Physical Science Chapter 4 Atomic Structure 4 1















- Slides: 23

Physical Science Chapter 4 Atomic Structure

4. 1 Studying Atoms � Democritus and Aristotle ◦ Democritus thought all matter consisted of extremely tiny particles that could not be divided. (Cut aluminum foil in half) ◦ Also thought matter in liquids was round and smooth; in solids rough and prickly ◦ Aristotle thought there was no limit to the number of times matter could be divided.

Dalton’s Atomic Theory � Developed a theory to explain why the elements in a compound always behave in the same way. � Main Points ◦ 1) All elements are composed of atoms. ◦ 2) All atoms of the same element have the same mass, and atoms of different elements have different masses. ◦ 3) Compounds contain atoms of more than one element. ◦ 4) In a compound, atoms of different elements always combine in the same way.

Thomson’s Model � Used a cathode ray tube to show evidence for subatomic, charged particles.

Thomson continued � Plum pudding model (chocolate chip ice cream. � Negatively charged particles evenly spaced throughout a solid mass of positive charge.

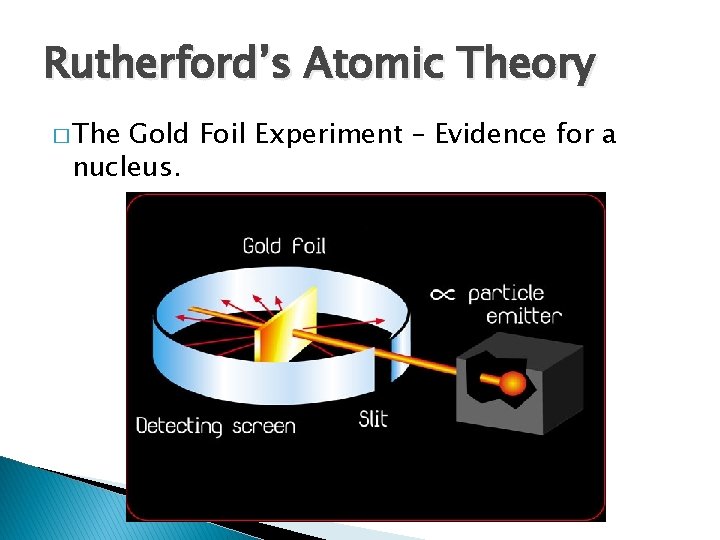
Rutherford’s Atomic Theory � The Gold Foil Experiment – Evidence for a nucleus.

Chapter 4 Lesson 2 “Structure of the Atom”

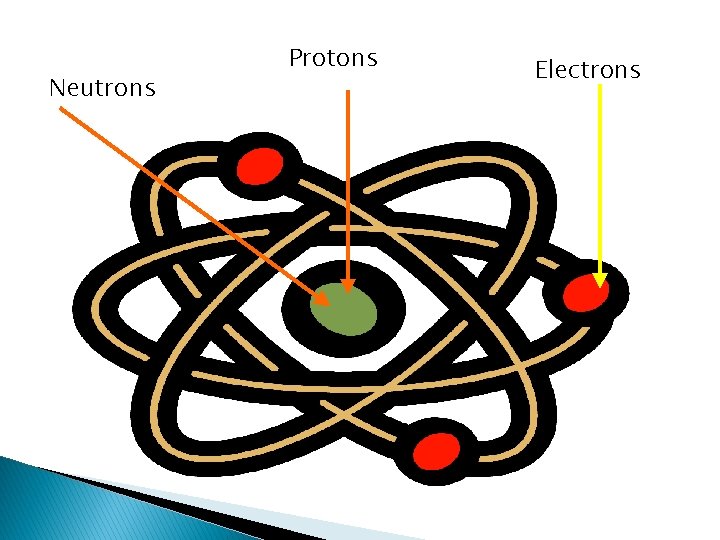
I. Subatomic Particles A. An atom 1. smallest part of matter 2. Ripping paper example B. Areas of an atom 1. Nucleus – center of an atom -holds protons and neutrons a. Protons (P) – positively charged particle found in the nucleus. -has a mass of 1 -has a charge of 1+

b. Neutrons (N) – non-charged particle found in the nucleus. -has a mass of 1 -has no charge “ 0” 2. Electron Cloud – area around the nucleus which contains electrons a. Electrons (e) – negatively charged particle found outside of the nucleus -has a mass of 0 -has a charge of 1 -moves around the outside of the nucleus

Neutrons Protons Electrons

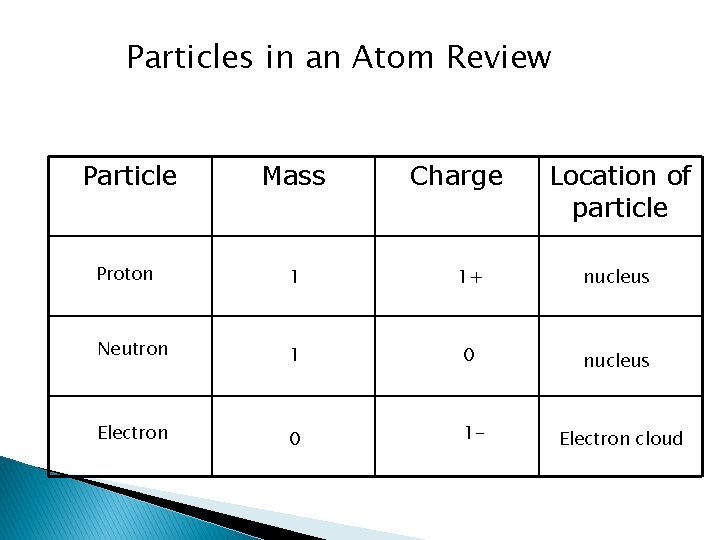
Particles in an Atom Review Particle Mass Charge Location of particle Proton 1 1+ nucleus Neutron 1 0 nucleus Electron 0 1 - Electron cloud

II. Atomic Number A. Def – the # of protons in an atom’s nucleus. 1. Every carbon atom has 6 protons… 2. Every neon atom has 10 protons… B. On P. T. – located right above the chemical symbol *Protons dictate the type of element we have* *Electrons always equal the # of protons*


III. Mass Number A. Def – The sum of protons and neutrons in the nucleus of an atom. 1. On P. T. – bottom number -round up/down 2. Ex: What is the mass number of the following elements? Carbon 12 Oxygen 16 Iron 56 Calcium 40 Sodium 23 Zinc 65

3. Mass # = Protons + Neutrons 4. How many Neutrons on average are found in the following elements? Boron 6 Fluorine 10 Silicon 14 Copper 35 Silver 61 Gold 118

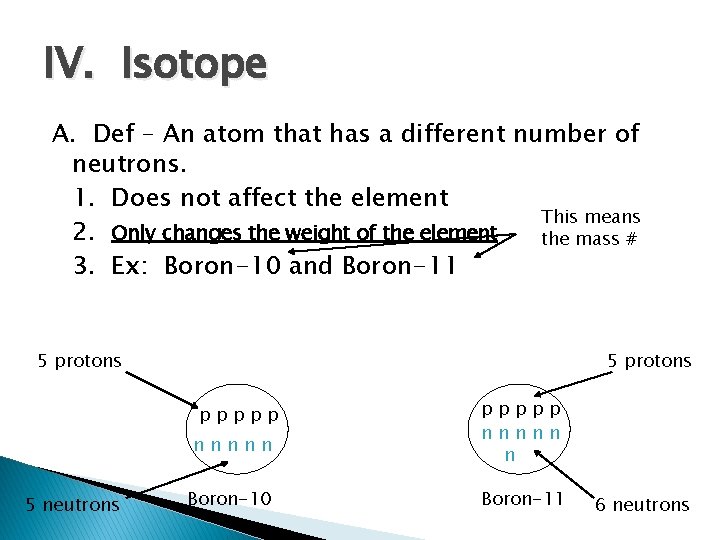
IV. Isotope A. Def – An atom that has a different number of neutrons. 1. Does not affect the element This means 2. Only changes the weight of the element the mass # 3. Ex: Boron-10 and Boron-11 5 protons ppppp nnnnn 5 neutrons Boron-10 ppppp nnnnn n Boron-11 6 neutrons

4. How many neutrons do the following isotopes have? Oxygen-14 6 Titanium-50 28 Magnesium-22 10 Potassium-43 24 Aluminum-23 10 Iodine-130 77

III. Average Atomic Mass A. Def – average mass of all isotopes of one element combined. 1. Found on P. T. underneath chemical symbol. 2. Ex: Carbon = 12. 011 “Carbon has 3 known isotopes. Carbon-12, 13, and 14. Carbon-12 makes up 99% of all carbon on earth. Carbon-13 and 14 make up 1%. ”

Chapter 4 Lesson 3 “Modern Atomic Theory” Chemistry Timeline…pg. 114 -115

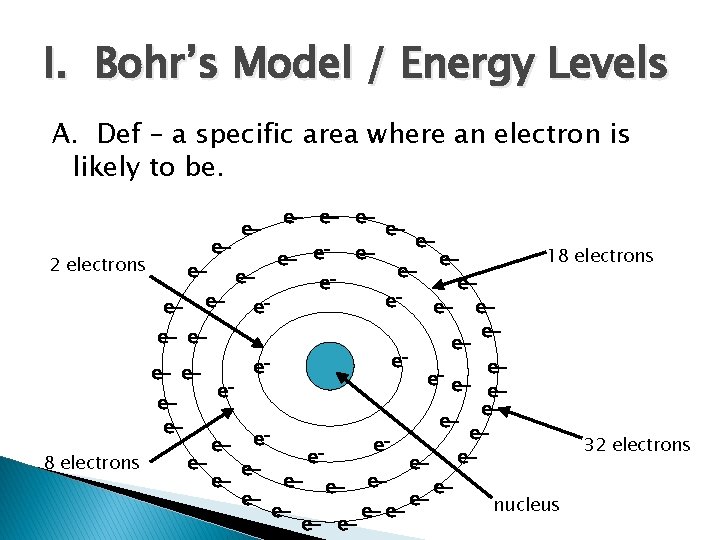
I. Bohr’s Model / Energy Levels A. Def – a specific area where an electron is likely to be. e- e- ee- e- 18 electrons eeeeee e- eee- eeee ee- eeee- e e 32 electrons e e 8 electrons eeeee- e- eeeenucleus ee- e 2 electrons

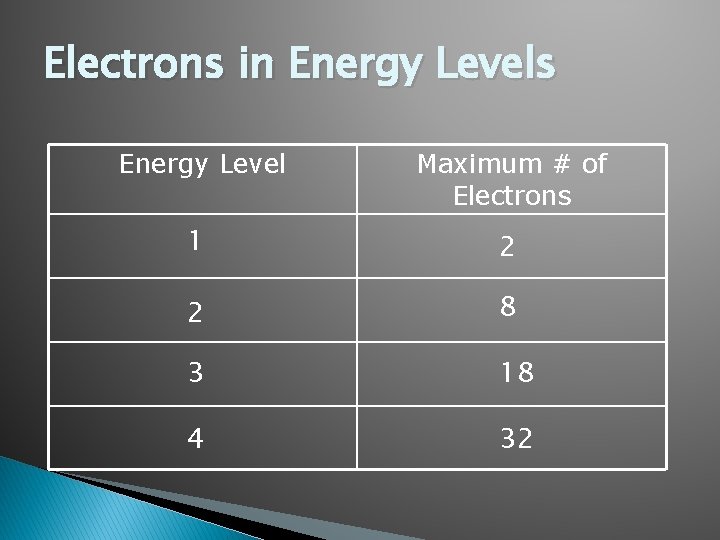
Electrons in Energy Levels Energy Level Maximum # of Electrons 1 2 2 8 3 18 4 32

In Class Assignment 1. Draw a picture of the following atoms with the correct number of e’s, p’s, and n’s… a. Na d. He b. Al e. C c. K f. S 2. Assign: Read pg. 118 and 119 3. What is the difference between a ground state and an excited state?
