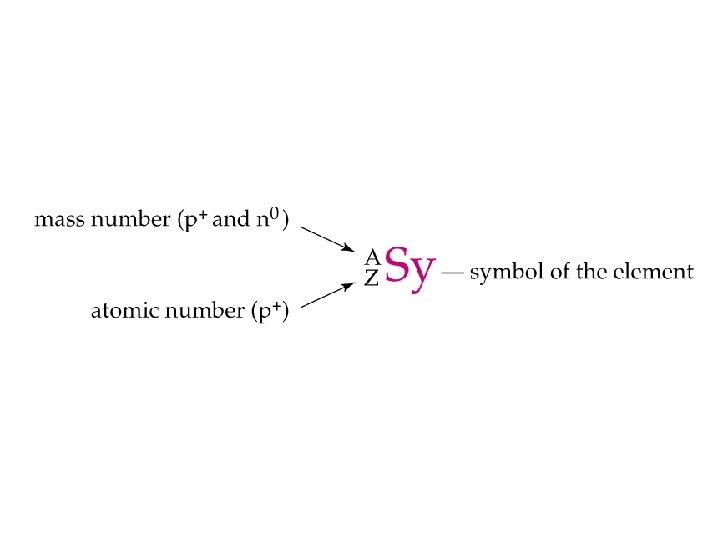
NUCLEAR CHEMISTRY Nuclear Particles PROTON NEUTRON Mass Charge























- Slides: 33

NUCLEAR CHEMISTRY Nuclear Particles: PROTON NEUTRON Mass Charge Symbol 1 amu +1 H+, H, p 1 amu © Copyright 1994 -2001 R. J. Rusay 0 n


Nuclear Decay / Radioactivity Unstable nuclei “decay” i. e. they lose particles which lead to other elements and isotopes. ð The elements and isotopes produced may also be unstable and go through further decay. ð Nuclear decay: reactions conserve mass. ð © Copyright 1994 -2001 R. J. Rusay

Nuclear Particles emitted from unstable nucleii ð Emitted Particles: Mass Charge Symbol ð alpha particle 4 amu ð beta particle very small ð gamma very small ð positron very small © Copyright 1994 -2001 R. J. Rusay +2 -1 0 +1

Nuclear Penetrating Power alpha particle: low ð beta particle: moderate ð gamma: high ð ð X-rays? Water © Copyright 1994 -2001 R. J. Rusay

Alpha Decay–Heavy Elements ð 238 U 234 Th + + e t 1/2 = 4. 48 x 10 9 years ð 210 Po + + e 206 Pb t 1/2 = 138 days ð 256 Rf 252 No + +e t 1/2 = 7 ms For Cobalt 60 predict the alpha decay product:

Beta Decay–Electron Emission ð ð n 14 C p+ + + Energy 14 N + + Energy t 1/2= 5730 years ð 90 Sr 247 Am ? ð 90 Y t 1/2= 30 years 247 Cm t 1/2= 22 min ð 131 I + + Energy 131 Xe ? ? t 1/2 = 8 days + + Energy + + Energy

Electron Capture–Positron Emission p+ + e- n + Energy = Electron capture 51 Cr + e- 51 V + Energy t 1/2 = 28 days p+ n + e+ + Energy = 7 Positron Be 7 Li emission + + Energy t 1/2 = 53 days 177 Ta ? + e- 177 Hf + Energy 144 Gd ? + 144 Eu + Energy

Nuclear Decay Predict the Particle or element: ð Thallium 206 decays to Lead 206. What particle is emitted? ð Cesium 137 goes through Beta decay. What element is produced and what is its mass? ð Thorium 230 decays to Radium 226. What particle is emitted? © Copyright 1994 -2001 R. J. Rusay

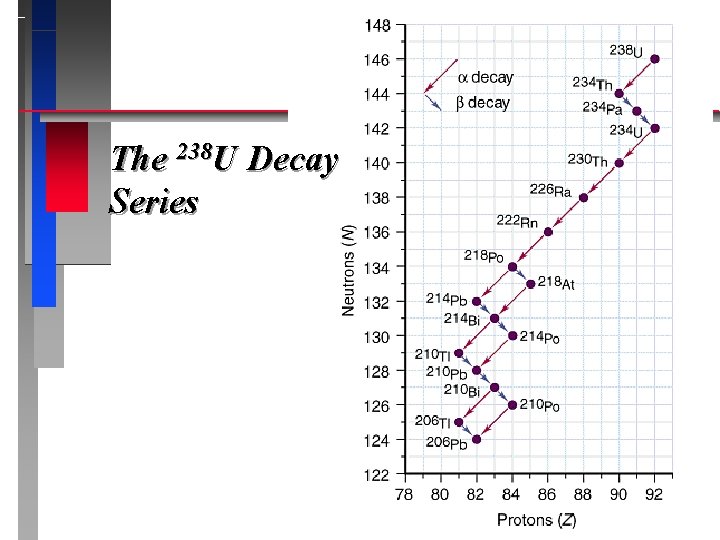
Nuclear Decay Series ð If the nuclei produced from radioactive decay are unstable, they continue to decay until a stable isotope results. ð An example is Radium which produces Lead © Copyright 1994 -2001 R. J. Rusay

The 238 U Decay Series

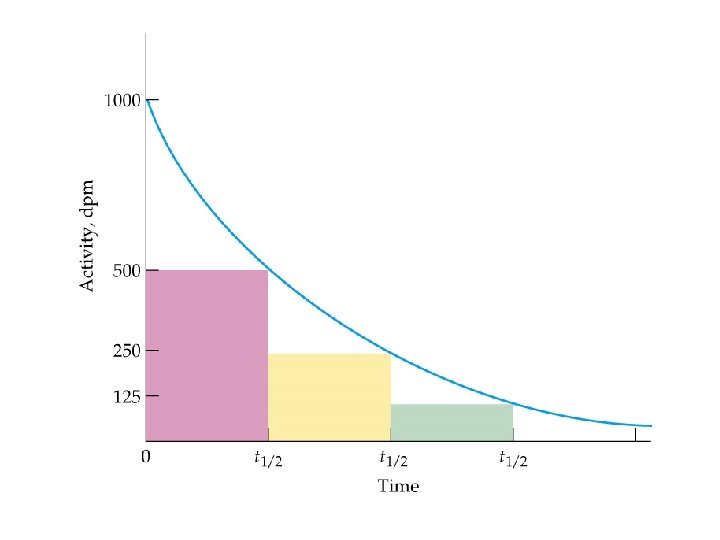
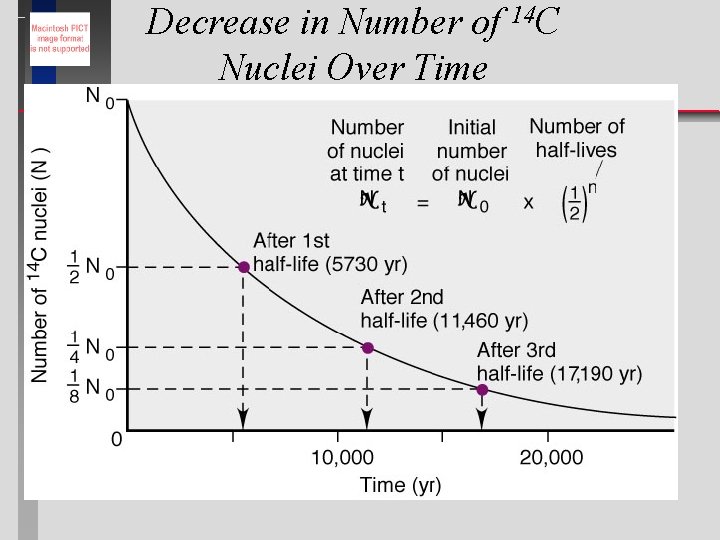
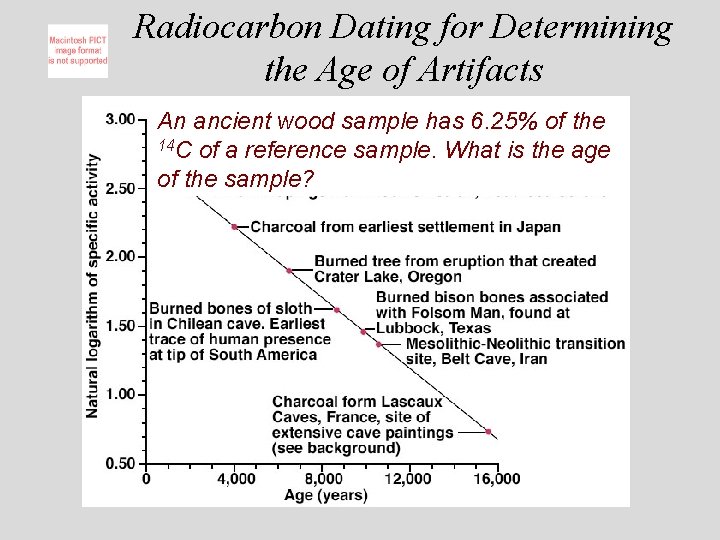
Radiodating Methods Three isotopes are currently used: Carbon-14 half life 5, 730 yrs Potassium-40 half life 1. 3 x 10 9 yrs Uranium-238 half life 4. 47 x 10 9 yrs ð The age of samples can be determined by measuring their disintegrations over time. ð © Copyright 1994 -2001 R. J. Rusay


Decrease in Number of 14 C Nuclei Over Time

Radiocarbon Dating for Determining the Age of Artifacts An ancient wood sample has 6. 25% of the 14 C of a reference sample. What is the age of the sample?

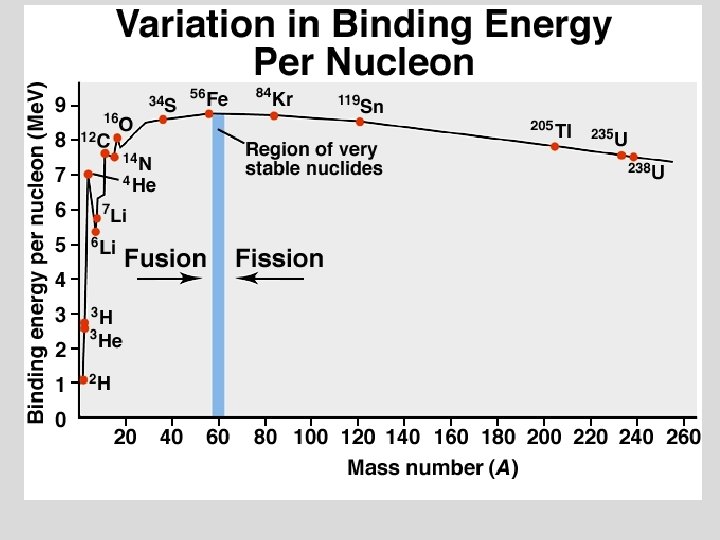
Nuclear Reactions The mass of the visible universe is 73% H 2 and 25% He. The remaining 2%, “heavy” elements, have atomic masses >4. ð The “heavy” elements are formed at very high temperatures (T>10 6 o. C) by FUSION, i. e. nuclei combining to form new elements. ð There is an upper limit to the production of heavy nuclei at A=92, Uranium. ð Heavy nuclei split to lighter ones by FISSION ð © Copyright 1994 -2001 R. J. Rusay

NUCLEAR STABILITY Patterns of Radioactive Decay Alpha decay ( ) –heavy isotopes ð Beta decay ( ) –neutron rich isotopes ð Positron emission ( )–proton rich isotopes ð Electron capture–proton rich isotopes x-rays ð Gamma-ray emission ( ð Spontaneous fission–very heavy isotopes ð


NUCLEAR ENERGY ð EINSTEIN’S EQUATION FOR THE CONVERSION OF MASS INTO ENERGY 2 ðE = mc ð m = mass (kg) c = Speed of light 8 c = 2. 998 x 10 m/s ð

Mass Energy Electron volt (ev) The energy an electron acquires when it moves through a potential difference of one volt: 1 ev = 1. 602 x 10 -19 J Binding energies are commonly expressed in units of megaelectron volts (Mev) 1 Mev = 106 ev = 1. 602 x 10 -13 J A particularly useful factor converts a given mass defect in atomic mass units to its energy equivalent in electron volts: 1 amu = 931. 5 x 106 ev = 931. 5 Mev

Nuclear Reactions ð ð ð Fission and Fusion reactions are highly exothermic (1 Mev / nucleon). This is 10 6 times larger than “chemical” reactions which are about 1 ev / atom. Nuclear fission was first used in a chain reaction: © Copyright 1994 -2001 R. J. Rusay

Nuclear Reactions: Fission: ð ð Fusion: Fission and Fusion reactions are highly exothermic (1 Mev / nucleon). This is 10 6 times larger than “chemical” reactions which are about 1 ev / atom.

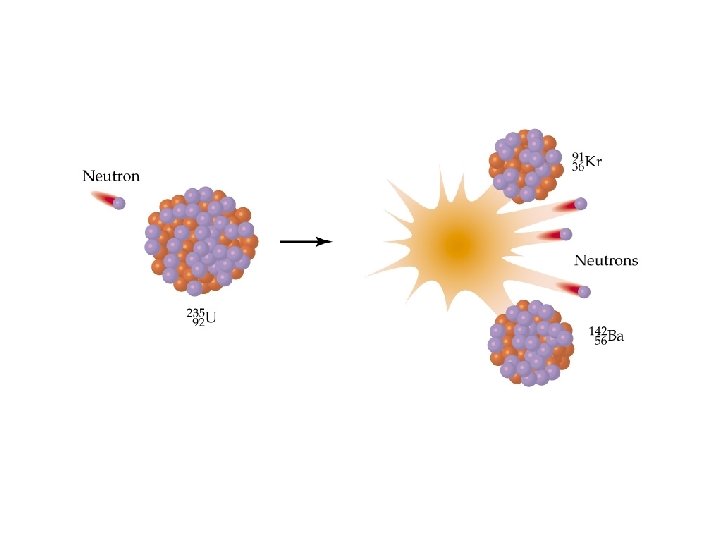
Nuclear Reactions ð Nuclear fission was first used in a chain reaction:



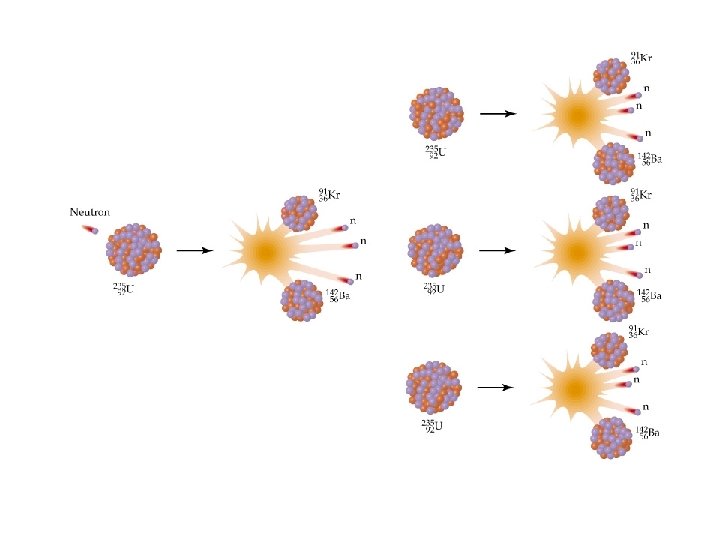
Nuclear Reactions / Fission The Fission Chain Reaction proceeds geometrically: 1 neutron -> 3 -> 9 -> 27 -> 81 etc. ð 1 Mole of U-235 (about 1/2 lb) produces 2 x 1010 k. J which is equivalent to the combustion of 800 tons of Coal! ð Commercial nuclear reactors use fission to produce electricity. . Fission bombs were used in the destruction of Hiroshima and Nagasaki, Japan, in August 1945. ð © Copyright R. J. Rusay 1994 -2001

The Nuclear Dawn August 6, 1945

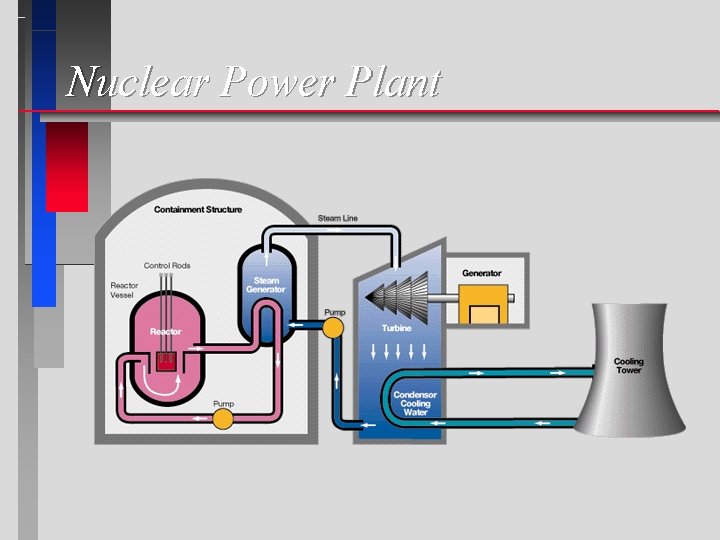
Nuclear Power Plant

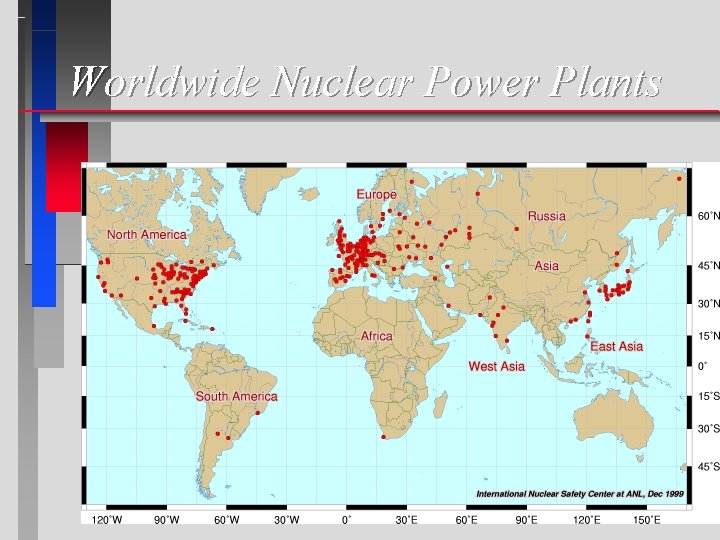
Worldwide Nuclear Power Plants

Chernobyl, Ukraine April, 1986 and April, 2001

Nuclear Reactions / Fusion ð ð Fusion has been described as the chemistry of the sun and stars. It too has been used in weapons. It has not yet found a peaceful commercial application ðThe application has great promise in producing relatively “clean” abundant energy through the combination of Hydrogen isotopes particularly from 2 H, deuterium and 3 H, tritium: (NIF/National Ignition Facility, LLNL) © Copyright 1994 -2001 R. J. Rusay

National Ignition Facility Lawrence Livermore National Laboratory

National Ignition Facility Lawrence Livermore National Laboratory © Copyright 1994 -2001 R. J. Rusay