Physical Science Chapter 6 Thermal Energy Heat Transfer







- Slides: 13

Physical Science Chapter 6 Thermal Energy & Heat Transfer

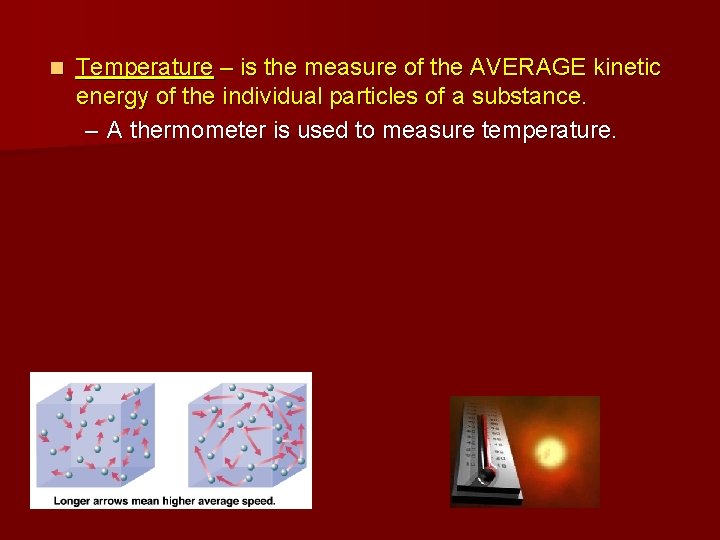
n Temperature – is the measure of the AVERAGE kinetic energy of the individual particles of a substance. – A thermometer is used to measure temperature.

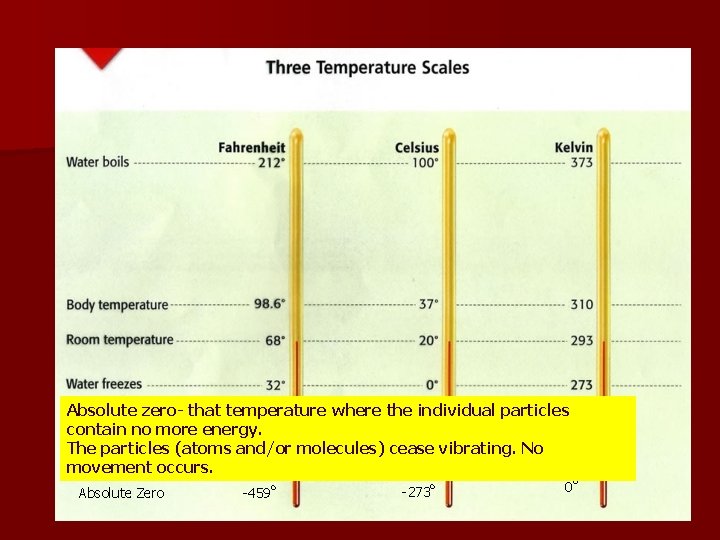
Absolute zero- that temperature where the individual particles contain no more energy. The particles (atoms and/or molecules) cease vibrating. No movement occurs. Absolute Zero -459 o -273 o 0 o

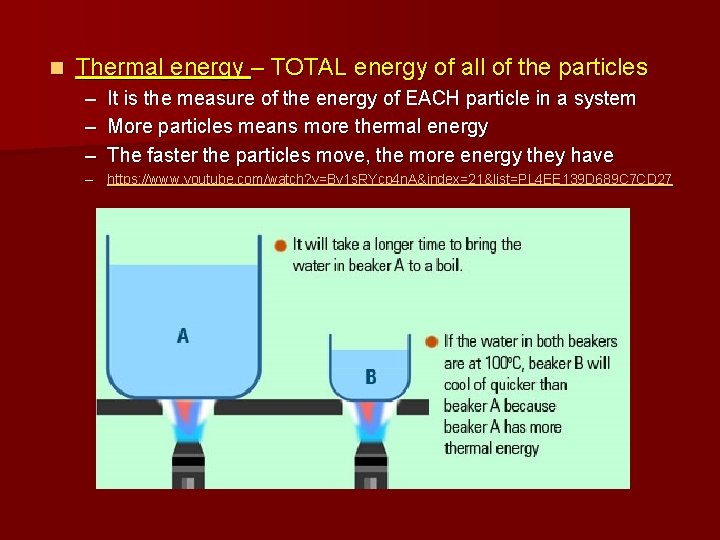
n Thermal energy – TOTAL energy of all of the particles – It is the measure of the energy of EACH particle in a system – More particles means more thermal energy – The faster the particles move, the more energy they have – https: //www. youtube. com/watch? v=By 1 s. RYcp 4 n. A&index=21&list=PL 4 EE 139 D 689 C 7 CD 27

Which has more thermal energy?

n Heat – thermal energy that moves from a warmer object to a cooler object, trying to reach thermodynamic equilibrium.

Heat of Reactions Endothermic – Heat is absorbed from the surroundings into the reaction – Ice melting, evaporation, cooking, photosynthesis – Usually feel cold: taking heat away from you. n Exothermic – Heat is released from the reaction into the surroundings. – Making ice, explosions, nuclear fusion and fission – Usually feel hot: giving you heat. n

Heat Transfer Heat is always trying to reach Thermodynamic Equilibrium n Heat is transferred (moves) in only one direction: from a warmer object to a cooler object. n – Hot coffee cools to room temp because the heat of the coffee is transferred to the cooler temperature of the room. – A cold glass of Iced tea soon warms up to the surrounding room temperature because the warmer temperature of the room’s surroundings is transferred to the colder glass of iced tea thereby warming it up. – Heat is transferred in one of three ways: § Conduction § Convection § Radiation

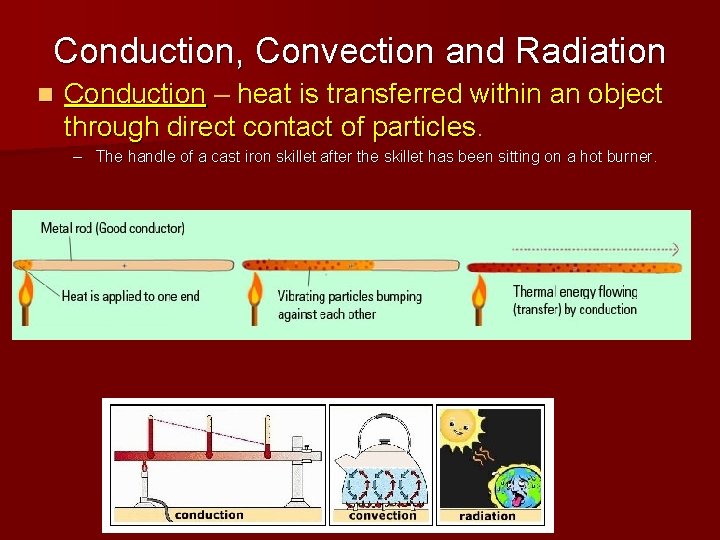
Conduction, Convection and Radiation n Conduction – heat is transferred within an object through direct contact of particles. – The handle of a cast iron skillet after the skillet has been sitting on a hot burner.

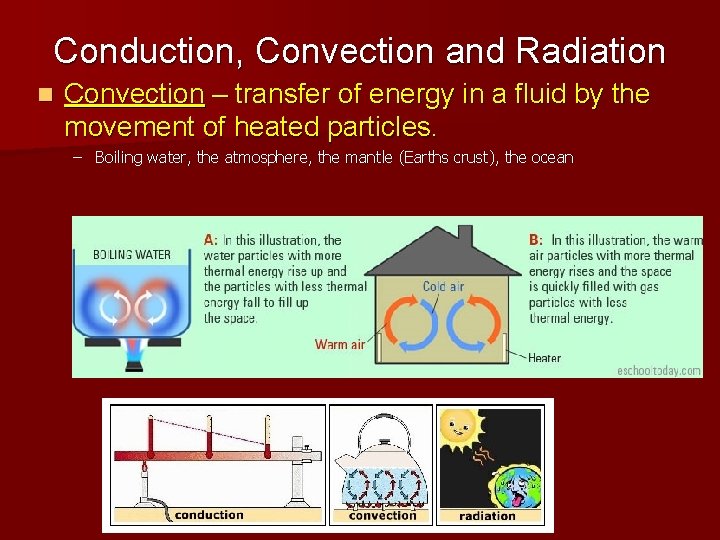
Conduction, Convection and Radiation n Convection – transfer of energy in a fluid by the movement of heated particles. – Boiling water, the atmosphere, the mantle (Earths crust), the ocean

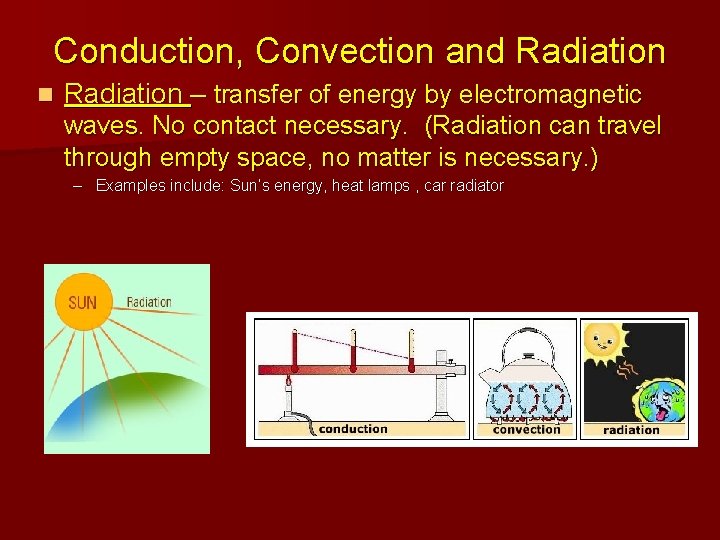
Conduction, Convection and Radiation n Radiation – transfer of energy by electromagnetic waves. No contact necessary. (Radiation can travel through empty space, no matter is necessary. ) – Examples include: Sun’s energy, heat lamps , car radiator

Heat Transfer n Conductor well. – a material that transfers heat – metal, tile, glass n Insulator – a material that does not transfer heat well. – air, carpet, wood

Song or Rap or Rhyming Poem The poem must use the assigned vocabulary terms or concepts. There must be a title reflecting the major concept of the poem. 24 pts The song must use the assigned vocabulary or concepts. 1. Assigned vocabulary and concepts 8 pts : Conduction, Convection, Radiation, Conductor, Insulator, Temperature, Thermal Energy, Heat 2. There must be an illustration (4 color minimum) showing an understanding of the notes from today 8 pts. 3. There must be a 2 to 3 paragraph explanation after the song/poem and illustration to explain how it covers the assigned concept and vocabulary listed above 8 pts. https: //www. youtube. com/watch? v=7 Y 3 mf. AGVn 1 c