Synthetic Iron Oxides An Indicator of Reduction In
















- Slides: 37

Synthetic Iron Oxides: An Indicator of Reduction In Soils (IRIS) What do we now know? 2006 NEMartin C. Rabenhorst Cooperative Soil Survey Conference University of Maryland Environmental Science & Technology

Why document reduction in soils? n Field Indicators of Hydric Soils q Applying the Technical Standard for Hydric Soils n n n Demonstrate that a soil is “hydric” in the absence of Field Indicators q n To test existing Field Indicators To evaluate proposed Field Indicators Also using the Technical Standard for Hydric Soils Demonstrate occurrence of redox processes q In constructed or restored wetlands n q Performance criteria Other research endeavors

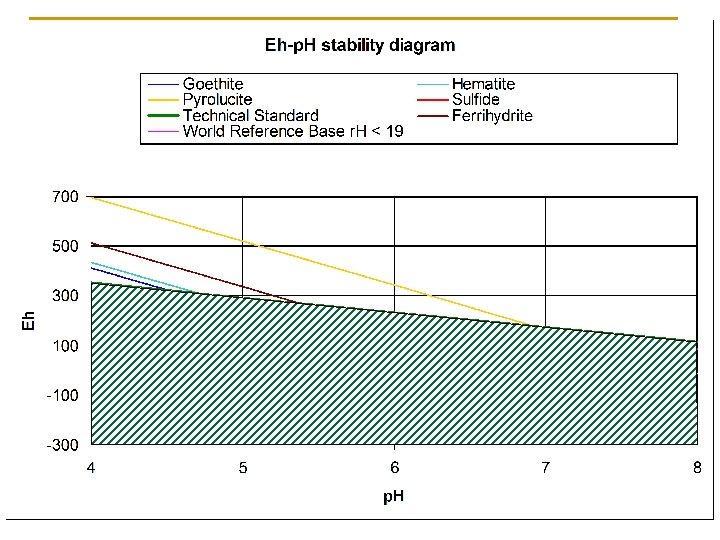
Available Technologies for Documenting Reduction in Soils n Eh measurements with Platinum electrodes (and p. H) q q n Use of alpha-dipyridyl q q q n Plot on Eh-p. H diagrams Time consuming and a bit tedious, equipment issues Reacts with ferrous Fe+2 Difficult to obtain (hazardous) Now available as test papers www. gallard. com Both provide only a “snapshot in time” of the soil conditions

www. gallard. com n Dipyridyl paper item# 90725 Pack Size: box of 200 strips Hazard Class /UN Number: Not Restricted Storage Temp: Room Temperature Impregnated with a, a’-dipyridyl (= 2, 2’-bipyridine), the paper reacts when Fe 2+ ions are present in mineral acid solutions, yielding a deep red very stable complex cation. Dipyridyl paper is specific for iron(II) (Fe 2+ ) and permits the detection of minute quantities of Fe 2+ , even in the presence of substantial amounts of Fe 3+. Limit of sensitivity: 2 mg/L Fe 2+ n $26. 90 per box of 200 n n n


IRIS (Indicator of Reduction in Soils) Tubes n Fe Oxide paint is applied to ½ inch schedule 40 PVC tubing while the tube is on a lathe device to ensure an even distribution of the paint. n. Jenkinson, B. 2002. Indicators of Reduction in Soils (IRIS): A visual method for the identification of hydric soils. Ph. D. Diss. Purdue Univ. , West Lafayette, IN

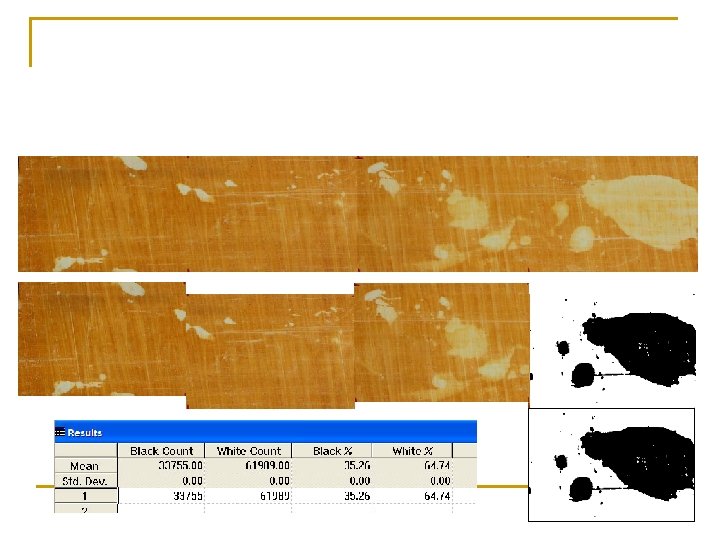
n n n Pilot hole made for each IRIS tube IRIS Tubes inserted into the soil Under anaerobic conditions, microbes oxidize OM utilize Fe oxides on IRIS tubes as e- acceptors As Fe(III) in paint is reduced to Fe(II), it dissolves Zones where Fe paint has been removed is visible and can be documented (quantified)

n n Originally we made composite photos of tubes after taking three photos while rotating each tube 120 o Then estimated paint removal q q Visually Using image analysis

New Approach to Collecting the Data from IRIS Tubes Can collect data on 0 -40 cm soil zone



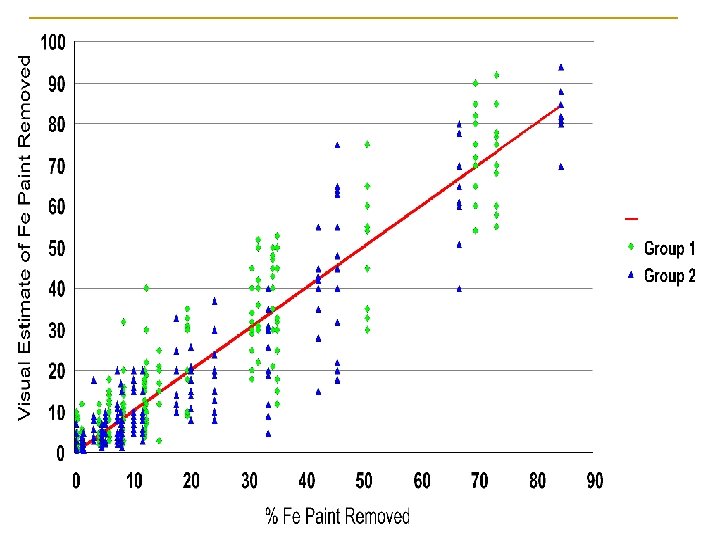
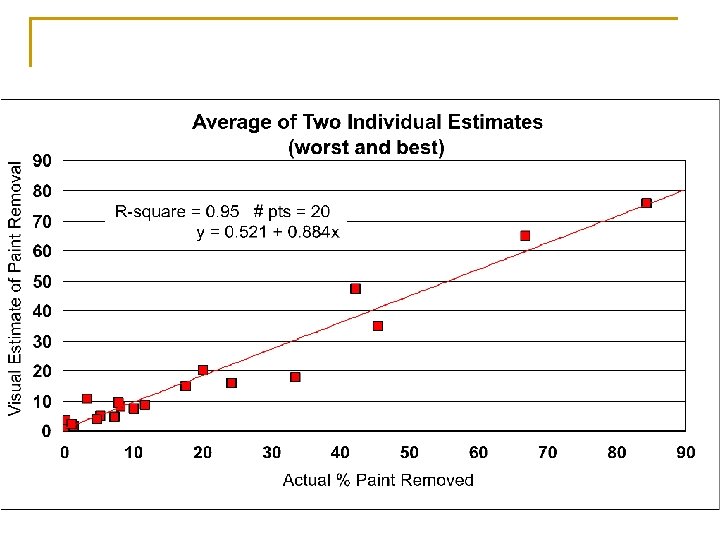
How effective are visual estimates of IRIS paint removal?


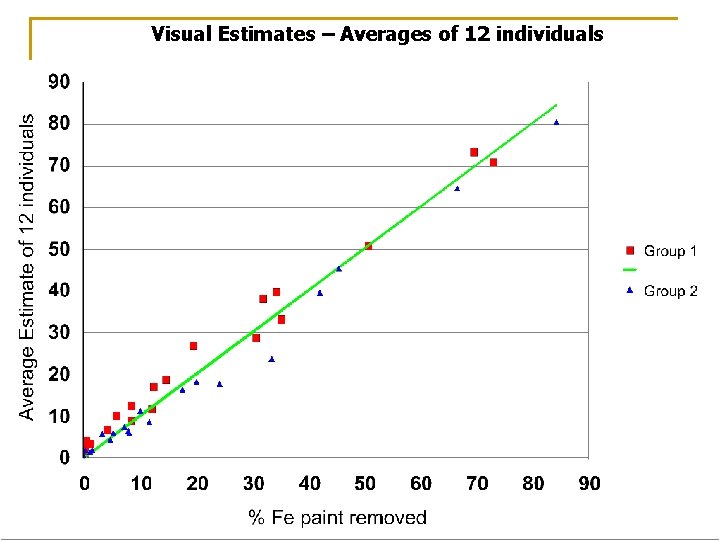
Visual Estimates – Averages of 12 individuals



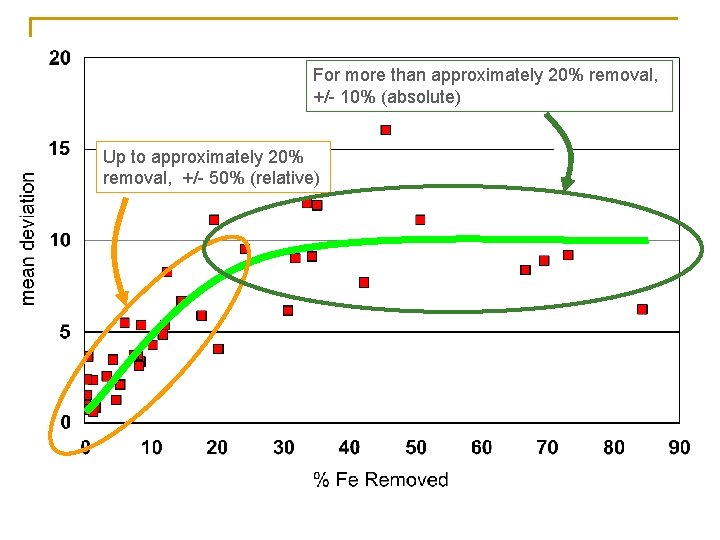
For more than approximately 20% removal, +/- 10% (absolute) Up to approximately 20% removal, +/- 50% (relative)

Guides for estimating percent removal Stoops, G. 2003. Guidelines for Analysis and Description of Soil and Regolith Thin Sections. Soil Sci. Soc. Am.

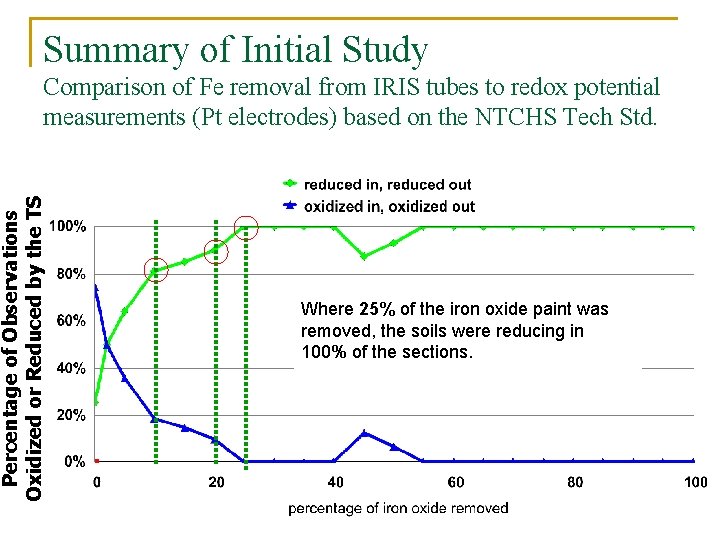
Summary of Initial Study Percentage of Observations Oxidized or Reduced by the TS Comparison of Fe removal from IRIS tubes to redox potential measurements (Pt electrodes) based on the NTCHS Tech Std. Where 10% of the iron oxide paint was Where 25% of the iron oxide paint was removed, the soils were reducing in 82% removed, the soils were reducing in 89% removed, the soils were reducing in of the sections while in 18% of the sections while in 11% of the 100% of the sections. cases, the soil was oxidized.

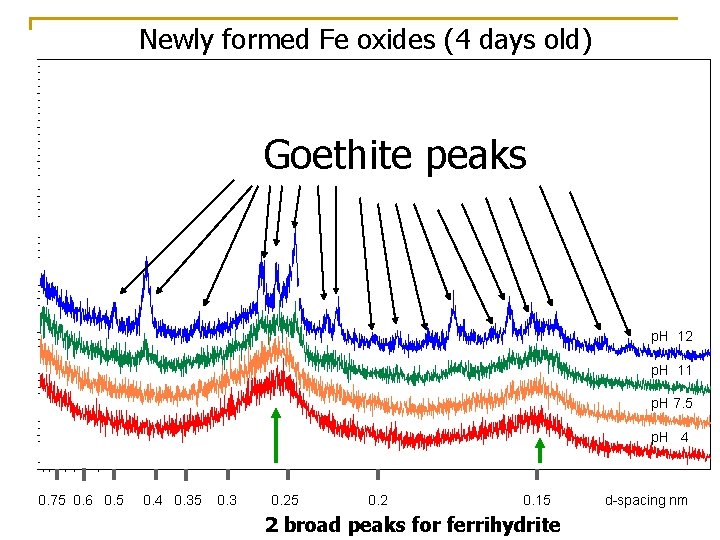
When initially synthesized, the paint is primarily ferrihydrite Fe 5 HO 8*4 H 2 O Poorly crystalline – probably similar to newly formed Fe oxides in wet soils

Paint of 4 ages were located in the lab left over from previous studies 1 2 3 4 14 d 120 d 270 d 625 d Color changes reflect changes in mineralogy

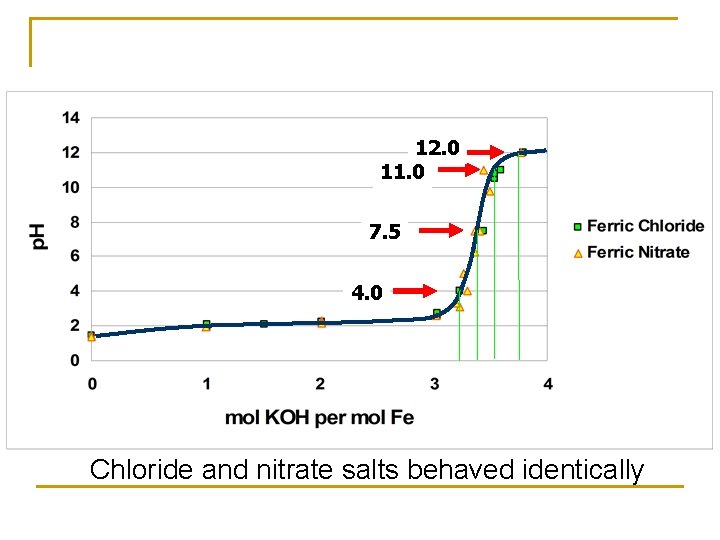
Problem with Newly Synthesized Paint n n We began to notice that newly synthesized paint would not adhere well to the PVC, but that sometimes paint 2 -3 weeks old would adhere better Therefore, we postulated that there might be some “aging” (mineralogical alteration) required for the paint to be useable. q q We knew from earlier observations that old paint stored in the lab changes to more crystalline forms Schwertmann and Cornell (2000) indicate that p. H away from the ZPC causes more rapid alteration of ferrihydrite to other crystalline phases

12. 0 11. 0 7. 5 4. 0 Chloride and nitrate salts behaved identically

Newly formed Fe oxides (4 days old) Goethite peaks p. H 12 p. H 11 p. H 7. 5 p. H 4 0. 75 0. 6 0. 5 0. 4 0. 35 0. 3 0. 25 0. 2 0. 15 d-spacing nm 2 broad peaks for ferrihydrite

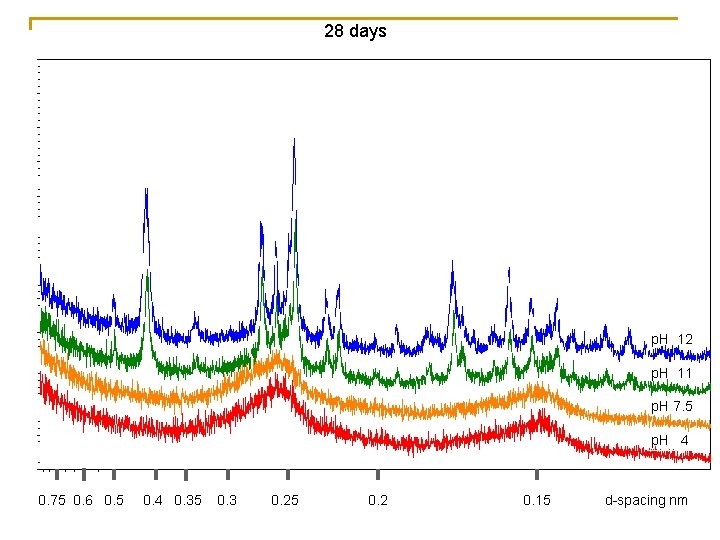
28 days p. H 12 p. H 11 p. H 7. 5 p. H 4 0. 75 0. 6 0. 5 0. 4 0. 35 0. 3 0. 25 0. 2 0. 15 d-spacing nm

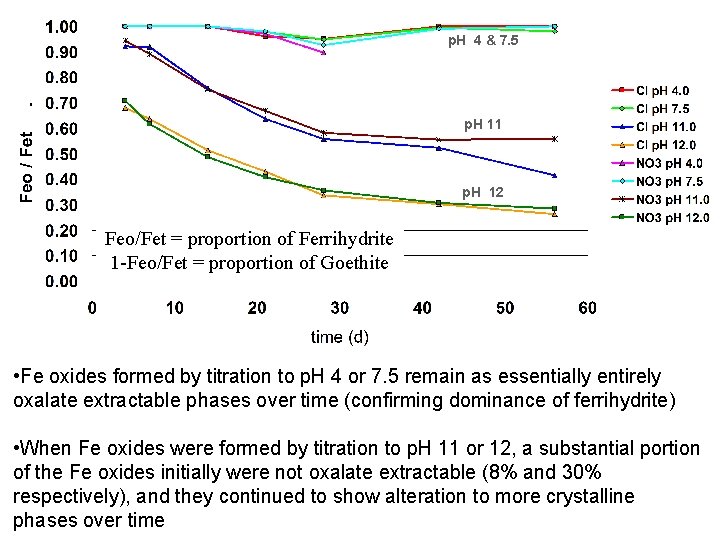
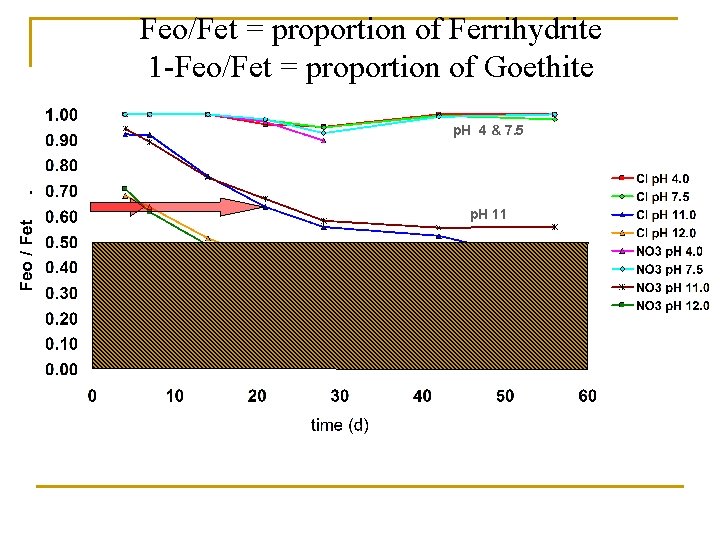
p. H 4 & 7. 5 p. H 11 p. H 12 Feo/Fet = proportion of Ferrihydrite 1 -Feo/Fet = proportion of Goethite • Fe oxides formed by titration to p. H 4 or 7. 5 remain as essentially entirely oxalate extractable phases over time (confirming dominance of ferrihydrite) • When Fe oxides were formed by titration to p. H 11 or 12, a substantial portion of the Fe oxides initially were not oxalate extractable (8% and 30% respectively), and they continued to show alteration to more crystalline phases over time

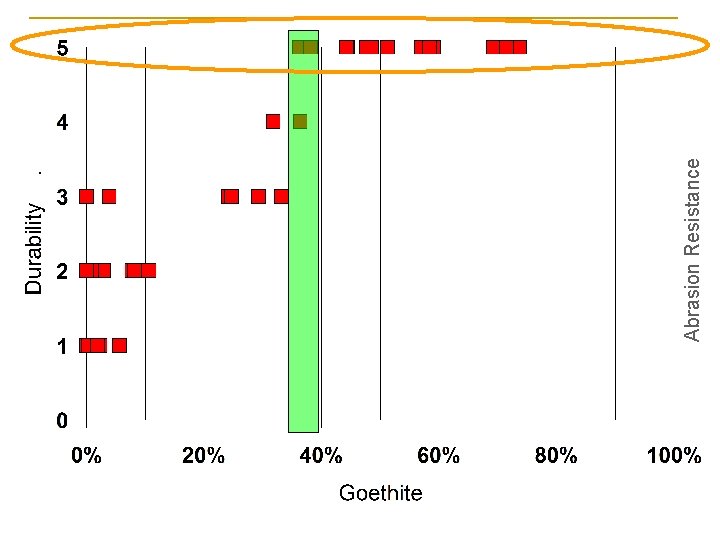
Abrasion Resistance and Durability n n n 1 - paint wipes off when applying very slight pressure 2 - paint wipes off when applying slight pressure 3 - paint wipes off when applying moderate pressure 4 - paint wipes off only when applying firm pressure 5 - paint does not wipe off when applying firm pressure.

Abrasion Resistance

Feo/Fet = proportion of Ferrihydrite 1 -Feo/Fet = proportion of Goethite p. H 4 & 7. 5 p. H 11 p. H 12

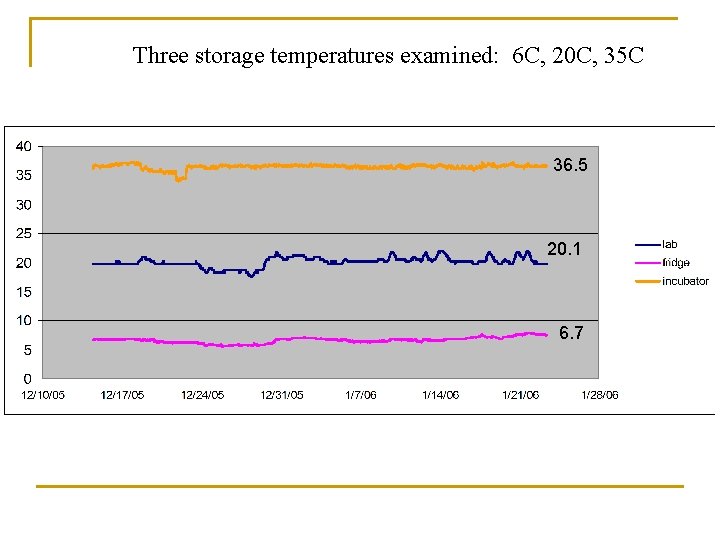
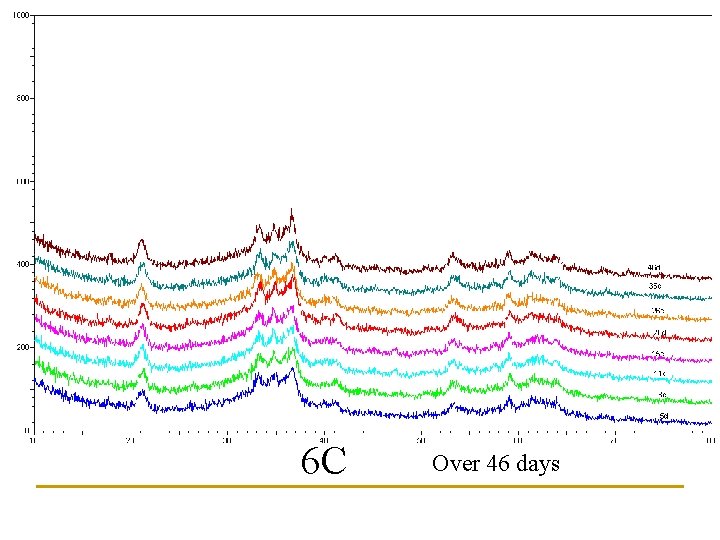
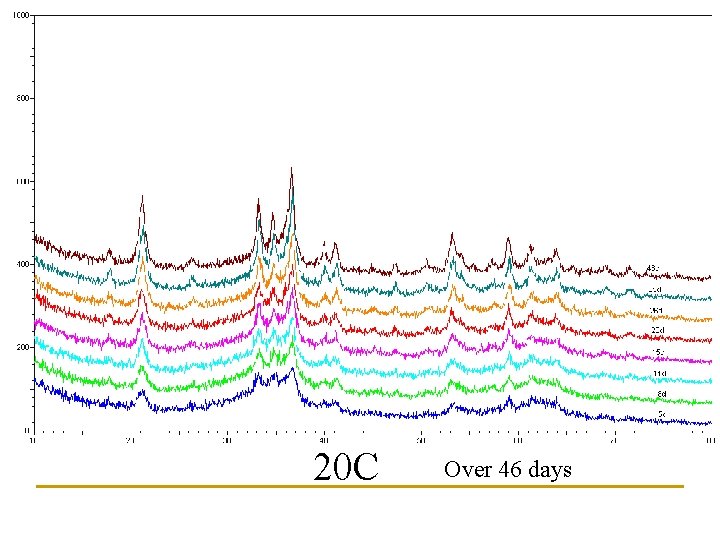
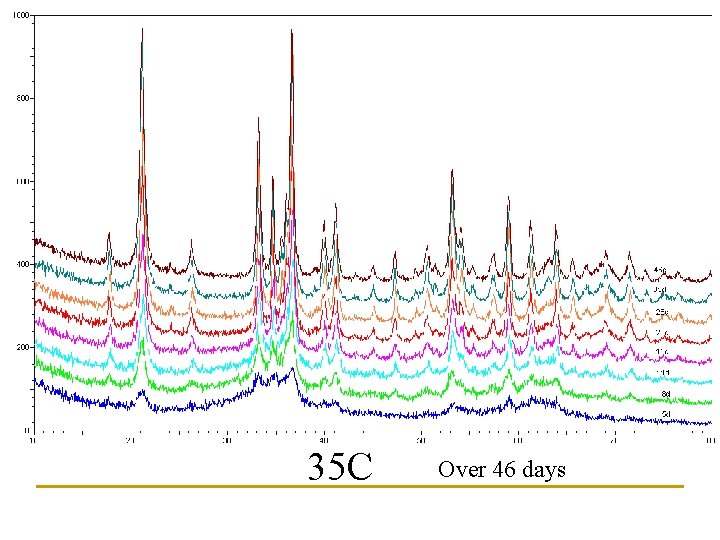
Newly Synthesized Paint What about storage? Effects of Temperature?

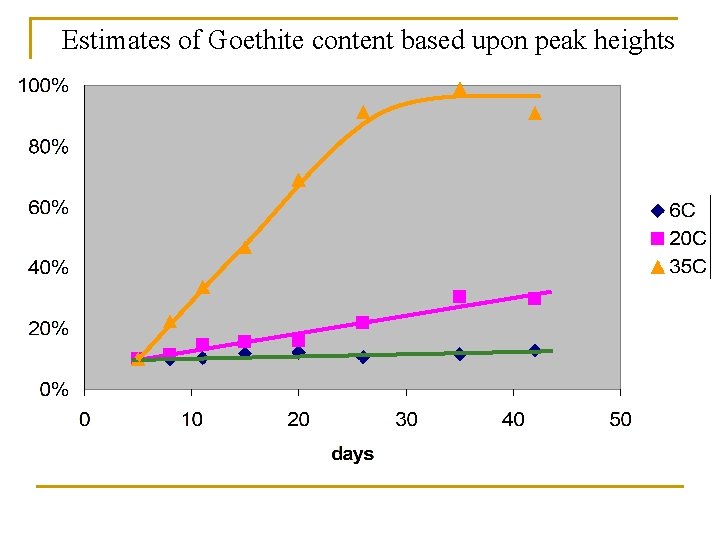
Three storage temperatures examined: 6 C, 20 C, 35 C 36. 5 20. 1 6. 7

6 C Over 46 days

20 C Over 46 days

35 C Over 46 days

Estimates of Goethite content based upon peak heights

Summary n What we now know: q q n We understand basically how to interpret the tubes We have developed a more rapid method for scanning and analysis of the tubes We know how to routinely make paint that will adhere well to the tubes and why We know that refrigeration will sustain the “pot life” of paint (retard mineralogical change) What we still don’t know: q q How significant are mineralogical differences in paint performance (40% Gt vs 70% Gt)? What the variations in colors on IRIS tubes represent?

Quick (7 day) IRIS Tube Paint Recipe and Construction Procedure 1 page handout available