Amino Acids Proteins and Enzymes Types of Proteins
































- Slides: 62

Amino Acids Proteins, and Enzymes Types of Proteins Amino Acids The Peptide Bond 1

Types of Proteins ﻭﻇﺎﺋﻒ ﺍﻟﺒﺮﻭﺗﻴﻨﺎﺕ • • Type Structural Contractile Transport Storage Hormonal Enzyme Protection Examples tendons, cartilage, hair, nails muscles hemoglobin milk insulin, growth hormone catalyzes reactions in cells immune response 2

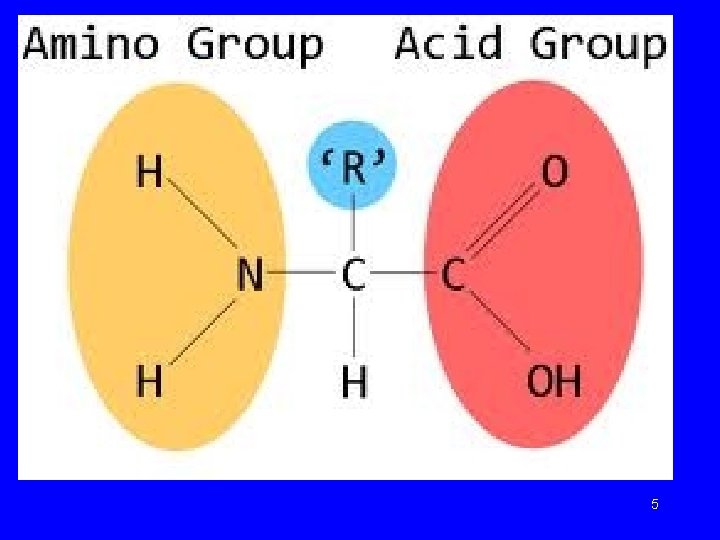
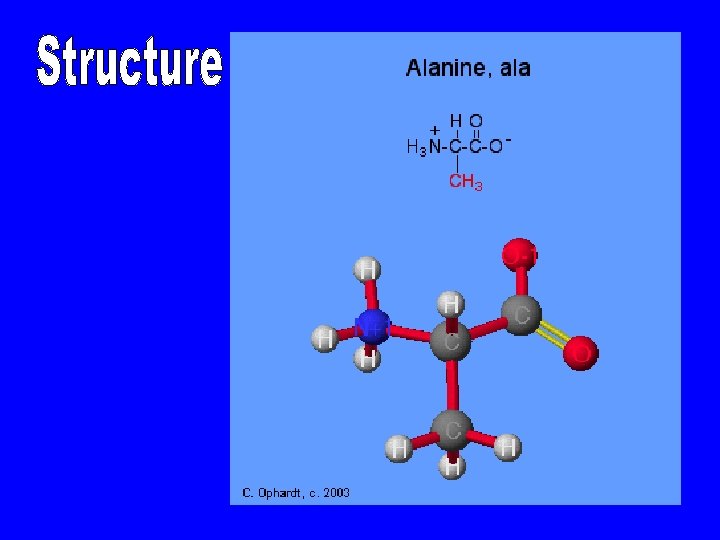
Amino Acids • • Building blocks of proteins ﻭﺣﺪﺍﺕ ﺑﻨﺎﺀ ﺍﻟﺒﺮﻭﺗﻴﻦ Carboxylic acid group ﺗﺤﻮﻱ ﻋﻠﻰ Amino group Side group R gives unique characteristics R side chain I H 2 H—C —COOH I H 3

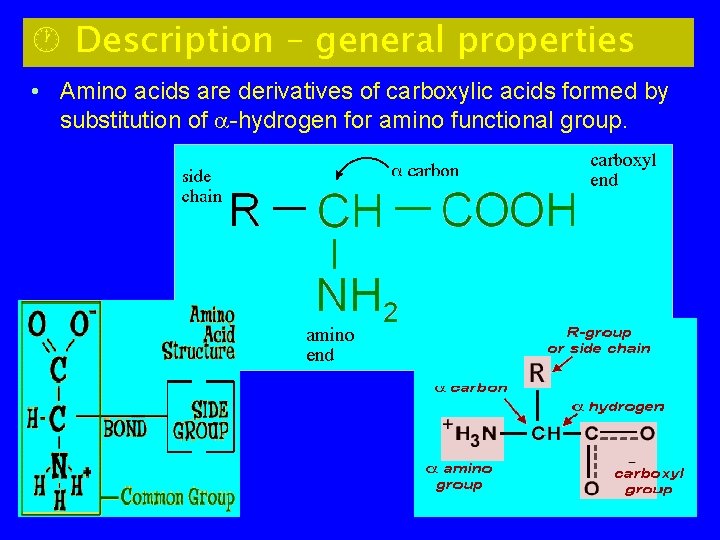
· Description – general properties • Amino acids are derivatives of carboxylic acids formed by substitution of -hydrogen for amino functional group.

5

-carbon

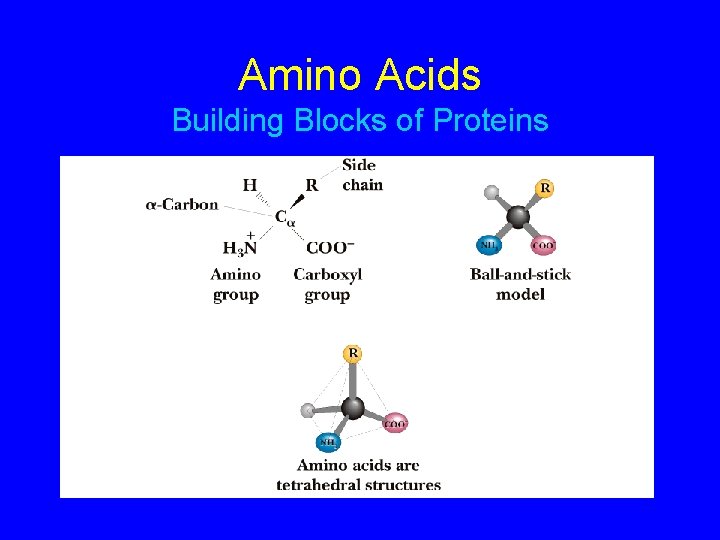
Amino Acids Building Blocks of Proteins

Amino Acids • Amino acids are the structural building blocks (monomers) of proteins. ﺍﻷﺤﻤﺎﺽ ﻣﻦ ﺃﻮﻟﻴﺔ ﻭﺣﺪﺍﺕ ﺍﻟﺒﺮﻭﺗﻴﻨﺎﺕ ﻟﺒﻨﺎﺀ ﺍﻻﻣﻴﻨﻴﺔ • There are twenty different kinds of amino acids used in proteins. ﺑﻨﺎﺀ ﻓﻲ ﺍﻣﻴﻨﻲ ﺣﺎﻣﺾ ﻋﺸﺮﻳﻦ ﺍﻟﺒﺮﻭﺗﻴﻨﺎﺕ • Proteins are referred to as heteropolymers due the variety of amino acids involved in their structure. ﻋﻠﻰ ﻳﺤﺘﻮﻱ ﻣﺘﻌﺪﺩ ﺑﻮﻟﻴﻤﺮ ﺍﻟﺒﺮﻭﺗﻴﻨﺎﺕ ﺍﻟﺘﺮﻛﻴﺐ ﻣﺨﺘﻠﻔﺔ ﺍﻣﻴﻨﻴﺔ ﺃﺤﻤﺎﺽ

BASICS Lets start with the basics. All amino acids have a common structural unit that is built around the alpha carbon (click 1). Lets call this the “core” structure. The figure shows the core with one of the bonds on the -carbon unassigned. A group in this location is represented by the letter R (click 1). COOH +H N C H 3 R R R groups are the only variable groups in the structure. Consider R the only unknown and focus on this group to learn the structures. Hence, Rule (1) is amino acids are composed of a core group and an R group. Rule (2) is the R group gives an amino acid its structural identity and, later as we will see, its unique biochemical properties.

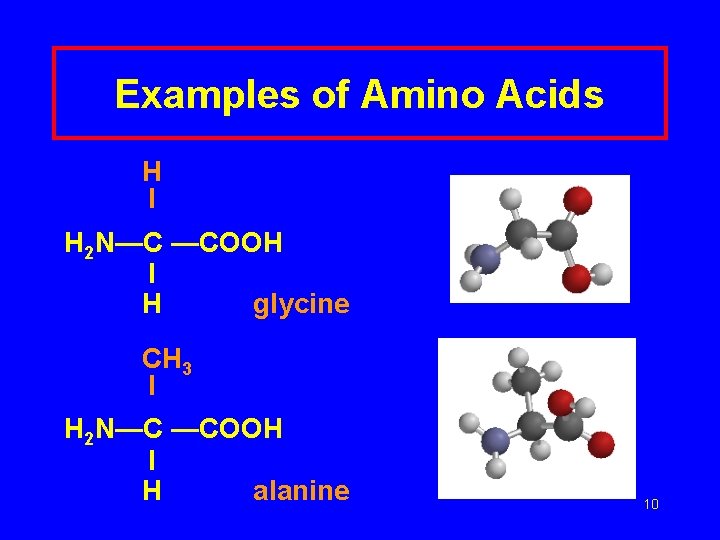
Examples of Amino Acids H I H 2 N—C —COOH I H glycine CH 3 I H 2 N—C —COOH I H alanine 10

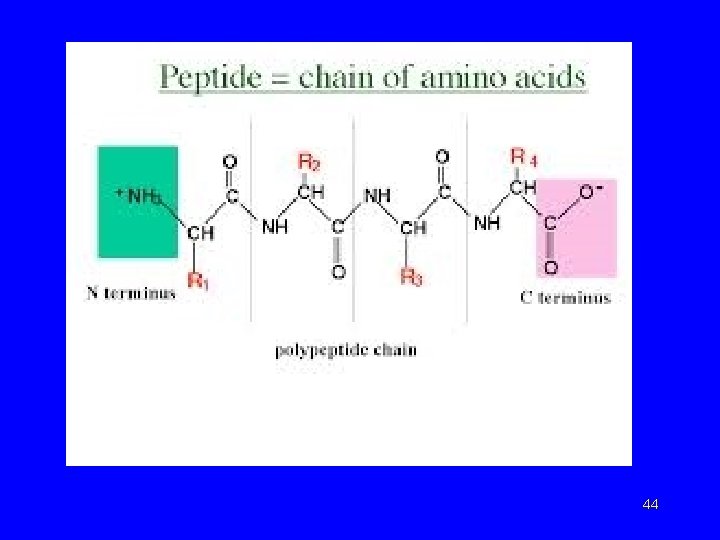
Amino Acids (cont’d. ) • The repeating sequence of atoms along a proteins is referred to as the polypeptide backbone. Attached to this repetitive chain are the different amino acid side chains (Rgroups) which are not involved in the peptide bond but which give each amino acid its unique property.

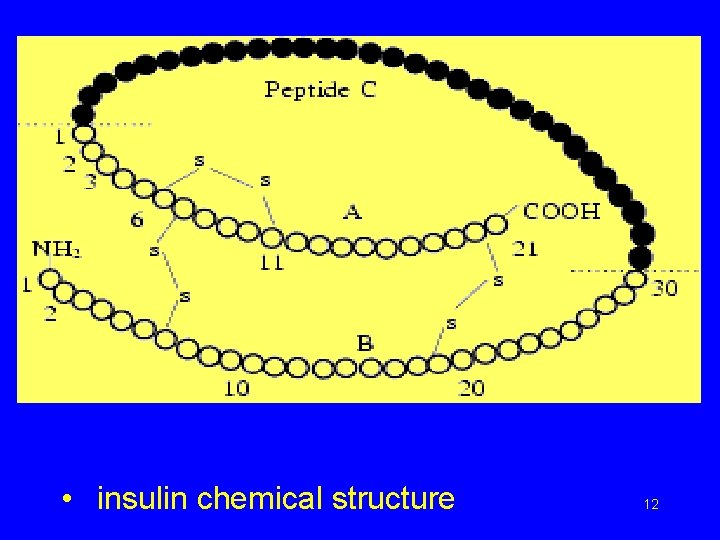
• insulin chemical structure 12

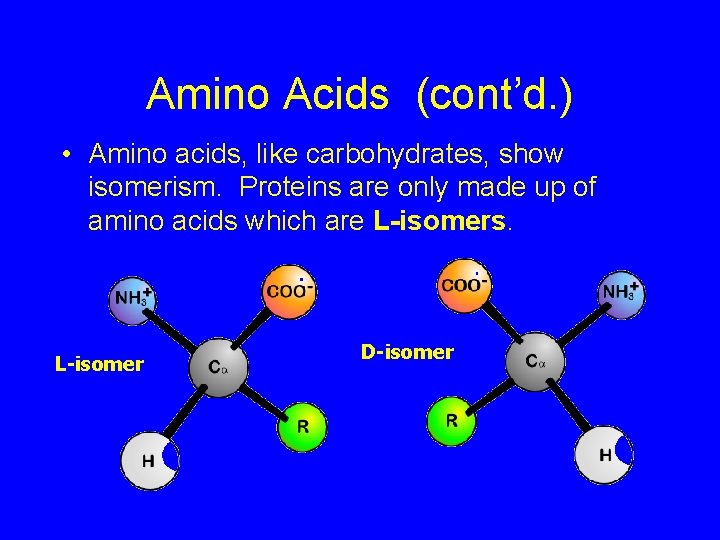
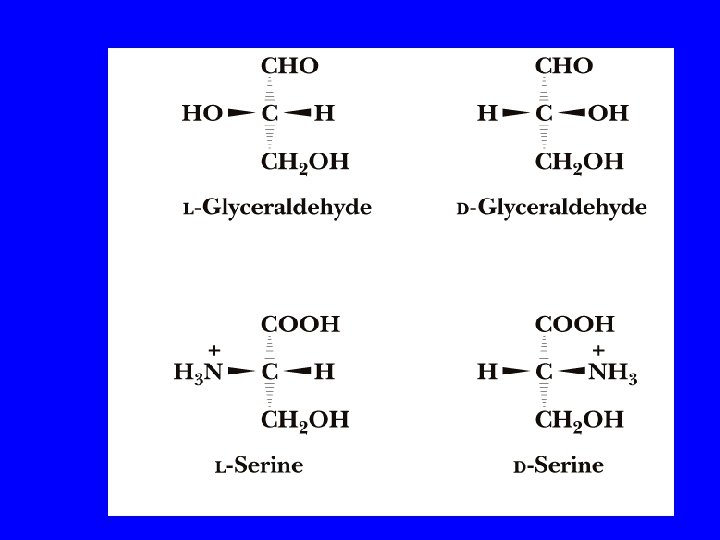
Amino Acids (cont’d. ) • Amino acids, like carbohydrates, show isomerism. Proteins are only made up of amino acids which are L-isomers. L-isomer D-isomer

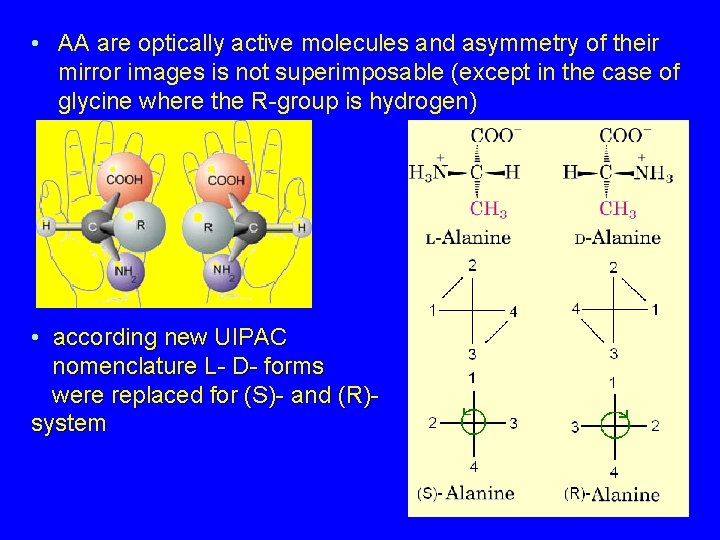
• AA are optically active molecules and asymmetry of their mirror images is not superimposable (except in the case of glycine where the R-group is hydrogen) • according new UIPAC nomenclature L- D- forms were replaced for (S)- and (R)- system


Types of Amino Acids Nonpolar R = H, CH 3, alkyl groups, aromatic O Polar ll R = –CH 2 OH, –CH 2 SH, –CH 2 C–NH 2, (polar groups with –O-, -SH, -N-) Polar/Acidic R = –CH 2 COOH, or -COOH Polar/ Basic R = –CH 2 NH 2 16

Properties of Amino Acids • High melting points, over 200 C • More soluble in water than in ether. • Larger dipole moments than simple acids or simple amines. • Less acidic than most carboxylic acids, less basic than most amines. p. Kb = 12 => Chapter 24 17

Complete Proteins • • Provide all the essential amino acids. Examples: those in meat, fish, milk, eggs. Plant proteins are generally incomplete. Vegetarians should eat many different kinds of plants, or supplement diet with milk or eggs. ﻣﺼﺎﺩ ﺍﻟﻐﺬﺍﺋﻴﺔ ﺍﻟﺒﺮﻭﺗﻴﻨﺎﺕ ﺭ => Chapter 24 18

19

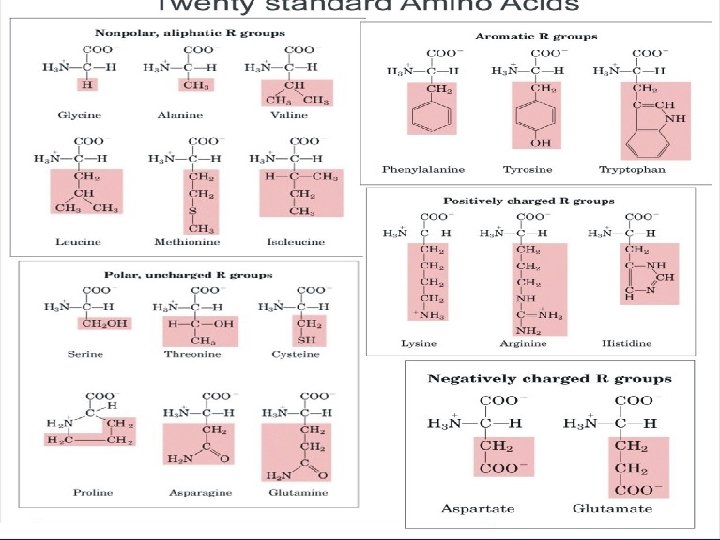
20 Common Amino Acids You should know names, structures, p. Ka values, 3 -letter and 1 -letter codes • Non-polar amino acids • Polar, uncharged amino acids • Acidic amino acids • Basic amino acids

Amino Acids (cont’d. ) • Amino acids are grouped according to whether their side chains are: • acidic • basic • uncharged polar • non polar

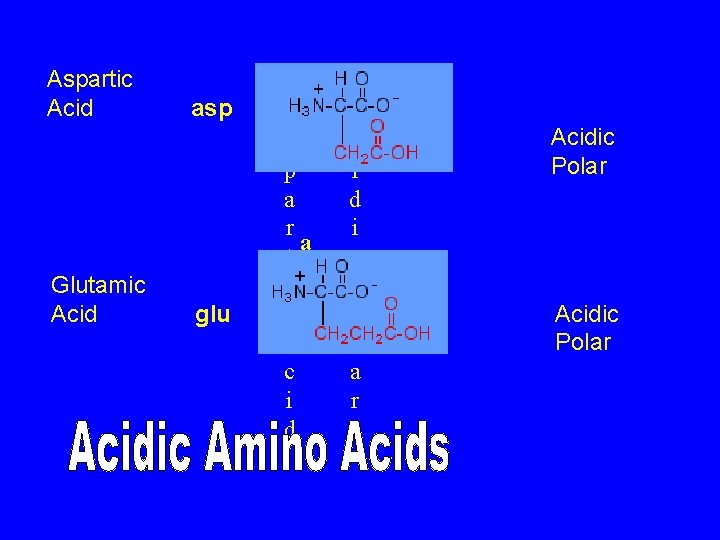
Aspartic Acid Glutamic Acid asp glu A A c s i p d a i r a t c s i D P p c o l A a c r i d Acidic Polar

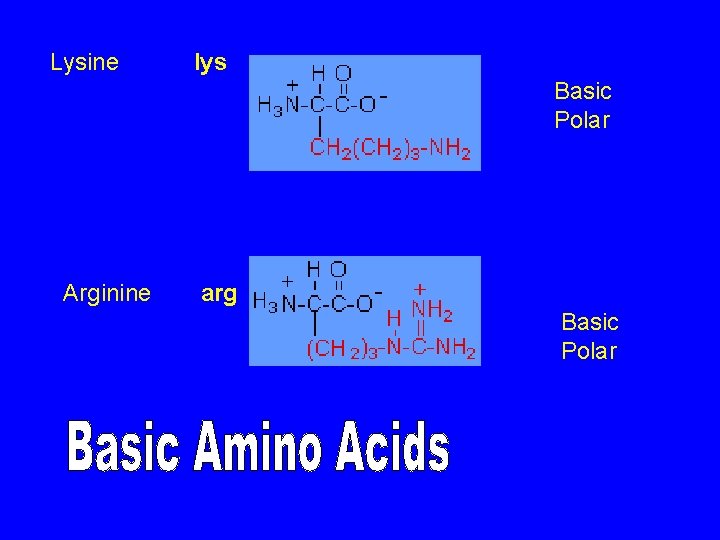
Lysine lys Arginine arg Basic Polar

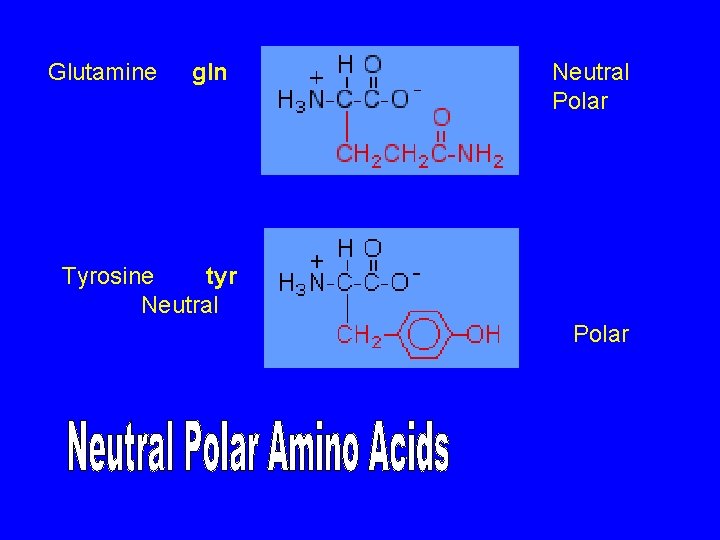
Glutamine gln Neutral Polar Tyrosine tyr Neutral Polar

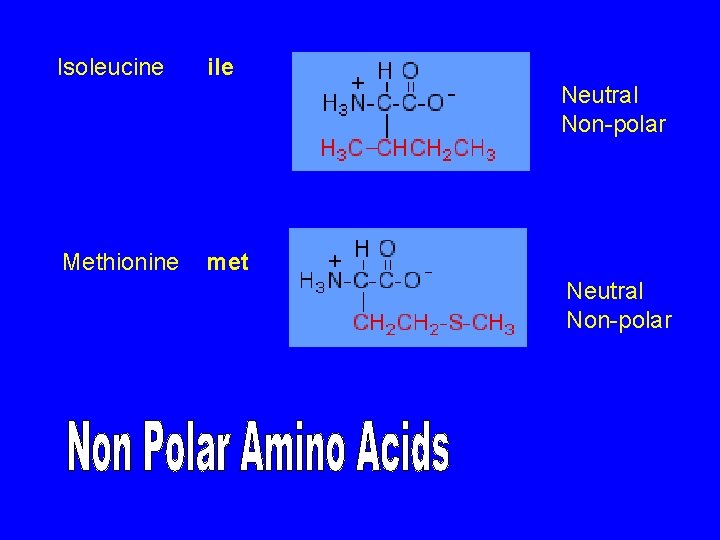
Isoleucine ile Neutral Non-polar Methionine met Neutral Non-polar

Amino Acids (cont’d. ) • The type of side chain is very important as it affects the solubility of the amino acid. • Hydrophobic features include long non-polar (uncharged) chains or complex aromatic rings. • Hydrophilic features include additional carboxyl groups or amino groups not involved in peptide bonding which are ionised in solution.

Essential Amino Acids • 10 amino acids not synthesized by the body • arg, his, ile, leu, lys, met, phe, thr, trp, val • Must obtain from the diet • All in diary products • 1 or more missing in grains and vegetables 27

Essential Amino Acids • • • Arginine (Arg) Threonine (Thr) Lysine (Lys) Valine (Val) Phenylalanine (Phe) • • • Tryptophan (Trp) Methionine (Met) Histidine (His) Leucine (Leu) Isoleucine (Ile) => Chapter 24 28

Amino Acids (cont’d. ) • At neutral p. H’s amino acids exist in an ionised form and have both acidic and basic properties. This is because the carboxylic group donates hydrogen ions to the solution (acidic) whereas the amino group (NH 2) attracts hydrogen ions from the solution.

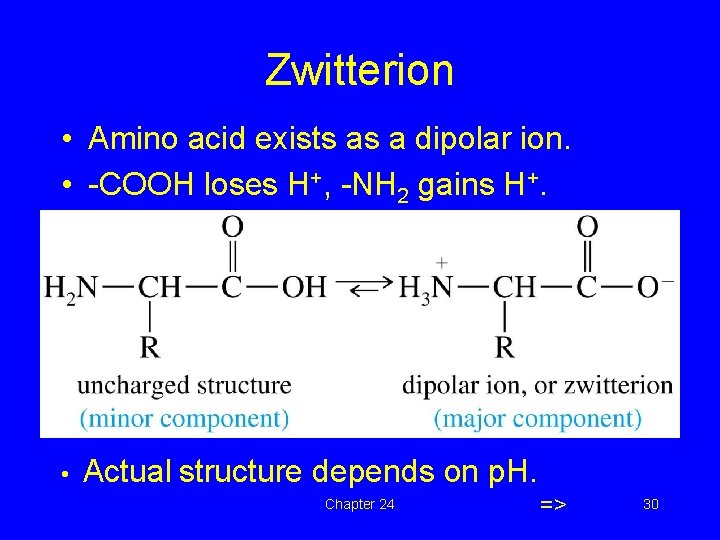
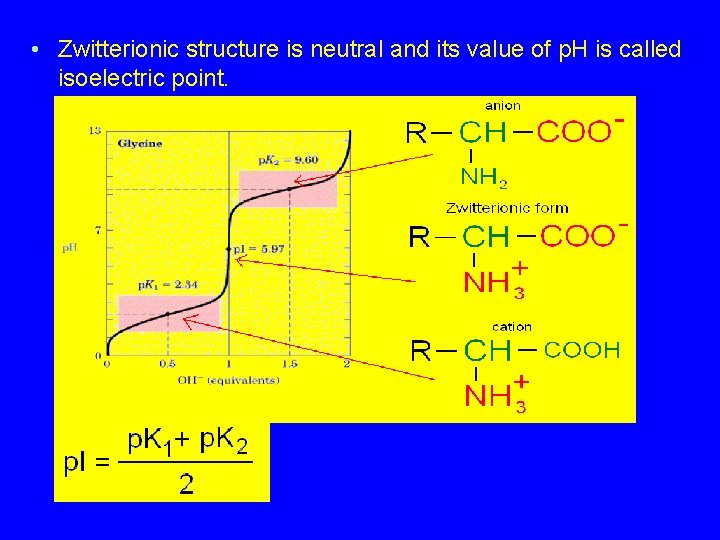
Zwitterion • Amino acid exists as a dipolar ion. • -COOH loses H+, -NH 2 gains H+. • Actual structure depends on p. H. Chapter 24 => 30

Amino Acids as Acids and Bases • Ionization of the –NH 2 and the –COOH group • Zwitterion has both a + and – charge • Zwitterion is neutral overall + NH 2–COOH glycine H 3 N–CH 2–COO– Zwitterion of glycine 31

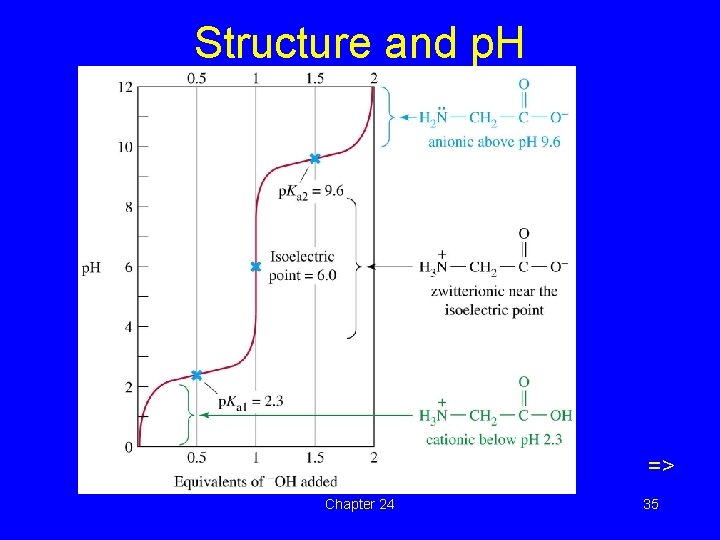
Isoelectric Point • p. H at which amino acids exist as the zwitterion (neutral). • Depends on structure of the side chain. • Acidic amino acids, isoelectric p. H ~3. • Basic amino acids, isoelectric p. H ~9. • Neutral amino acids, isoelectric p. H is slightly acidic, 5 -6. => Chapter 24 32

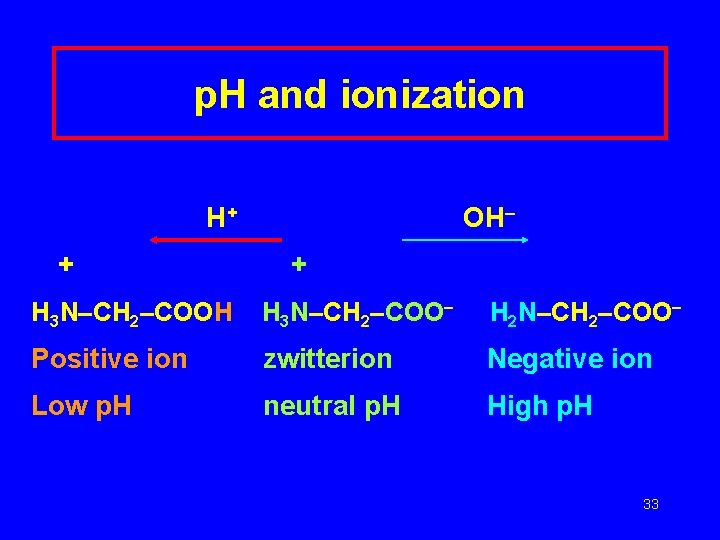
p. H and ionization H+ + OH– + H 3 N–CH 2–COOH H 3 N–CH 2–COO– H 2 N–CH 2–COO– Positive ion zwitterion Negative ion Low p. H neutral p. H High p. H 33

• Zwitterionic structure is neutral and its value of p. H is called isoelectric point.

Structure and p. H => Chapter 24 35

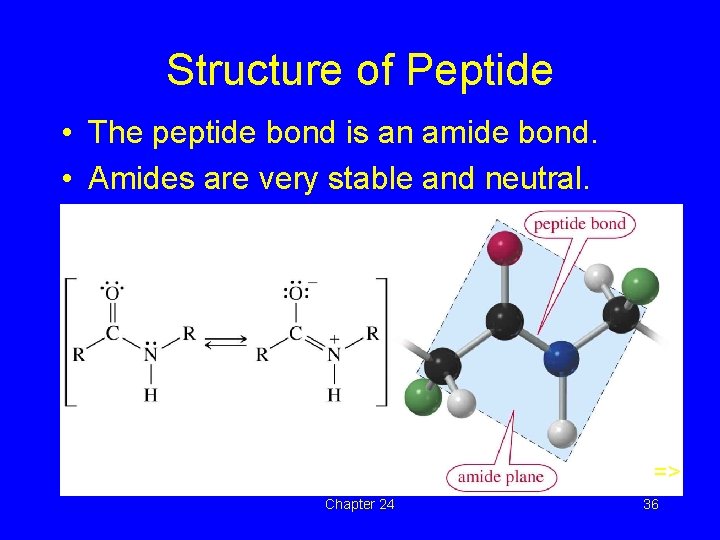
Structure of Peptide • The peptide bond is an amide bond. • Amides are very stable and neutral. => Chapter 24 36

Peptide Bond Formation • The amino group of one molecule condenses with the acid group of another. • Polypeptides usually have molecular weight less than 5000. • Protein molecular weight 6000 -40, 000. => Chapter 24 37

Learning Check AA 1 Identify each as (1) polar or (2) nonpolar A. NH 2–COOH (Glycine) CH 3 | CH–OH | B. NH 2–CH–COOH (Serine) 38

Solution AA 1 Identify each as (1) polar or (2) nonpolar A. (2) NH 2–COOH (Glycine) CH 3 | CH–OH | B. (1) NH 2–CH–COOH (Serine) 39

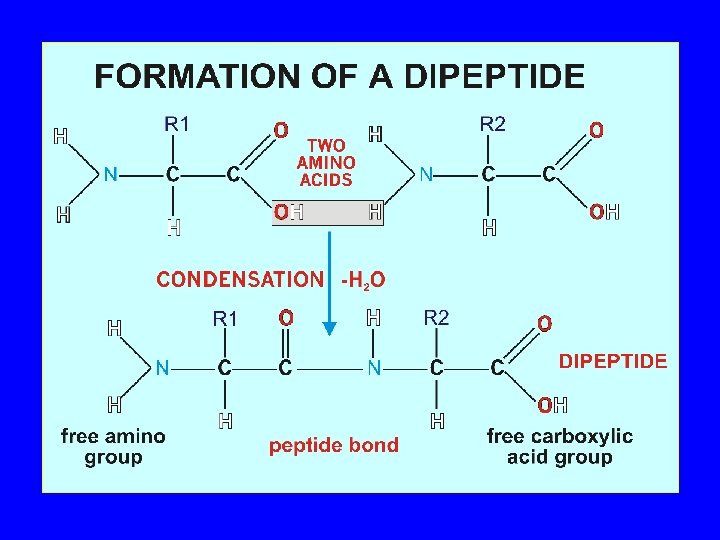
Amino Acids (cont’d. ) • Amino acids link together by covalent peptide bonds. This involves a condensation /dehydration reaction. These bonds are very strong. When this takes place the charged amino and carboxylic groups disappear.

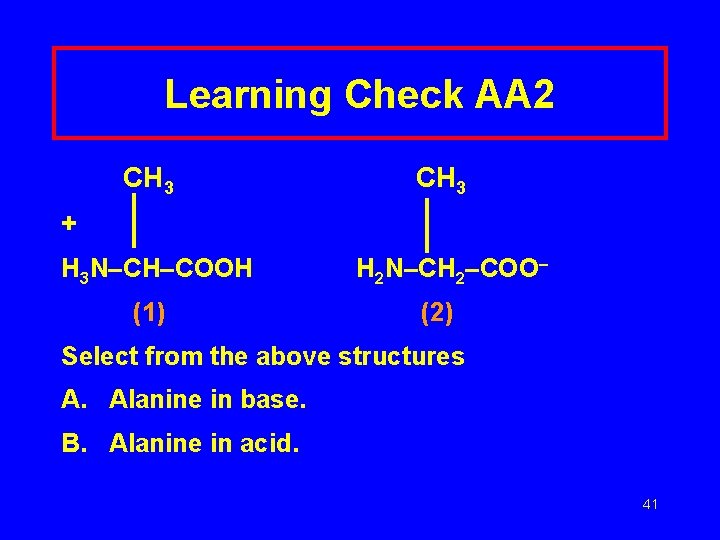
Learning Check AA 2 CH 3 + H 3 N–CH–COOH (1) H 2 N–CH 2–COO– (2) Select from the above structures A. Alanine in base. B. Alanine in acid. 41

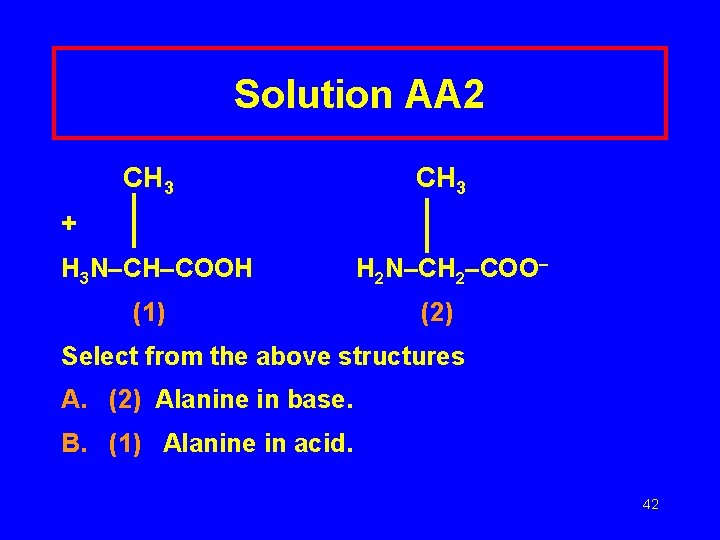
Solution AA 2 CH 3 + H 3 N–CH–COOH (1) H 2 N–CH 2–COO– (2) Select from the above structures A. (2) Alanine in base. B. (1) Alanine in acid. 42

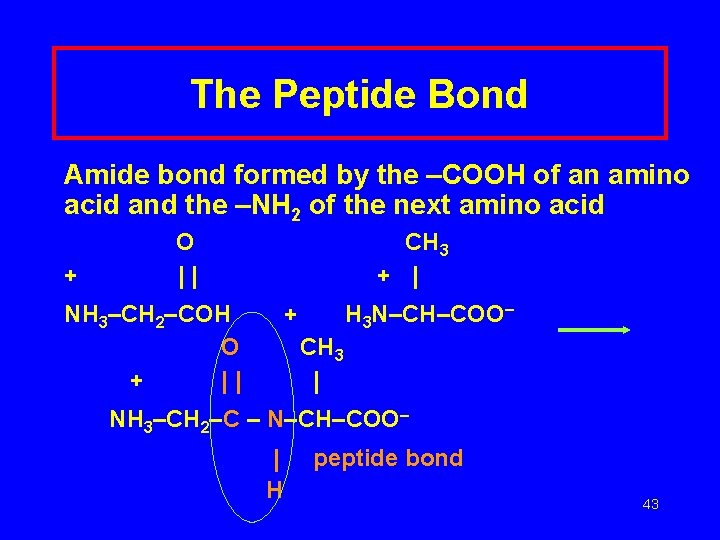
The Peptide Bond Amide bond formed by the –COOH of an amino acid and the –NH 2 of the next amino acid + O || CH 3 + | NH 3–CH 2–COH O + || + H 3 N–CH–COO– CH 3 | NH 3–CH 2–C – N–CH–COO– | H peptide bond 43

44

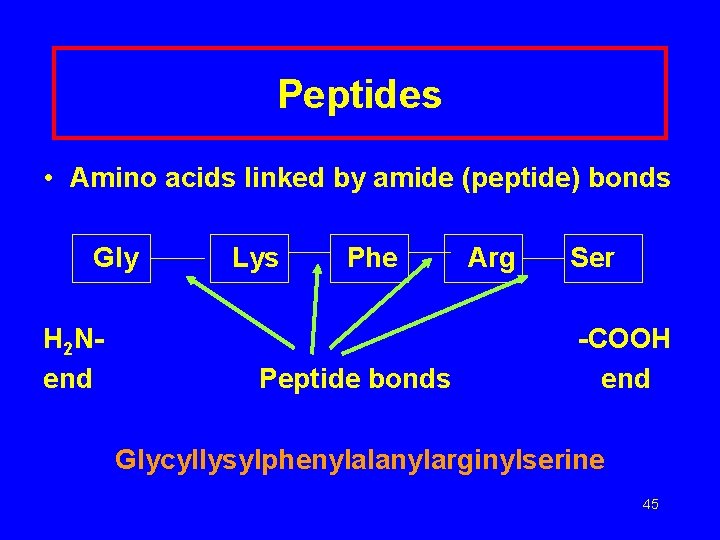
Peptides • Amino acids linked by amide (peptide) bonds Gly H 2 Nend Lys Phe Peptide bonds Arg Ser -COOH end Glycyllysylphenylalanylarginylserine 45

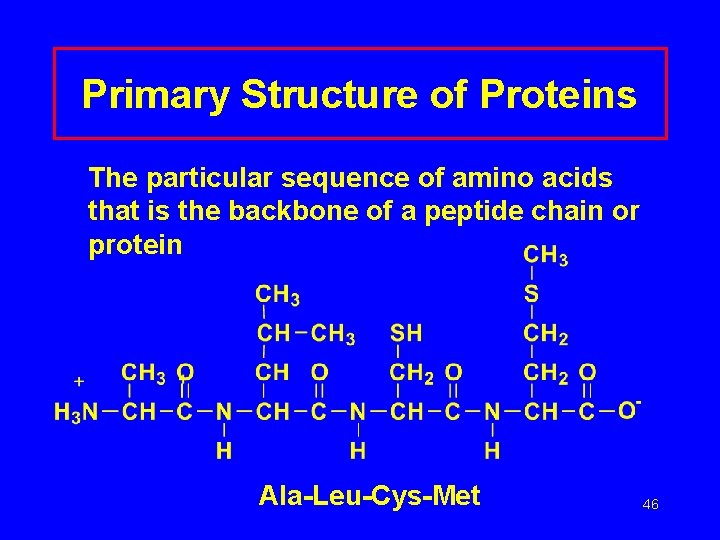
Primary Structure of Proteins The particular sequence of amino acids that is the backbone of a peptide chain or protein Ala-Leu-Cys-Met 46

Learning Check AA 3 What are the possible tripeptides formed from one each of leucine, glycine, and alanine? 47

Solution AA 3 Tripeptides possible from one each of leucine, glycine, and alanine Leu-Gly-Ala Leu-Ala-Gly Ala-Leu-Gly Ala-Gly-Leu Gly-Ala-Leu Gly-Leu-Ala 48

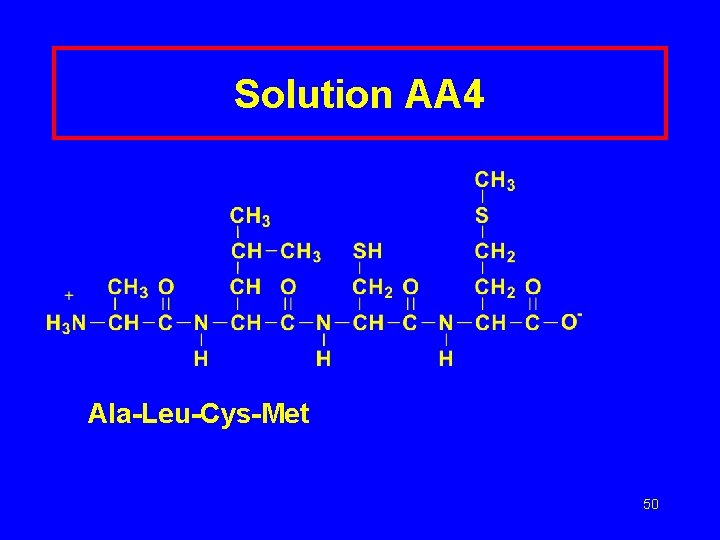
Learning Check AA 4 Write three-letter abbreviations for the following tetrapeptide: 49

Solution AA 4 Ala-Leu-Cys-Met 50

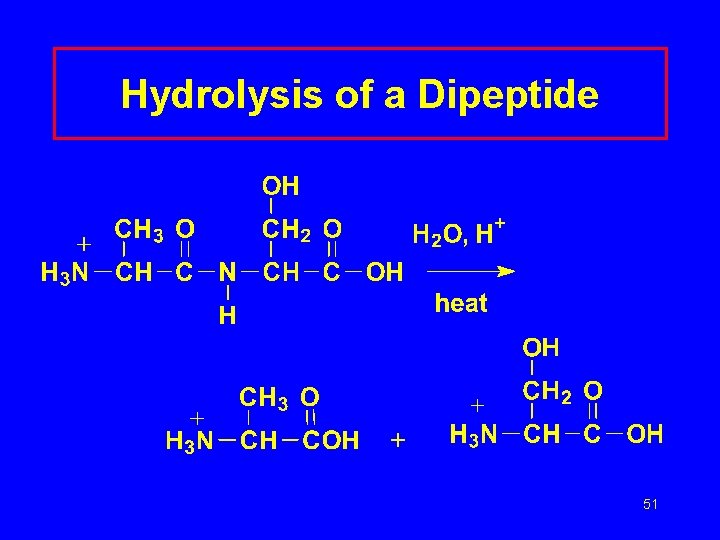
Hydrolysis of a Dipeptide 51

Learning Check P 4 What are the products of the complete hydrolysis of Ala-Ser-Val? 52

Solution P 4 The products of the complete hydrolysis of Ala-Ser-Val are alanine serine valine 53

Test Your Knowledge. Click to see the answer. Q: What amino acid has the shortest carbon chain in its R group? A: Glycine. It has no carbon in its R group. Q: What structural feature is common to alanine, serine and cyst A: All three have a single carbon in their R groups. Q: Which amino acid has the longest straight chain of carbons in its R group? A: Lysine. It has 4. Leucine and isoleucine have 4 but their chains are branched Q: What R group structural feature is common to phenylalanine, tyrosine, tryptophan, and histidine? A: All four have rings that are attached to the core via a –CH 2 Q: What structural feature is common to isoleucine and threonine A: Both have an asymmetric carbon in their R group

Peptide Bond Formation • The amino group of one molecule condenses with the acid group of another. • Polypeptides usually have molecular weight less than 5000. • Protein molecular weight 6000 -40, 000. => Chapter 24 55


Classification of Proteins • Simple: hydrolyze to amino acids only. • Conjugated: bonded to a nonprotein group, such as sugar, nucleic acid, or lipid. • Fibrous: long, stringy filaments, insoluble in water, function as structure. • Globular: folded into spherical shape, function as enzymes, hormones, or transport proteins. => Chapter 24 57

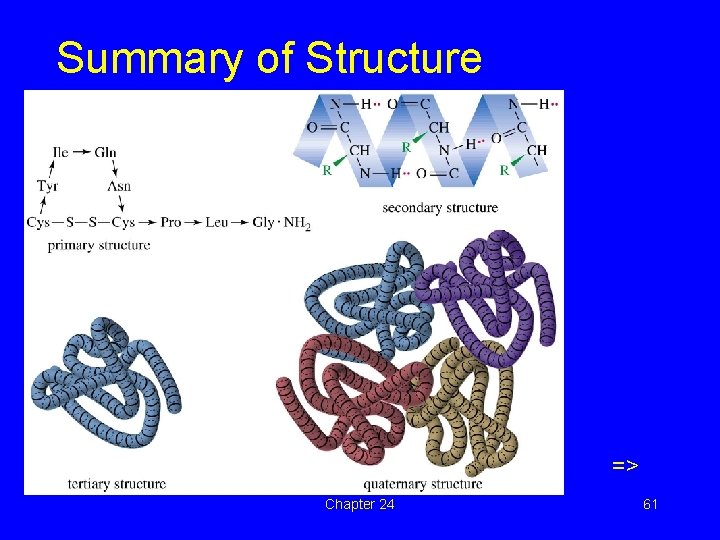
Levels of Protein Structure • Primary: the sequence of the amino acids in the chain and the disulfide links. • Secondary: structure formed by hydrogen bonding. Examples are helix and pleated sheet. • Tertiary: complete 3 -D conformation. • Quaternary: association of two or more peptide chains to form protein. => Chapter 24 58

Alpha Helix Each carbonyl oxygen can hydrogen bond with an N-H hydrogen on the next turn of the coil. => Chapter 24 59

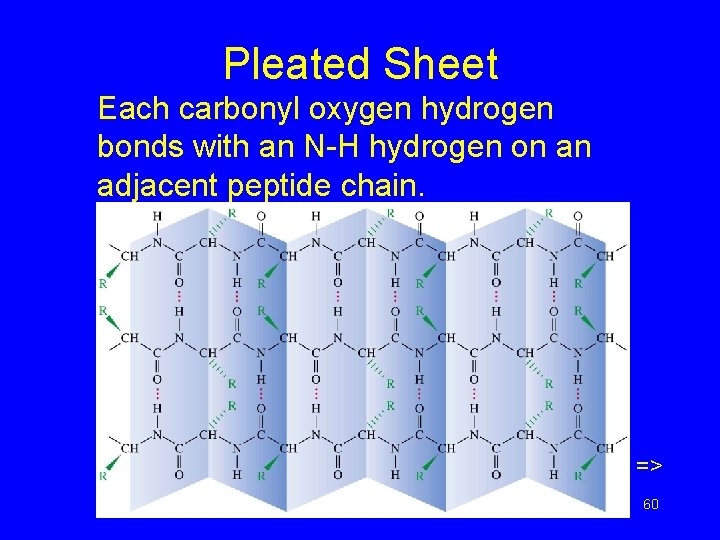
Pleated Sheet Each carbonyl oxygen hydrogen bonds with an N-H hydrogen on an adjacent peptide chain. => Chapter 24 60

Summary of Structure => Chapter 24 61

Denaturation • Disruption of the normal structure of a protein, such that it loses biological activity. • Usually caused by heat or changes in p. H. • Usually irreversible. A cooked egg cannot be “uncooked. ” => Chapter 24 62