Stoichiometry The Mole Stoichiometry The study of the




















- Slides: 21

Stoichiometry: The Mole

Stoichiometry The study of the quantitative aspects of chemical reactions.

“The Mole” • A counting unit • 1 mole = 602 billion trillion: 602, 000, 000, 000 • 6. 02 X 1023 (in scientific notation) • 1 dozen eggs = 12 • 1 “mole” of eggs = 6. 02 x 1023 eggs • 1 dozen cars = 12 cars • 1 “mole” of cars = 6. 02 x 1023 cars Note that the NUMBER is always the same, but the MASS is very different! Mole is abbreviated “mol”

A Mole of Particles Contains 6. 02 x 1023 particles 1 mole C = 6. 02 x 1023 C atoms 1 mole H 2 O = 6. 02 x 1023 H 2 O molecules Note that a particle could be an atom OR a molecule!

Avogadro’s Number as a Conversion Factor 6. 02 x 1023 particles 1 mole or 1 mole 6. 02 x 1023 particles

Learning Check 1. Number of atoms in 0. 500 mole of Al a) 500 Al atoms b) 6. 02 x 1023 Al atoms c) 3. 01 x 1023 Al atoms 2. Number a) 1. 0 b) 3. 0 c) 1. 1 of moles of S in 1. 8 x 1024 S atoms mole S atoms x 1048 mole S atoms

Molar Mass • The Mass of 1 mole (in grams) • Equal to the numerical value of the average atomic mass (get from periodic table) 1 mole of C atoms = 12. 0 g 1 mole of Mg atoms = 24. 3 g 1 mole of Cu atoms = 63. 5 g

Learning Check! Find the molar mass: (usually we round to the tenths place) A. 1 mole of Br atoms = B. 1 mole of Sn atoms= 79. 9 g/mole 118. 7 g/mole

Chemical Formulas of Compounds • Formulas give the relative numbers of atoms or moles of each element in a formula unit - always a whole number ratio (the law of definite proportions). NO 2 2 atoms of O for every 1 atom of N 1 mole of NO 2 : 2 moles of O atoms to every 1 mole of N atoms • If we know or can determine the relative number of moles of each element in a compound, we can determine a formula for the compound.

Molar Mass of Molecules and Compounds Mass in grams of 1 mole sum of the atomic masses 1 mole of Ca. Cl 2 = 111. 1 g/mol 1 mole Ca x 40. 1 g/mol + 2 moles Cl x 35. 5 g/mol 1 mole of N 2 O 4 = 111. 1 g/mol Ca. Cl 2 = 92. 0 g/mol

Learning Check! A. Molar Mass of K 2 O = ? Grams/mole B. Molar Mass of antacid Al(OH)3 = ? Grams/mole

Learning Check! The artificial sweetener aspartame (Nutra-Sweet) formula C 14 H 18 N 2 O 5 is used to sweeten diet foods, coffee and soft drinks. How many moles of aspartame are present in 225 g of aspartame?

Calculations molar mass Grams Avogadro’s number Moles particles You can convert Grams to Particles, but you always must go through moles first.

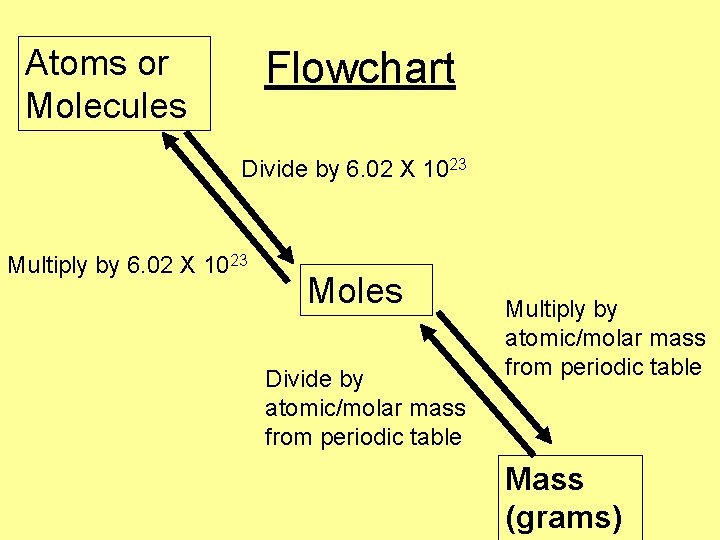
Atoms or Molecules Flowchart Divide by 6. 02 X 1023 Multiply by 6. 02 X 1023 Moles Divide by atomic/molar mass from periodic table Multiply by atomic/molar mass from periodic table Mass (grams)

Atoms/Molecules and Grams How many atoms of Cu are present in 35. 4 g of Cu? 35. 4 g Cu 1 mol Cu 63. 5 g Cu 6. 02 X 1023 atoms Cu 1 mol Cu = 3. 4 X 1023 atoms Cu

Learning Check! How many atoms of K are present in 78. 4 g of K?

Learning Check! What is the mass (in grams) of 1. 20 x 1024 molecules of glucose (C 6 H 12 O 6)?

Using Equations • Chemical equations also show mole ratios. • In the equation: – 2 Li. OH + CO 2 Li 2 CO 3 + H 2 O – For every 2 moles of Li. OH and 1 mole of CO 2, 1 mole of Li 2 CO 3 and 1 mole of H 2 O are produced.

Mole-Mass Conversions • Most of the time in chemistry, the amounts are given in grams instead of moles • We still go through moles and use the mole ratio, but now we also use molar mass to get to grams • Example: How many grams of chlorine are required to react completely with 5. 00 moles of sodium to produce sodium chloride? 2 Na + Cl 2 2 Na. Cl 5. 00 moles Na 1 mol Cl 2 2 mol Na 70. 90 g Cl 2 1 mol Cl 2 = 177 g Cl 2

Learning Check • For each of the following equations, calculate the moles of product that would be produced by reacting 4. 0 moles of oxygen, O 2. Show your work. – 4 Fe + 3 O 2 Fe 2 O 3 – 2 Mg + O 2 2 Mg. O – 2 Cu + O 2 2 Cu. O

Learning Check • Given the following reaction: – N 2 + O 2 2 NO a) How many moles of NO can form when 40. 0 g of N 2 react with excess oxygen? b) How many grams of NO can form, using the amounts in part A?