1 Razi University KermanshahIran Physical Organic Chemistry Part1














- Slides: 23

1

Razi University Kermanshah-Iran Physical Organic Chemistry Part-1 (Sessions 1 and 2) (For M. S. Students of Organic Chemistry) Prof. Avat Arman Taherpour Department of Organic Chemistry, Faculty of Chemistry, Razi University, P. O. Box: 67149 -67346, Kermanshah, Iran 1398 -99 -2 2

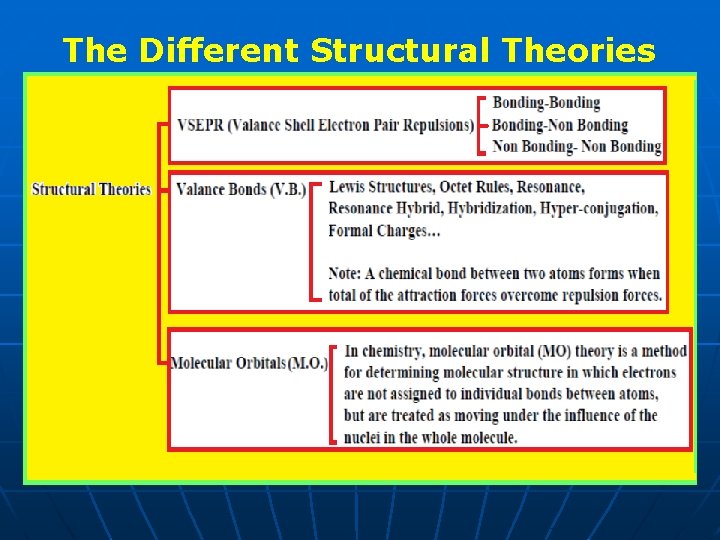
The Different Structural Theories

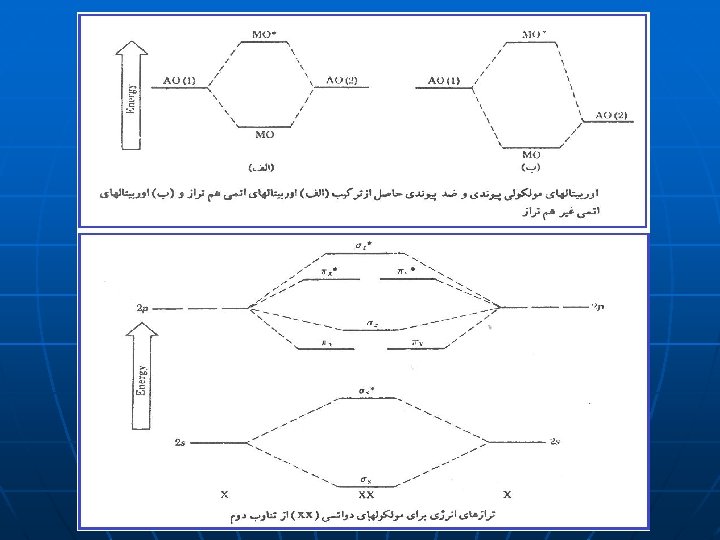
4

5

6

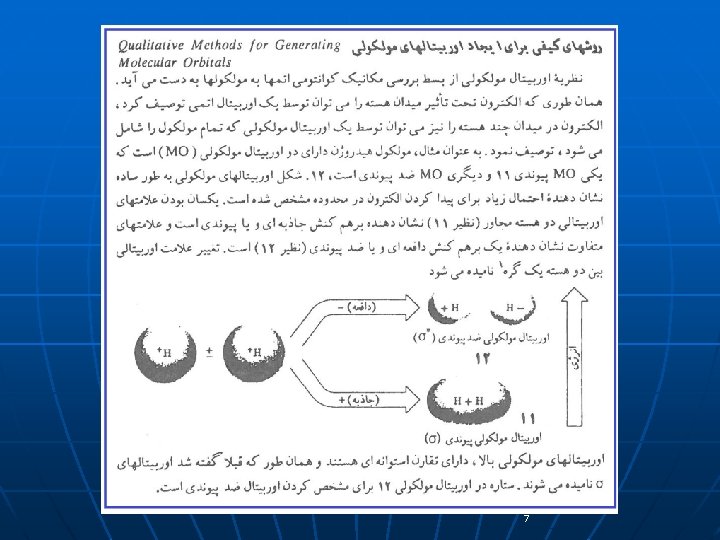
7

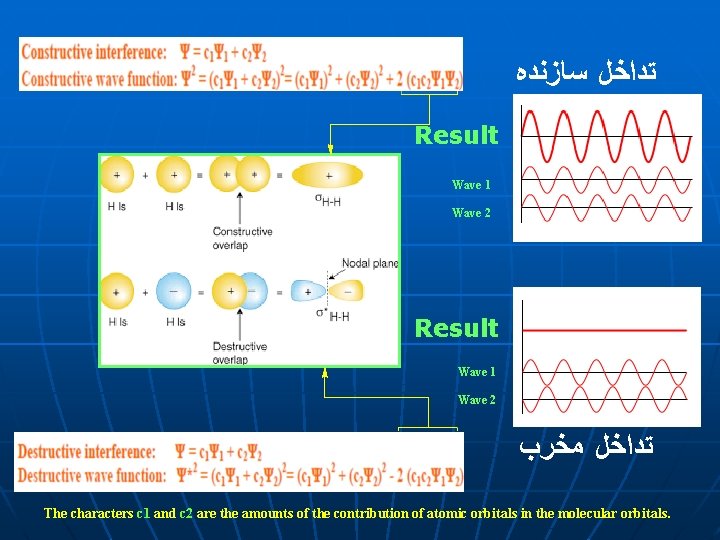
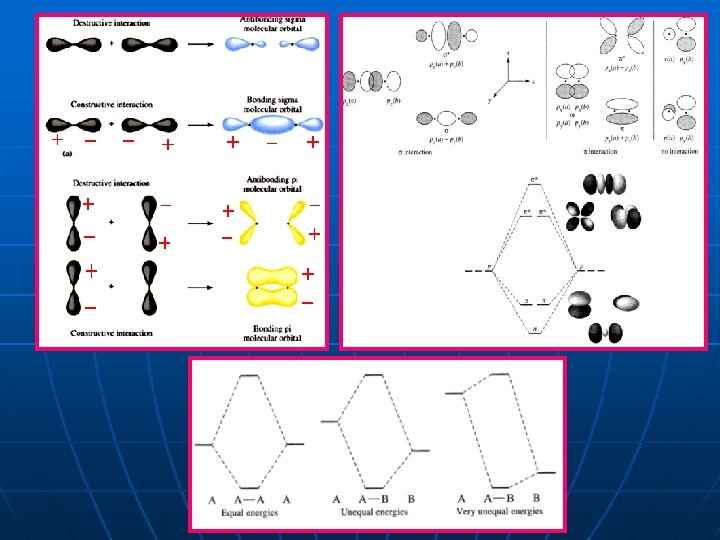
ﺗﺪﺍﺧﻞ ﺳﺎﺯﻧﺪﻩ Result Wave 1 Wave 2 ﺗﺪﺍﺧﻞ ﻣﺨﺮﺏ The characters c 1 and c 2 are the amounts of the contribution of atomic orbitals in the molecular orbitals.

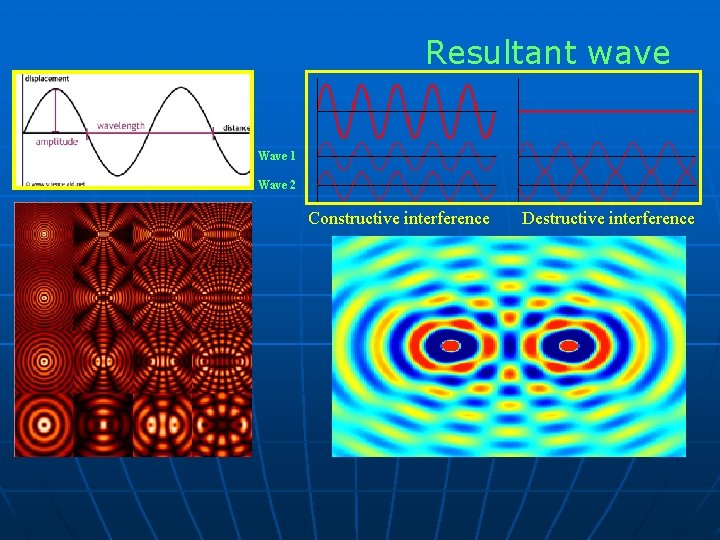
Resultant wave Wave 1 Wave 2 Constructive interference Destructive interference

10


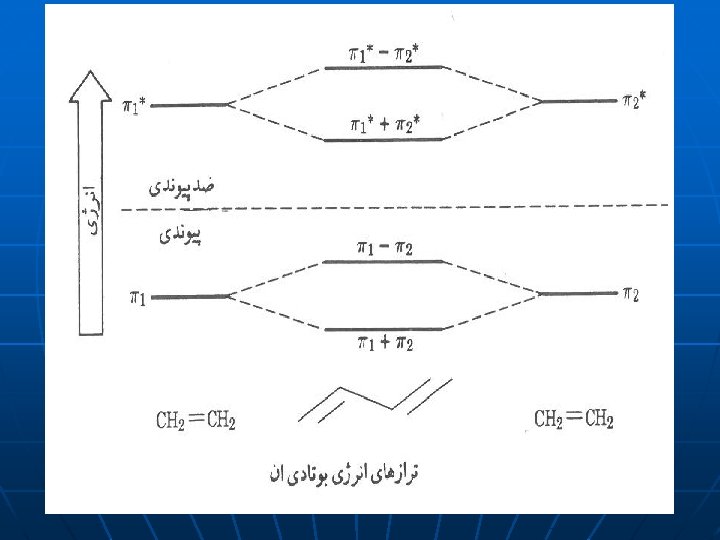
12

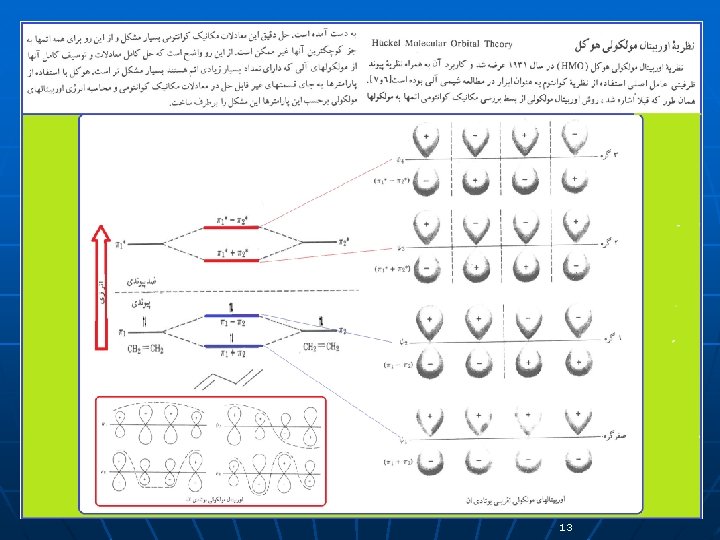
13

Uncertainty Principle Heisenberg: In quantum mechanics, the uncertainty principle, also known as Heisenberg's uncertainty principle, is any of a variety of mathematical inequalities asserting a fundamental limit to the precision with which certain pairs of physical properties of a particle known as complementary variables, such as position x and momentum p, can be known simultaneously. Introduced first in 1927, by the German physicist "Werner Heisenberg", it states that the more precisely the position of some particle is determined, the less precisely its momentum can be known, and vice versa. The formal inequality relating the standard deviation of position Δx and the standard deviation of momentum Δm. v was derived by Earle Hesse Kennard later that year and by Hermann Weyl in 1928: Δx. Δm. v ≈ ћ/2 (ħ= 6. 62606957 E-34 m 2 kg/s, is the reduced Planck constant). 1) Heisenberg, W. (1927), "Über den anschaulichen Inhalt der quantentheoretischen Kinematik und Mechanik", Zeitschrift für Physik (in German) 43 (3– 4): 172– 198. 2) Kennard, E. H. (1927), "Zur Quantenmechanik einfacher Bewegungstypen", Zeitschrift für Physik (in German) 44 (4– 5): 326. 3) Weyl, H. (1928), Gruppentheorie und Quantenmechanik, Leipzig: Hirzel, Germany. : ﺍﺻﻞ ﻋﺪﻡ ﻗﻄﻌیﺖ ﻫﺎیﺰﻧﺒﺮگ ﺍیﻦ ﺍﺻﻞ ﻣﺪﻋی ﺍﺳﺖ. ﺑﻪ ﻋﻨﻮﺍﻥ ﺍﺻﻞ ﻋﺪﻡ ﻗﻄﻌیﺖ ﻫﺎیﺰﻧﺒﺮگ ﺷﻨﺎﺧﺘﻪ ﺷﺪﻩ ﺍﺳﺖ ، ﺍﺻﻞ ﻋﺪﻡ ﻗﻄﻌیﺖ ، ﺩﺭ ﻣکﺎﻧیک کﻮﺍﻧﺘﻮﻣی کﻪ ﻫﺮ یک ﺍﺯ ﺍﻧﻮﺍﻉ ﻧﺎﺑﺮﺍﺑﺮﻫﺎی ﺭیﺎﺿی ﺑﺎ ﺟﻔﺖ ﺧﺎﺻی ﺍﺯ ﻭیژگی ﻓیﺰیکی ﺫﺭﺍﺕ ﺷﻨﺎﺧﺘﻪ ﺷﺪﻩ ﺑﻪ ﻋﻨﻮﺍﻥ ﻣﺘﻐیﺮﻫﺎی . کﻪ ﻣی ﺗﻮﺍﻥ ﺑﻪ ﻃﻮﺭ ﻫﻤﺰﻣﺎﻥ ﺷﻨﺎﺳﺎیی کﺮﺩ P ﻭ ﺍﻧﺪﺍﺯﻩ ﺣﺮکﺖ X ﻣﺎﻧﻨﺪ ﻣﻮﻗﻌیﺖ. ﺩﺭ ﺩﻗﺖ ﻣﺤﺪﻭﺩیﺖ ﺍﺳﺎﺳی ﺩﺍﺭﺩ ، ﻣکﻤﻞ ﺍیﻦ ﺍﺻﻞ ﺑیﺎﻥ ﻣی. ﺗﻮﺳﻂ ﻓیﺰیکﺪﺍﻥ آﻠﻤﺎﻧی "ﻭﺭﻧﺮ ﻫﺎیﺰﻧﺒﺮگ" ﻣﻌﺮﻓی ﺷﺪﻩ ﺍﺳﺖ ،1927 ﺍیﻦ ﺍﺻﻞ ﺍﻭﻟیﻦ ﺑﺎﺭ ﺩﺭ ﺳﺎﻝ . ﻣﻨﺠﺮ ﺑﻪ ﺩﻗﺖ کﻤﺘﺮﺩﺭ ﺍﻧﺪﺍﺯﻩ ﺣﺮکﺖ آﻦ ﺫﺭﻩ ﻣی ﺷﻮﺩ ﻭ ﺑﺎﻟﻌکﺲ ، ﺗﻌییﻦ ﺩﻗیﻘﺘﺮ ﻣﻮﻗﻌیﺖ ﻣکﺎﻧی ﺑﺮﺧی ﺍﺯ ﺫﺭﺍﺕ ، کﻨﺪ کﻪ E. H. ( ﺗﻮﺳﻂ Δm. v ) ( ﻭ ﺍﻧﺤﺮﺍﻑ ﺍﺳﺘﺎﻧﺪﺍﺭﺩ ﺍﺯ ﺍﻧﺪﺍﺯﻩ ﺣﺮکﺖ Δx) ﺍیﻦ ﻧﺎﺑﺮﺍﺑﺮی ﺩﺭ ﺍﻧﺤﺮﺍﻑ ﺍﺳﺘﺎﻧﺪﺍﺭﺩ ﺍﺯ ﻣﻮﻗﻌیﺖ ﻣکﺎﻧی : ﺑﻪ ﺩﺳﺖ آﻤﺪﻩ 1928 ﺩﺭ ﺳﺎﻝ H. Weyl ﻭ پﺲ ﺍﺯ ﺍﻭ ﺩﺭ ﻫﻤﺎﻥ ﺳﺎﻝ ﺗﻮﺳﻂ ،Kennard



17

18

19

20

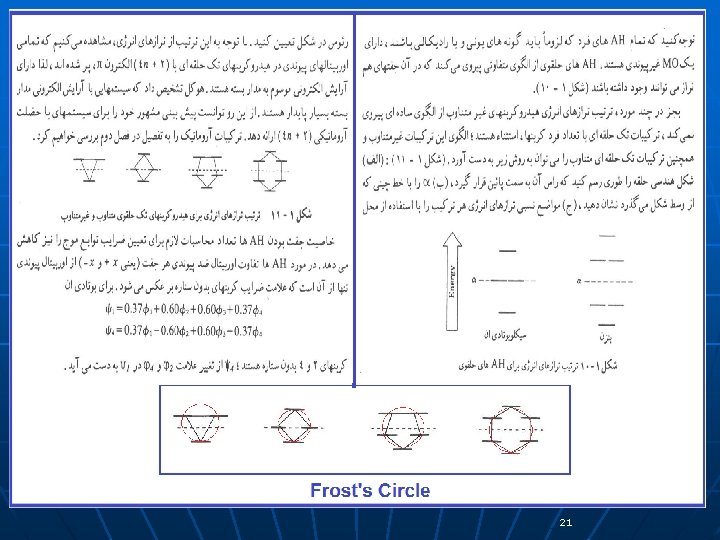
21

22

End of Sessions 1 and 2 23