Lab 8 Titration Curve Potentiometic Titration of amino

Lab (8): Titration Curve Potentiometic Titration of amino acids KAU-Faculty of Science- Biochemistry department Analytical biochemistry lab (Bioc 343) 2012 T. A Nouf Alshareef nf. shareef@hotmail. com

Isoelectric point (p. I): • is p. H at which amino acid or protein has no net charge (zero) • Solubility at p. I is minimum (i. e. protein precipitate) • amino acid at this point is zwitter ion. Important of p. I: • We can separate protein according to p. I (salting in/salting out). • We can use it in separation method as: Ø ion exchange chromatography Ø protein electrophoresis choose st. phase choose buffer p. H

• Proteins behave as poly-ionic macromolecules • Amino acids are amphoteric molecules: (contains at least one amino and one carboxyl group, and they have acid-base properties) when reacts with acids, it acts as a base: Acid media when reacts with bases and acts as acid: base media • Since a-carboxyl and a-amino groups are weak acids and bases, respectively, buffering action by these groups will occur.

• Proteins can be separated according to their p. I in a process known as isoelectric focusing. ü at p. H below p. I proteins carry a net positive charge. ü at p. H Above p. I proteins carry a net negative charge. • p. H of electrophoresis gel is determined by buffer used for that gel: - If p. H of buffer is above p. I of protein >>>protein will migrate to +ve pole (Negative charge is attracted to positive pole) - If p. H of buffer is below p. I >>>>>protein will migrate to –ve pole of gel (Positive charge is attracted to negative pole). - If protein is run with a buffer p. H that is equal to p. I, it will not migrate. • This also true for individual amino acids.

Titration curve • To determine p. I of amino acid or protein, we will titrate it with base and then with acid • A curve will be drown between p. H and volume of acid or base. • From the curve we can determine: - p. Ka - p. Kb • So, p. I = halfway between two points of strongest buffering capacity. PI = (p. K 1 + p. K 2) 2
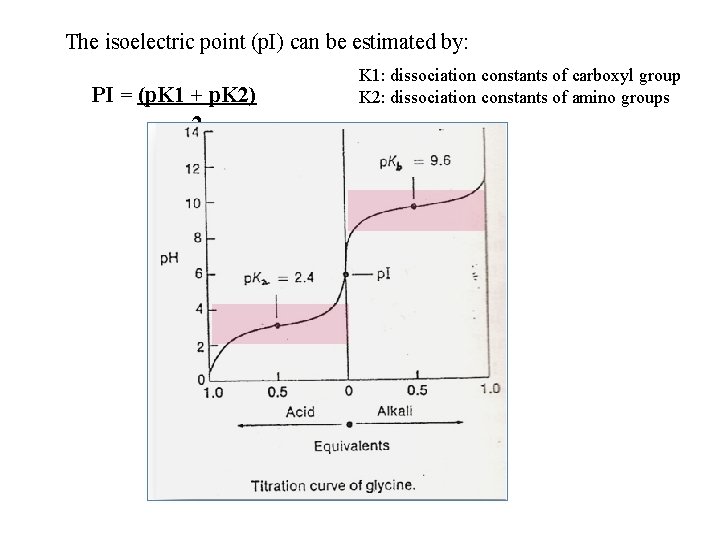
The isoelectric point (p. I) can be estimated by: PI = (p. K 1 + p. K 2) 2 K 1: dissociation constants of carboxyl group K 2: dissociation constants of amino groups
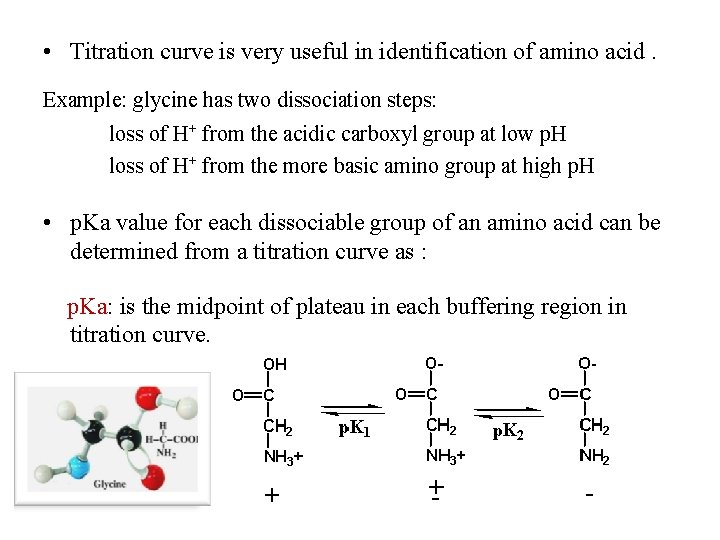
• Titration curve is very useful in identification of amino acid. Example: glycine has two dissociation steps: loss of H+ from the acidic carboxyl group at low p. H loss of H+ from the more basic amino group at high p. H • p. Ka value for each dissociable group of an amino acid can be determined from a titration curve as : p. Ka: is the midpoint of plateau in each buffering region in titration curve.

Lab practice: Determination of p. I of amino acids Procedure: • 20 ml of your selected amino acid solution into the a beaker. • Assemble: titration stand, burette and p. H meter. • Record the initial p. H of the amino acid solution and the initial reading of your burette (Which should be 0. 0 ml)

• Turn on the stirrer, check that the magnetic bar does not collide with the electrodes. Base titration: • Titrate with base by dropwise addition of the 0. 5 N Na. OH. • Record the volume addition of Na. OH and p. H of the solution throughout the titration until a p. H > 12 is reached. Acid titration: • Repeat the titration with 0. 25 N H 2 SO 4. • Record the volume of added H 2 SO 4 and p. H of the solution throughout the titration until a p. H below 1. 5 is reached.
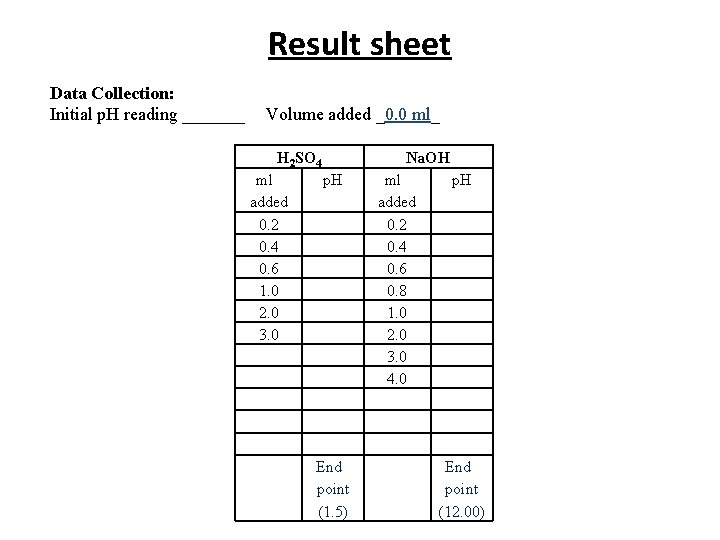
Result sheet Data Collection: Initial p. H reading _______ Volume added _0. 0 ml_ H 2 SO 4 ml added 0. 2 0. 4 0. 6 1. 0 2. 0 3. 0 Na. OH p. H End point (1. 5) ml added 0. 2 0. 4 0. 6 0. 8 1. 0 2. 0 3. 0 4. 0 p. H End point (12. 00)

Answer the following questions: • Plot p. H versus ml of Na. OH and H 2 SO 4 solutions added. • Use p. H as the dependent variable (Y-axis) and ml of Na. OH or H 2 SO 4 as the independent variable (X-axis) Determine the p. I of your amino acid from curve and by calculation?
- Slides: 11