AS Chemistry Polymers Nylon demo You have been

AS Chemistry Polymers

• Nylon demo

• You have been given a hydrocarbon cut it out so it is in one piece • Then fold it like in the pictures, but do not stick it together • What molecule do you think you have been given? Explain your answer. Carbon atoms Hydrogen atoms
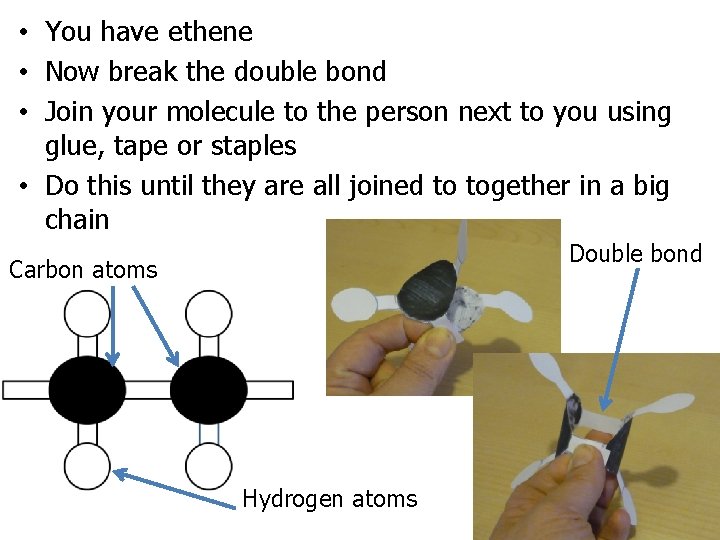
• You have ethene • Now break the double bond • Join your molecule to the person next to you using glue, tape or staples • Do this until they are all joined to together in a big chain Double bond Carbon atoms Hydrogen atoms

Polymers from Alkenes Objective: to know about the addition polymerisation of alkenes Success criteria: • describe the addition polymerisation of alkenes • deduce the repeat unit of an addition polymer obtained from a given monomer • identify the monomer that would produce a given section of an addition polymer

Polymerisation • • Polymers are long chain molecules built up from a whole lot of identical small molecules called monomers. Monomers are: – Always unsaturated (double bond present) – Based on a particular alkene • Addition polymerisation: – Is the process by which these monomers are joined. – Results in long saturated chains.
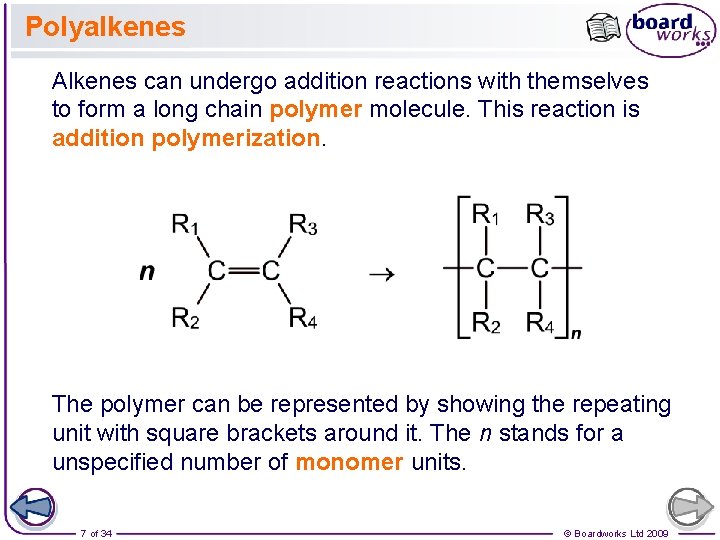
Polyalkenes Alkenes can undergo addition reactions with themselves to form a long chain polymer molecule. This reaction is addition polymerization. The polymer can be represented by showing the repeating unit with square brackets around it. The n stands for a unspecified number of monomer units. 7 of 34 © Boardworks Ltd 2009
Polymerization of ethene 8 of 34 © Boardworks Ltd 2009
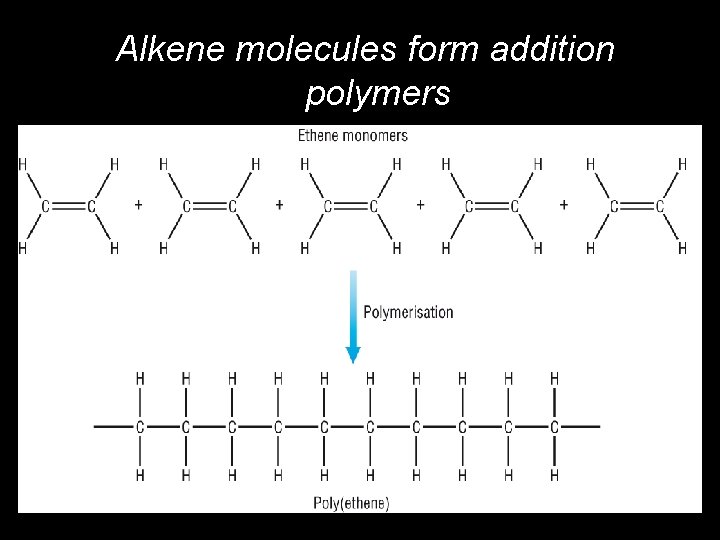
Alkene molecules form addition polymers © Pearson Education Ltd 2008 This document may have been altered from the original
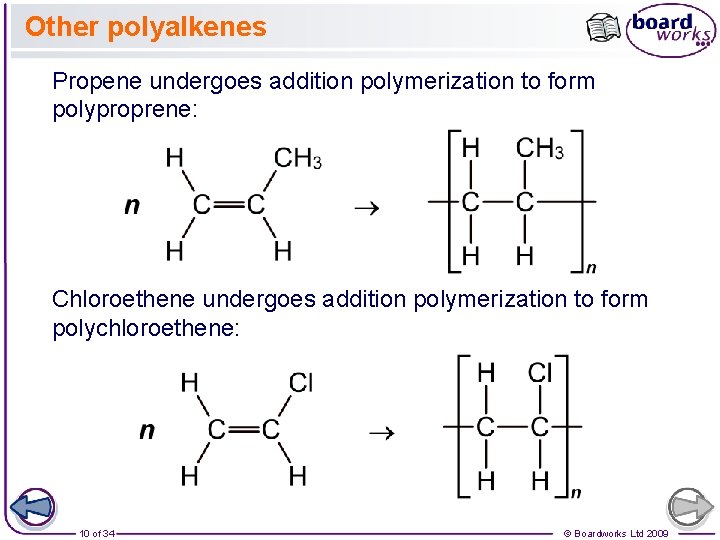
Other polyalkenes Propene undergoes addition polymerization to form polyproprene: Chloroethene undergoes addition polymerization to form polychloroethene: 10 of 34 © Boardworks Ltd 2009
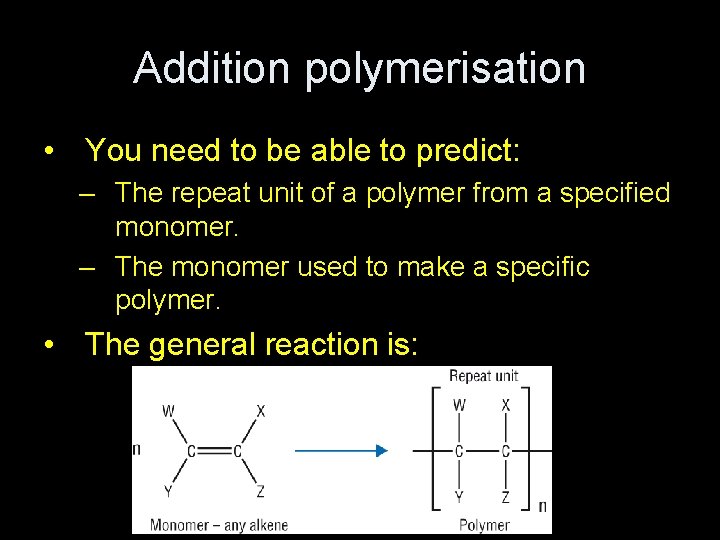
Addition polymerisation • You need to be able to predict: – The repeat unit of a polymer from a specified monomer. – The monomer used to make a specific polymer. • The general reaction is:
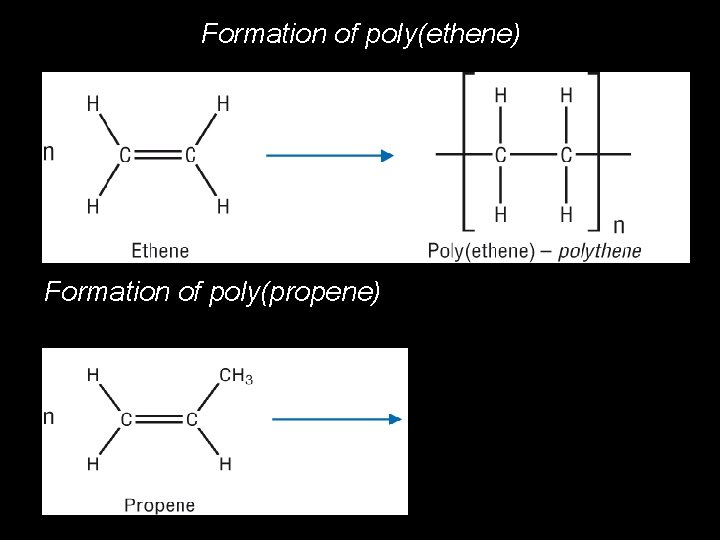
Formation of poly(ethene) Week 16 Formation of poly(propene) © Pearson Education Ltd 2008 This document may have been altered from the original
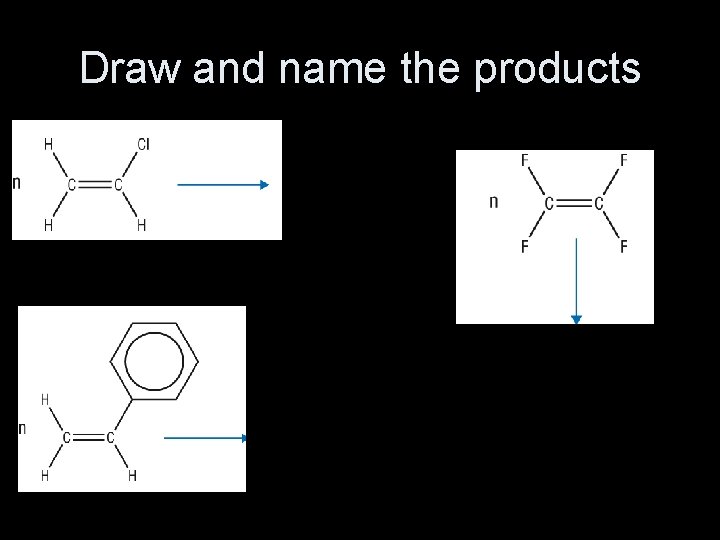
Draw and name the products

Week 16 Poly(tetrafluoroethene) (Teflon or PTFE) © Pearson Education Ltd 2008 This document may have been altered from the original

Week 16 Draw the monomer used to make Orlon © Pearson Education Ltd 2008 This document may have been altered from the original
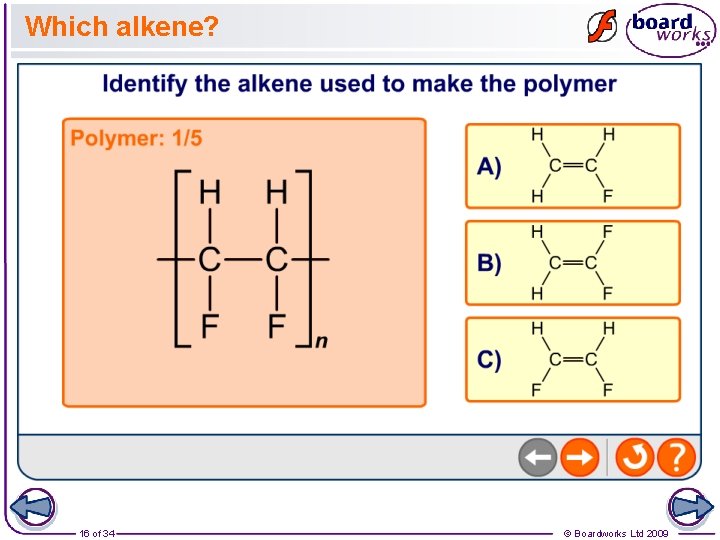
Which alkene? 16 of 34 © Boardworks Ltd 2009
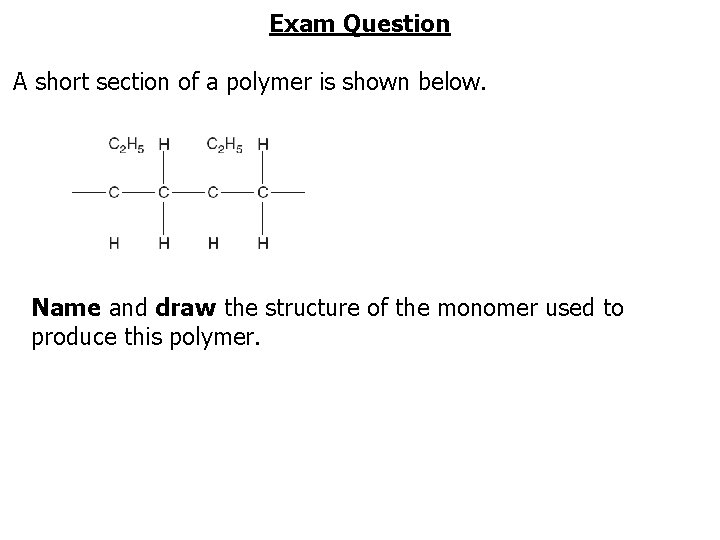
Exam Question A short section of a polymer is shown below. Name and draw the structure of the monomer used to produce this polymer.
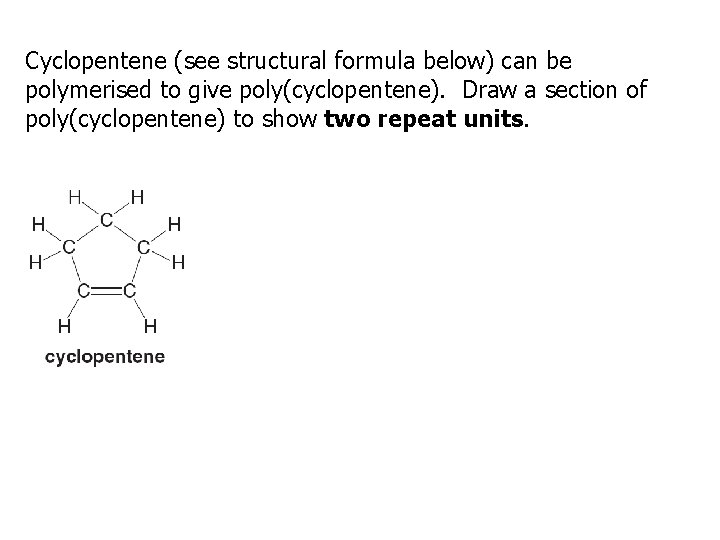
Cyclopentene (see structural formula below) can be polymerised to give poly(cyclopentene). Draw a section of poly(cyclopentene) to show two repeat units.

Another exam question

Polymers from Alkenes Objective: to know about the addition polymerisation of alkenes Success criteria: • describe the addition polymerisation of alkenes • deduce the repeat unit of an addition polymer obtained from a given monomer • identify the monomer that would produce a given section of an addition polymer
- Slides: 20