Nuclear Chemistry http www mhhe comphysscichemistryessentialchemistryflashradioa 7 swf



















- Slides: 28

Nuclear Chemistry

http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/radioa 7. swf


Radioactive decay movie n http: //streaming. discoveryeducation. co m/index. cfm

Radioactivity: n n Decay of, and emission of electromagnetic radiation and particles from the nuclei of certain elements that spontaneously desintegrate. Atoms with Atomic Number greater than 83 (unstable)

ALPHA DECAY: Alpha emitter (+): He, α (4 amu, charge 2+) n n n Ra → Rn + He Atomic number decreases by 2. (2 p) The mass number decreases by 4. (2 p+2 n)

Beta Decay: A beta particle is a high speed electron. n Pb → Bi + e n Th → Pa + e n n Atomic number increases by 1 (1 p) Mass number remains the same.

GAMMA RADIATION (γ) Are not particles: no mass, no charge. Similar to X-rays, but greater energy.

Positron Emission: o amu. , 1+; e. , moderate power. n p → n n K → Ar n n + e Atomic number decreases by 1 Atomic mass remains the same.

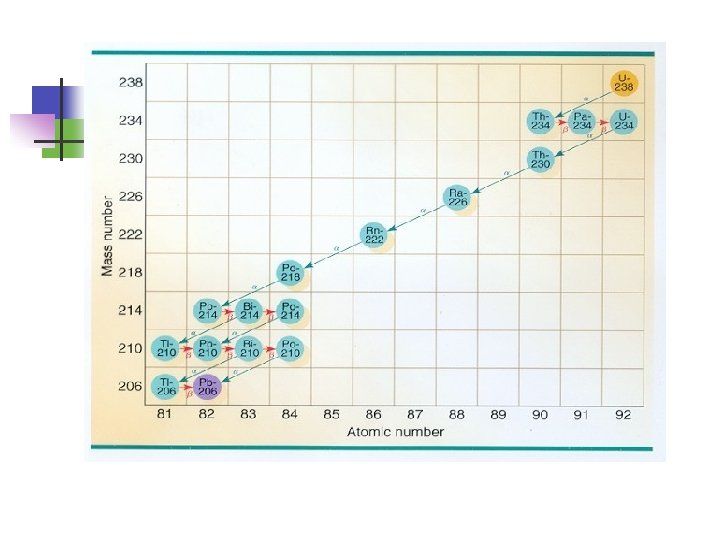
Decay series of 235 U (based on data from the U. S. Department of Energy): 235 U --> 231 Th + 24 α + γ 231 Th --> 231 Pr + β- + γ 231 Pr --> 227 Ac + 24 α + γ 227 Ac --> 227 Th + β- or 227 Ac --> 223 Fr + 24 α 227 Th --> 223 Ra + 24 α + γ or 227 Ac --> 223 Ra + β- + γ 223 Ra --> 219 Rn + 24 α 219 Rn --> 215 Po + 24 α + γ 215 Po --> 211 Pb + 24 α 211 Pb --> 211 Bi + β- + γ 211 Bi --> 207 Tl + 24 α + γ 207 Tl --> 207 Pb + βAs is clear, the final product of uranium-235 decay is lead.

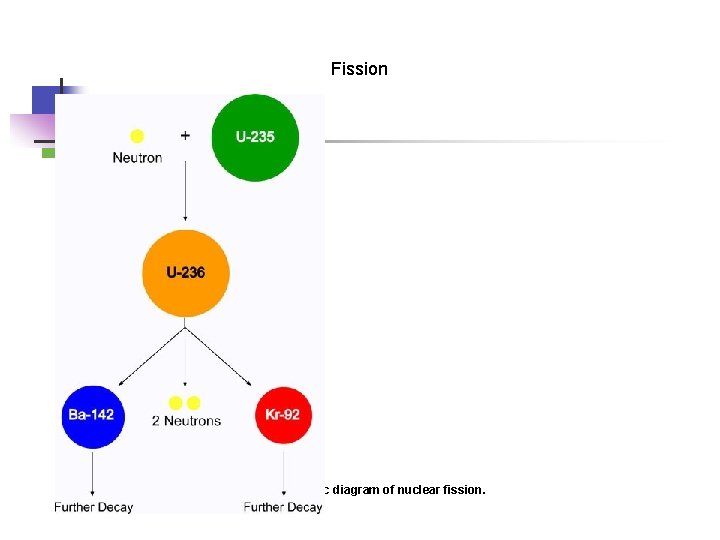
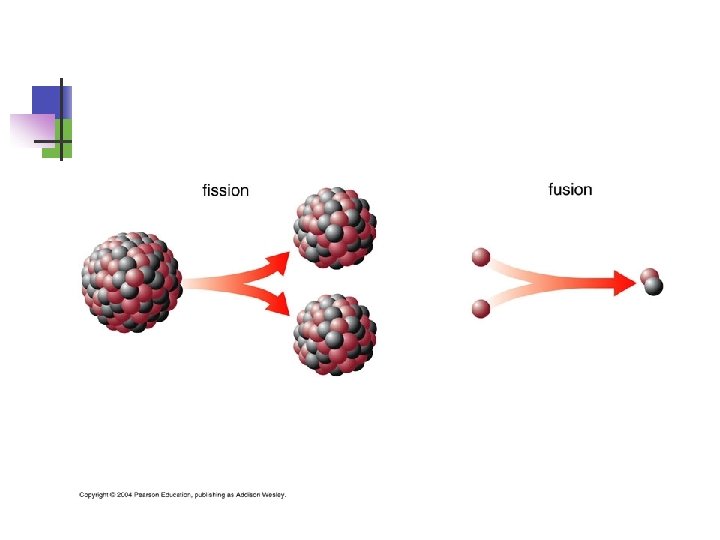
Fission Figure 6: A basic diagram of nuclear fission.





– Nuclear Decay. Practice Write equations for the following nuclear decay reactions. Make sure that both mass numbers and atomic numbers are balanced on each side. 1. Decay of polonium-218 by alpha ( ) emission. 2. Decay of carbon-14 by beta ( -) emission. 3. Decay of chlorine-32 by positron ( +) emission. 4. Decay of promethium-142 by electron capture.

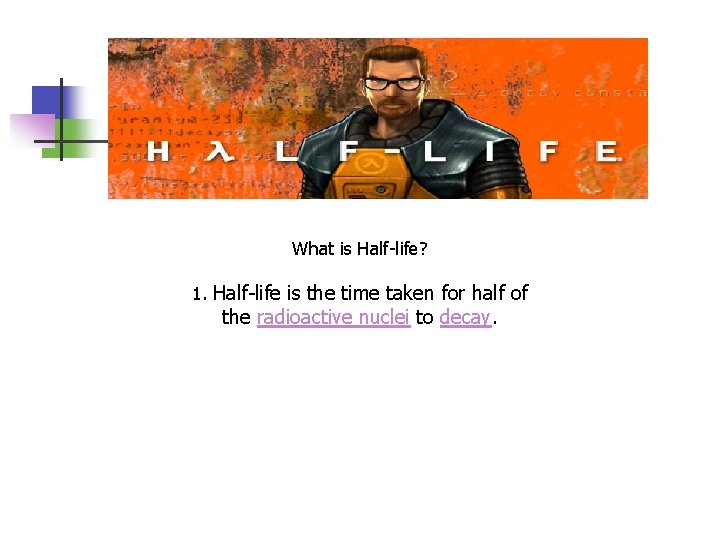
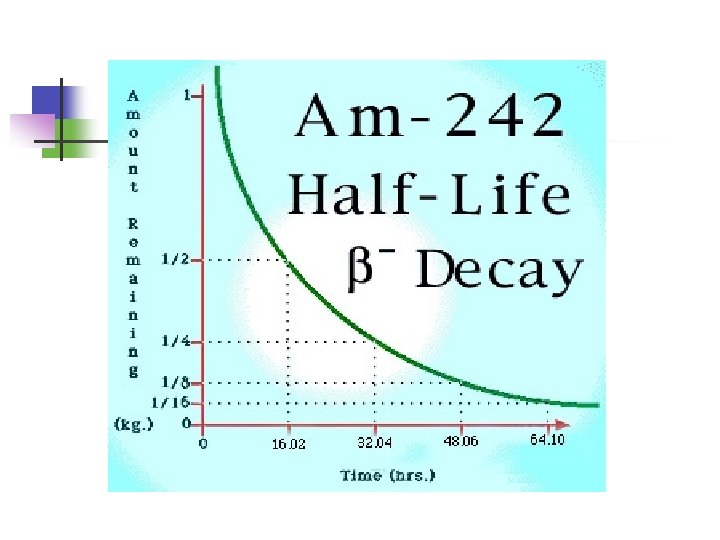
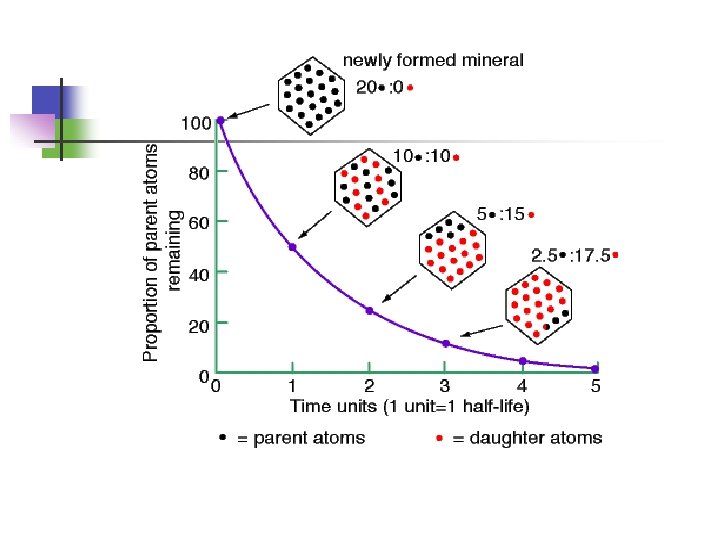
What is Half-life? 1. Half-life is the time taken for half of the radioactive nuclei to decay.

Half-life The half-life of phosphorous-30 is 2. 5 min. If you start with 35 g of phosphorus-30, how many grams would remain after 20. 0 min? The half-life of polonium-210 is 138. 4 days. How many milligrams of polonium-210 remain after 415. 2 days if you start with 2. 0 mg of the isotope? 20. 0 g of a radioactive isotope are present at 1: 00 p. m. , and 5. 0 g remain at 2: 00 p. m. How many half-lives have gone by? _____________ How long is the half-life of the isotope? _____________ Predict how many grams will be left at 2: 30 p. m. _____________

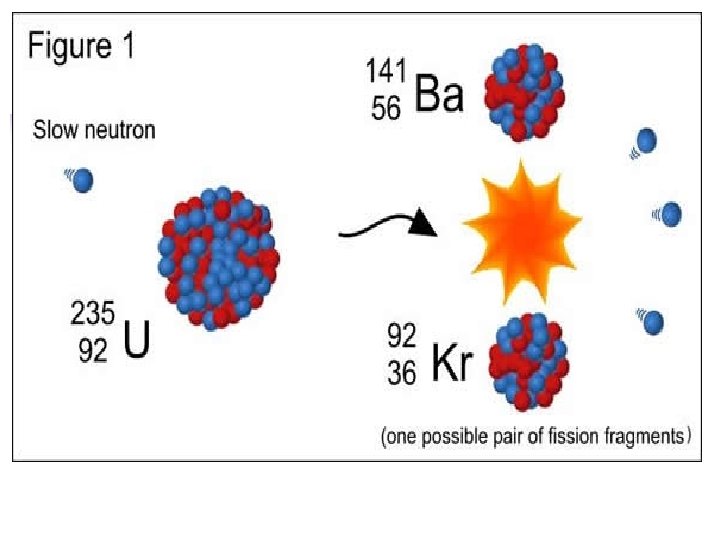
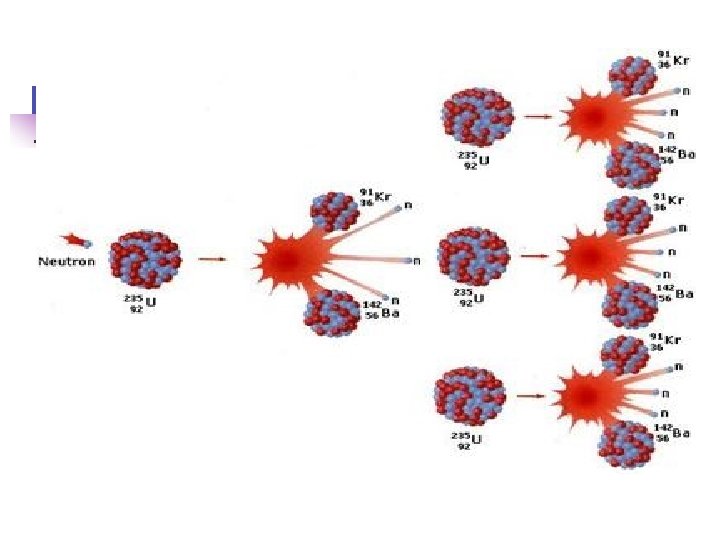
Nuclear equations Q. Write the 14 0 14 beta decay for C 6 C -1 e + 7 N -14 209 4 205 Q. Write the + Po He Pb 84 2 82 alpha decay for 209 Po Q. Complete this fission reaction 94 1 235 1 139 92 U + 0 n 3 0 n + 56 Ba + 36 Kr In all cases, charge and mass must be balanced

Discovery of Radioactivity is the release of energy or particles when an atom disintegrates (demo) n Radioactivity was discovered when minerals were exposed to film through an opaque cover n The 3 types of radioactivity can be shown by passing emissions through an electrical field: n Lead block + Radioactive substance – Phosphorescent zinc sulfide detection screen Strong magnetic or electrostatic field

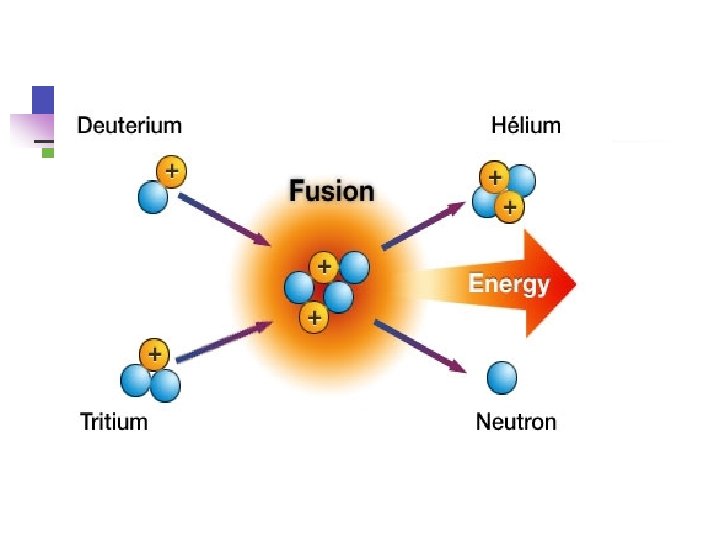
Types of Radioactivity Types of radiation: 1) , 2) , 3) 1. Alpha ( ) particles are symbolized as 42 He 2. Beta ( ) particles (essentially electrons) are 0– 1 e 3. Gamma ( ) rays are symbolized as 00 n You can determine the composition of each: : mass of 4 u, charge of +2 (2 p+, 2 n 0, 0 e–) n Other symbols: proton = 11 p, neutron = 10 n n There are different terms to describe the different types of nuclear reactions n “alpha decay” means an particle is given off. n Other: beta decay, fusion (meaning to bring together), fission (meaning to break apart)

Risk of Radioactivity n n n Biological exposure; (mutations, cancer) Radioactive waste ( Isotopes w/long half live) Danger of accidents (Chernobyl, Three Mile Island, Fukushima Daiichi plant. )

Chernobyl https: //www. youtube. com/watch? v=i. LJu. Q QDPY 8 Y#t=161. 939396

Benefits n n n Nuclear power- electricity. Tracers: I-131, Carbon-14(short half lives) Food: used in food preservation Geology and Archeology: Dating Rocks: U-238/Pb-206 Dating Fossils; Carbon dating C-14/C-12



