NMR Nuclear Magnetic Resonance M Proton NMR Symmetry

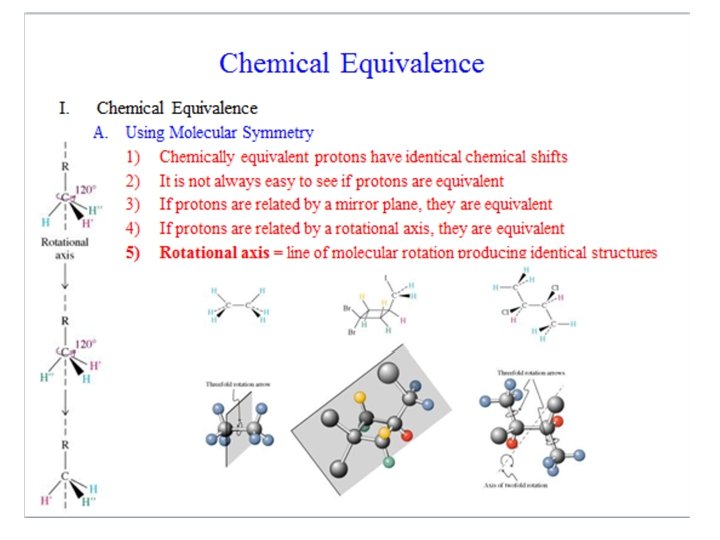





- Slides: 46

NMR Nuclear Magnetic Resonance M Proton NMR: Symmetry Index NMR-basics H-NMR


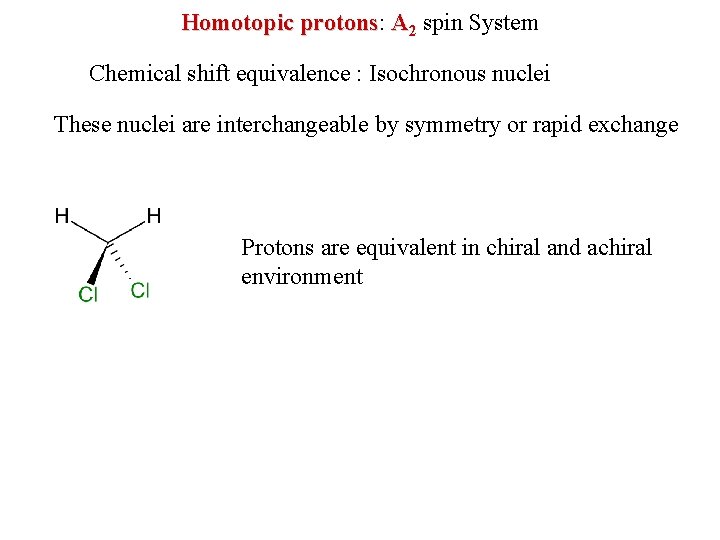
Homotopic protons: protons A 2 spin System Chemical shift equivalence : Isochronous nuclei These nuclei are interchangeable by symmetry or rapid exchange Protons are equivalent in chiral and achiral environment

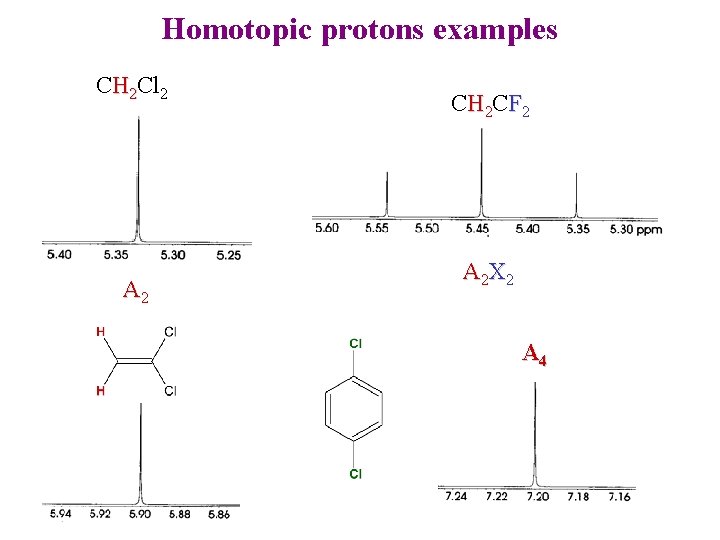
Homotopic protons examples CH 2 Cl 2 A 2 CH 2 CF 2 A 2 X 2 A 4

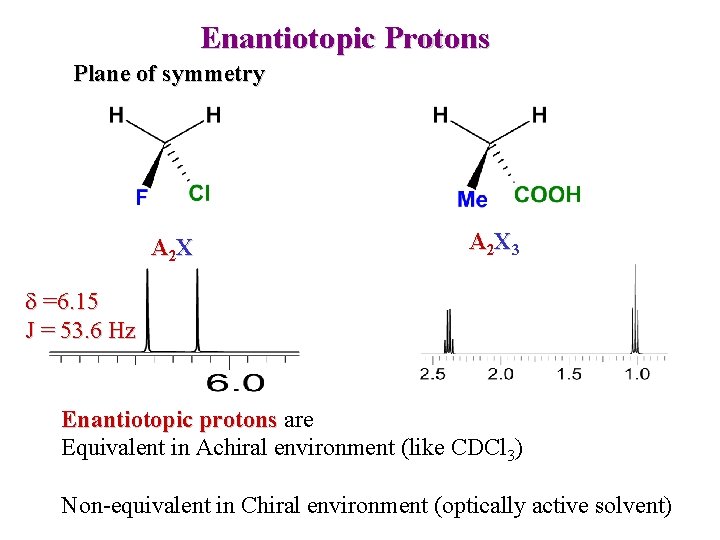
Enantiotopic Protons Plane of symmetry A 2 X 3 d =6. 15 J = 53. 6 Hz Enantiotopic protons are Equivalent in Achiral environment (like CDCl 3) Non-equivalent in Chiral environment (optically active solvent)

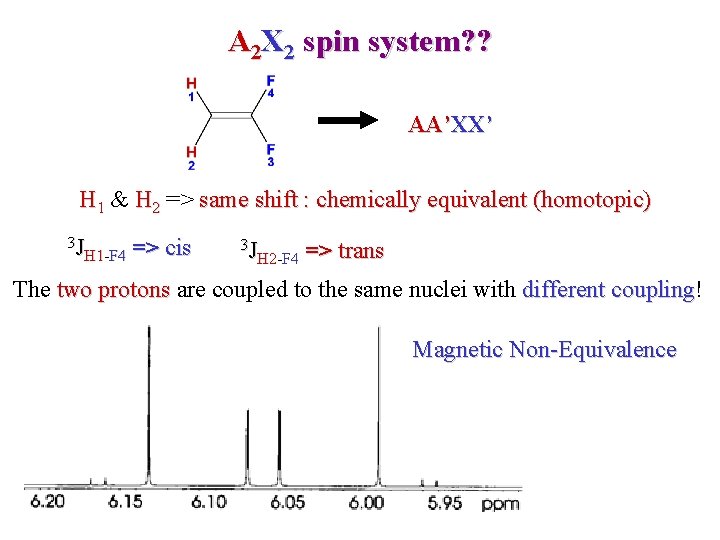
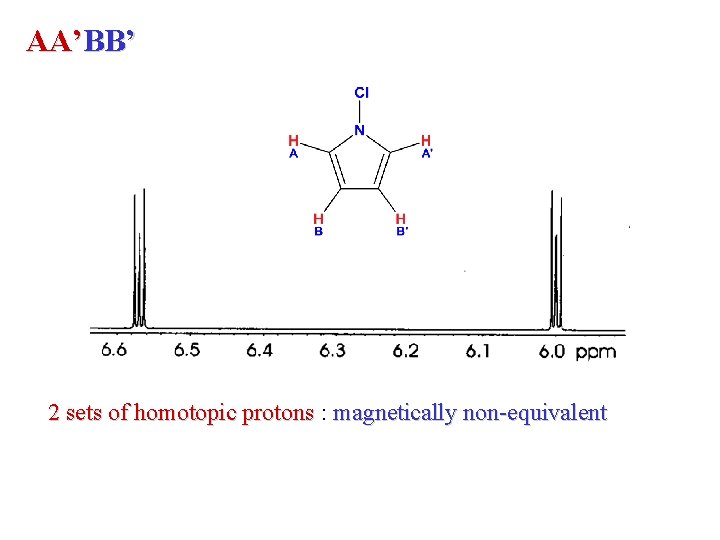
A 2 X 2 spin system? ? AA’XX’ H 1 & H 2 => same shift : chemically equivalent (homotopic) 3 J H 1 -F 4 => cis 3 J H 2 -F 4 => trans The two protons are coupled to the same nuclei with different coupling! coupling Magnetic Non-Equivalence

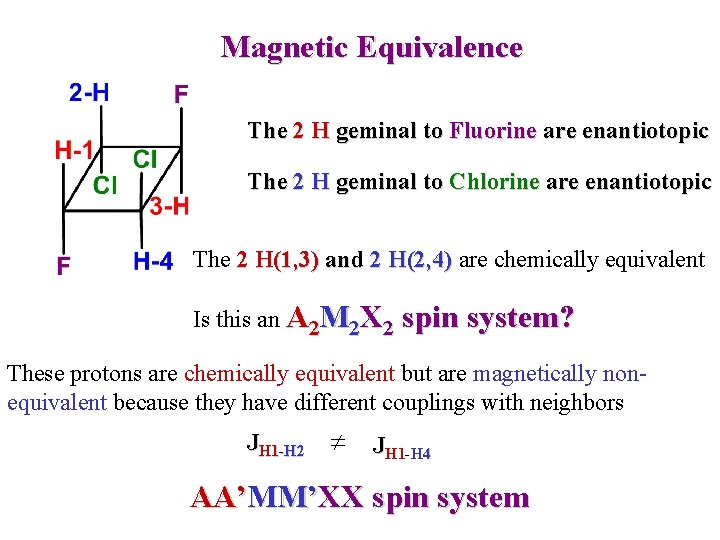
Magnetic Equivalence The 2 H geminal to Fluorine are enantiotopic The 2 H geminal to Chlorine are enantiotopic The 2 H(1, 3) and 2 H(2, 4) are chemically equivalent Is this an A 2 M 2 X 2 spin system? These protons are chemically equivalent but are magnetically nonequivalent because they have different couplings with neighbors JH 1 -H 2 JH 1 -H 4 AA’MM’XX spin system

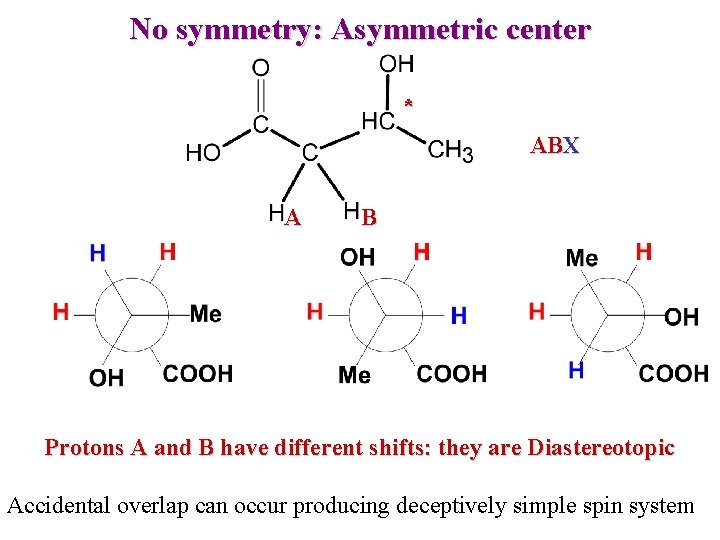
No symmetry: Asymmetric center * ABX A B Protons A and B have different shifts: they are Diastereotopic Accidental overlap can occur producing deceptively simple spin system

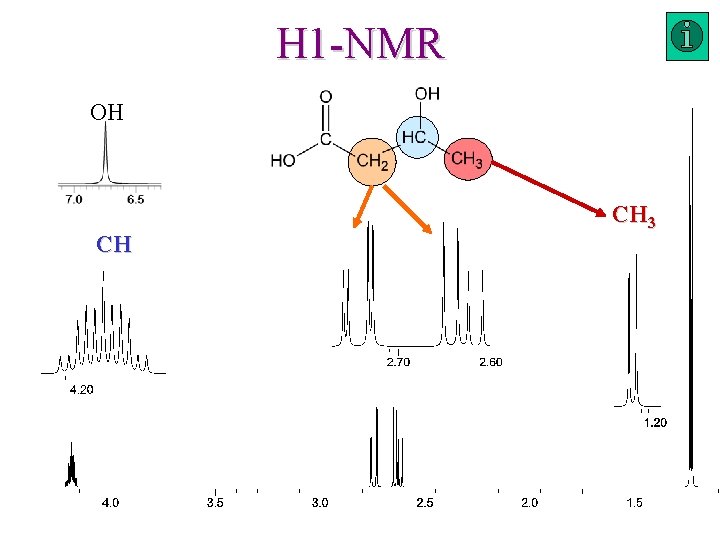
H 1 -NMR OH CH CH 3

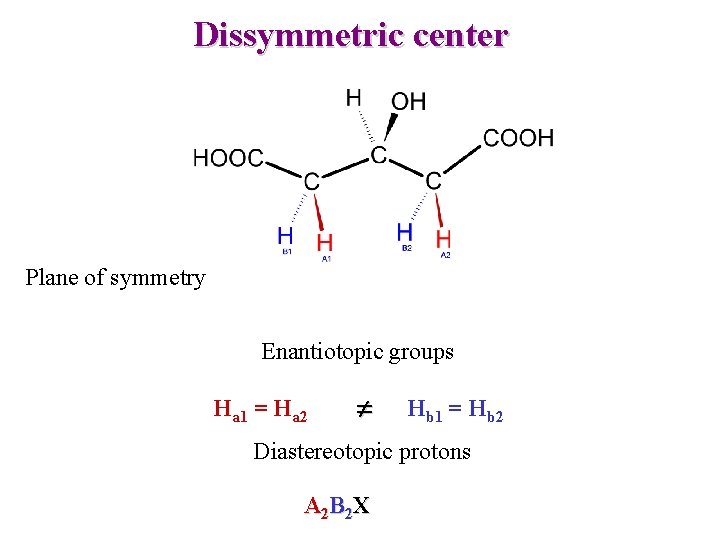
Dissymmetric center Plane of symmetry Enantiotopic groups Ha 1 = Ha 2 Hb 1 = Hb 2 Diastereotopic protons A 2 B 2 X

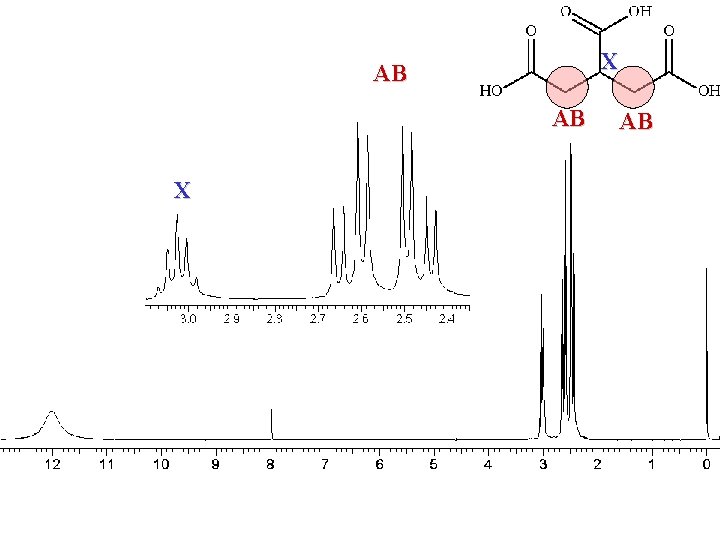
X AB AB X AB

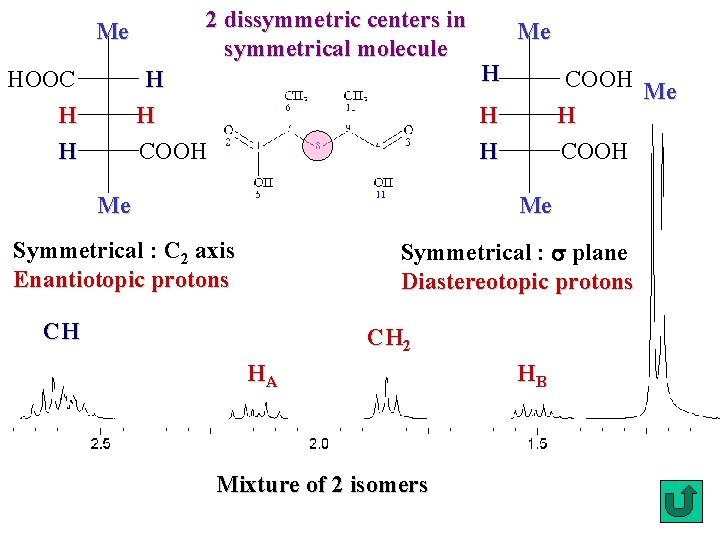
2 dissymmetric centers in symmetrical molecule Me H H HOOC H H Me H COOH Me H H H COOH Me Symmetrical : s plane Diastereotopic protons Symmetrical : C 2 axis Enantiotopic protons CH CH 2 HA Mixture of 2 isomers HB

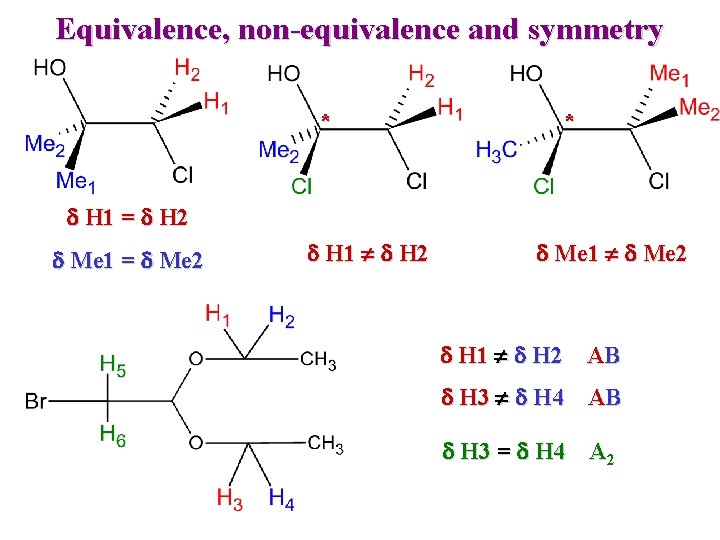
Equivalence, non-equivalence and symmetry * * d H 1 = d H 2 d Me 1 = d Me 2 d H 1 d H 2 d Me 1 d Me 2 d H 1 d H 2 AB d H 3 d H 4 AB d H 3 = d H 4 A 2

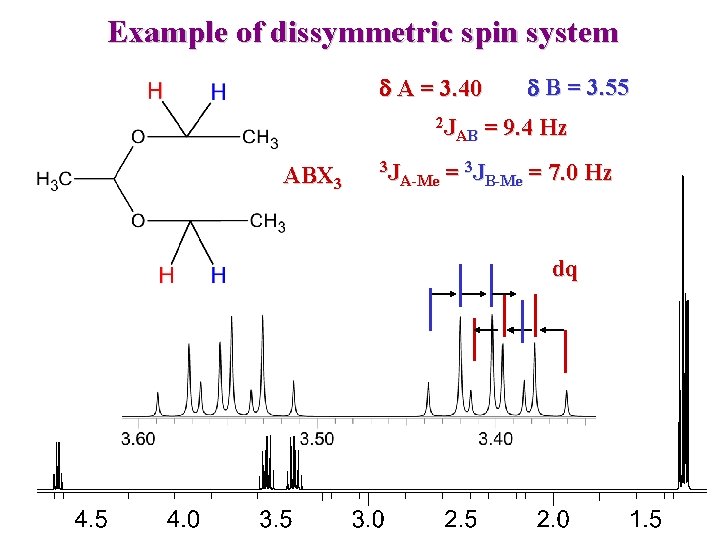
Example of dissymmetric spin system d A = 3. 40 2 J AB ABX 3 3 J A-Me d B = 3. 55 = 9. 4 Hz = 3 JB-Me = 7. 0 Hz dq

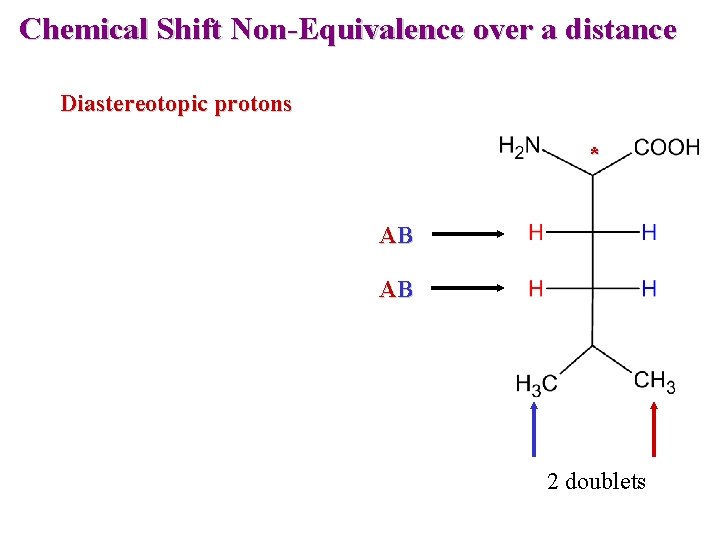
Chemical Shift Non-Equivalence over a distance Diastereotopic protons * AB AB 2 doublets

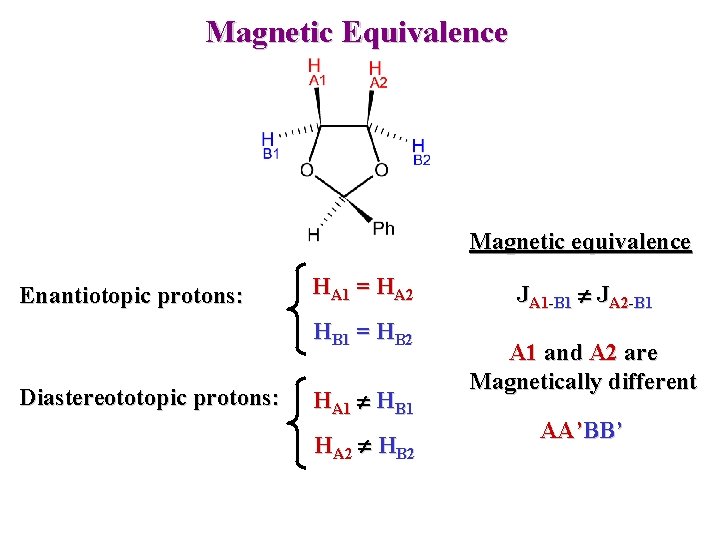
Magnetic Equivalence Magnetic equivalence Enantiotopic protons: HA 1 = HA 2 HB 1 = HB 2 Diastereototopic protons: HA 1 HB 1 HA 2 HB 2 JA 1 -B 1 JA 2 -B 1 A 1 and A 2 are Magnetically different AA’BB’

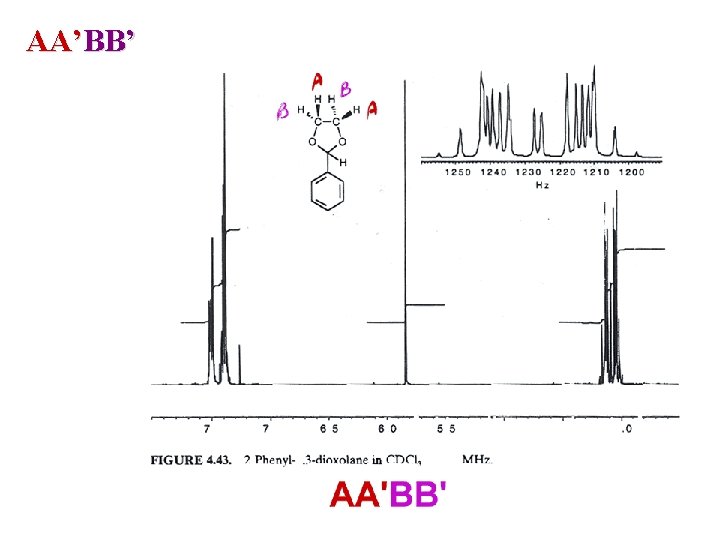
AA’BB’

AA’BB’ 2 sets of homotopic protons : magnetically non-equivalent

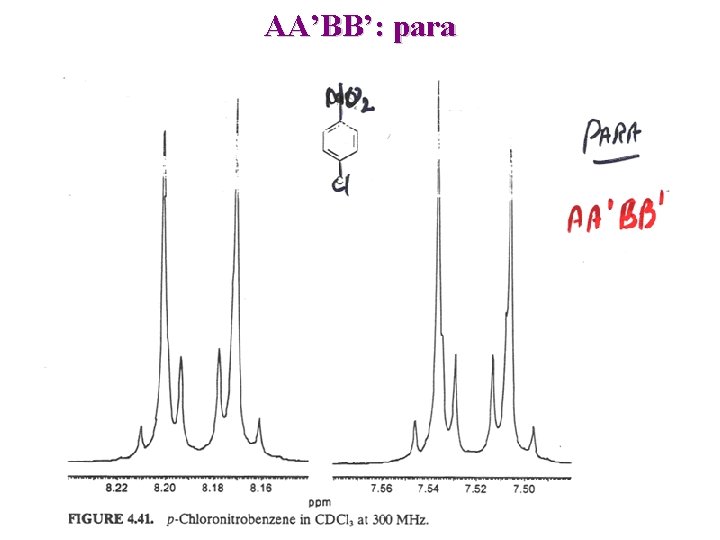
AA’BB’: para

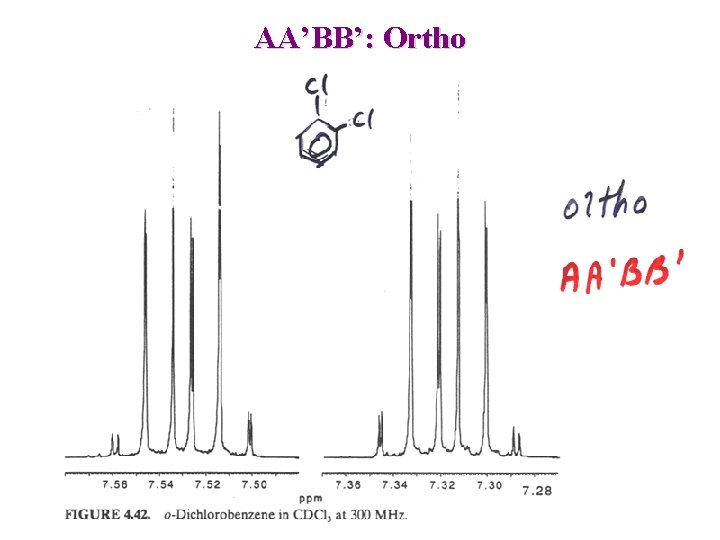
AA’BB’: Ortho

Spin System: Pople Notation • Each Chemical Shift is designated by a letter • Dn -> Difference in Shift in Hz • J -> Coupling in Hz • If the ratio Dn/J is Small (<8), Letters used to designate the shift are close AB, ABC … This represent case of second order spectra: These spectra must be simulated with the help of quantum mechanic equations. Such programs are available on Nuts or Mestrec or Spectrometer software. This case is also called strongly coupled • If the ratio Dn/J is large (>8), Letters used to designate the shift are far AM, AX … This case give rise to first order type spectra Is is also refer to as weakly coupled case

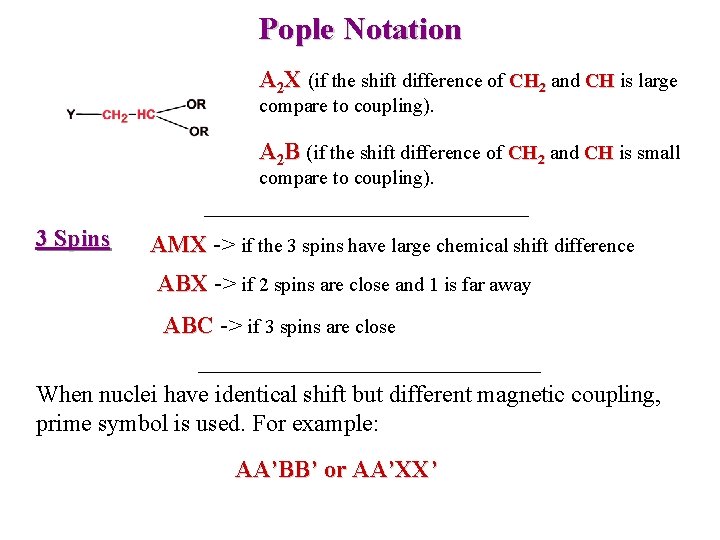
Pople Notation A 2 X (if the shift difference of CH 2 and CH is large compare to coupling). A 2 B (if the shift difference of CH 2 and CH is small compare to coupling). 3 Spins AMX -> if the 3 spins have large chemical shift difference ABX -> if 2 spins are close and 1 is far away ABC -> if 3 spins are close When nuclei have identical shift but different magnetic coupling, prime symbol is used. For example: AA’BB’ or AA’XX’

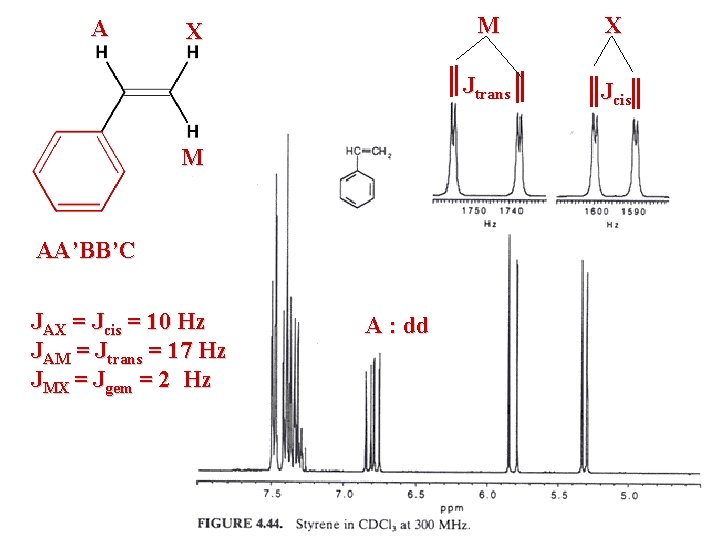
A X M AA’BB’C JAX = Jcis = 10 Hz JAM = Jtrans = 17 Hz JMX = Jgem = 2 Hz A : dd M X Jtrans Jcis

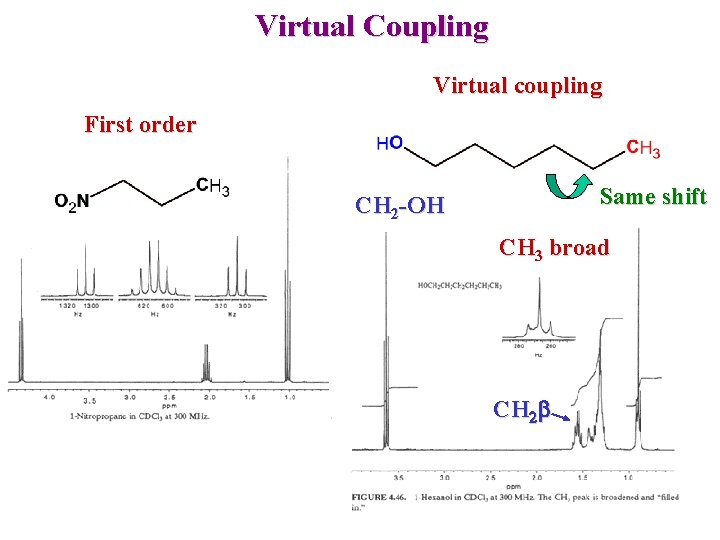
Virtual Coupling Virtual coupling First order Same shift CH 2 -OH CH 3 broad CH 2 b

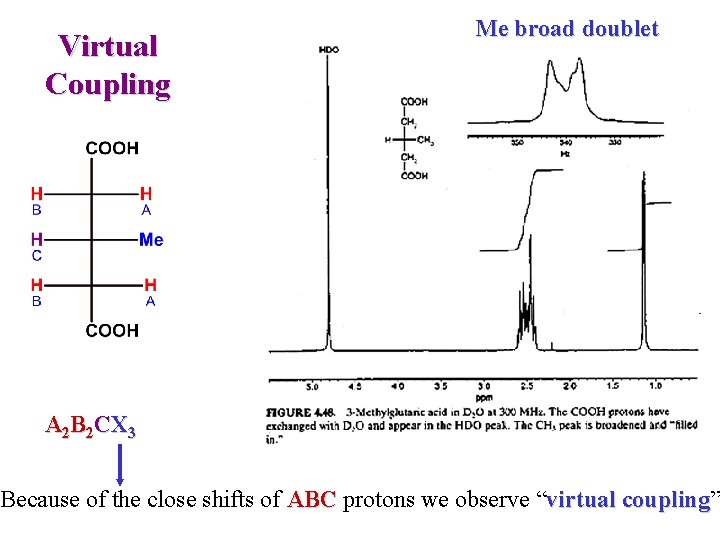
Virtual Coupling Me broad doublet A 2 B 2 CX 3 Because of the close shifts of ABC protons we observe “virtual coupling” coupling

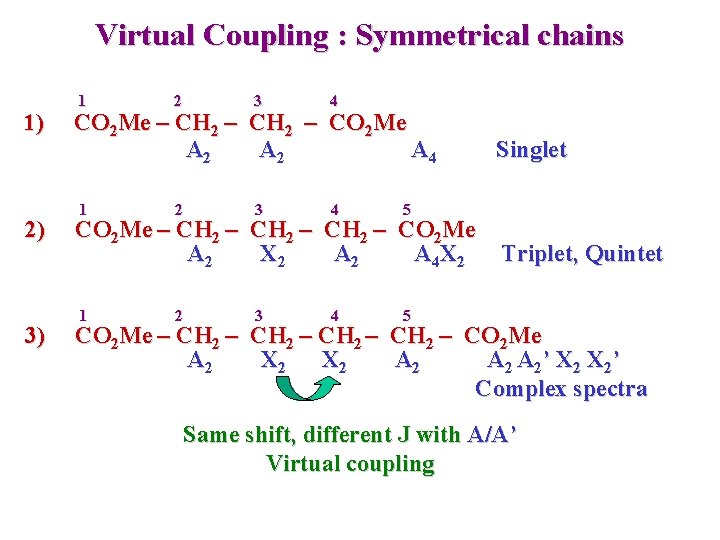
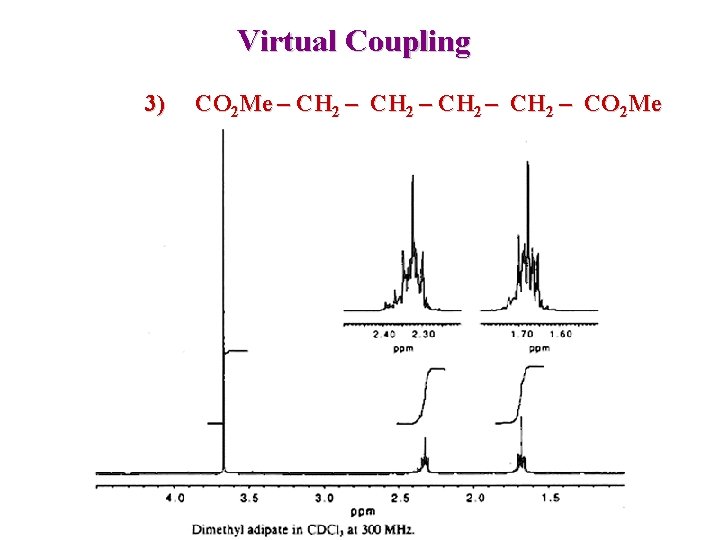
Virtual Coupling : Symmetrical chains 1) 2) 3) 1 2 3 4 CO 2 Me – CH 2 – CO 2 Me A 2 A 4 1 2 3 4 5 CO 2 Me – CH 2 – CO 2 Me A 2 X 2 A 4 X 2 1 2 3 4 Singlet Triplet, Quintet 5 CO 2 Me – CH 2 – CO 2 Me A 2 X 2 A 2 A 2’ X 2’ Complex spectra Same shift, different J with A/A’ Virtual coupling

Virtual Coupling 3) CO 2 Me – CH 2 – CO 2 Me

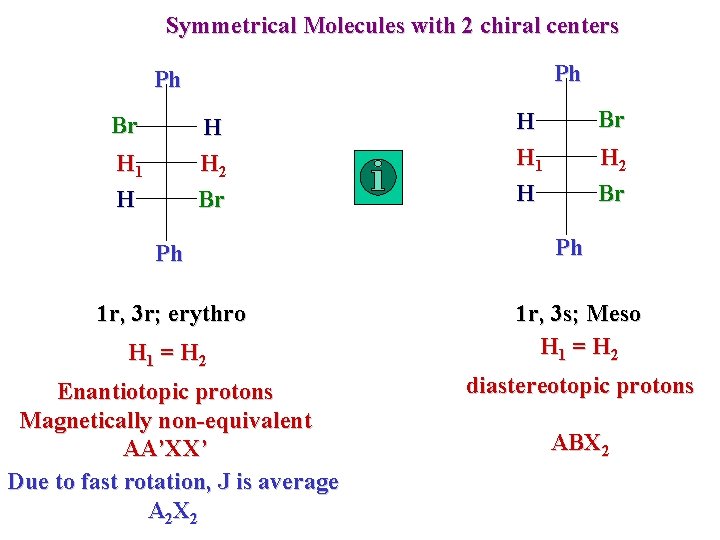
Symmetrical Molecules with 2 chiral centers Ph Ph Br H H 2 H 1 H Br Ph 1 r, 3 r; erythro H 1 = H 2 Enantiotopic protons Magnetically non-equivalent AA’XX’ Due to fast rotation, J is average A 2 X 2 Br H H 1 H H 2 Br Ph 1 r, 3 s; Meso H 1 = H 2 diastereotopic protons ABX 2

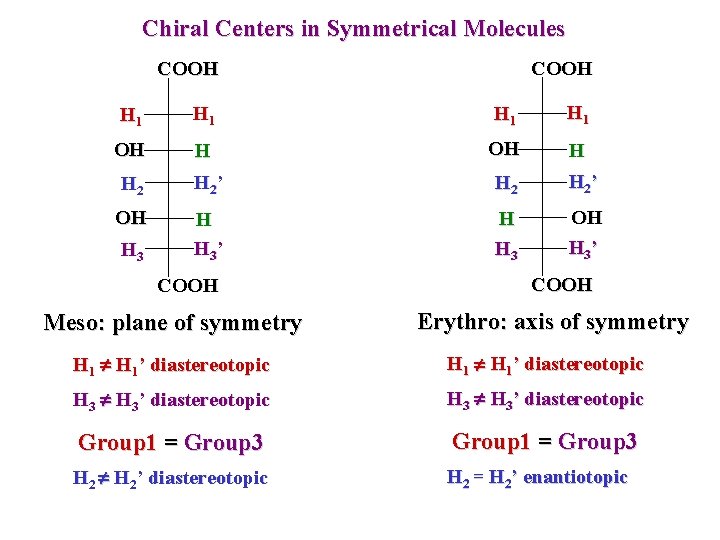
Chiral Centers in Symmetrical Molecules COOH H 1 H 1 OH H H 2 H 2 ’ OH H H 3 ’ H H 3 OH H 3 ’ H 3 COOH Meso: plane of symmetry COOH Erythro: axis of symmetry H 1’ diastereotopic H 3’ diastereotopic Group 1 = Group 3 H 2’ diastereotopic H 2 = H 2’ enantiotopic

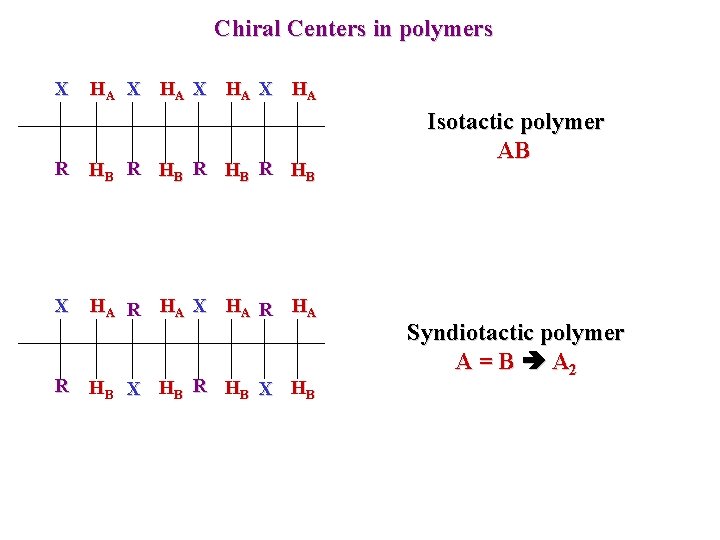
Chiral Centers in polymers X HA R HB X HA R HB X HB Isotactic polymer AB Syndiotactic polymer A = B A 2

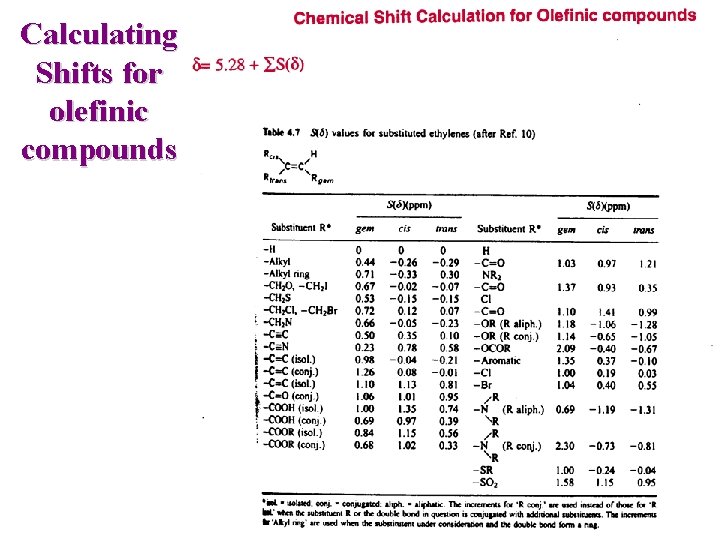
Calculating Shifts for simple aliphatic compounds d = 0. 23 + SSi(d) CH 3 Cl d(calc)=2. 76 d(exp. )=3. 1 CH 2 Cl 2 d(calc)=5. 29 d(exp. )=5. 3 CHCl 3 d(calc)=7. 82 d(exp. )=7. 27

Calculating Shifts for aliphatic compounds d = 0. 933 + SSi(d) 1 2 3 e. g. CH 3 -CO-CO-CH 3 Subst. Effect - C 2 -C 3 =O (at C 2) =O (at C 3) -CR 3 (at C 3) value +0. 244 +1. 021 +0. 004 -0. 038 S S i (d ) +1. 231 d = 0. 933 + 1. 231 = 2. 164 Experimental = 2. 23

Calculating Shifts for olefinic compounds

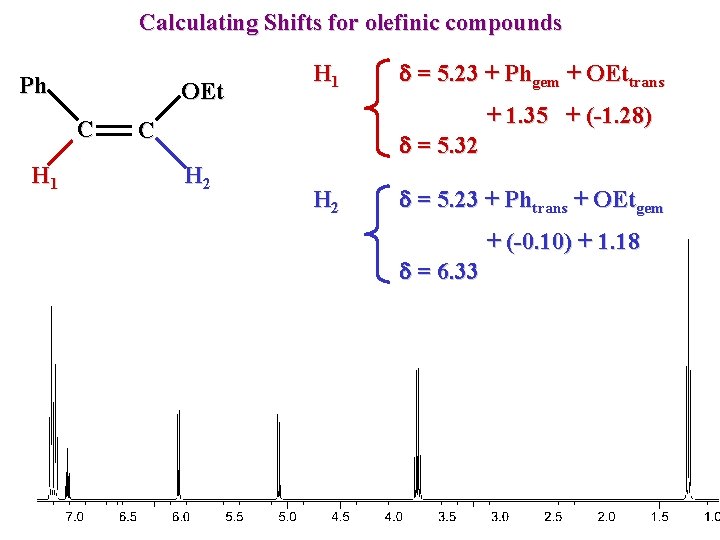
Calculating Shifts for olefinic compounds Ph OEt C H 1 C d = 5. 23 + Phgem + OEttrans d = 5. 32 H 2 + 1. 35 + (-1. 28) d = 5. 23 + Phtrans + OEtgem d = 6. 33 + (-0. 10) + 1. 18

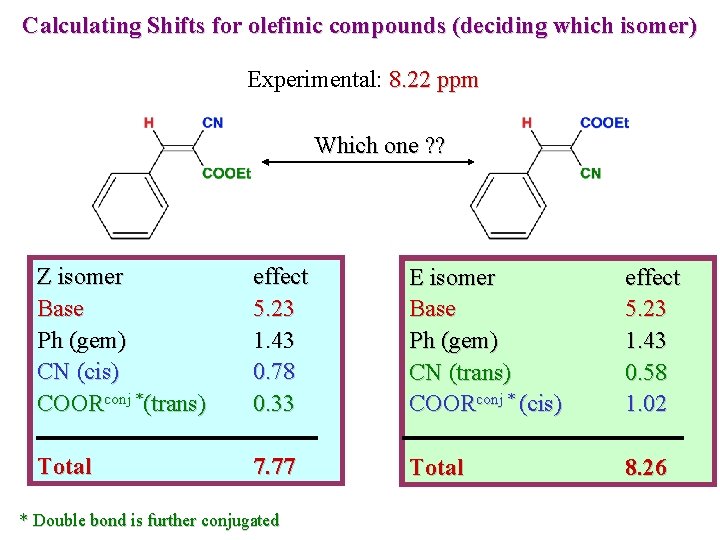
Calculating Shifts for olefinic compounds (deciding which isomer) Experimental: 8. 22 ppm Which one ? ? Z isomer Base Ph (gem) CN (cis) COORconj *(trans) effect 5. 23 1. 43 0. 78 0. 33 E isomer Base Ph (gem) CN (trans) COORconj * (cis) effect 5. 23 1. 43 0. 58 1. 02 Total 7. 77 Total 8. 26 * Double bond is further conjugated

Calculating Shifts for aromatic compounds

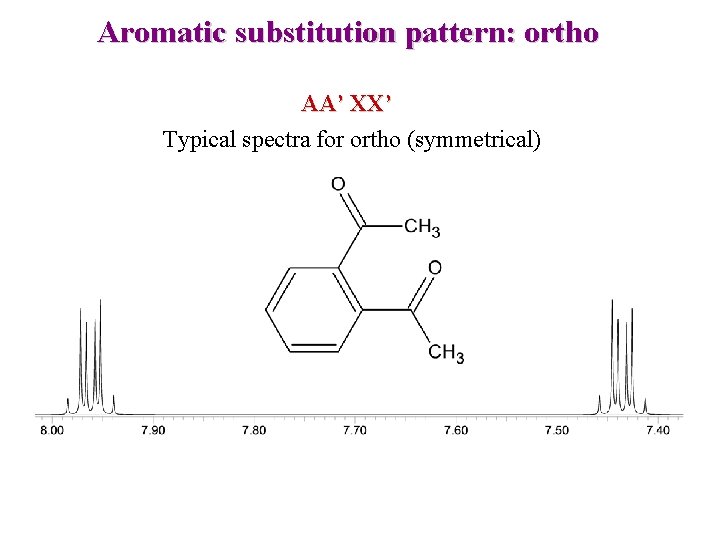
Aromatic substitution pattern: ortho AA’ XX’ Typical spectra for ortho (symmetrical)

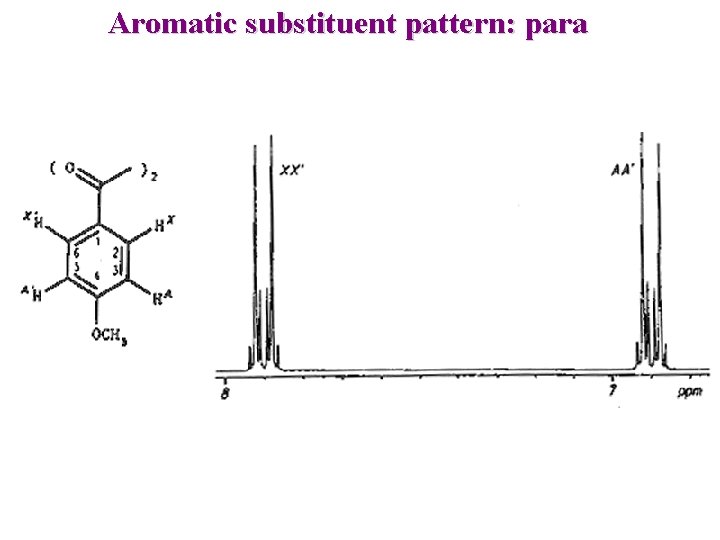
Aromatic substituent pattern: para

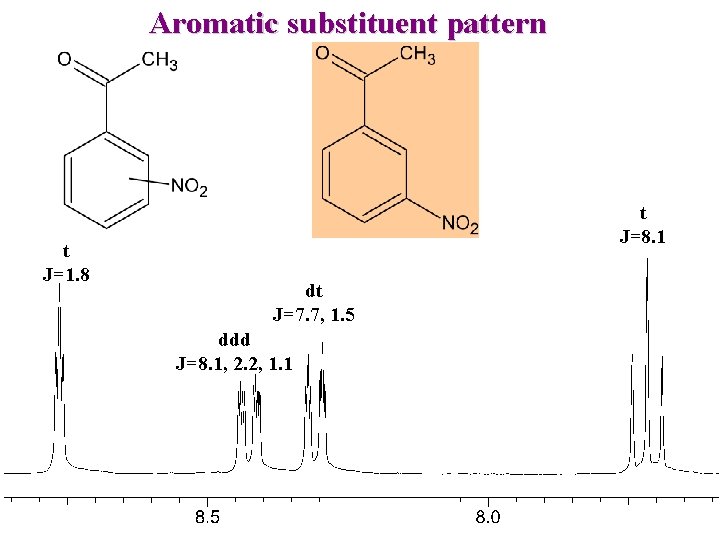
Aromatic substituent pattern t J=1. 8 t J=8. 1 dt J=7. 7, 1. 5 ddd J=8. 1, 2. 2, 1. 1

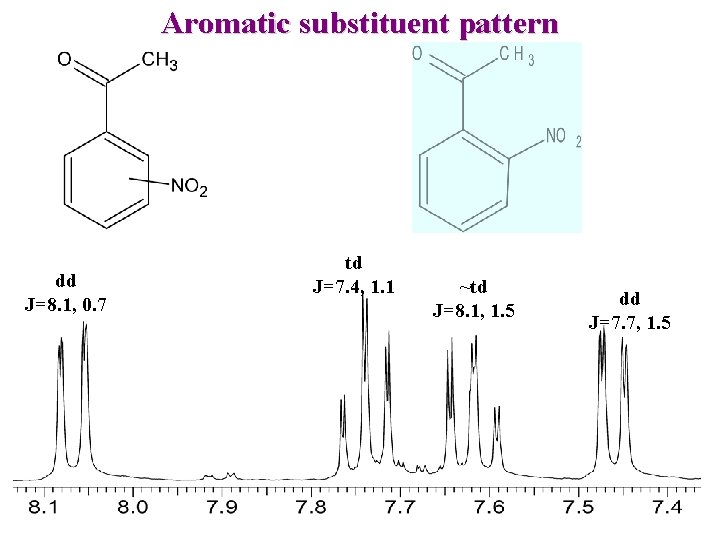
Aromatic substituent pattern dd J=8. 1, 0. 7 td J=7. 4, 1. 1 ~td J=8. 1, 1. 5 dd J=7. 7, 1. 5

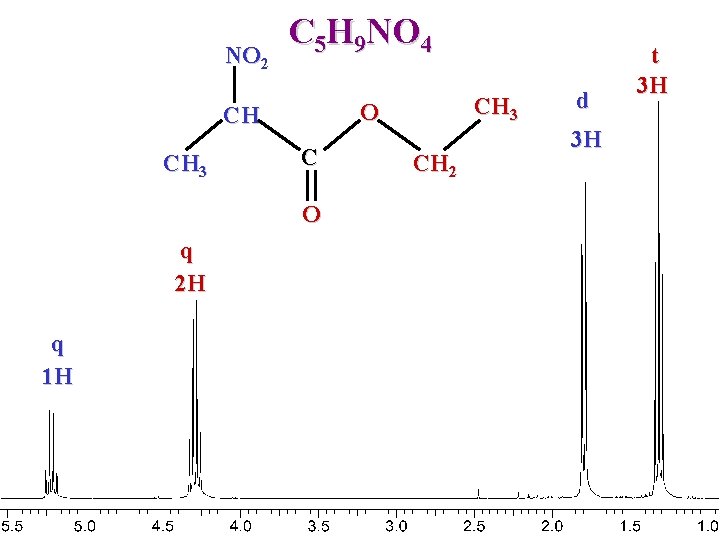
NO 2 C 5 H 9 NO 4 CH 3 C O q 2 H q 1 H CH 3 O CH CH 2 d 3 H t 3 H

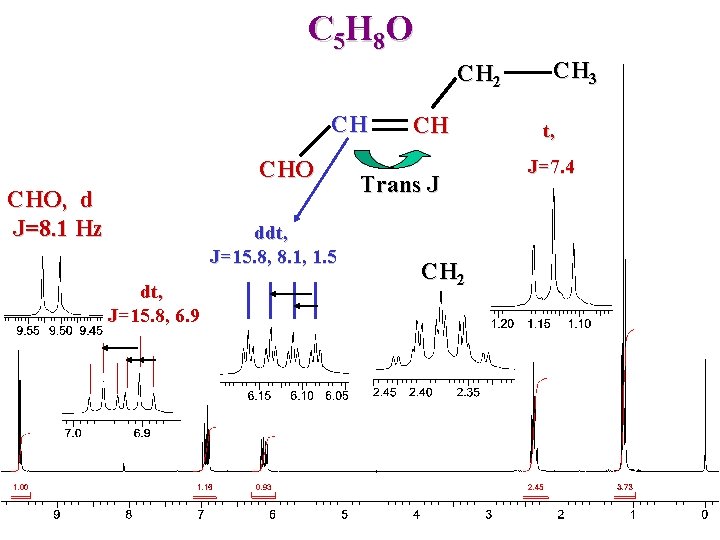
C 5 H 8 O CH 2 CH CHO, d J=8. 1 Hz ddt, J=15. 8, 8. 1, 1. 5 dt, J=15. 8, 6. 9 CH Trans J CH 2 CH 3 t, J=7. 4

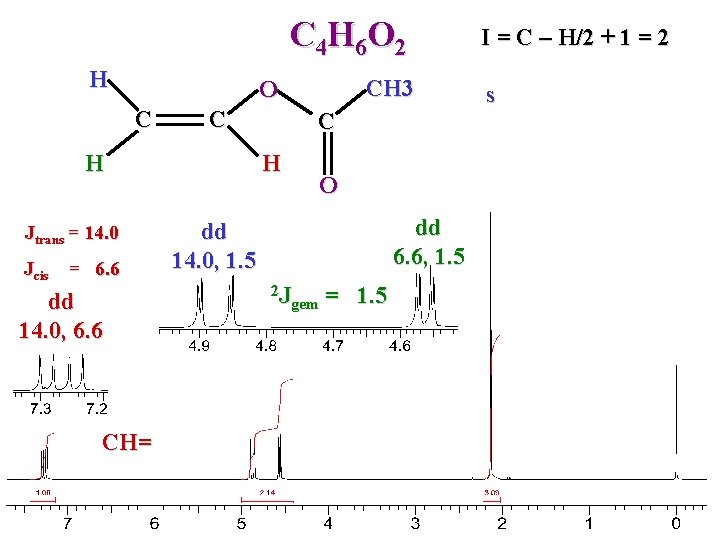
C 4 H 6 O 2 H CH 3 O C C H Jtrans = 14. 0 Jcis = 6. 6 dd 14. 0, 6. 6 CH= C H O dd 6. 6, 1. 5 dd 14. 0, 1. 5 2 J gem = 1. 5 I = C – H/2 + 1 = 2 s


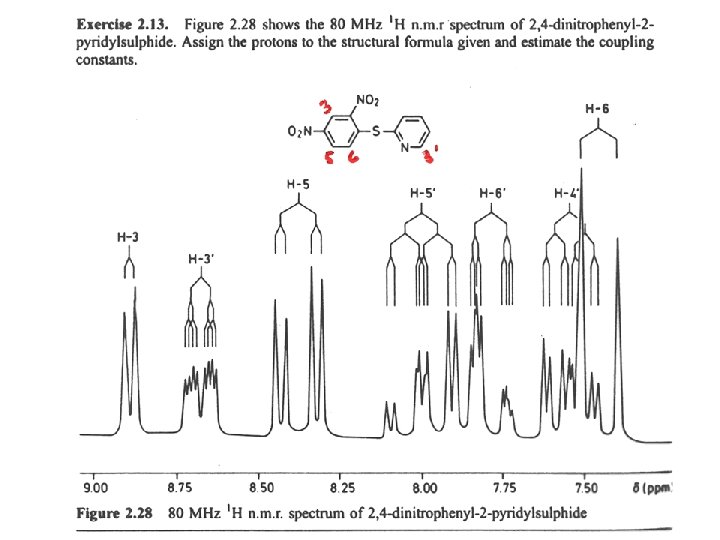
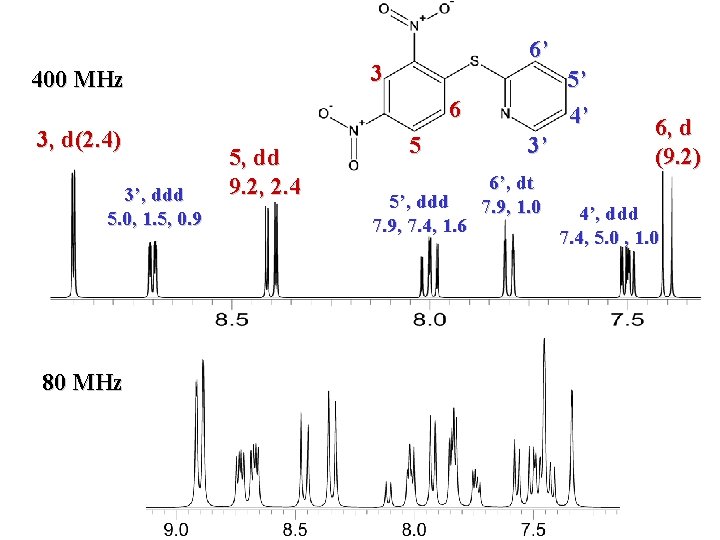
6’ 3 400 MHz 5’ 4’ 6 3, d(2. 4) 3’, ddd 5. 0, 1. 5, 0. 9 80 MHz 5, dd 9. 2, 2. 4 5 5’, ddd 7. 9, 7. 4, 1. 6 3’ 6’, dt 7. 9, 1. 0 6, d (9. 2) 4’, ddd 7. 4, 5. 0 , 1. 0

NEXT Proton and Heteronuclear NMR Index NMR-basics H-NMR NMR-Symmetry Heteronuclear-NMR