ATOMS Proton Neutron Electron SUBATOMIC PARTICLES PARTICLE CHARGE


ATOMS - + + + - - • Proton (+) • Neutron • Electron(-)

SUBATOMIC PARTICLES PARTICLE CHARGE LOCATION PROTON + Nucleus NEUTRON NONE Nucleus - Outside Nucleus ELECTRON (in motion)

SUBATOMIC PARTICLES

Basic Atomic Structure Movie
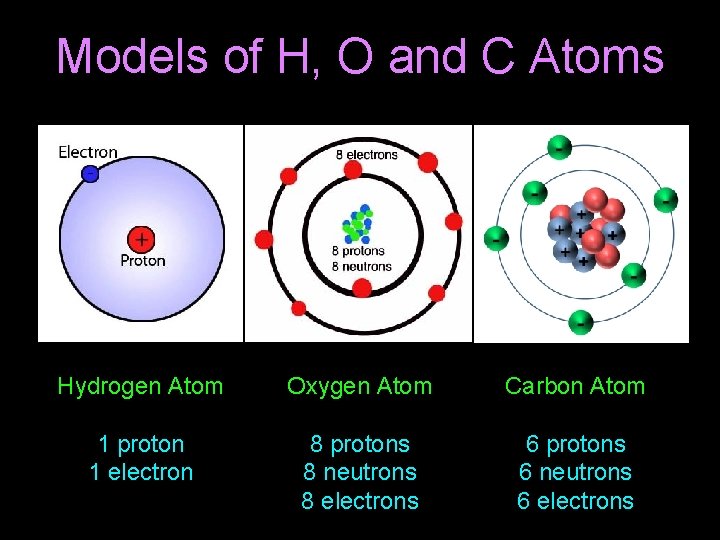
Models of H, O and C Atoms Hydrogen Atom Oxygen Atom Carbon Atom 1 proton 1 electron 8 protons 8 neutrons 8 electrons 6 protons 6 neutrons 6 electrons
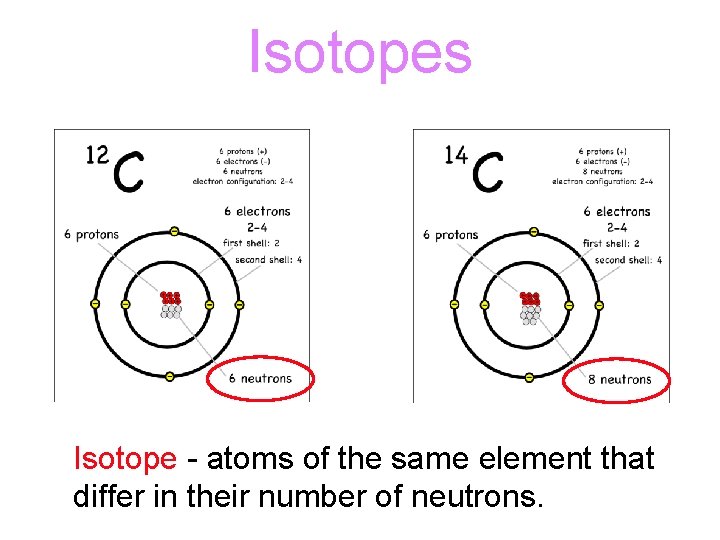
Isotopes Isotope - atoms of the same element that differ in their number of neutrons.
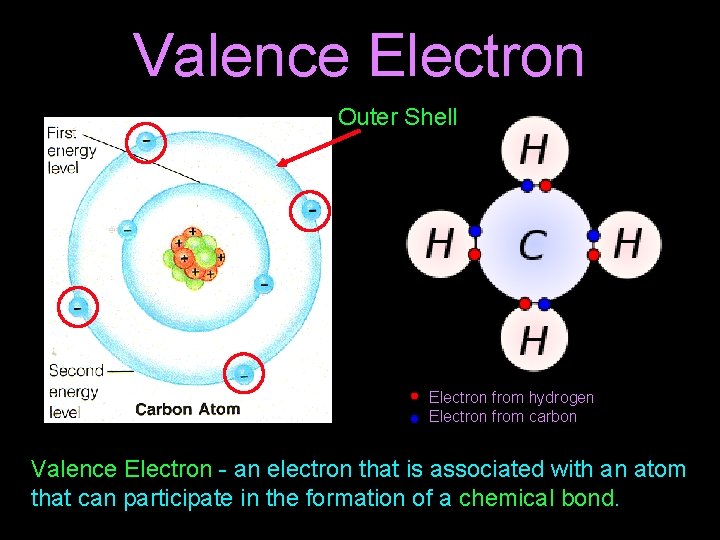
Valence Electron Outer Shell Electron from hydrogen Electron from carbon Valence Electron - an electron that is associated with an atom that can participate in the formation of a chemical bond.

Stop Here

Chemical Compound - a substance formed by the chemical combination of two or more elements in definite proportions.
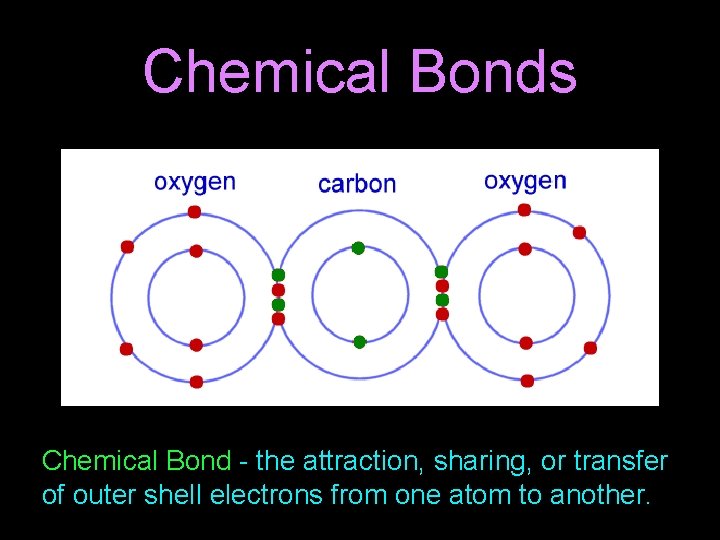
Chemical Bonds Chemical Bond - the attraction, sharing, or transfer of outer shell electrons from one atom to another.
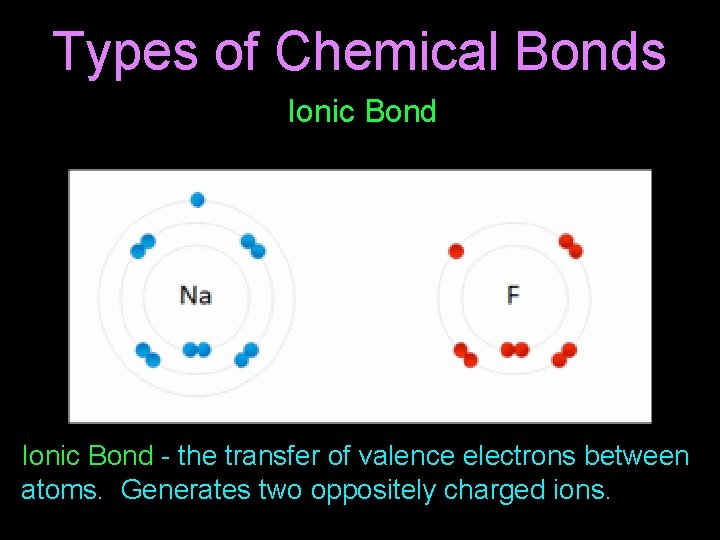
Types of Chemical Bonds Ionic Bond - the transfer of valence electrons between atoms. Generates two oppositely charged ions.
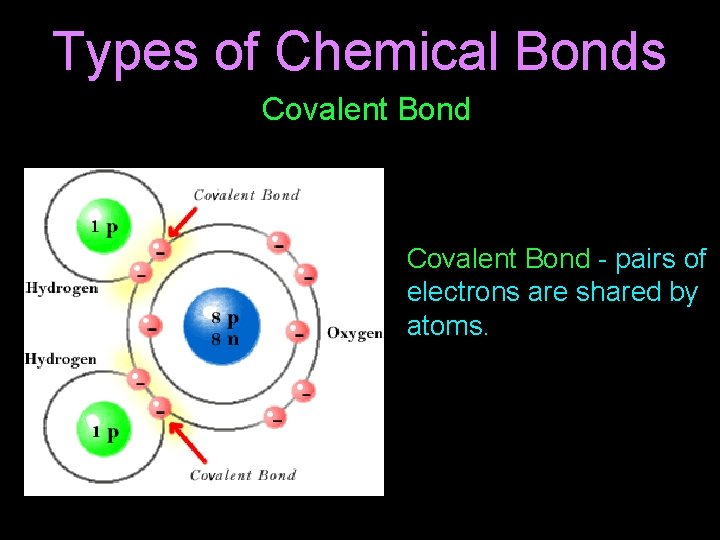
Types of Chemical Bonds Covalent Bond - pairs of electrons are shared by atoms.
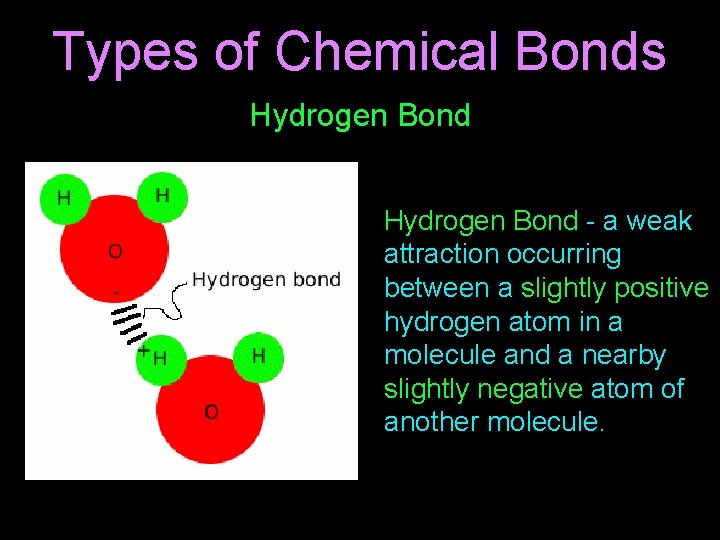
Types of Chemical Bonds Hydrogen Bond - a weak attraction occurring between a slightly positive hydrogen atom in a molecule and a nearby slightly negative atom of another molecule.

Water
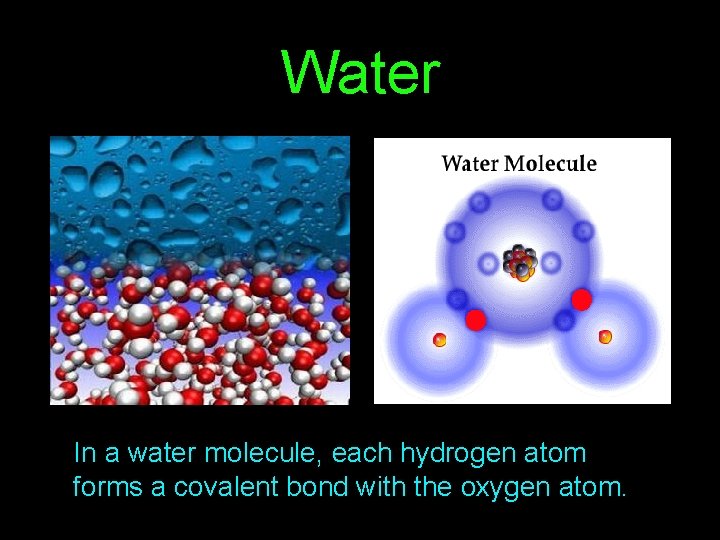
Water In a water molecule, each hydrogen atom forms a covalent bond with the oxygen atom.

Water is a Polar Molecule Water has positive charges on one side and negative charges on the other side

Properties of Water • • • Water is polar Water resists temperature changes Water expands when it freezes Water sticks well to itself (cohesion) Water sticks well to other things (adhesion) Water has a high level of surface tension

What is Surface Tension? Surface tension is the cohesive force between liquid molecules.

Stop Here

Sinkin’ Lincoln Lab Objectives: • Measure the surface tension of different liquids. • Identify independent and dependent variables. • Plot a graph with the data collected. • Learn about polar molecules and surface tension.
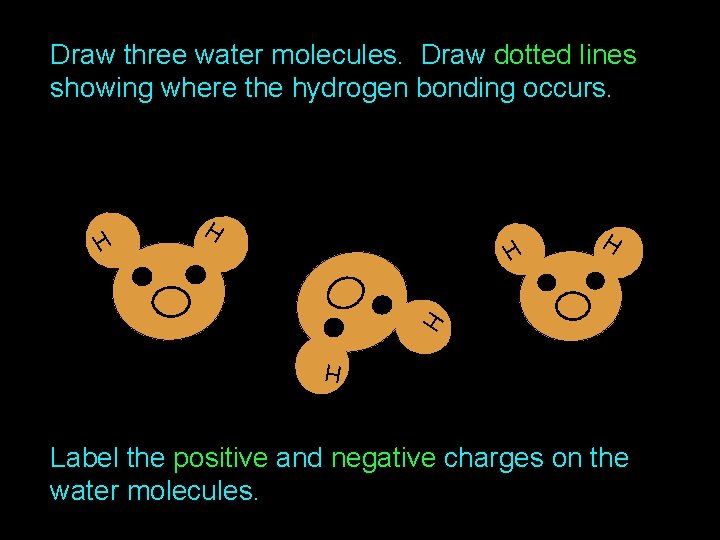
Draw three water molecules. Draw dotted lines showing where the hydrogen bonding occurs. H H H Label the positive and negative charges on the water molecules.

Stop Here
- Slides: 23