH–COOH l l Ornithine NH 2 O ll H 2 N–C–NH 2 Arginase l O=C–NH 2 Citrulline Arginino-succinate synthetase HOOC–CH 2–CH–COOH l NH 2 Aspartic acid ATP AMP+PPi CH 2–CH–COOH l l NH NH 2 l NH 2–C=NH Citrulline Ornithine Urea CH 2–CH–COOH l l NH NH 2 H 2 O Arginine Arginino succinate Arginino succinase HOOC–C–H Fumarate ll H–C–COOH CH 2–CH–COOH l l NH NH 2 l NH–C=NH l HOOC–CH 2–CH–COOH

Carbamoyl- phosphate synthetase I is different from carbamoyl phosphate synthetase II CPS II Mitochondria Cytoplasm Pathway Urea synthesis Pyrimidine synthesis +ve effector Inhibitor N-acetyl glutamate Nil CTP Source of N Ammonia Glutamine Site
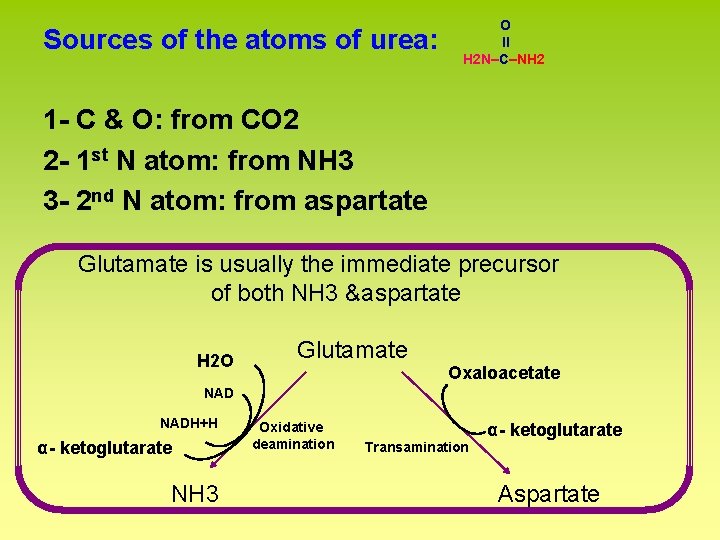
Sources of the atoms of urea: O ll H 2 N–C–NH 2 1 - C & O: from CO 2 2 - 1 st N atom: from NH 3 3 - 2 nd N atom: from aspartate Glutamate is usually the immediate precursor of both NH 3 &aspartate H 2 O Glutamate Oxaloacetate NADH+H α- ketoglutarate NH 3 Oxidative deamination α- ketoglutarate Transamination Aspartate

Overall reaction: NH 3 + CO 2 + Aspartate Urea + fumarate There in no net gain or loss of ornithine, citrulline , argininosuccinate or arginine. Ornithine is regenerated with each turn of the urea cycle. The release of the high energy phosphate of carbamoyl phosphate as inorganic phosphate drives the reaction in the forward direction.
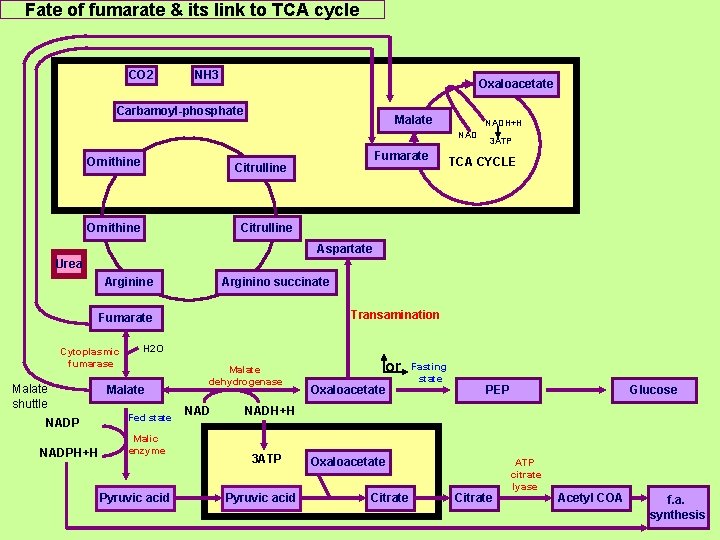
Fate of fumarate & its link to TCA cycle CO 2 NH 3 Oxaloacetate Carbamoyl-phosphate Malate NADH+H NAD Ornithine Fumarate Citrulline Ornithine 3 ATP TCA CYCLE Citrulline Aspartate Urea Arginine Arginino succinate Transamination Fumarate Cytoplasmic fumarase Malate shuttle NADPH+H H 2 O Malate Fed state Malic enzyme Pyruvic acid Malate dehydrogenase NAD or Oxaloacetate Fasting state PEP Glucose NADH+H 3 ATP Pyruvic acid Oxaloacetate Citrate ATP citrate lyase Acetyl COA f. a. synthesis

Bioenergetics: Urea cycle consumes four "high-energy" phosphate bonds (3 ATP hydrolyzed to 2 ADP and one AMP). 1 ATP ADP + Pi ~P 1 ATP ADP + Pi Adenosine ~ P 1 ATP AMP + Pi ~P ►However. One NADH+H molecule is produced by oxidative deamination of glutamate to NH 3 and α-ketoglutarate. Glutamate provides the NH 3 used in the initial synthesis of carbamoyl phosphate. ► Also fumarate in the cycle may be converted to malate in the cytosol. Malate then oxidized to oxaloacetate gives 1 NADH+H equivalent to 3 ATP obtained from 3 ADP, So the net energy expenditure is only one high energy phosphate. The two NADH+H produced can provide energy for the formation of 5 ATP, a net production of one high energy phosphate bond for the urea cycle. However, if gluconeogenesis is underway in the cytosol, the latter reducing equivalent is used to drive the reversal of the glyceraldehyde 3 -p dehydrogenase step instead of generating ATP. So the net energy expenditure is only one high energy phosphate.
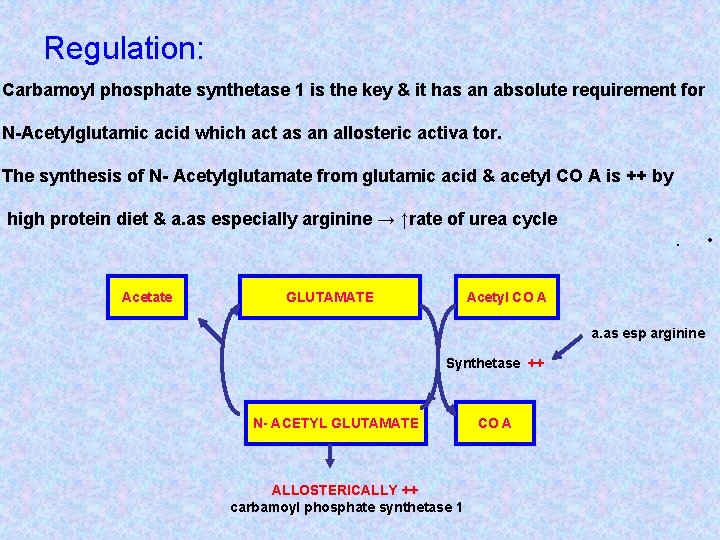
Regulation: Carbamoyl phosphate synthetase 1 is the key & it has an absolute requirement for N-Acetylglutamic acid which act as an allosteric activa tor. The synthesis of N- Acetylglutamate from glutamic acid & acetyl CO A is ++ by high protein diet & a. as especially arginine → ↑rate of urea cycle. Acetate GLUTAMATE Acetyl CO A a. as esp arginine Synthetase ++ N- ACETYL GLUTAMATE ALLOSTERICALLY ++ carbamoyl phosphate synthetase 1 CO A •

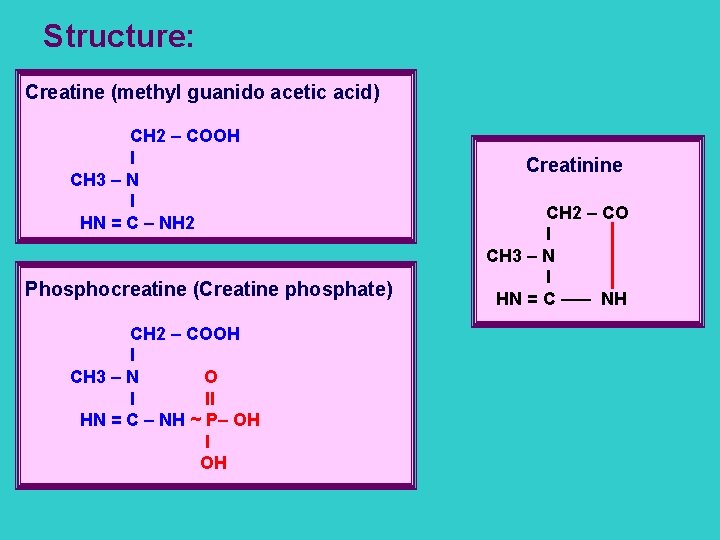
Structure: Creatine (methyl guanido acetic acid) CH 2 – COOH l CH 3 – N l HN = C – NH 2 Phosphocreatine (Creatine phosphate) CH 2 – COOH l CH 3 – N O l ll HN = C – NH ~ P– OH l OH Creatinine CH 2 – CO l CH 3 – N l HN = C ––– NH
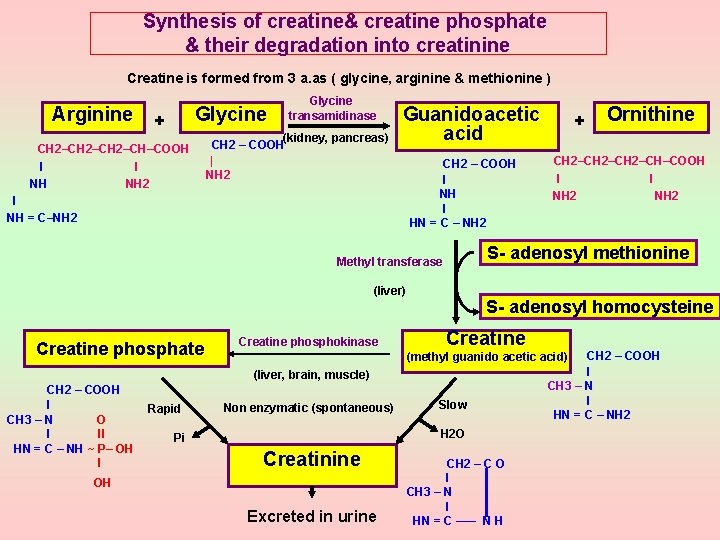
Synthesis of creatine& creatine phosphate & their degradation into creatinine Creatine is formed from 3 a. as ( glycine, arginine & methionine ) Arginine Glycine + CH 2–CH–COOH l l NH NH 2 Glycine transamidinase (kidney, pancreas) CH 2 – COOH | NH 2 Guanidoacetic acid l NH = C–NH 2 CH 2 – COOH l NH l HN = C – NH 2 (liver) Creatine phosphokinase Rapid CH 2–CH–COOH l l NH 2 S- adenosyl homocysteine Creatine CH 2 – COOH l CH 3 – N l HN = C – NH 2 (methyl guanido acetic acid) (liver, brain, muscle) CH 2 – COOH l CH 3 – N O l ll HN = C – NH ~ P– OH l Non enzymatic (spontaneous) Slow H 2 O Pi Creatinine OH Excreted in urine Ornithine S- adenosyl methionine Methyl transferase Creatine phosphate + CH 2 – C O l CH 3 – N l HN = C ––– N H

Importance of creatine: ►Creatine is converted to creatine phosphate using ATP as a phosphate donor. Creatine phosphate acts as a store of high energy phosphate , so it is used as a source of energy in time of need (as it can give the phosphate group to ADP to form ATP). ►Creatine is present mainly in the muscle (98%) , but also in the brain & blood. Both the muscle & the brain contain large amounts of creatine phosphate. ►During muscular relaxation, creatine becomes converted to creatine phosphate to be used as a source of energy during muscular contraction. (During ms contraction, ATP is mostly derived from glycolysis for which ms glycogen acts as a substrate, However before ATP from glycolysis become available, phosphocreatine acts as a temporary source of ATP. So it is especially important during the early stages of ms exercise(1 st few minutes), where the largest quantities of creatine –P are found ). Level of creatine in the blood& urine: ►Plasma creatine level = 0. 4 mg % Whole blood creatine level = 2 - 4 mg % ► Normally little creatine is excreted in urine ( normally < 100 mg /day ). This is more in females & children due to low androgens which increase the uptake of creatine by ms.

Creatinuria ►Def. : It is the presence of large amounts of creatine in urine (Normal level of creatine excreted in urine is < 100 mg / day. ) ►Causes: 1 - Physiological creatinuria : ■ in children: due to - ↓androgens → ↓ uptake of creatine by the muscle - small mass of the ms ■ in female: due to - ↓ free androgen especially during pregnancy. - after labour due to involution of the uterus. 2 - Pathological creatinuria : ■ in males with ↓ androgens( hypogonadism ) ■ in muscle atrophy as in myopathies. ■ ↑ protein catabolism in --hormonal disturbances as D. M. , hyperthyrodism and cushing syndrome, --also in fever and starvation

Creatinine levels in serum& urine: ►Normal serum creatinine level = 0. 5 - 1. 5 mg%. ↑ serum creatinine level Causes: 1 - Impairment of kidney function : about 50% of kidney function must be lost before serum creatinine is ↑. 2 - ↑ ms mass : as acromegally & gigantism. ↓ serum creatinine level Causes: ↓ ms mass as in - myasthenia gravis - muscular dystrophy - debilitation

► Normal urine creatinine level = 1 - 1. 5 mg %. - It is more in male due to greater ms mass ---- male = 1. 5 g / day. ---- female = 1 gm / day. - There is a steady production of constant amount of creatinine that is proportional to the total amount of phosphocreatine & creatine in the body , which is in turn proportional to the ms mass of the individual. So, the serum & urine creatinine is constant for an individual & approximately proportional to the ms mass. Creatinine excretion rate (creatinine coefficient) : • It is the amount of creatinine measured in mg / kg body weight / day. • It is said to be remarkably constant (21 in male & 16 in female). (Why? ) So, creatinine excretion rate (creatinine coeffecient) can be used to check the accuracy of 24 hr collection of urine. If found lower than expected, it indicates that part of the urine was discarded.

Creatinine clearance - - - The volume of serum or plasma that would be cleared of creatinine by one minute's excretion of urine. value that reflects the body's ability to excrete creatinine; it is used to diagnose and monitor renal function. Urinary creatinine is 100% endogenous in origin i. e. not affected by diet being synthesized in the body & no diurnal variation. So it can be used for calculation of urinary output of substance amount of creatinine in urine. Also creatinine is chiefly filtered by the kidney, though a small amount is actively secreted. There is little-to-no tubular reabsorption of creatinine. If the filtering of the kidney is deficient, blood levels rise. As a result, creatinine levels in blood and urine may be used to calculate the creatinine clearance (Cr. Cl), which reflects the glomerular filtration rate (GFR). The GFR is clinically important because it is a measurement of renal function. However, in cases of severe renal dysfunction, the creatinine clearance rate will be "overestimated" because the active secretion of creatinine will account for a larger fraction of the total creatinine cleared. Ketoacids, cimetidine and trimethoprim reduce creatinine tubular secretion and therefore increase the accuracy of the GFR estimate, particularly in severe renal dysfunction. (In the absence of secretion, creatinine behaves like inulin. ) Creatinine clearance (CCr) can be calculated if values for creatinine's urine concentration (UCr), urine flow rate (V), and creatinine's plasma concentration (PCr) are known. Since the product of urine concentration and urine flow rate yields creatine's excretion rate, creatinine clearance is also said to be its excretion rate (UCr×V) divided by its plasma concentration. This is commonly represented mathematically as : Creatinine clearance(ml/min)= Urinary creatinine conc. X urine volume in ml Plasma creatinine conc X time of collection/min


NPN Compounds Def: Are nitrogenous compounds not precipitated by protein ppting reagents. Include: a. as Urea Creatine NH 3 Uric acid Creatinine
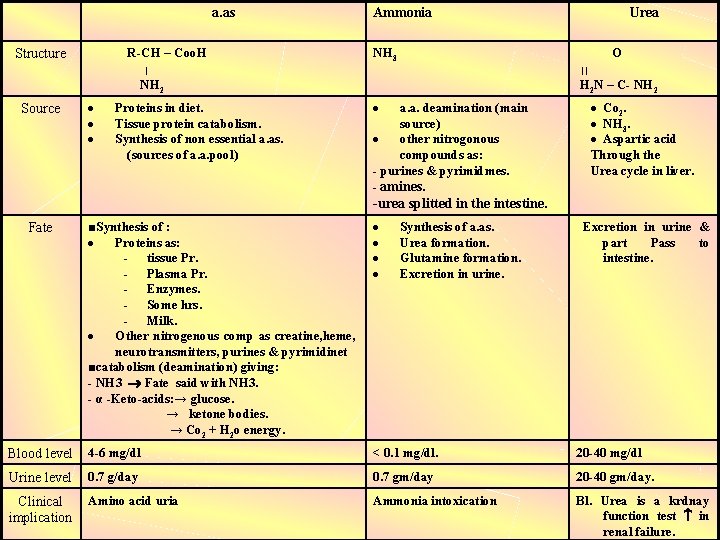
a. as R-CH – Coo. H Structure ׀ Ammonia NH 3 O ׀׀ H 2 N – C- NH 2 Source Proteins in diet. Tissue protein catabolism. Synthesis of non essential a. as. (sources of a. a. pool) Urea a. a. deamination (main source) other nitrogonous compounds as: - purines & pyrimidmes. Co 2. NH 3. Aspartic acid Through the Urea cycle in liver. - amines. -urea splitted in the intestine. ■Synthesis of : Proteins as: - tissue Pr. - Plasma Pr. - Enzymes. - Some hrs. - Milk. Other nitrogenous comp as creatine, heme, neurotransmitters, purines & pyrimidinet ■catabolism (deamination) giving: - NH 3 Fate said with NH 3. - α -Keto-acids: → glucose. → ketone bodies. → Co 2 + H 2 o energy. Blood level 4 -6 mg/dl < 0. 1 mg/dl. 20 -40 mg/dl Urine level 0. 7 g/day 0. 7 gm/day 20 -40 gm/day. Clinical implication Amino acid uria Ammonia intoxication Bl. Urea is a krdnay function test in renal failure. Fate Synthesis of a. as. Urea formation. Glutamine formation. Excretion in urine & part Pass to intestine.
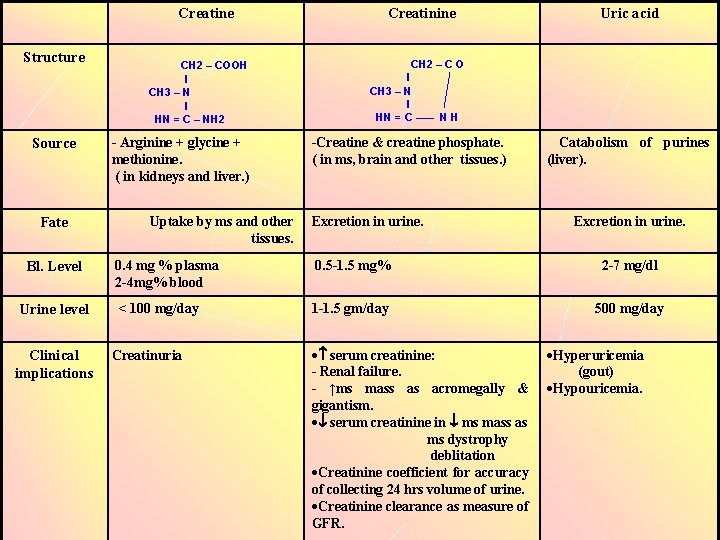
Creatine Structure Source Fate Bl. Level Urine level Clinical implications CH 2 – COOH l CH 3 – N l HN = C – NH 2 - Arginine + glycine + methionine. ( in kidneys and liver. ) Uptake by ms and other tissues. Creatinine Uric acid CH 2 – C O l CH 3 – N l HN = C ––– N H -Creatine & creatine phosphate. ( in ms, brain and other tissues. ) Excretion in urine. Catabolism of purines (liver). Excretion in urine. 0. 4 mg % plasma 2 -4 mg% blood 0. 5 -1. 5 mg% 2 -7 mg/dl < 100 mg/day 1 -1. 5 gm/day 500 mg/day Creatinuria serum creatinine: - Renal failure. - ↑ms mass as acromegally & gigantism. serum creatinine in ms mass as ms dystrophy deblitation Creatinine coefficient for accuracy of collecting 24 hrs volume of urine. Creatinine clearance as measure of GFR. Hyperuricemia (gout) Hypouricemia.

Heterocyclic a. as 1 - Tryptophan Synthesis: essential a. a. Catabolism: Both glucogenic and ketogenic. Tryptophan pyrrolase (oxygenase) N- formyl kynurenine Acetoacetic acid -keto adipic acidα α-aminomuconate semialdehyde Quinolinic acid NAD&NADP Nicotinic acid 2 -Acrolyl 3 - amino fumarate 3 -Hydroxyanthranilic acid Hydroxykynurenine Kynurenine NAD&NADP. 3 -Melatonin 4 -Indole &skatole 1 -Nicotinic acid: • It is produced from tryptophan in the liver since the enzyme picolinate carboxylase has low activity in the liver so some 2 -acrolyl 3 aminofumarate is cyclized in non enzymatic reaction to niacin. • Every 60 mg tryptophan gives 1 mg niacin. 50%of niacin is formed from tryptophan while the rest are obtained from diet. Pellagra: n maize eating areas or↓ absorption in hartnup disease hesis from tryptophan in case of carcinoid syndrome. 3 - Isoniazid drug administration in T. B. treatment leading to vitamin B 6 deficiency which is important in formation of niacin from tryptophan • pellagra is characterized by 3 Ds diarrhea, dermatitis and dementia
- Slides: 80