CAMPBELL BIOLOGY IN FOCUS Urry Cain Wasserman Minorsky

CAMPBELL BIOLOGY IN FOCUS Urry • Cain • Wasserman • Minorsky • Jackson • Reece 3 Carbon and the Molecular Diversity of Life Lecture Presentations by Kathleen Fitzpatrick and Nicole Tunbridge © 2014 Pearson Education, Inc.


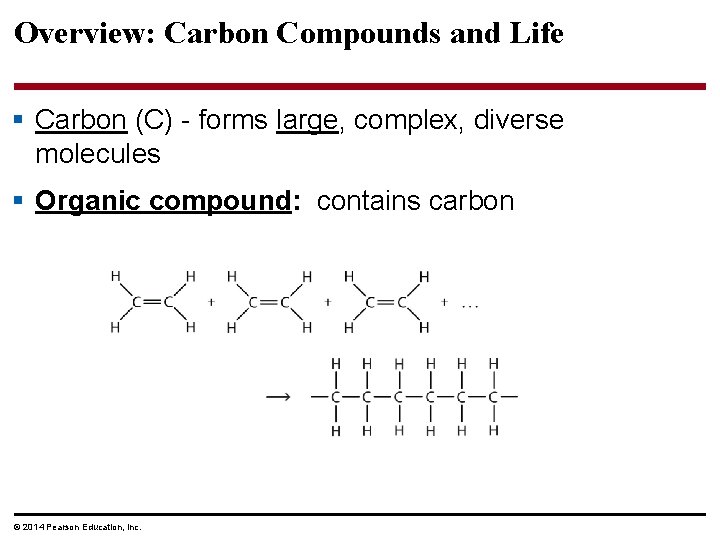
Overview: Carbon Compounds and Life § Carbon (C) - forms large, complex, diverse molecules § Organic compound: contains carbon © 2014 Pearson Education, Inc.
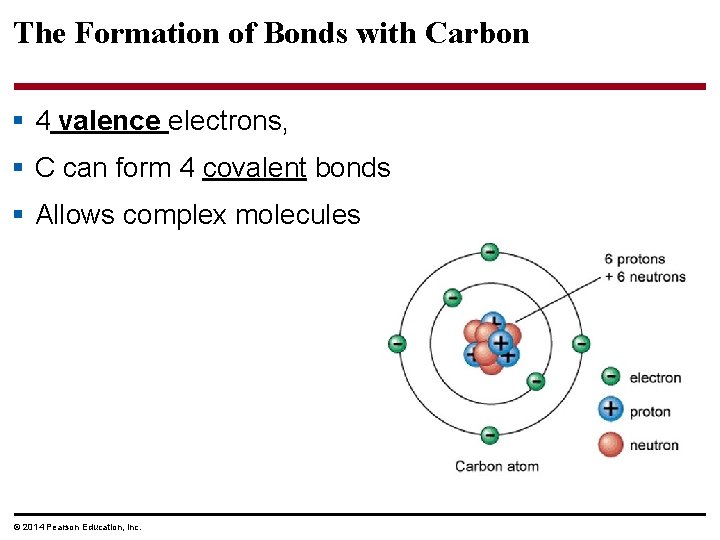
The Formation of Bonds with Carbon § 4 valence electrons, § C can form 4 covalent bonds § Allows complex molecules © 2014 Pearson Education, Inc.
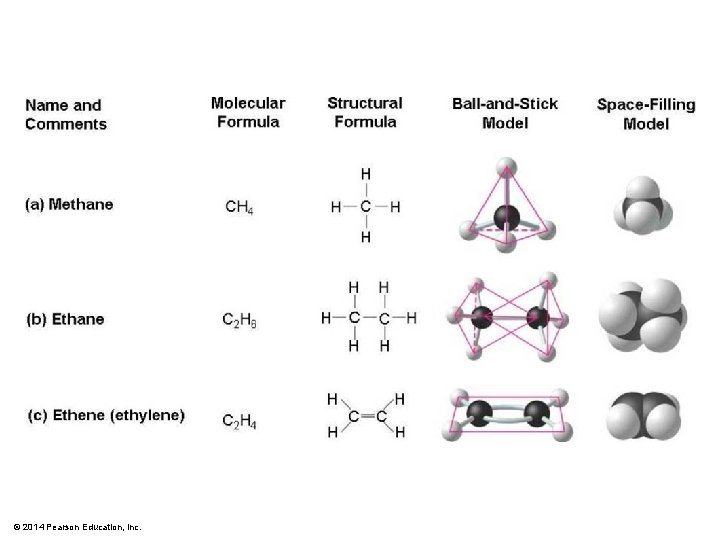
© 2014 Pearson Education, Inc.
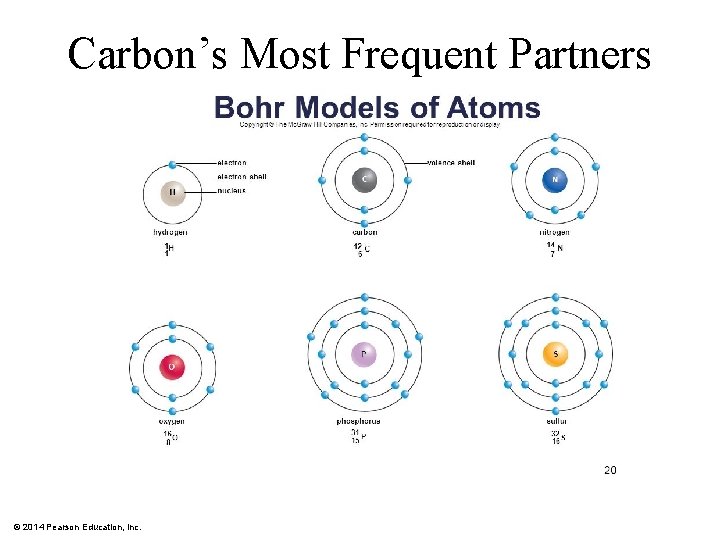
Carbon’s Most Frequent Partners © 2014 Pearson Education, Inc.

Molecular Diversity Arising from Variation in Carbon Skeletons § Carbon chains form skeletons of most organic molecules § Chains may be straight, branched, or in rings © 2014 Pearson Education, Inc.
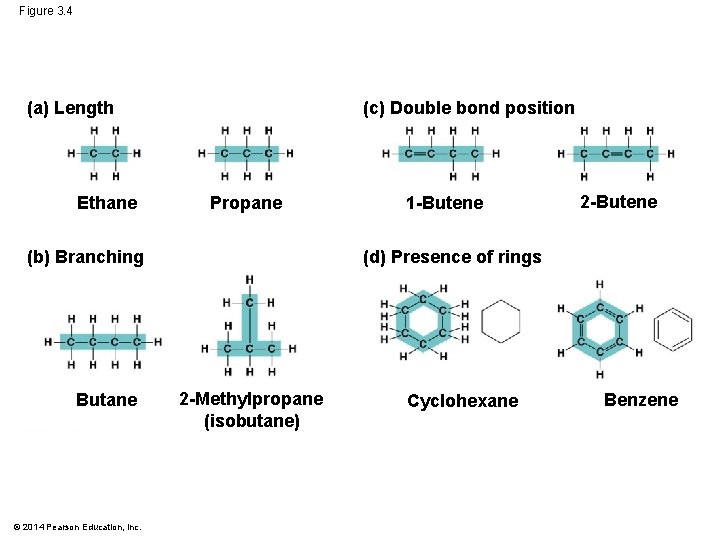
Figure 3. 4 (a) Length Ethane (c) Double bond position Propane (b) Branching Butane © 2014 Pearson Education, Inc. 1 -Butene 2 -Butene (d) Presence of rings 2 -Methylpropane (isobutane) Cyclohexane Benzene
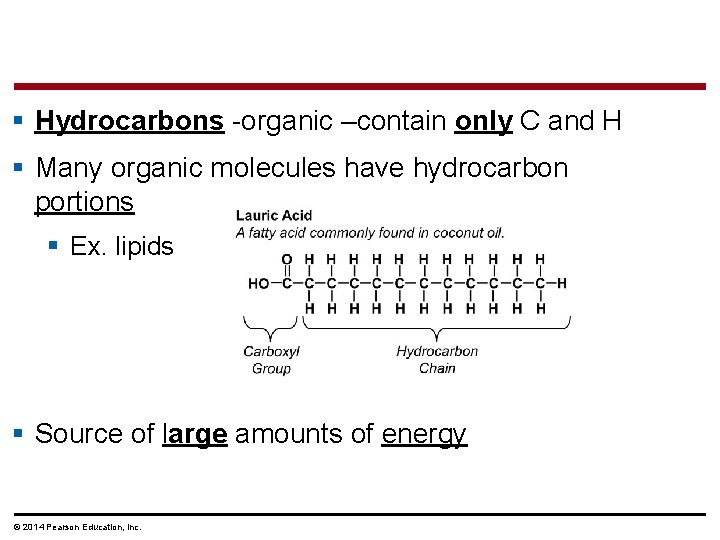
§ Hydrocarbons -organic –contain only C and H § Many organic molecules have hydrocarbon portions § Ex. lipids § Source of large amounts of energy © 2014 Pearson Education, Inc.
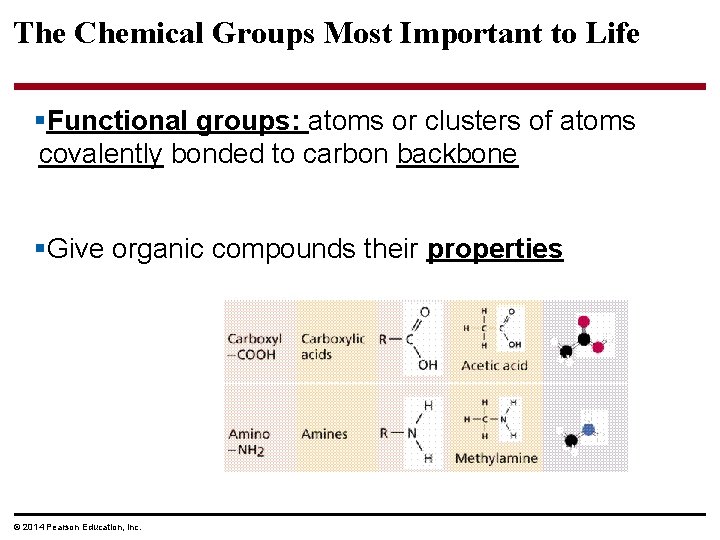
The Chemical Groups Most Important to Life §Functional groups: atoms or clusters of atoms covalently bonded to carbon backbone §Give organic compounds their properties © 2014 Pearson Education, Inc.

§ The seven functional groups that are most important in the chemistry of life: § Hydroxyl group - OH § Carbonyl group - C=O § Carboxyl group - COOH § Amino group - NH 3+ § Sulfhydryl group - SH § Phosphate group - PO 3§ Methyl group - CH 3 © 2014 Pearson Education, Inc.
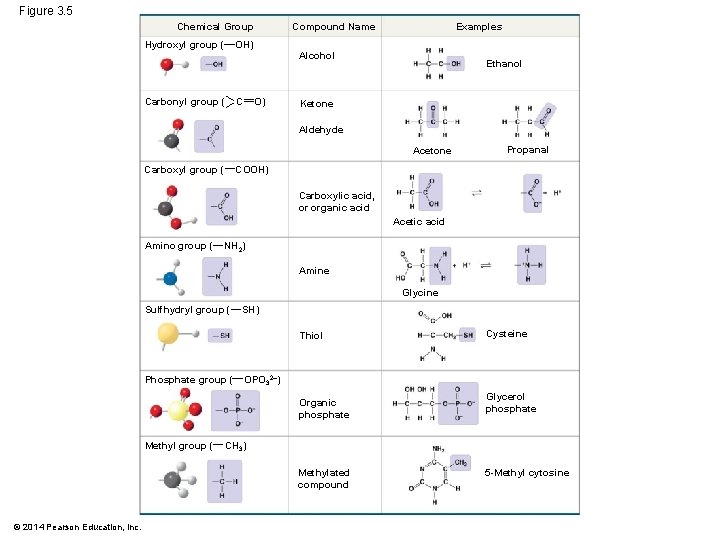
Figure 3. 5 Chemical Group Hydroxyl group ( Compound Name OH) Carbonyl group ( C Examples Alcohol O) Ethanol Ketone Aldehyde Acetone Carboxyl group ( Propanal COOH) Carboxylic acid, or organic acid Acetic acid Amino group ( NH 2) Amine Glycine Sulfhydryl group ( SH) Thiol Phosphate group ( OPO 32–) Organic phosphate Methyl group ( Glycerol phosphate CH 3) Methylated compound © 2014 Pearson Education, Inc. Cysteine 5 -Methyl cytosine
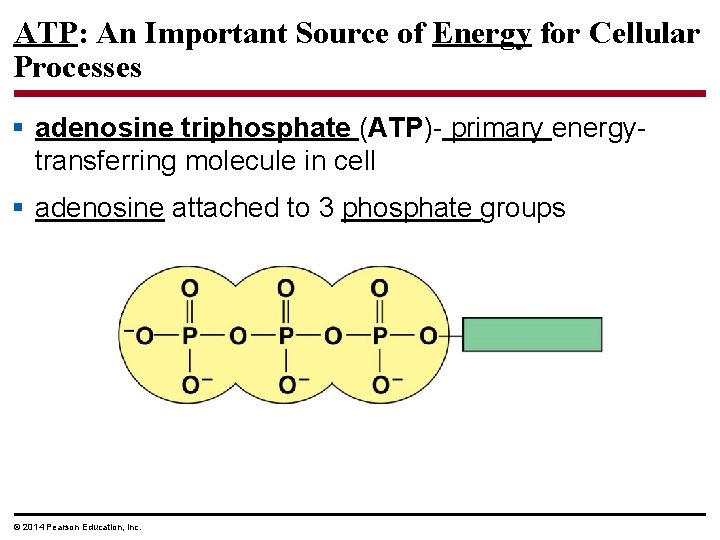
ATP: An Important Source of Energy for Cellular Processes § adenosine triphosphate (ATP)- primary energytransferring molecule in cell § adenosine attached to 3 phosphate groups © 2014 Pearson Education, Inc.
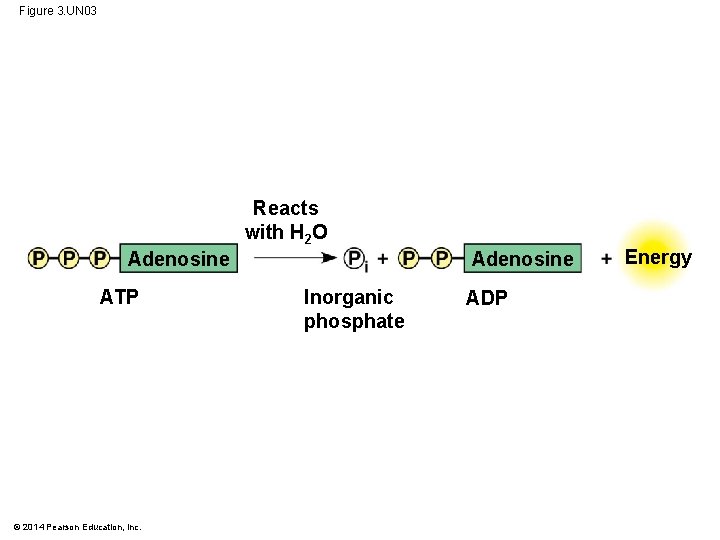
Figure 3. UN 03 Reacts with H 2 O Adenosine ATP © 2014 Pearson Education, Inc. Inorganic phosphate ADP Energy
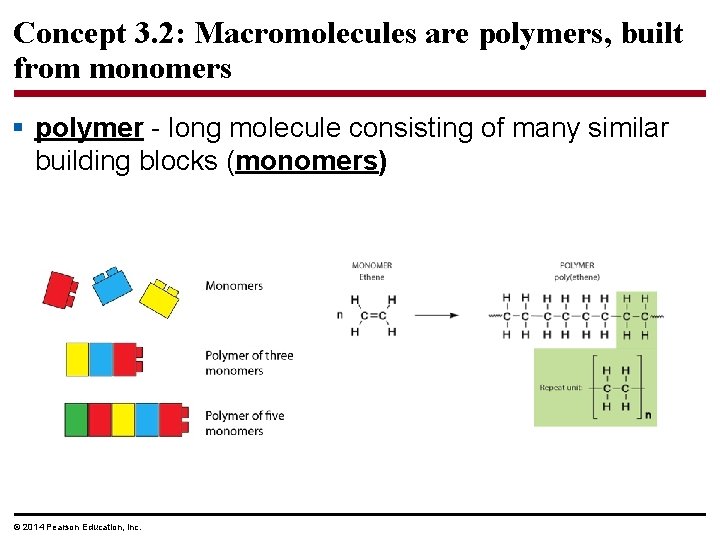
Concept 3. 2: Macromolecules are polymers, built from monomers § polymer - long molecule consisting of many similar building blocks (monomers) © 2014 Pearson Education, Inc.

How Do Macromolecules Form? §Dehydration synthesis: occurs when two monomers bond together through the loss of a water molecule
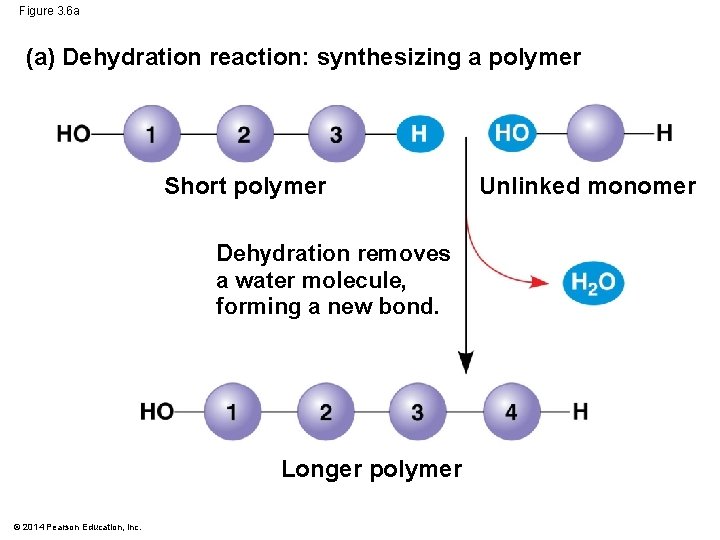
Figure 3. 6 a (a) Dehydration reaction: synthesizing a polymer Short polymer Dehydration removes a water molecule, forming a new bond. Longer polymer © 2014 Pearson Education, Inc. Unlinked monomer
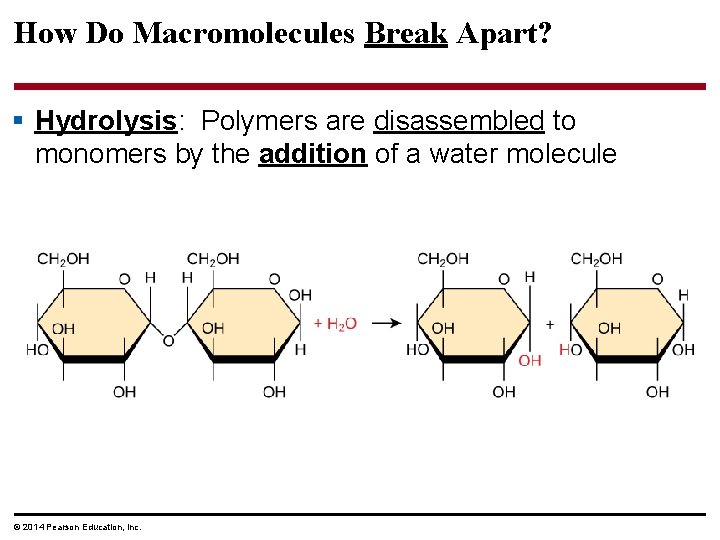
How Do Macromolecules Break Apart? § Hydrolysis: Polymers are disassembled to monomers by the addition of a water molecule © 2014 Pearson Education, Inc.
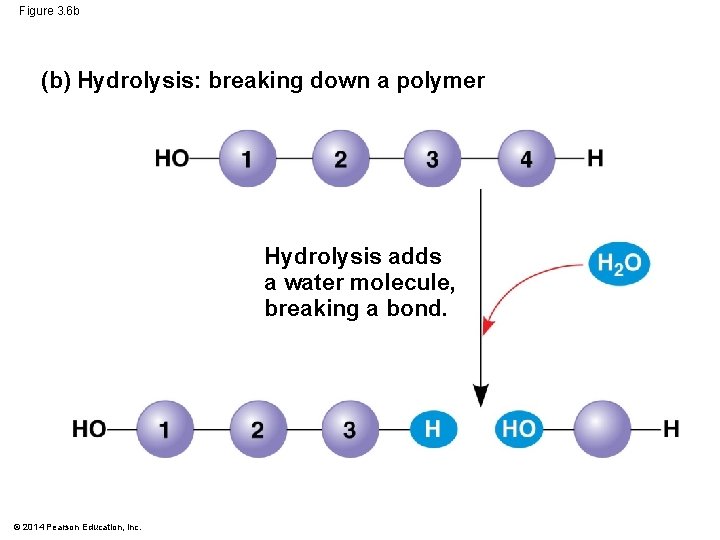
Figure 3. 6 b (b) Hydrolysis: breaking down a polymer Hydrolysis adds a water molecule, breaking a bond. © 2014 Pearson Education, Inc.
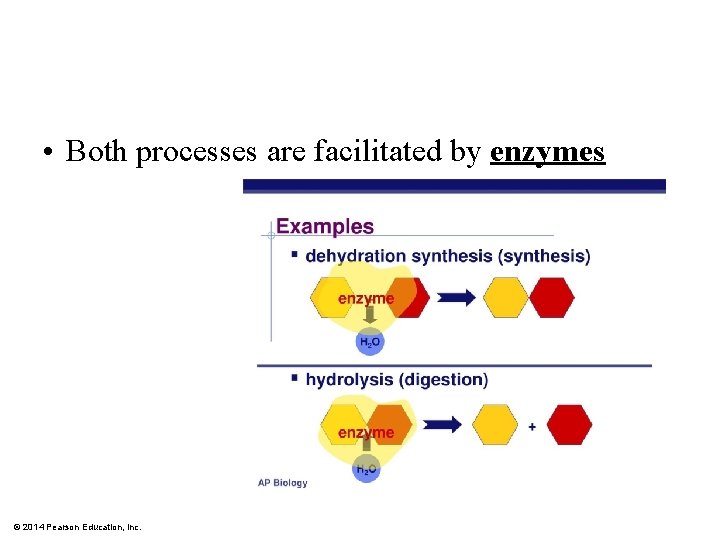
• Both processes are facilitated by enzymes © 2014 Pearson Education, Inc.

§ 4 Classes of Organic Compounds § Carbohydrates § Lipids § Proteins § Nucleic acids § Biomolecules (8 min) https: //www. youtube. com/watch? v=YO 244 P 1 e 9 QM &t=262 s © 2014 Pearson Education, Inc.

Concept 3. 3: Carbohydrates serve as fuel and building material § Carbohydrates: sugars and polymers of sugars © 2014 Pearson Education, Inc.
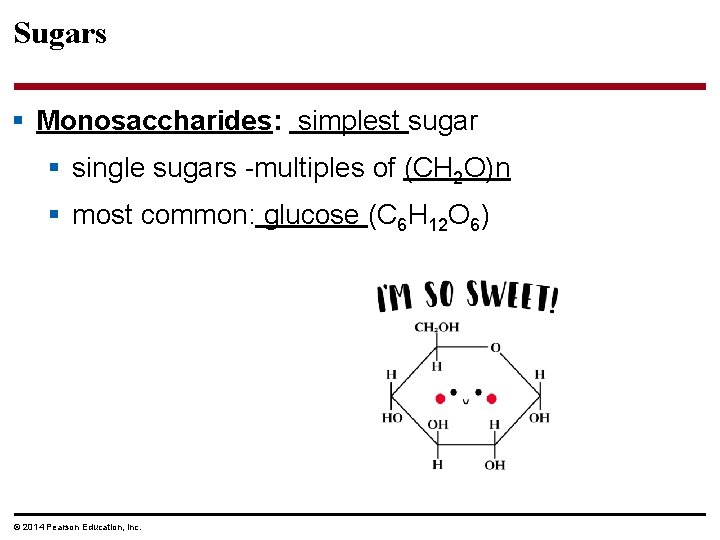
Sugars § Monosaccharides: simplest sugar § single sugars -multiples of (CH 2 O)n § most common: glucose (C 6 H 12 O 6) © 2014 Pearson Education, Inc.
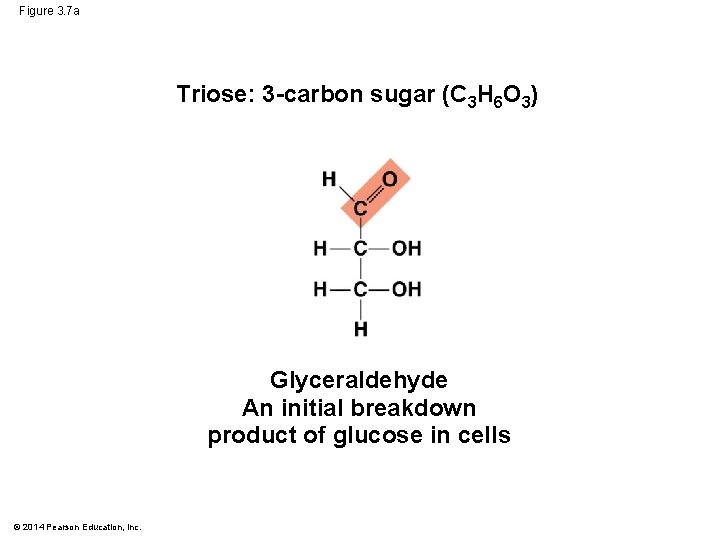
Figure 3. 7 a Triose: 3 -carbon sugar (C 3 H 6 O 3) Glyceraldehyde An initial breakdown product of glucose in cells © 2014 Pearson Education, Inc.
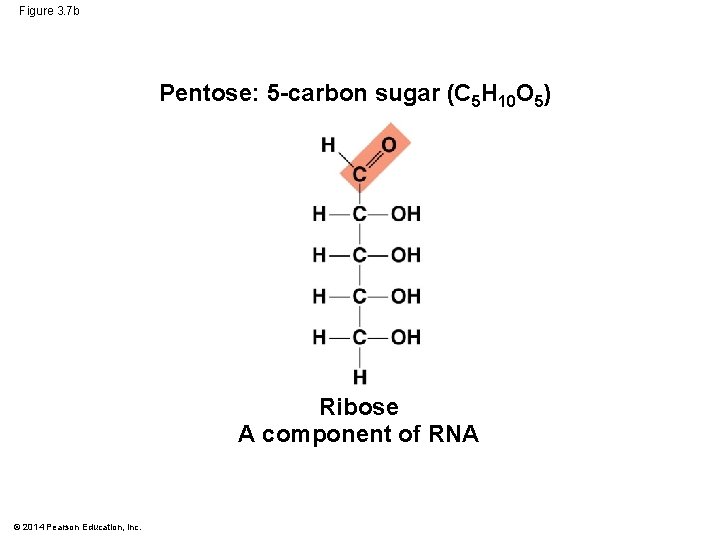
Figure 3. 7 b Pentose: 5 -carbon sugar (C 5 H 10 O 5) Ribose A component of RNA © 2014 Pearson Education, Inc.
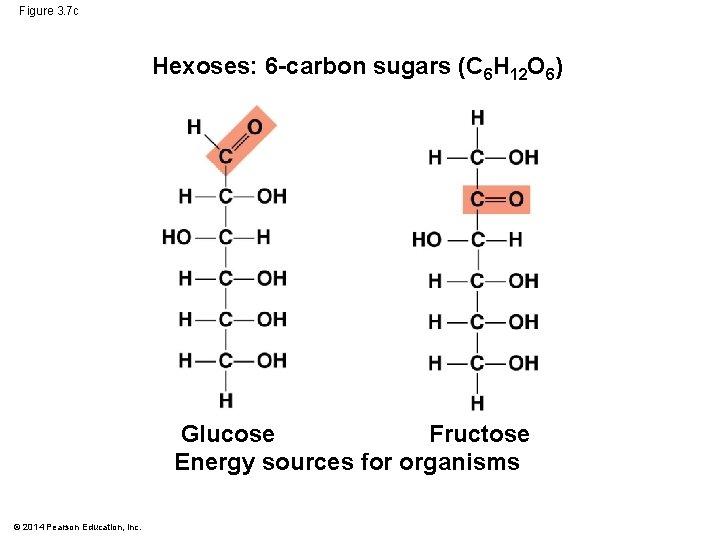
Figure 3. 7 c Hexoses: 6 -carbon sugars (C 6 H 12 O 6) Glucose Fructose Energy sources for organisms © 2014 Pearson Education, Inc.
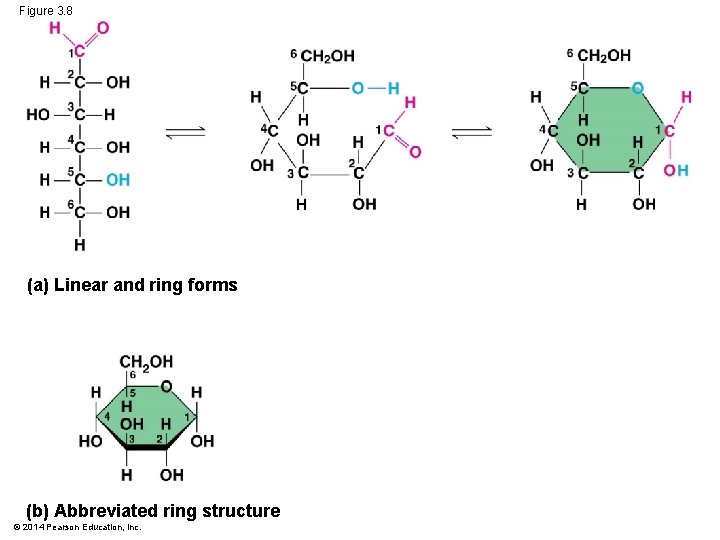
Figure 3. 8 (a) Linear and ring forms (b) Abbreviated ring structure © 2014 Pearson Education, Inc.
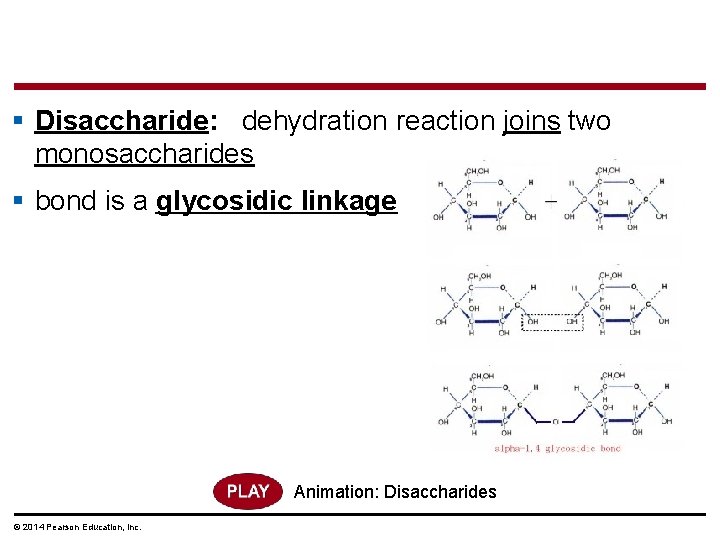
§ Disaccharide: dehydration reaction joins two monosaccharides § bond is a glycosidic linkage Animation: Disaccharides © 2014 Pearson Education, Inc.
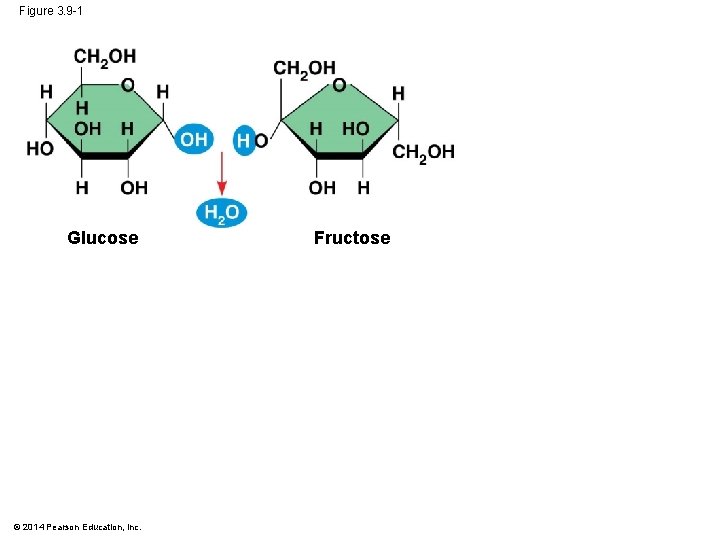
Figure 3. 9 -1 Glucose © 2014 Pearson Education, Inc. Fructose
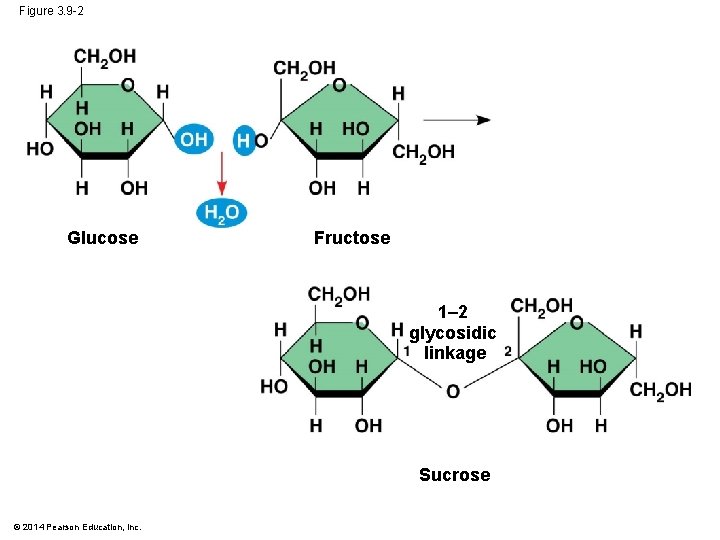
Figure 3. 9 -2 Glucose Fructose 1– 2 glycosidic linkage Sucrose © 2014 Pearson Education, Inc.
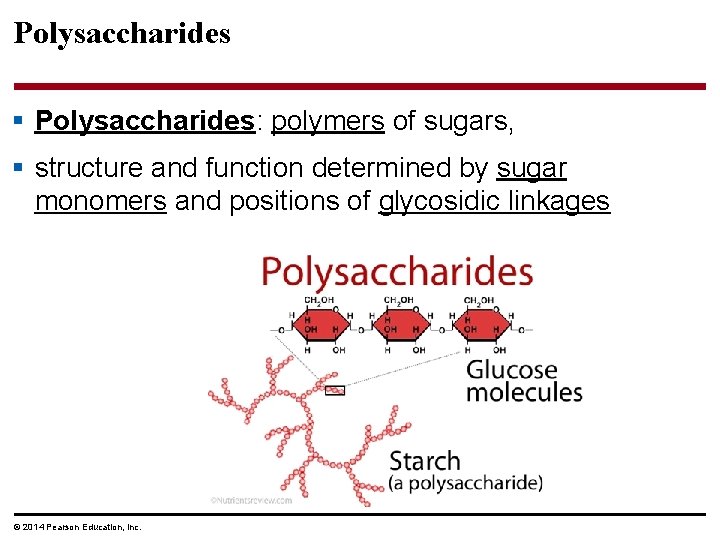
Polysaccharides § Polysaccharides: polymers of sugars, § structure and function determined by sugar monomers and positions of glycosidic linkages © 2014 Pearson Education, Inc.
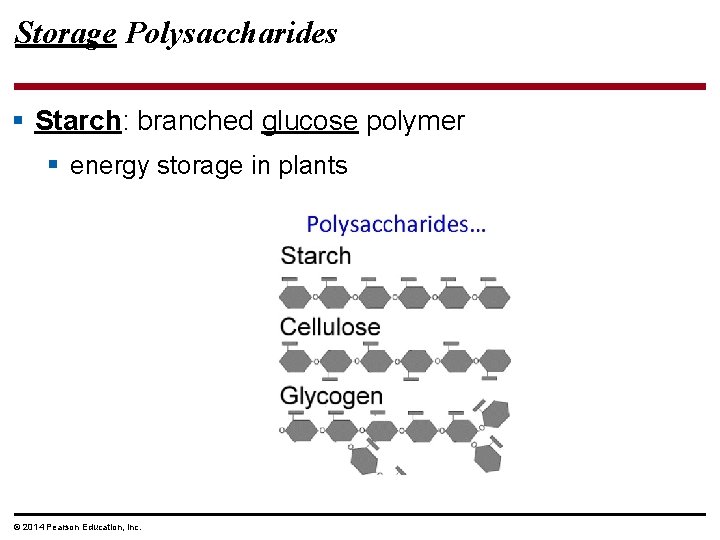
Storage Polysaccharides § Starch: branched glucose polymer § energy storage in plants © 2014 Pearson Education, Inc.
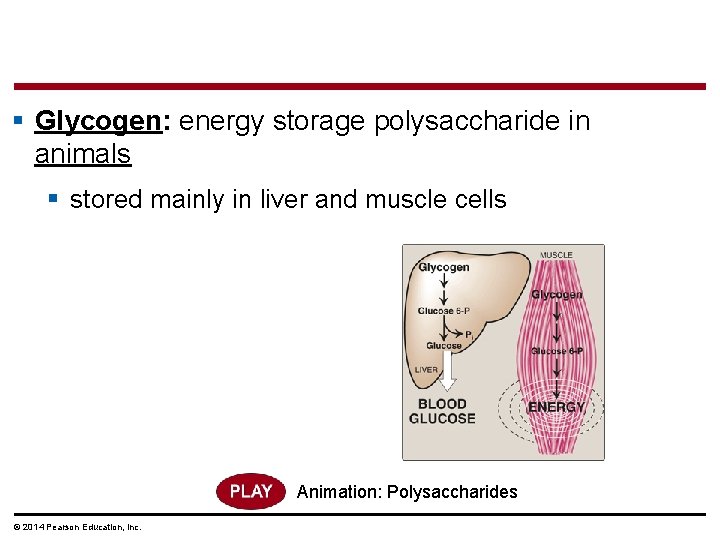
§ Glycogen: energy storage polysaccharide in animals § stored mainly in liver and muscle cells Animation: Polysaccharides © 2014 Pearson Education, Inc.
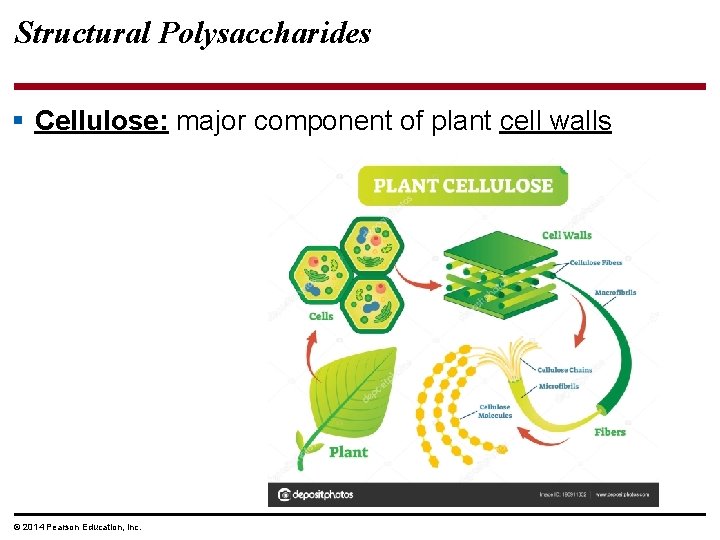
Structural Polysaccharides § Cellulose: major component of plant cell walls © 2014 Pearson Education, Inc.
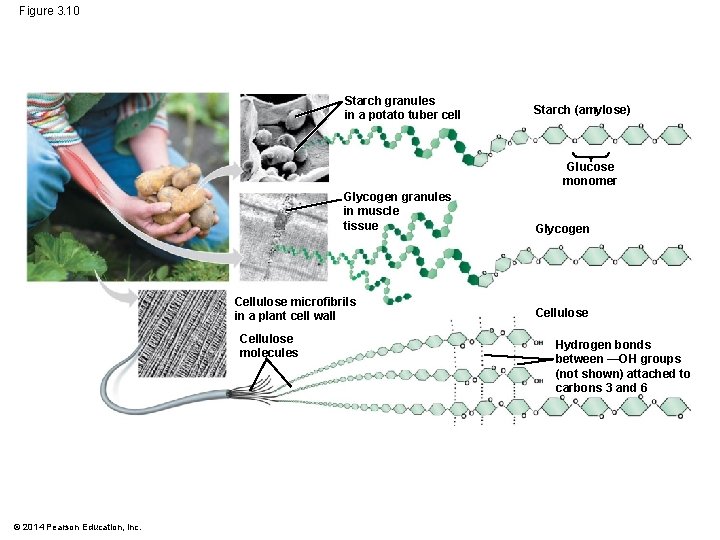
Figure 3. 10 Starch granules in a potato tuber cell Starch (amylose) Glucose monomer Glycogen granules in muscle tissue Cellulose microfibrils in a plant cell wall Cellulose molecules © 2014 Pearson Education, Inc. Glycogen Cellulose Hydrogen bonds between —OH groups (not shown) attached to carbons 3 and 6

§ Chitin: structural polysaccharide in exoskeletons of arthropods and fungal cell walls © 2014 Pearson Education, Inc.
Concept 3. 4: Lipids are a diverse group of hydrophobic molecules § Lipids most biologically important are fats, phospholipids, and steroids © 2014 Pearson Education, Inc.

Fats § Fats energy storage in animals § Molecular building blocks: § glycerol bonded to 3 fatty acids § joined by an ester linkage, creating a triacylglycerol (triglyceride) Animation: Fats © 2014 Pearson Education, Inc.
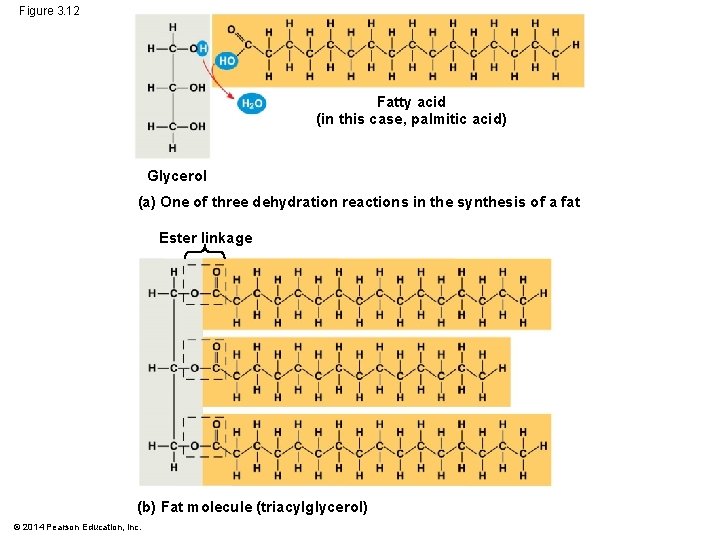
Figure 3. 12 Fatty acid (in this case, palmitic acid) Glycerol (a) One of three dehydration reactions in the synthesis of a fat Ester linkage (b) Fat molecule (triacylglycerol) © 2014 Pearson Education, Inc.
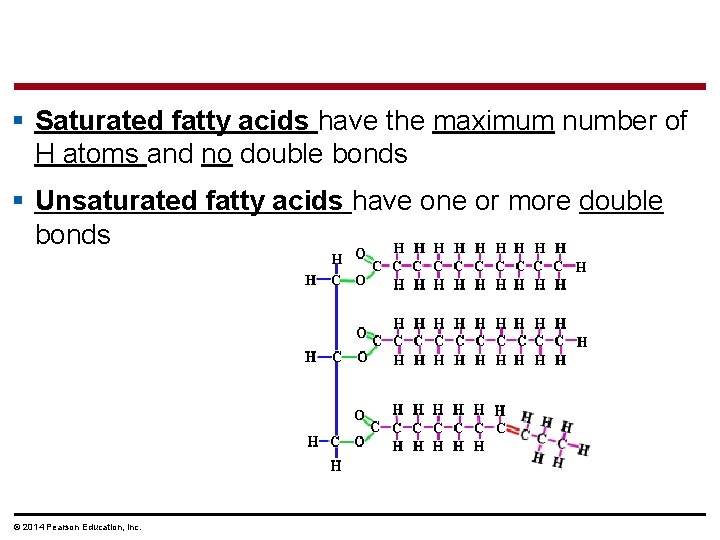
§ Saturated fatty acids have the maximum number of H atoms and no double bonds § Unsaturated fatty acids have one or more double bonds © 2014 Pearson Education, Inc.
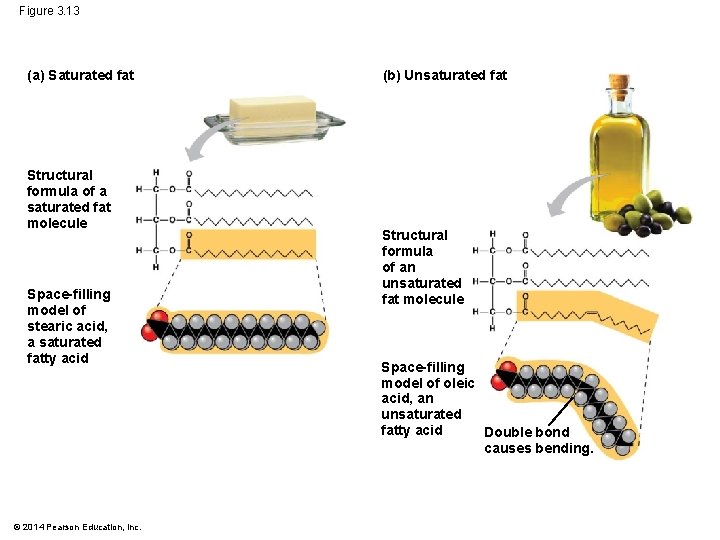
Figure 3. 13 (a) Saturated fat Structural formula of a saturated fat molecule Space-filling model of stearic acid, a saturated fatty acid © 2014 Pearson Education, Inc. (b) Unsaturated fat Structural formula of an unsaturated fat molecule Space-filling model of oleic acid, an unsaturated fatty acid Double bond causes bending.

Phospholipids § phospholipid, 2 fatty acids and a phosphate group are attached to glycerol § fatty acid tails are hydrophobic, phosphate group forms a hydrophilic head § Phospholipids are major constituents of cell membranes © 2014 Pearson Education, Inc.
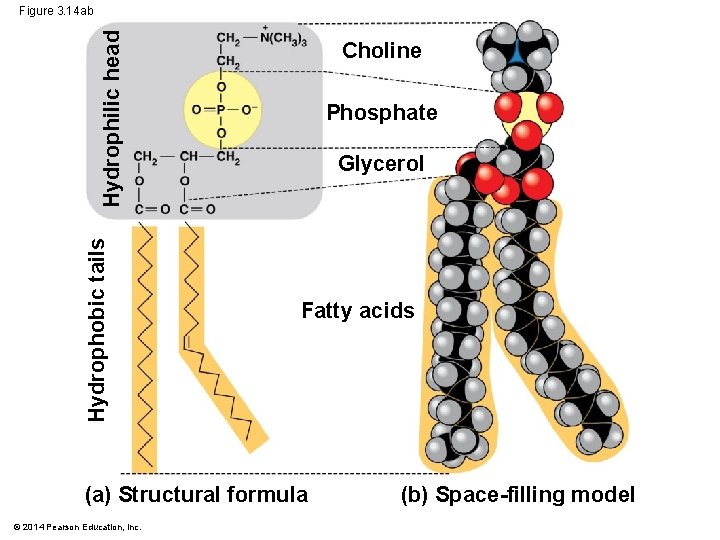
Hydrophobic tails Hydrophilic head Figure 3. 14 ab Choline Phosphate Glycerol Fatty acids (a) Structural formula © 2014 Pearson Education, Inc. (b) Space-filling model

§ phospholipids in water self-assemble into a bilayer, hydrophobic tails pointing toward interior © 2014 Pearson Education, Inc.
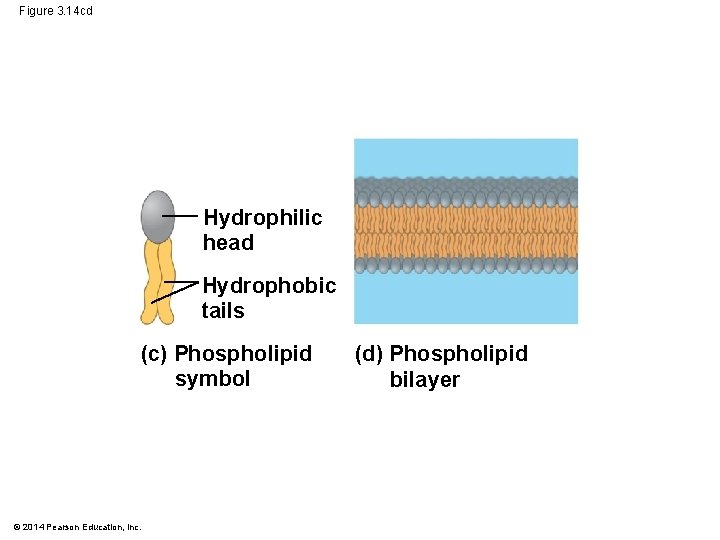
Figure 3. 14 cd Hydrophilic head Hydrophobic tails (c) Phospholipid symbol © 2014 Pearson Education, Inc. (d) Phospholipid bilayer
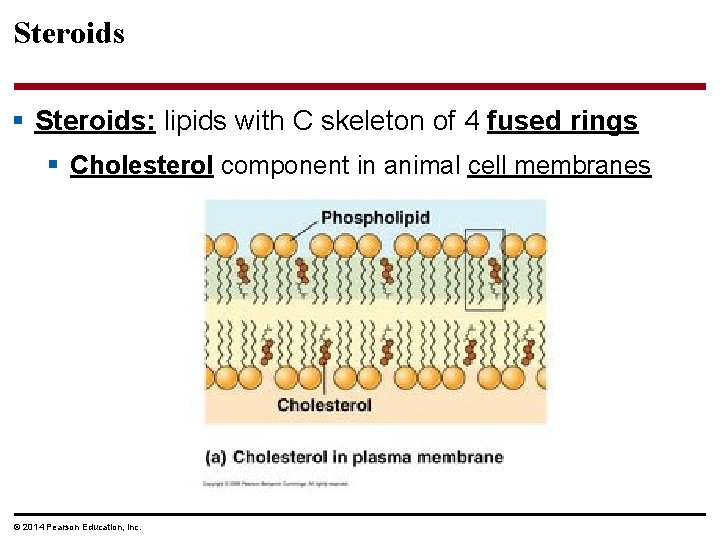
Steroids § Steroids: lipids with C skeleton of 4 fused rings § Cholesterol component in animal cell membranes © 2014 Pearson Education, Inc.
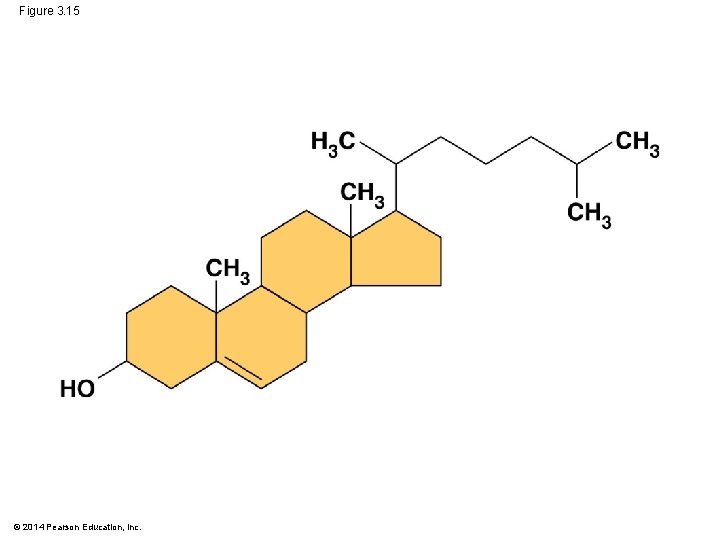
Figure 3. 15 © 2014 Pearson Education, Inc.

Concept 3. 5 Proteins § Protein - molecule made of one or more polypeptides § Polypeptides: unbranched polymers built from the same set of 20 amino acids § Account for more than 50% of a cell’s dry mass, have numerous functions § Human has tens of thousands of different proteins, each with a specific structure and function

Amino Acids § Amino acids consist of: § A central carbon atom (alpha) § a carboxyl (-COOH) group § an amino (-NH 2) group § an R group § The identity of the amino acid is determined by R group © 2014 Pearson Education, Inc.
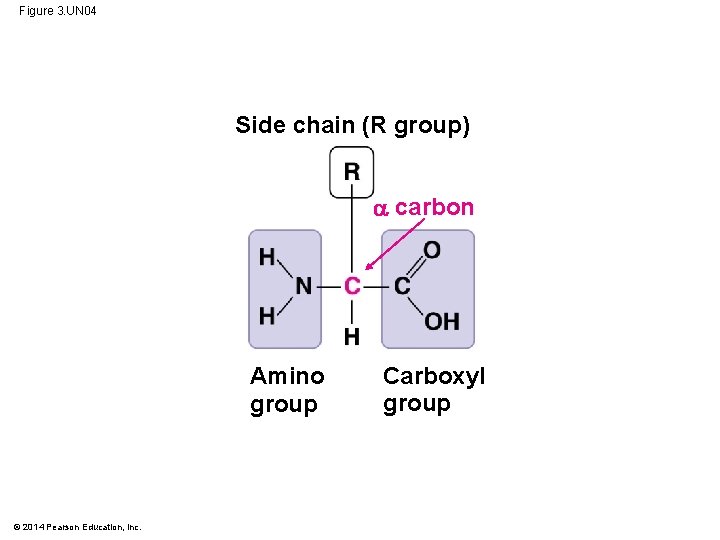
Figure 3. UN 04 Side chain (R group) carbon Amino group © 2014 Pearson Education, Inc. Carboxyl group

• The properties of the side chain determine an amino acid’s chemical behavior (acidic, basic, polar, nonpolar) © 2014 Pearson Education, Inc.
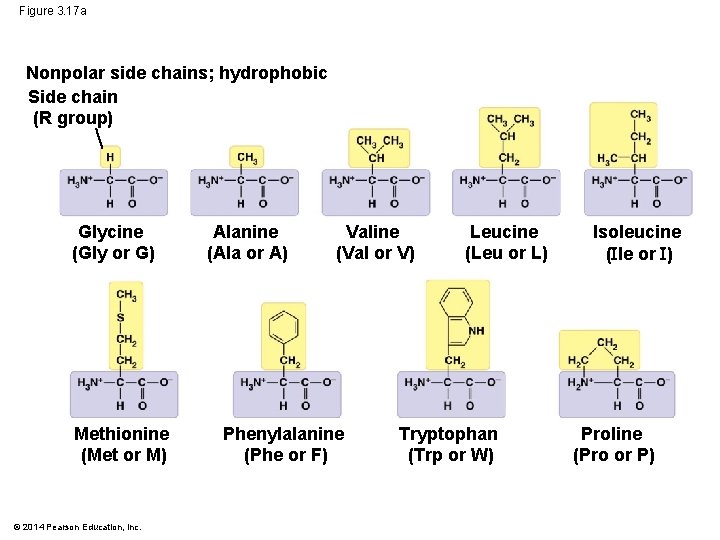
Figure 3. 17 a Nonpolar side chains; hydrophobic Side chain (R group) Glycine (Gly or G) Methionine (Met or M) © 2014 Pearson Education, Inc. Alanine (Ala or A) Valine (Val or V) Phenylalanine (Phe or F) Leucine (Leu or L) Tryptophan (Trp or W) Isoleucine ( le or ) Proline (Pro or P)
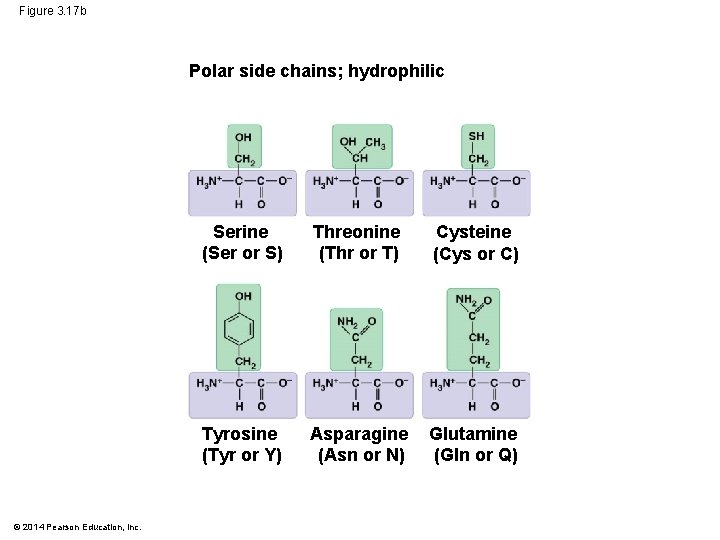
Figure 3. 17 b Polar side chains; hydrophilic © 2014 Pearson Education, Inc. Serine (Ser or S) Threonine (Thr or T) Cysteine (Cys or C) Tyrosine (Tyr or Y) Asparagine (Asn or N) Glutamine (Gln or Q)
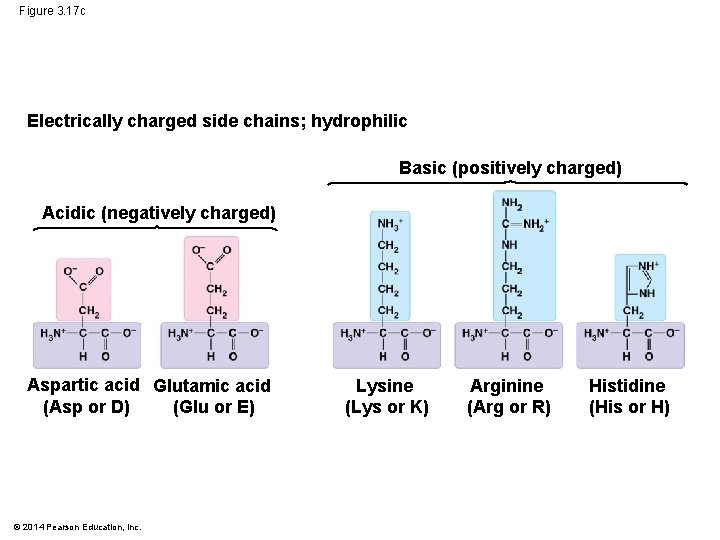
Figure 3. 17 c Electrically charged side chains; hydrophilic Basic (positively charged) Acidic (negatively charged) Aspartic acid Glutamic acid (Asp or D) (Glu or E) © 2014 Pearson Education, Inc. Lysine (Lys or K) Arginine (Arg or R) Histidine (His or H)

Polypeptides § Amino acids linked by peptide bonds (covalent) § Process? § Dehydration synthesis © 2014 Pearson Education, Inc.
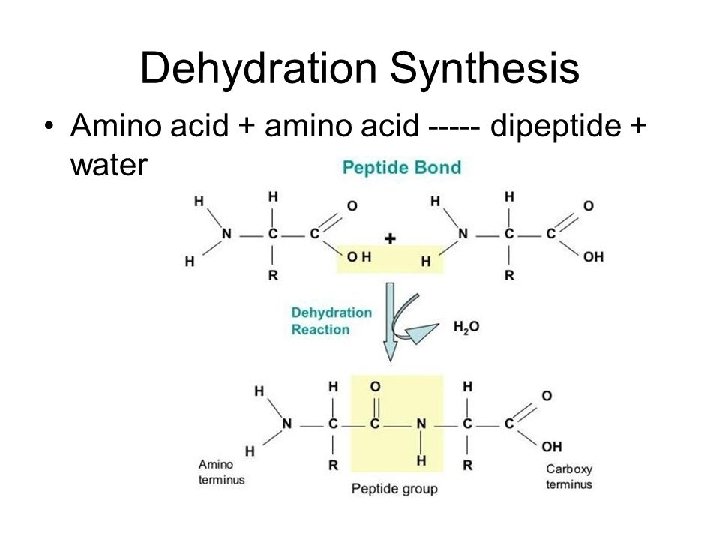
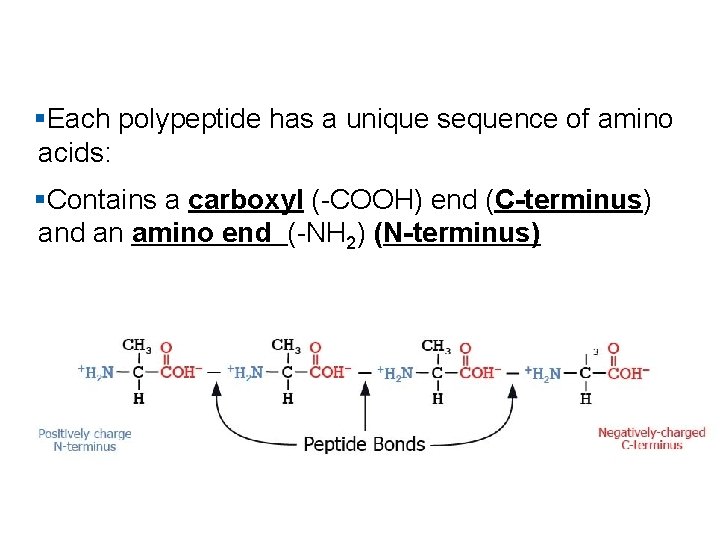
§Each polypeptide has a unique sequence of amino acids: §Contains a carboxyl (-COOH) end (C-terminus) and an amino end (-NH 2) (N-terminus)
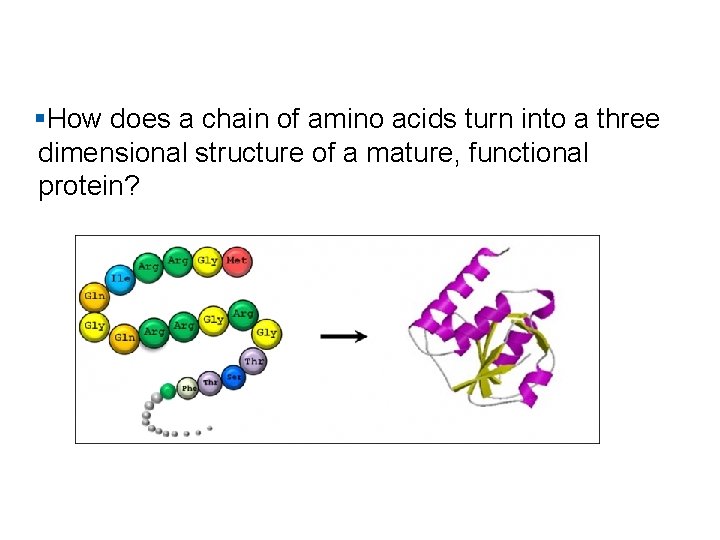
§How does a chain of amino acids turn into a three dimensional structure of a mature, functional protein?
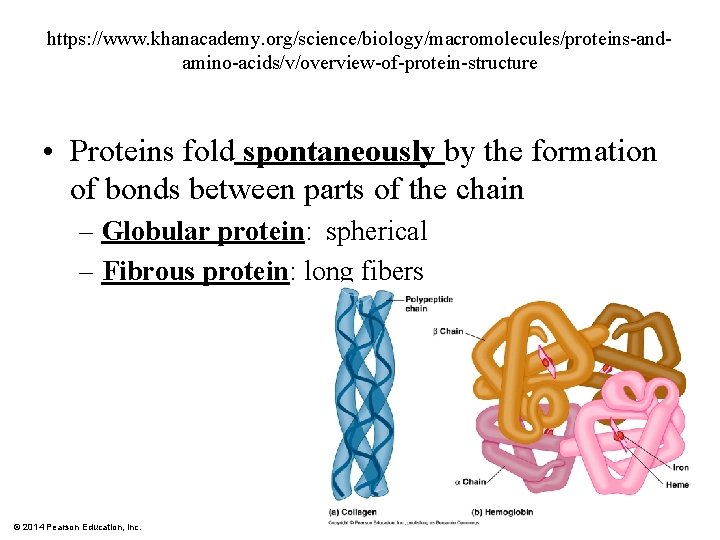
https: //www. khanacademy. org/science/biology/macromolecules/proteins-andamino-acids/v/overview-of-protein-structure • Proteins fold spontaneously by the formation of bonds between parts of the chain – Globular protein: spherical – Fibrous protein: long fibers © 2014 Pearson Education, Inc.
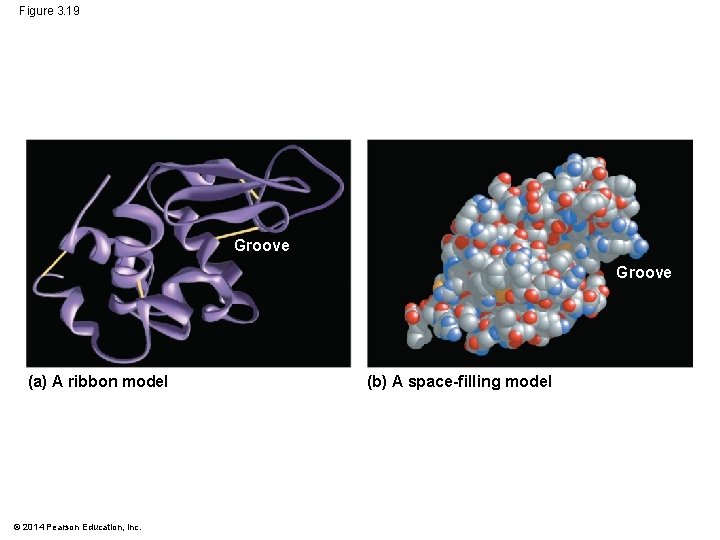
Figure 3. 19 Groove (a) A ribbon model © 2014 Pearson Education, Inc. (b) A space-filling model
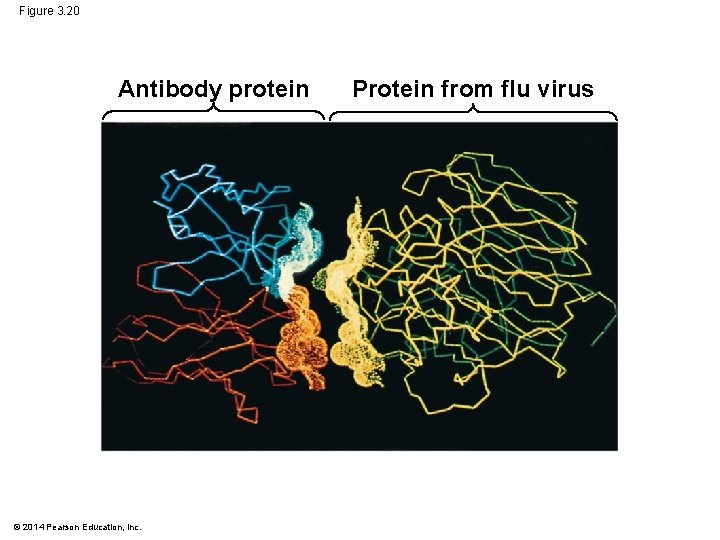
Figure 3. 20 Antibody protein © 2014 Pearson Education, Inc. Protein from flu virus

4 Levels of Protein Structure § Primary § Secondary § Tertiary § Quaternary © 2014 Pearson Education, Inc.
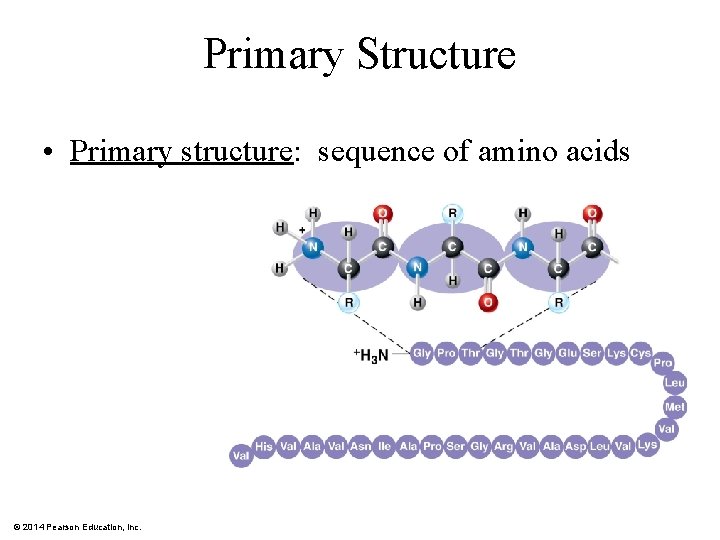
Primary Structure • Primary structure: sequence of amino acids © 2014 Pearson Education, Inc.

Secondary Structure: contains coils and folds §Result of hydrogen bonds between sections of the polypeptide backbone §Ex. Alpha helix : coil held together by hydrogen bonds between every fourth amino acid § Beta pleated sheets: two or more segments of the polypeptide chain laying side by side are connected by hydrogen bonds
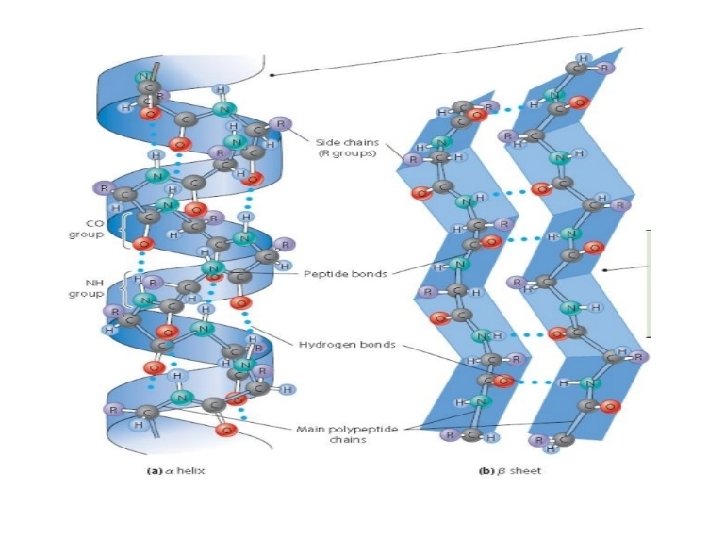

Figure 3. 21 c © 2014 Pearson Education, Inc.
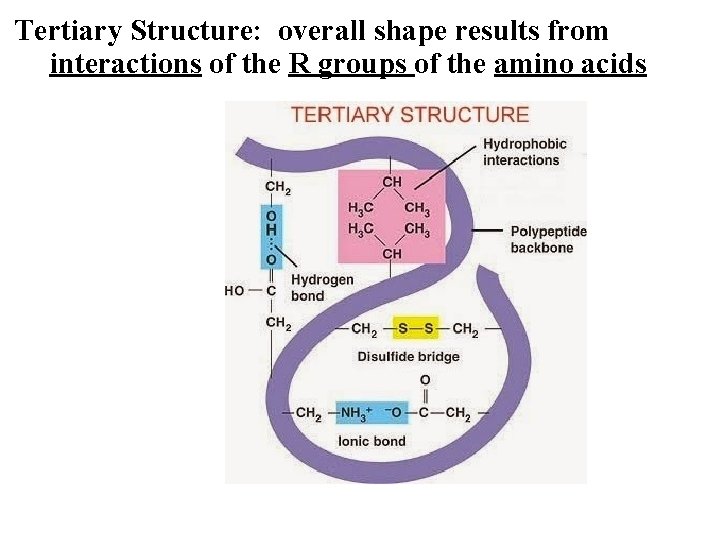
Tertiary Structure: overall shape results from interactions of the R groups of the amino acids
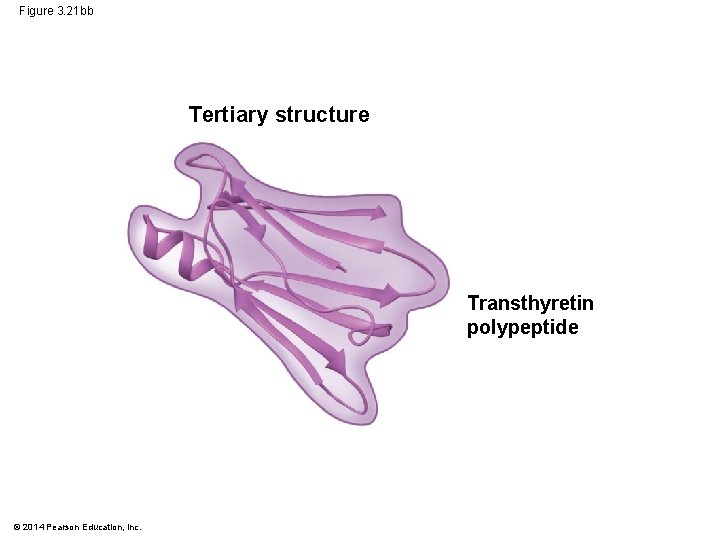
Figure 3. 21 bb Tertiary structure Transthyretin polypeptide © 2014 Pearson Education, Inc.
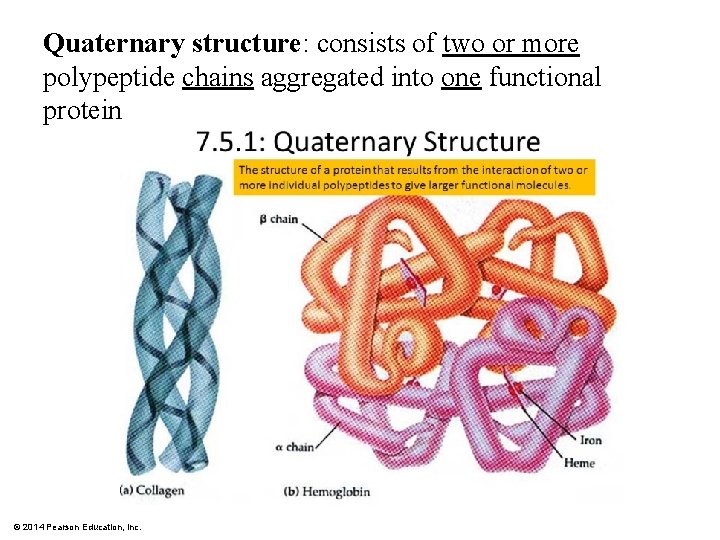
Quaternary structure: consists of two or more polypeptide chains aggregated into one functional protein © 2014 Pearson Education, Inc.
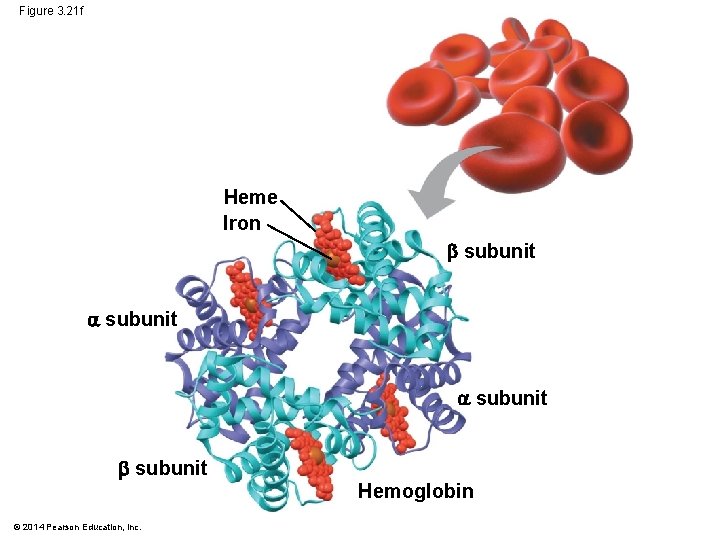
Figure 3. 21 f Heme Iron subunit Hemoglobin © 2014 Pearson Education, Inc.

Sickle-Cell Disease: A Change in Primary Structure § Primary structure is the sequence of amino acids on the polypeptide chain § A slight change in primary structure can affect a protein’s structure and ability to function § Sickle-cell disease, an inherited blood disorder, results from a single amino acid substitution in the protein hemoglobin © 2014 Pearson Education, Inc.
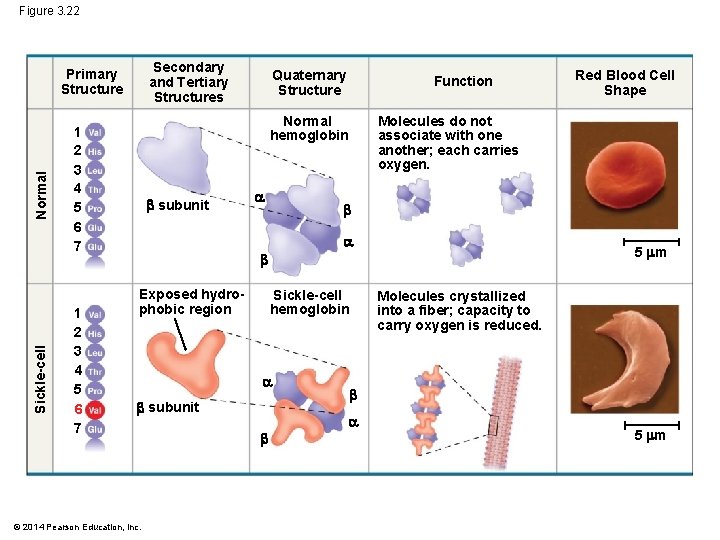
Figure 3. 22 Secondary and Tertiary Structures Sickle-cell Normal Primary Structure Function Normal hemoglobin 1 2 3 4 5 6 7 Quaternary Structure subunit Molecules do not associate with one another; each carries oxygen. 5 m Exposed hydrophobic region Sickle-cell hemoglobin subunit © 2014 Pearson Education, Inc. Red Blood Cell Shape Molecules crystallized into a fiber; capacity to carry oxygen is reduced. 5 m

Concept 3. 5: Proteins include a diversity of structures, resulting in a wide range of functions § Protein’s intricate 3 D structure determines its function § Protein functions include defense, storage, transport, cellular communication, movement, and structural support © 2014 Pearson Education, Inc.
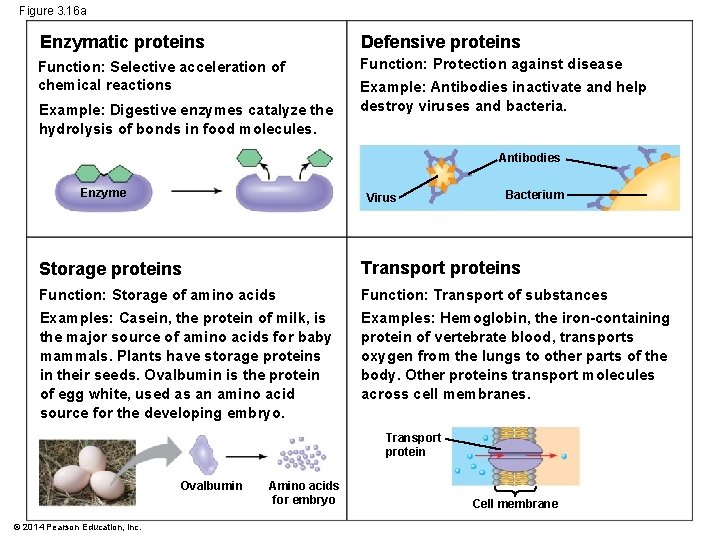
Figure 3. 16 a Enzymatic proteins Defensive proteins Function: Selective acceleration of chemical reactions Function: Protection against disease Example: Digestive enzymes catalyze the hydrolysis of bonds in food molecules. Example: Antibodies inactivate and help destroy viruses and bacteria. Antibodies Enzyme Virus Bacterium Storage proteins Transport proteins Function: Storage of amino acids Function: Transport of substances Examples: Casein, the protein of milk, is the major source of amino acids for baby mammals. Plants have storage proteins in their seeds. Ovalbumin is the protein of egg white, used as an amino acid source for the developing embryo. Examples: Hemoglobin, the iron-containing protein of vertebrate blood, transports oxygen from the lungs to other parts of the body. Other proteins transport molecules across cell membranes. Transport protein Ovalbumin © 2014 Pearson Education, Inc. Amino acids for embryo Cell membrane
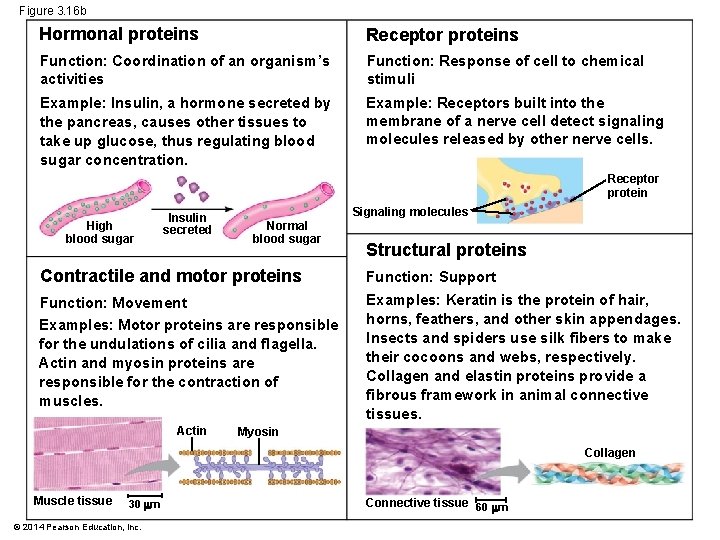
Figure 3. 16 b Hormonal proteins Receptor proteins Function: Coordination of an organism’s activities Function: Response of cell to chemical stimuli Example: Insulin, a hormone secreted by the pancreas, causes other tissues to take up glucose, thus regulating blood sugar concentration. Example: Receptors built into the membrane of a nerve cell detect signaling molecules released by other nerve cells. Receptor protein High blood sugar Insulin secreted Signaling molecules Normal blood sugar Structural proteins Contractile and motor proteins Function: Support Function: Movement Examples: Keratin is the protein of hair, horns, feathers, and other skin appendages. Insects and spiders use silk fibers to make their cocoons and webs, respectively. Collagen and elastin proteins provide a fibrous framework in animal connective tissues. Examples: Motor proteins are responsible for the undulations of cilia and flagella. Actin and myosin proteins are responsible for the contraction of muscles. Actin Myosin Collagen Muscle tissue 30 m © 2014 Pearson Education, Inc. Connective tissue 60 m
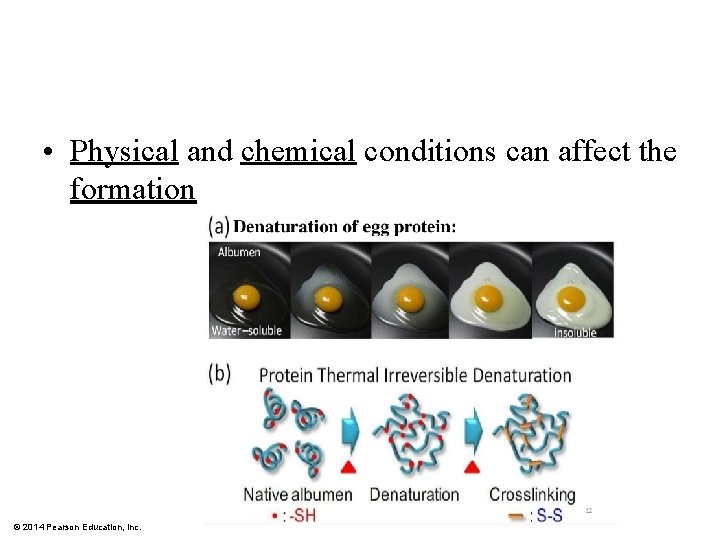
• Physical and chemical conditions can affect the formation © 2014 Pearson Education, Inc.

What Determines Protein Structure? § Denaturation - Alterations in p. H, temperature, salt concentration, or other factors can cause protein to change shape § Affects the chemical bonds and interactions § Protein is biologically inactive © 2014 Pearson Education, Inc.

Concept 3. 6: Nucleic acids store, transmit, and help express hereditary information §Quick Review §Gene: unit of inheritance § Consist of DNA § Nucleic Acids § Monomers of nucleic acids: neucleotides © 2014 Pearson Education, Inc.

§DNA: deoxyribonucleic acid § Provides direction for its own replication and RNA synthesis, and protein synthesis (through RNA) §RNA: ribonucleic acid § Protein synthesis
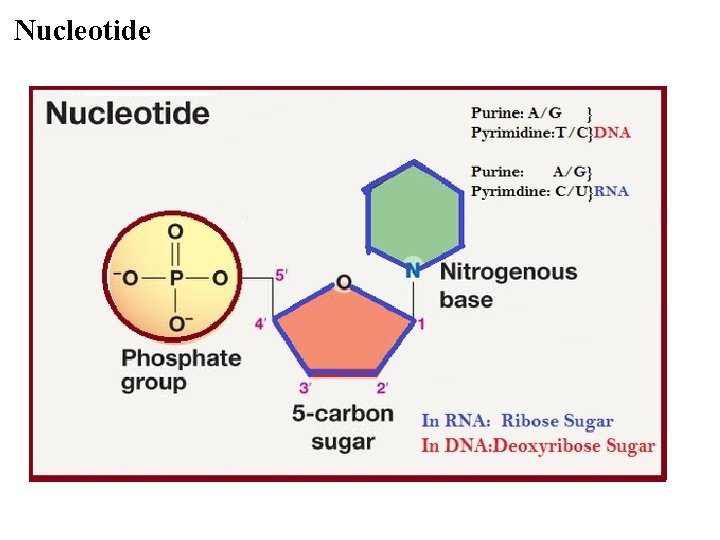
Nucleotide

Nucleotide §One or more phosphate groups §A five carbon sugar §Nitrogenous base § Pyrimidine § Cystine (C) § Thymine (T) § (in RNA U instead of T) §Purine § Adenine (A) § Guanine (G)

§A-T, C-G
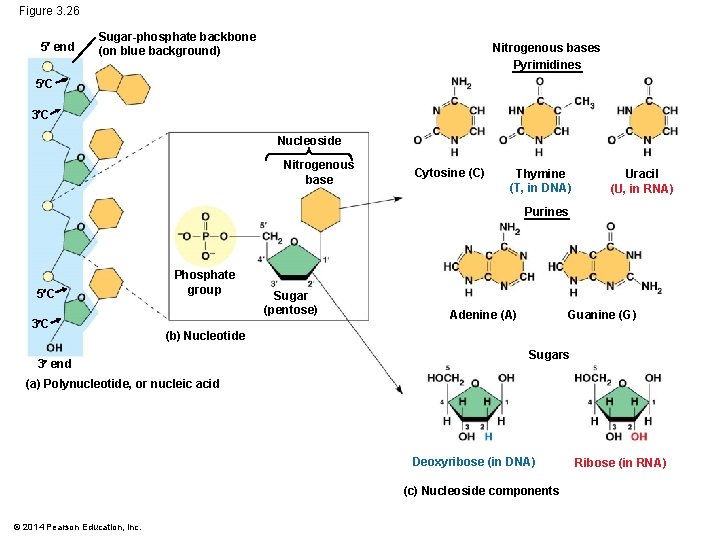
Figure 3. 26 5 end Sugar-phosphate backbone (on blue background) Nitrogenous bases Pyrimidines 5 C 3 C Nucleoside Nitrogenous base Cytosine (C) Thymine (T, in DNA) Uracil (U, in RNA) Purines 5 C 3 C Phosphate group Sugar (pentose) Adenine (A) Guanine (G) (b) Nucleotide 3 end Sugars (a) Polynucleotide, or nucleic acid Deoxyribose (in DNA) (c) Nucleoside components © 2014 Pearson Education, Inc. Ribose (in RNA)
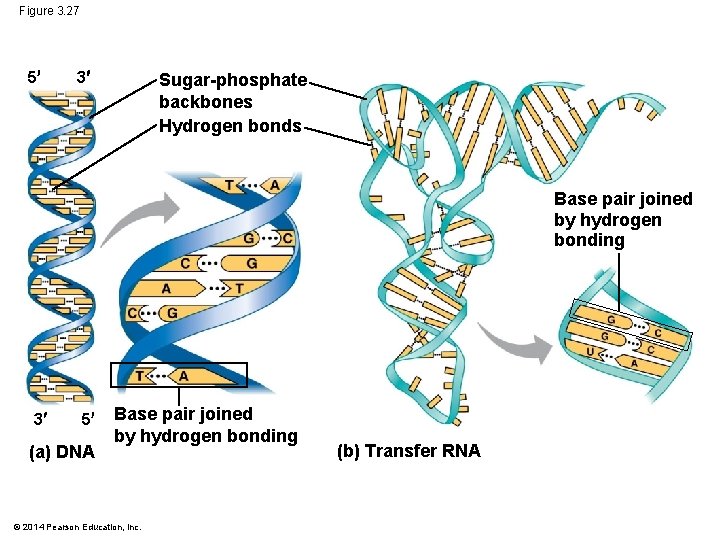
Figure 3. 27 5 3 Sugar-phosphate backbones Hydrogen bonds Base pair joined by hydrogen bonding 3 5 (a) DNA Base pair joined by hydrogen bonding © 2014 Pearson Education, Inc. (b) Transfer RNA
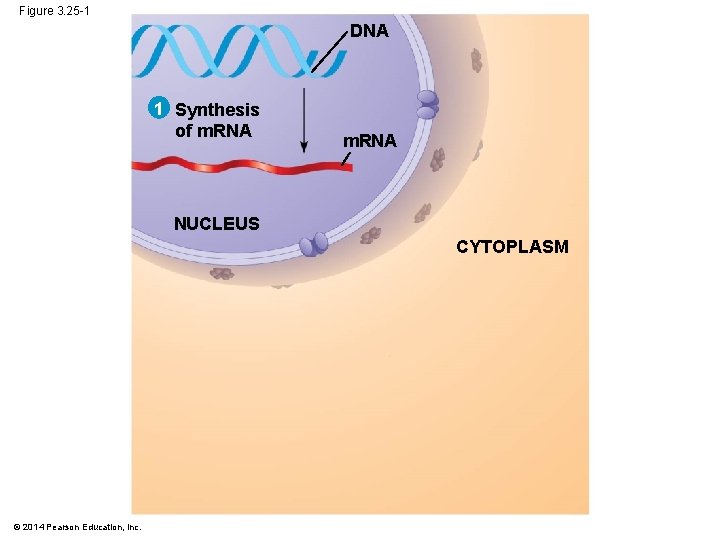
Figure 3. 25 -1 DNA 1 Synthesis of m. RNA NUCLEUS CYTOPLASM © 2014 Pearson Education, Inc.
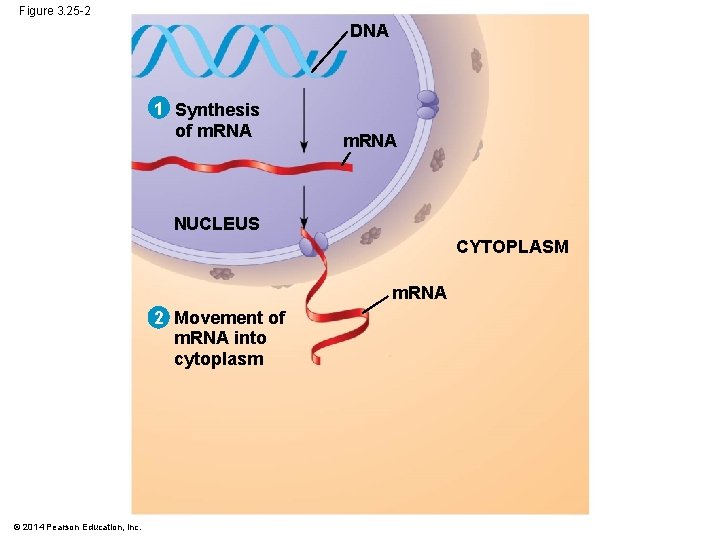
Figure 3. 25 -2 DNA 1 Synthesis of m. RNA NUCLEUS CYTOPLASM m. RNA 2 Movement of m. RNA into cytoplasm © 2014 Pearson Education, Inc.
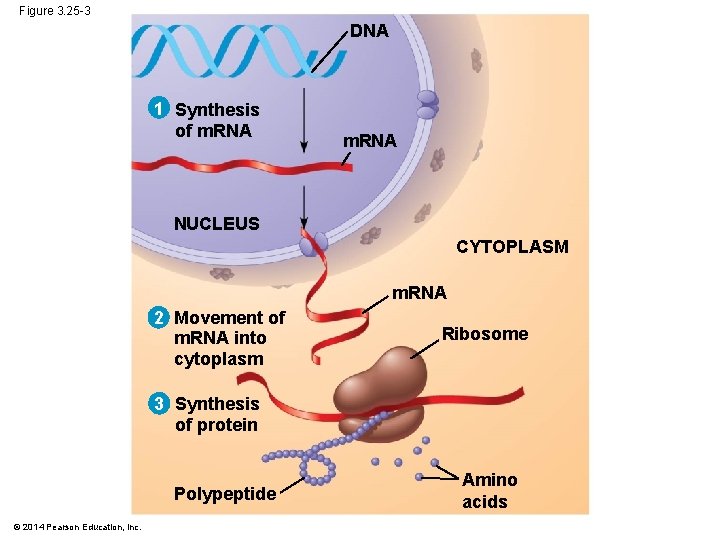
Figure 3. 25 -3 DNA 1 Synthesis of m. RNA NUCLEUS CYTOPLASM m. RNA 2 Movement of m. RNA into cytoplasm Ribosome 3 Synthesis of protein Polypeptide © 2014 Pearson Education, Inc. Amino acids
- Slides: 88