NUCLEAR CHEMISTRY Subatomic Particles Protons positive charge In

NUCLEAR CHEMISTRY
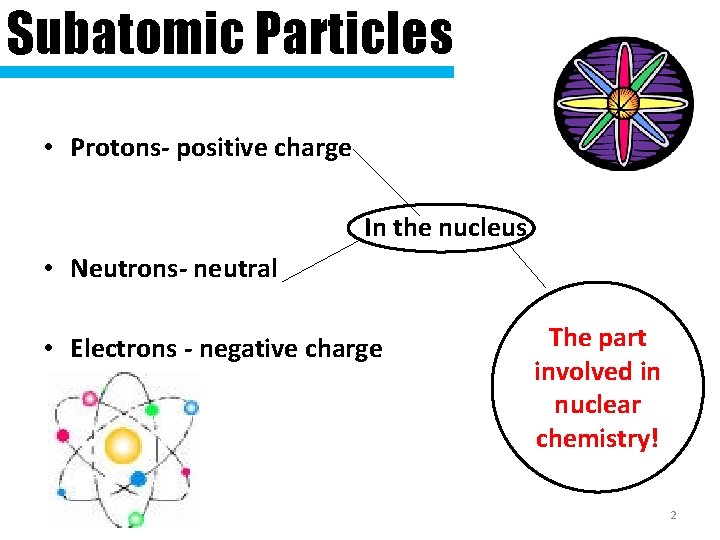
Subatomic Particles • Protons- positive charge In the nucleus • Neutrons- neutral • Electrons - negative charge The part involved in nuclear chemistry! 2

Examples: Nuclear Fission • Carbon-14 Dating • The nuclear bomb • Nuclear power • Nuclear medicine • Radon testing in basements • Chain Reactions

What holds the nucleus together? • Normally particles with same charge would repel each other – So why doesn’t the nucleus fly apart from protons repelling each other? ! • Strong Force

Where do the radioactive particles and energy • Sometimes there come from? are too many neutrons - makes the atom unstable • The atom flies apart and releases particles and energy from the nucleus
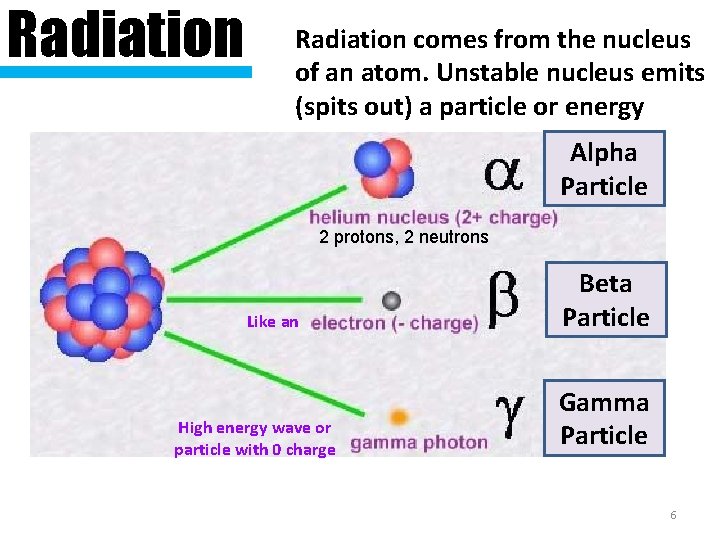
Radiation comes from the nucleus of an atom. Unstable nucleus emits (spits out) a particle or energy Alpha Particle 2 protons, 2 neutrons Like an High energy wave or particle with 0 charge Beta Particle Gamma Particle 6
Charge of Nuclear Particles
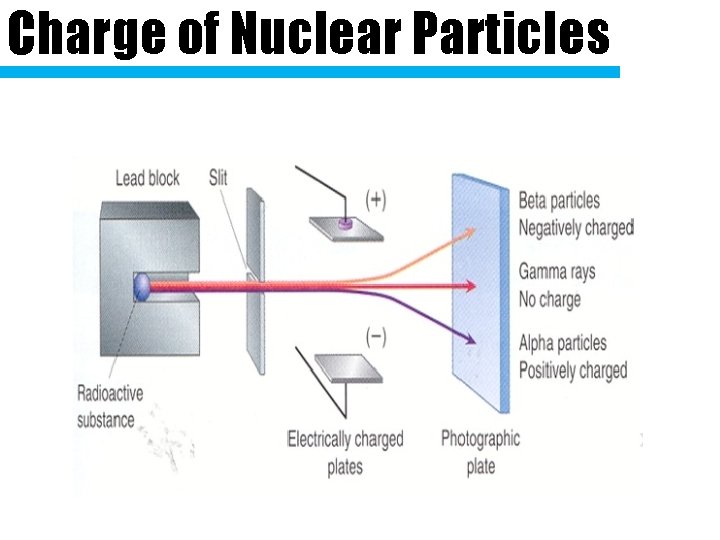
Charge of Nuclear Particles
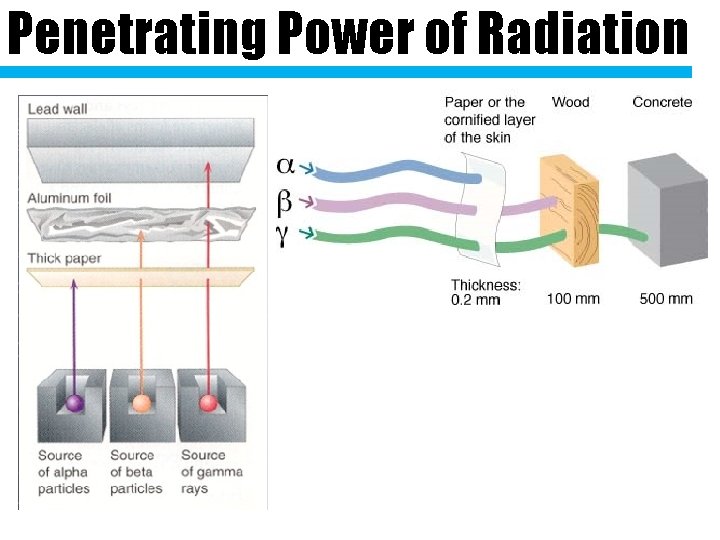
Penetrating Power of Radiation

Copy the symbols down Type What is it? Symbol Charge What Stops It 2 protons Alpha 2 neutrons Particle 2+ Paper Like an Beta Particle electron Aluminum, 1 wood, clothes (Helium nucleus) Gamma High speed energy waves Ray 0 Thick lead or concrete
Groups for Post Test Questions #1 23 26 6 18 33* #2 5 14 10 19 #3 4 16 9 1 Seat numbers with a * - move to any group that only has three people due to someone being absent #4 29 21 25 17 #5 20 28 3 7 #6 2 12 22 27 34* #7 30 11 32 15 #8 8 13 24 31 35* THE SALMON COLORED PAPERS MUST STAY IN THE CLASSROOM AT ALL TIMES! IF YOU TAKE IT OUT OF THE CLASSROOM YOU WILL RECEIVE A ZERO!
- Slides: 11