Atomic Structure and the Bohr Model Atomic Structure

Atomic Structure and the Bohr Model

Atomic Structure & Bohr Model • Objectives: SWBAT describe the structure of atoms (using Bohr model) in terms of protons, neutrons and electrons, and differentiate among these particles in terms of mass, electrical charges and location within the atom. • Essential Question: How does the atom’s subatomic particle’s mass and charge shape the structure of the atom?
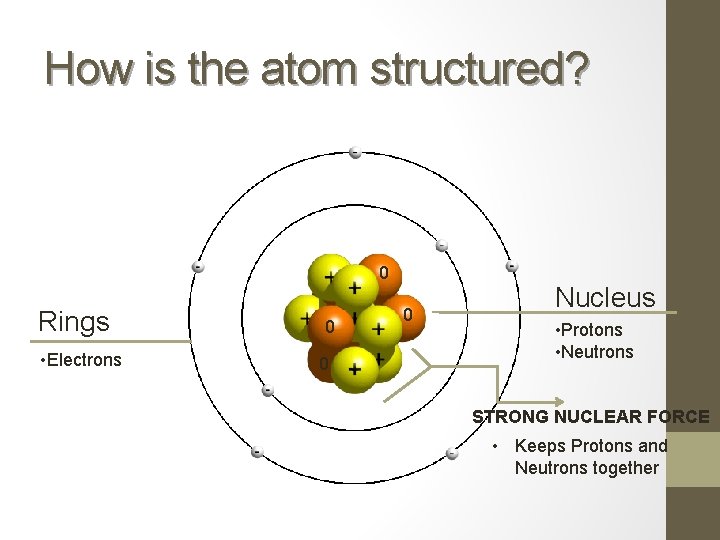
How is the atom structured? 0 Rings • Electrons 0 0 0 Nucleus • Protons • Neutrons STRONG NUCLEAR FORCE • Keeps Protons and Neutrons together
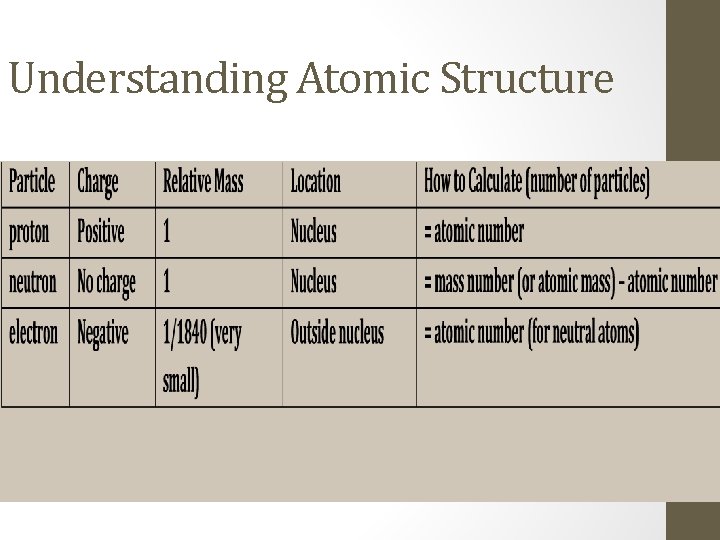
Understanding Atomic Structure

Understanding Atomic Structure • Atomic Number is the number of protons in an atom. • Mass Number or atomic mass is the total number of protons and neutrons in an atomic nucleus.

Understanding Atomic Structure • So, what type of particles are in an atom again? • Proton • What is its charge? • Where is it located? • Neutron • What is its charge? • Where is it located? • Electron • What is its charge? • Where is it located?
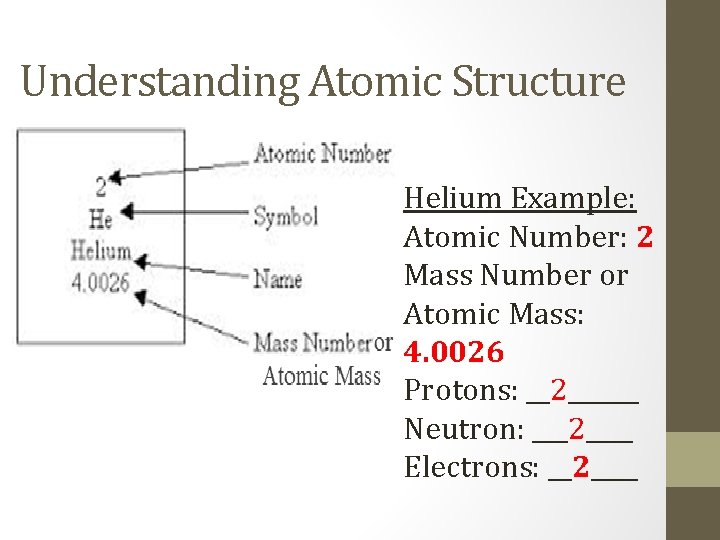
Understanding Atomic Structure Helium Example: Atomic Number: 2 Mass Number or Atomic Mass: 4. 0026 Protons: __2______ Neutron: ___2____ Electrons: __2____

Help me out! Sodium Example: Atomic Number: ______ Mass Number or Atomic Mass: _______ Protons: ____ Neutron: _______ Electrons: ______

Niels Bohr and Bohr Models • Niels Bohr: (1913) proposed that an electron is found only in specific circular paths, or orbits, around the nucleus. Electrons move around the nucleus at certain energy levels, represented as n = 1, n = 2, n =3, etc.
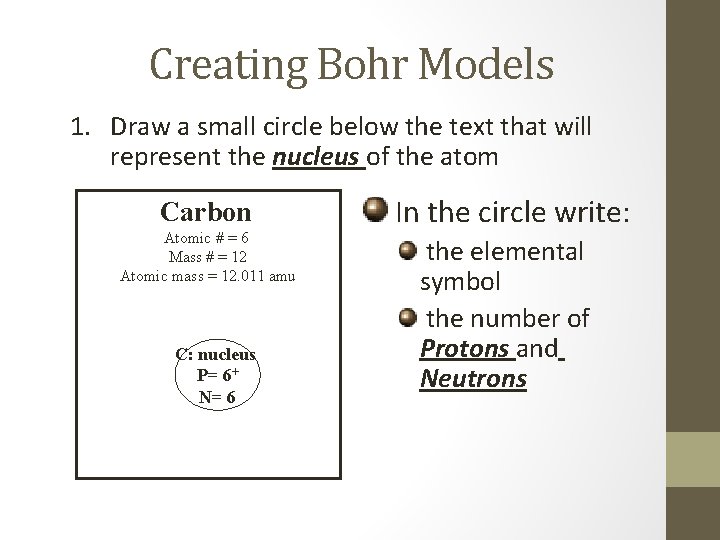
Creating Bohr Models 1. Draw a small circle below the text that will represent the nucleus of the atom Carbon Atomic # = 6 Mass # = 12 Atomic mass = 12. 011 amu C: nucleus P= 6+ N= 6 In the circle write: the elemental symbol the number of Protons and Neutrons
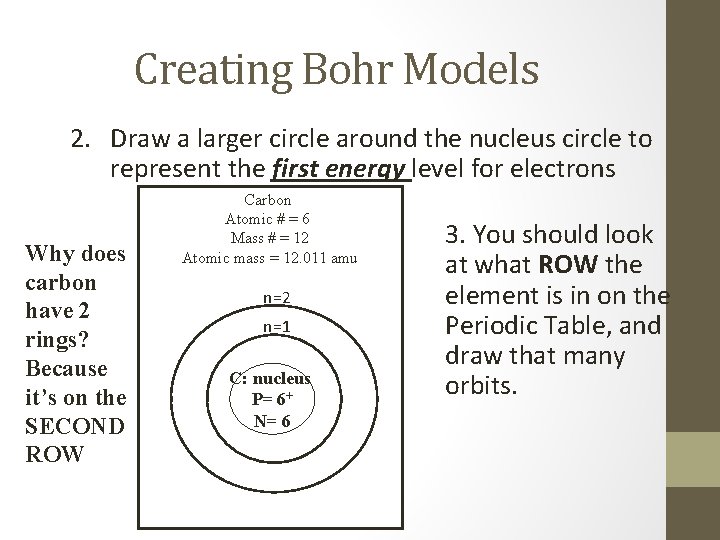
Creating Bohr Models 2. Draw a larger circle around the nucleus circle to represent the first energy level for electrons Why does carbon have 2 rings? Because it’s on the SECOND ROW Carbon Atomic # = 6 Mass # = 12 Atomic mass = 12. 011 amu n=2 n=1 C: nucleus P= 6+ N= 6 3. You should look at what ROW the element is in on the Periodic Table, and draw that many orbits.
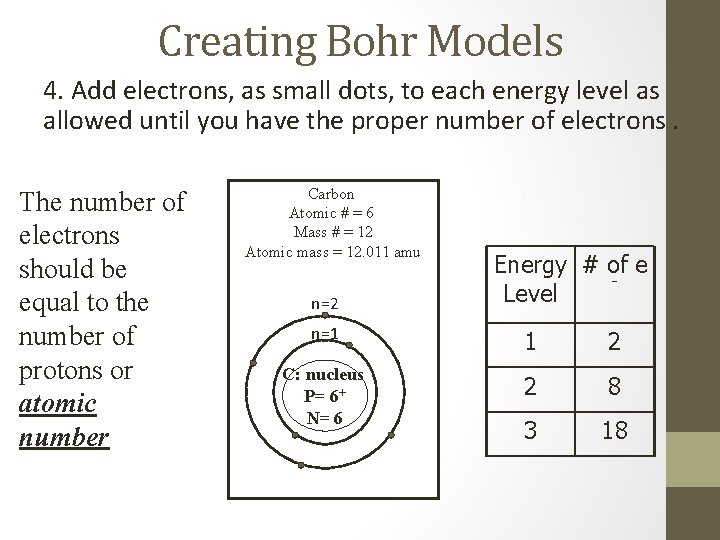
Creating Bohr Models 4. Add electrons, as small dots, to each energy level as allowed until you have the proper number of electrons. The number of electrons should be equal to the number of protons or atomic number Carbon Atomic # = 6 Mass # = 12 Atomic mass = 12. 011 amu n=2 Energy # of e Level n=1 1 2 C: nucleus P= 6+ N= 6 2 8 3 18
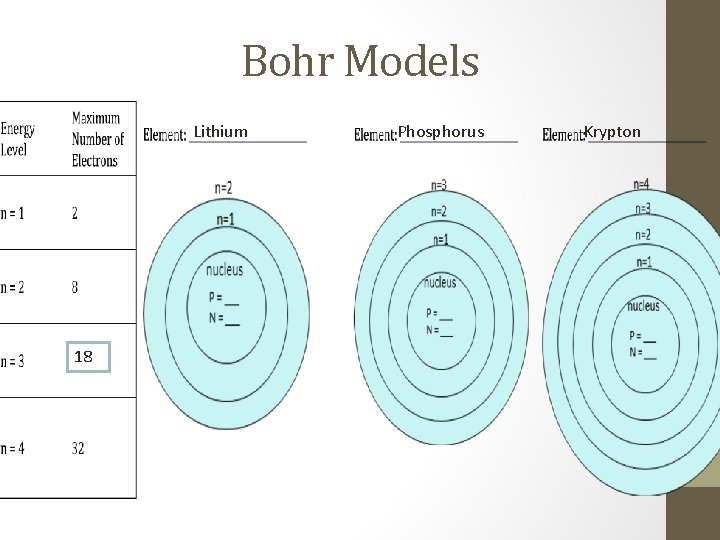
Bohr Models Lithium 18 Phosphorus Krypton
- Slides: 13