Atoms and Bonding Ch 2 3 Bettelheim text

Atoms and Bonding- Ch. 2 & 3 Bettelheim text **Recommended sites: kentchemistry. com Also, Janet Coonce videos on youtube • Atom- Composed of p+, n 0 and e • Know atomic theory!
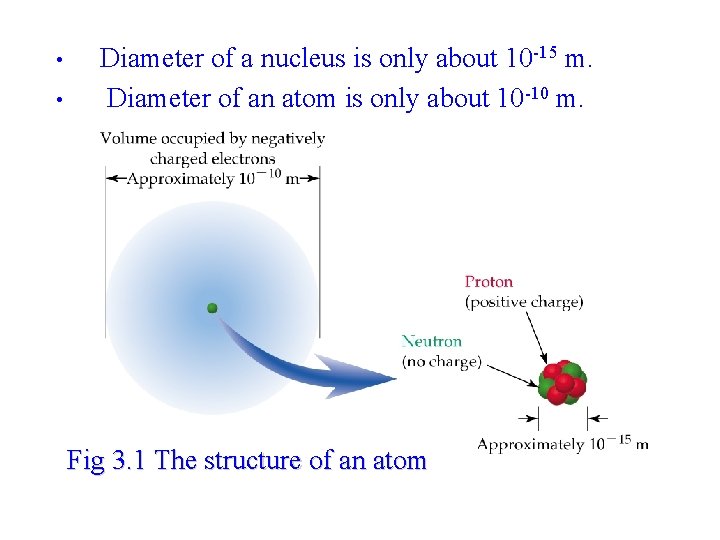
• • Diameter of a nucleus is only about 10 -15 m. Diameter of an atom is only about 10 -10 m. Fig 3. 1 The structure of an atom

Atomic number (Z) • • Always equals the p+ number In a neutral atom, p+ = e. Mass number: p+ and n 0 Isotopes- different number of neutrons, so different masses
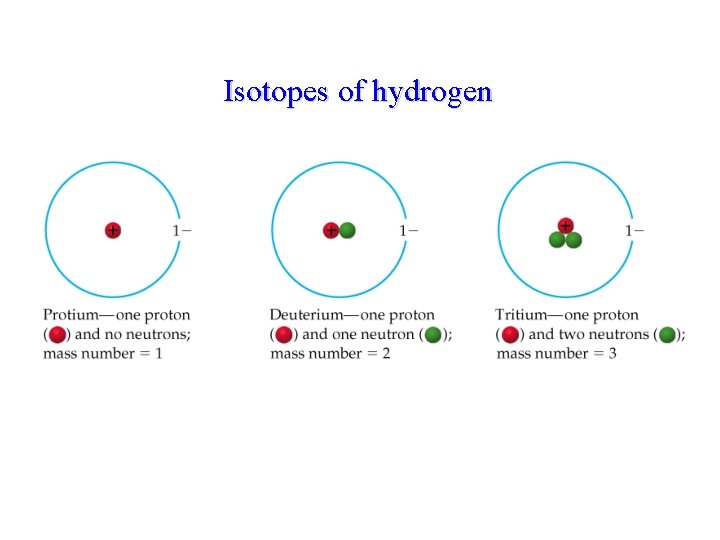
Isotopes of hydrogen

Periodic Table • Mendeleev-1869 -organized elements by atomic mass; forerunner of modern PT • Periods (rows) • Groups (columns)- same # of valence e • Know names of groups • Metals, metalloids and non-metals
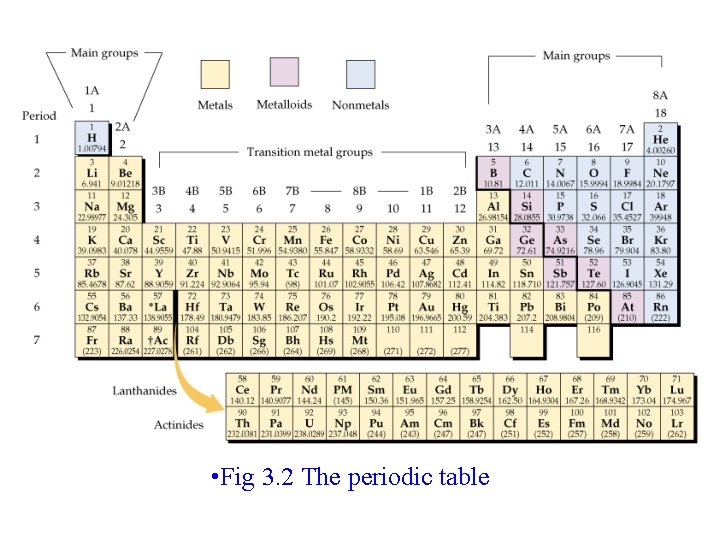
• Fig 3. 2 The periodic table

Periodicity • Atomic radius-decreases across periods and increases down groups • Know characteristics of groups • Properties of elements determined by earrangement • Electronic structure (electron configurations) • *note-shell=principle energy level • S=spherical orbitals; P=dumbbell shaped

Electronic structure con’t • The maximum # of e- that can occupy an energy level (shell) is 2 n 2

• • The first shell has only an s subshell The second shell has and p subshell The third shell has an s, p, and d subshell. The fourth shell has an s, p, d, and f subshell.


• Electronic configuration of a few elements are shown below:

Sublevel Diagram, Order in which Sublevels are Usually Filled 1 s 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 7 s 7 p

Electrons in Energy Levels, Sublevels, and Orbitals Principal Available Energy Sublevels Level (n) (s, p, d, f) Orbitals in Sublevel (boxes) Possible Electrons in Sublevel Possible Electrons in Energy Level 1 s 1 2 2 2 s p 1 3 2 6 8 3 s p d 1 3 5 2 6 10 18 4 s p d f 1 3 5 7 2 6 10 14 32

Electron configurations con’t • In class- do configurations for C, N and P • Also, draw the electron config for potassium and the potassium ion • How might an abbreviated configuration of potassium be written?
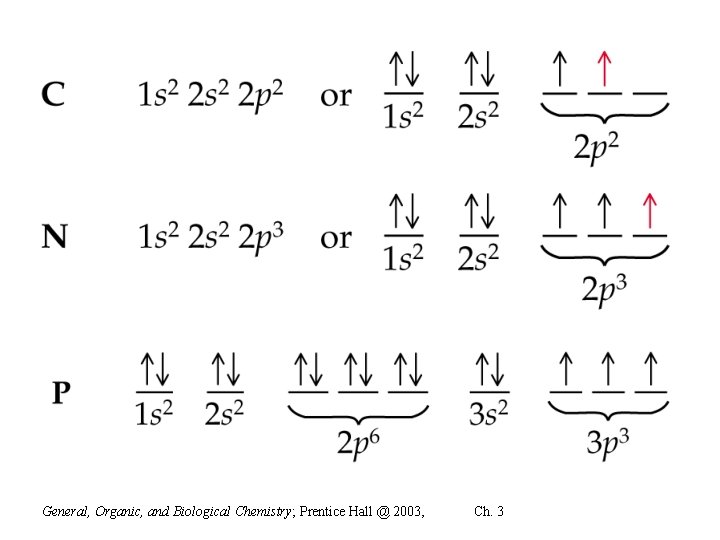
General, Organic, and Biological Chemistry; Prentice Hall @ 2003, Ch. 3

Valence Electrons - are the electrons in the outermost orbital of an atom - are associated with atom’s highest principal energy level (period) - only valence electrons determine chemical properties of an element.

Ionic Bonds • Typically, atoms from groups IA and IIA ionically bond with VIA an VIIA • Ion- Charged atom (lose or gain e-)

The symbol for cation is written by adding a positive charge as a superscript to the symbol for the element. For example, by losing an electron Na metal is converted to the sodium cation (Na+). General, Organic, and Biological Chemistry; Prentice Hall @ 2003, Ch. 4

• Gain of one or more electrons by a neutral atom gives a negatively charged ion called a anion. The symbol for the anion is written as by adding a negative charge as a superscript to the symbol for the element. For example, by gaining an electron chlorine atom becomes chloride ion, Cl-. General, Organic, and Biological Chemistry; Prentice Hall @ 2003, Ch. 4
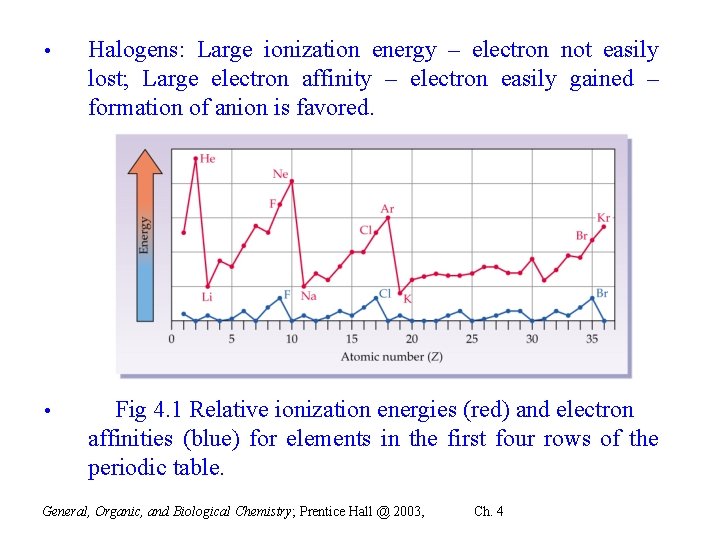
• Halogens: Large ionization energy – electron not easily lost; Large electron affinity – electron easily gained – formation of anion is favored. • Fig 4. 1 Relative ionization energies (red) and electron affinities (blue) for elements in the first four rows of the periodic table. General, Organic, and Biological Chemistry; Prentice Hall @ 2003, Ch. 4

Where do the electrons come from? Where do the electrons go? • Atoms with unfilled valence shells can become filled by transferring or sharing electrons with other atoms • When atoms come together by sharing or transferring electrons, a chemical bond results. • After a chemical bond is formed, the atoms involved in the bond each have a filled valence shell (octet).
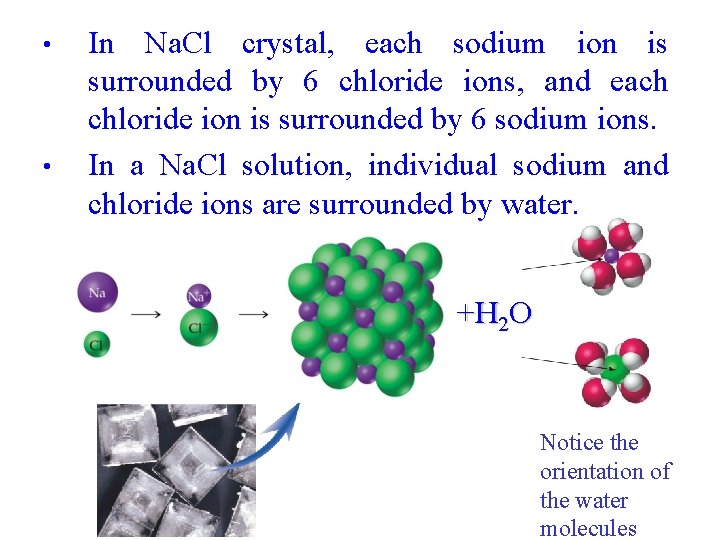
• • In Na. Cl crystal, each sodium ion is surrounded by 6 chloride ions, and each chloride ion is surrounded by 6 sodium ions. In a Na. Cl solution, individual sodium and chloride ions are surrounded by water. +H 2 O Notice the orientation of the water molecules

Ionic Bonds • • Opposite electrical charges attract each other. When sodium combines with chlorine, sodium transfer electron to chlorine forming Na+ and Clions. The oppositely charged Na+ and Cl- ions are held together by a ionic bond. In three dimensional crystals, all nearby oppositely charged ions attract each other. We can not separate attraction between only two oppositely charged ions -we speak of the whole crystal and crystal as being an ionic solid or ionic compound. (molecule)

Some Properties of Ionic Compounds • Ionic compounds are usually crystalline solids. • Different ions vary in size and charge. • Ions packed together in crystals in different ways. • The crystal pattern ensures that ions efficiently fills space and maximizes ionic bonding. • Ionic compounds have high melting and high boiling points.

• The formula of an ionic compound shows the lowest possible ratio of atoms in the compound.

Naming ionic compounds • We will NOT cover the “old” naming system • For binary ionic compounds, name the cation first and then the anion. However, you must change the ending of the anion to -ide • If a transition metal is the cation, a roman numeral must be given to indicate it’s oxidation number (charge) • Although polyatomic ions bond covalently, they may ionically bond with a cation or an anion • For test, know biological importance of Ca, Fe, K and Na • Polyatomic ions to know for this test: hydroxide, nitrate, carbonate, sulfate, phosphate, ammonium, cyanide, hydrogen carbonate (bicarbonate)

Acids and bases • One definition (Arrhenius): Acid-substance that donates H+ in water Base- donates OH • Hydronium ion H 30+ • Exp: HCl, Na. OH • p. H scale 0 -7 -14 • p. H + p. OH = 14 • Properties of acids and bases (Acids- react with metals; “sour”; Bases-slippery, bitter) • Acid + Hydroxide Base Salt and Water

Acids and bases • Problem with Arrhenius definition: Not all acid/ base reactions occur in water • Bronsted- Lowry definition: Acid is a proton (H+) donor and base is proton acceptor • NH 3 + HCl-> NH 4+ + Cl-

More acids and bases • Buffer-substance that can accept hydrogen or hydroxide ions in solution. Prevent drastic changes in p. H • Major buffers in the body: 1) bicarbonate 2)hydrogen phosphate • CO 2 + H 2 O H 2 CO 3 H+ + HCO 3 • For our lab: Ca. CO 3+HCl Ca. Cl 2+H 2 O+CO 2

Covalent Bonds • • • Covalent bond: A bonds formed by sharing electrons between atoms. Usually nonmetals Molecule: A group of atoms held together by covalent bonds. The nonmetals near the middle of the periodic table reach an electron octet by sharing an appropriate number of electrons. General, Organic, and Biological Chemistry; Prentice Hall @ 2003, Ch. 5

• Spherical 1 S orbital of two individual hydrogen atoms bends together and overlap to give an egg shaped region in the hydrogen molecule. The shared pair of electrons in a covalent bond is often represented as a line between atoms. *Remember, hydrogen doesn’t get a full octet, it only fills the first shell s orbital
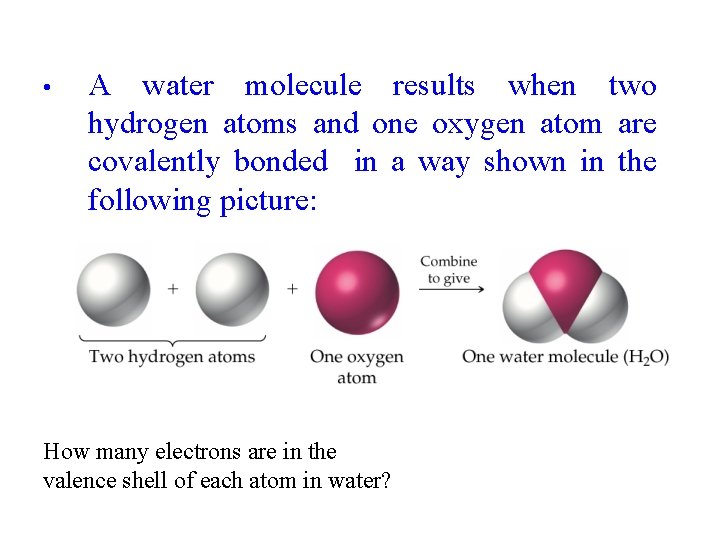
• A water molecule results when two hydrogen atoms and one oxygen atom are covalently bonded in a way shown in the following picture: How many electrons are in the valence shell of each atom in water?
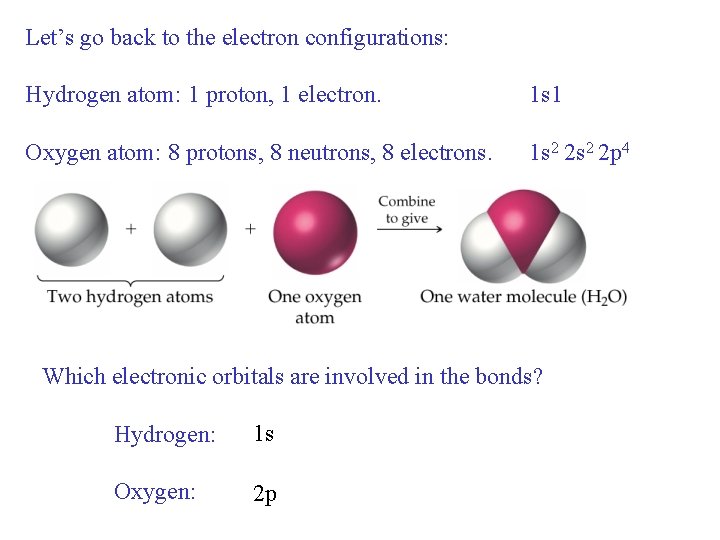
Let’s go back to the electron configurations: Hydrogen atom: 1 proton, 1 electron. 1 s 1 Oxygen atom: 8 protons, 8 neutrons, 8 electrons. 1 s 2 2 p 4 Which electronic orbitals are involved in the bonds? Hydrogen: 1 s Oxygen: 2 p
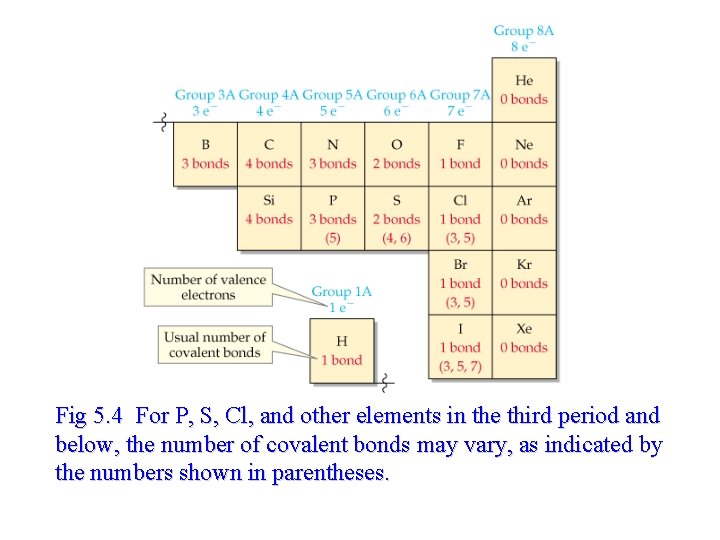
Fig 5. 4 For P, S, Cl, and other elements in the third period and below, the number of covalent bonds may vary, as indicated by the numbers shown in parentheses.

Numbers of Covalent Bonds - single bond– sharing of two electrons - longest bond length - double bond- sharing of four electrons - middle bond length - triple bond- sharing of six electrons - shortest bond length - strongest covalent bond - The electrons in the valence shell that are not shared are called lone pairs.

Drawing Lewis Structures • To draw Lewis structure, you need to know the connections among atoms. Also, common bonding patterns, shown below, for C, N, O, X (all halogens), and H simplifies writing Lewis structure. • • C forms 4 bonds (no lone pairs) N forms 3 (1 lone pair) O forms 2 (2 lone pairs) Halogens form 1 (3 lone pairs)

Method #1 - I suggest this method. Refer to the handouts “General Rules for Drawing Lewis Structures” and “How to Determine Formal Charge Method #2 - Book’s suggested rules for drawing molecules (Lewis structures)**note-these don’t always work…they are merely a guide. Some have expanded octets. 1. Determine the number of valence electrons in the molecule. 2. Determine the arrangement of atoms in the molecule (least electronegative –center) 3. Connect the atoms with single bonds 4. Give each peripheral atom an octet by adding lone pairs 5. Place all remaining electrons on central atom by adding lone pairs 6. If the central atom still doesn’t have an octet, take a lone pair from a peripheral atom to form a multiple bond

LEWIS DOT STRUCTURES 1. Arrange the symbols such that the least electronegative element is in the center and the other elements are surrounding the central atom. O C O 2. Give each of the elements their appropriate number of valence electrons (dots). Remember the number of valence electrons for a representative element is the same as the group number. . . : O : C: O: . . 3. Keep track of the total numbr of valence electrons for the compound by adding the valence electrons from each atom. If the compound is an ion then add electrons (dots) for each negative charge or subtract electrons (dots) for each positive charge. 4 for C and 6 for O (twice) = 16 electrons

LEWIS DOT STRUCTURES 4. Now move the dots around so that you have 8 dots (the octet rule) around each element (do not forget the exceptions) while at the same time keeping the dots in pairs. Electrons, at this point, exist as pairs (the buddy system). 5. EXCEPTIONS TO THE OCTET RULE: Group I, II, and III need only 2, 4, and 6 electrons, respectively, around that atom (if they are covalently bonding). 6. If there are too few pairs to give each atom eight electrons, change the single bonds between two atoms to either double or triple bonds by moving the unbonded pairs of electrons next to a bonding pair. . . : O: : C: : O:

LEWIS DOT STRUCTURES 7. Once the octet rule has been satisfied for each atom in the molecule then you may replace each pair of dots between two atoms with a dash. . . : O =C= O: 8. Now check your structure by a) count the total number of electrons to make sure you did not lose or gain electrons during the process. b) Use FORMAL CHARGE (FC) calculations as a guideline to the correct structure. A zero formal charge is usually a good indication of a stable structure. *Several formulas for this, but they all do the same thing: FC (X) = # of valence electrons - (1/2 bonding electrons + nonbonding electrons) Or: FC = (Valence – nonbonding e-) – Bonding e- / 2 For our example: FC(C) = 4 - (1/2 8 + 0) = 0 FC(O) = 6 - (1/ 4 + 4) = 0

FC Videos • Wayne Breslyn: how to calc formal charges – https: //www. youtube. com/watch? v=v. OFAPlq 4 y_k • Leah 4 sci: orgo basics- formal charges. (I personally like this one, because you don’t have to learn or use the formula!) – https: //www. youtube. com/watch? v=J 03 H 472 f. A KI *Another good site is: Organic Chemistry Tutor https: //www. youtube. com/watch? v=R_SIjij. Cou. E

The Basics: Drawing Lewis Structures. *Refer to my handout for more “rules” Step 1: Calculate the total number of valence electrons in the molecule or ion Step 2: Determine the central atom(s) of the molecule or ion – usually it’s the least electronegative atom. Step 3: Draw a tentative diagram for the molecule or ion. Remember: a) A hydrogen atom always forms one bond. Hydrogen is always a terminal atom in a Lewis diagram – an atom that is bonded to only one other atom. b) A carbon atom normally forms four bonds c) When several carbon atoms appear in the same molecule, they are often bonded to each other.

Warm Up Practice • Draw Lewis Structures – Water – Carbon monoxide (*We will go over naming covalent compounds tomorrow) – Ammonia (NH 3) – BH 3 – CCl 4 – PBr 3 – NH 4+

Draw the Lewis Structure for the following molecules. 1. H 2 O Oxygen has 6 valence electrons & Hydrogen has 1 valence electron for a total of 8 electrons. Which is correct? . . H: O: H. . or 2. CO Oxygen has 6 valence electrons & Carbon has 4 valence electrons for a total of 10 electrons. : C: : : O:

Draw the Lewis Structure for the following molecules. 3. BH 3 Boron has 3 valence electrons & Hydrogen has 1 valence electron for a total of 6 electrons H: B: H. . H 4. NH 3 Nitrogen has 5 valence electrons & Hydrogen has 1 valence electron for a total of 8 electrons . . H: N: H. . H

CW- Draw CCl 4
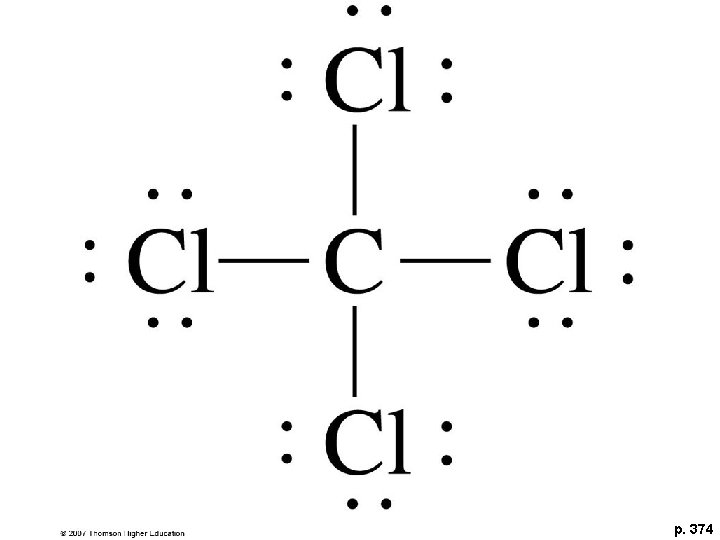
p. 374

CW- Draw PBr 3
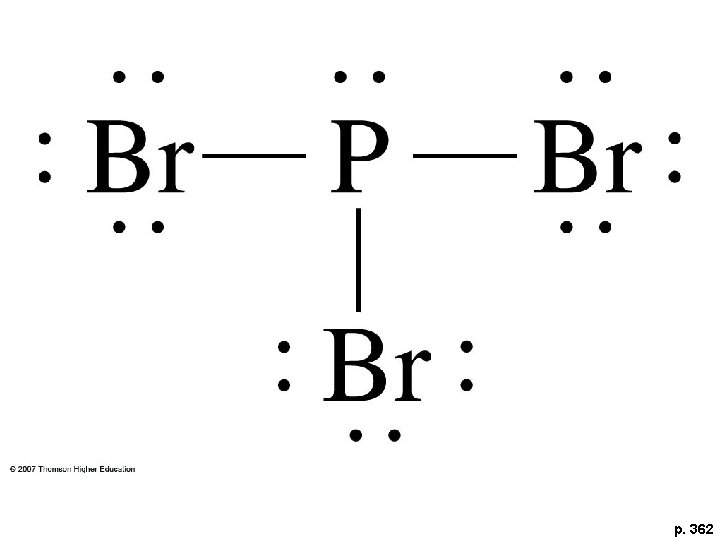
p. 362

Draw NH 4+ *Polyatomic Cation **Coordinate covalent bond- one atom donates both shared e-
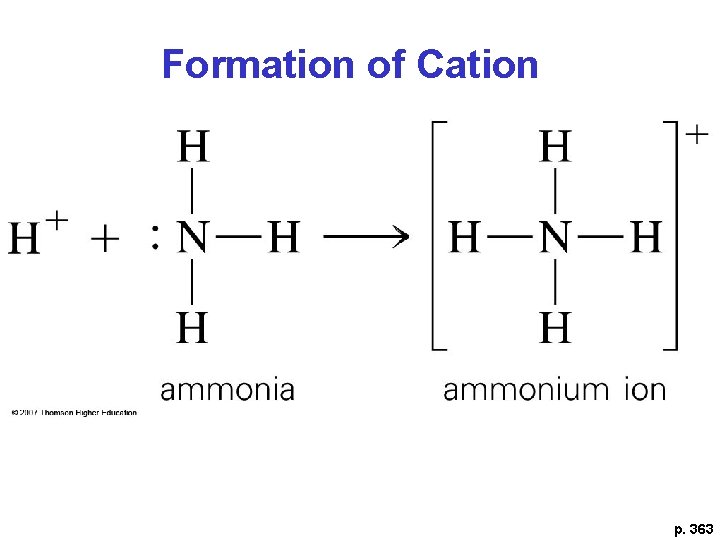
Formation of Cation p. 363

Explain the last example in terms of acid/base • NH 3 is the ? • H+ is the ?

The next few slides have…. • Some more “challenging” Lewis structures. Let’s try a couple…

Get out your pencils. 2 Draw SO 3 *sulfite polyatomic anion
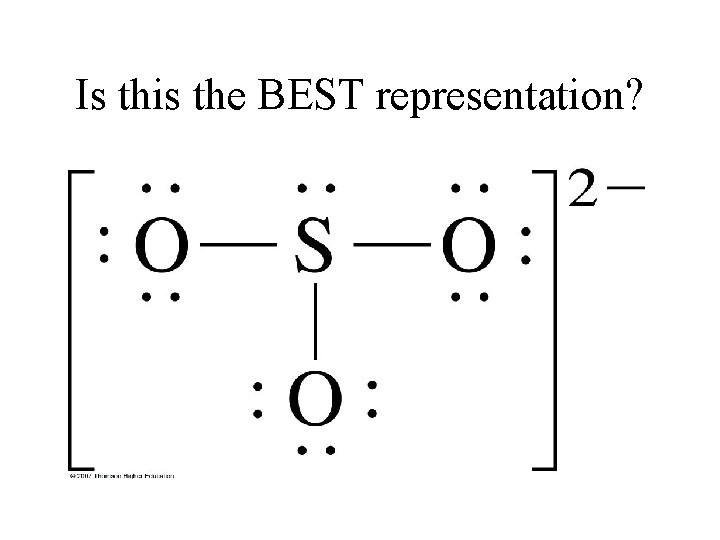
Is this the BEST representation?

Start with the “rules” (but remember, sometimes they don’t always quite work). • Octet: 8 x 4 = 32 • Valence: 6 x 4 = 24 + 2 = 26 – Must add 2 more because of 2 - overall charge • Subtract: 32 -26= 6 bonding e– That would mean 20 are Non-Bonding – So far, so good. Or is it? • Check formal charge of each atom.

Formal Charge • Each O would be 1 - (total of 3 -) • S would be 1+ • While this gives an overall 2 -, remember: – Try to get as many atoms to 0 as possible (*especially the ‘central’ atom) – How? Move 1 NB pair on one of the O atoms and make it a bonding pair *Watch Wayne Breslyn (youtube): https: //www. youtube. com/watch? v=Qi 4 GTd 1 LCc
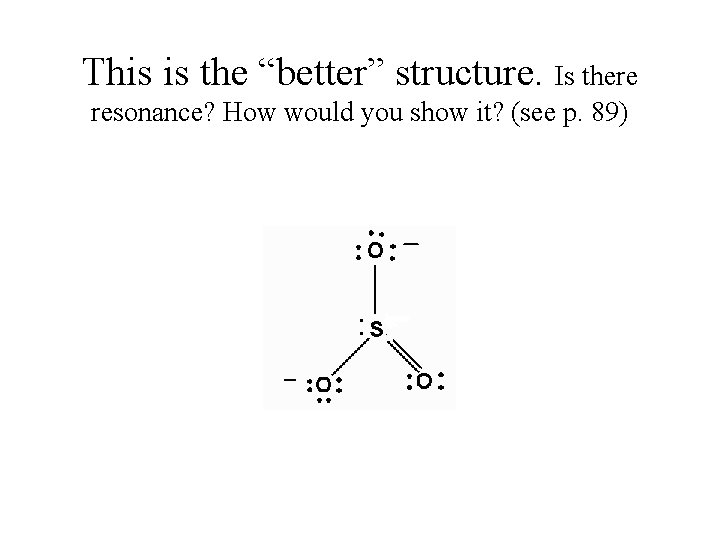
This is the “better” structure. Is there resonance? How would you show it? (see p. 89)

Draw: Carbonate ion *If it displays resonance, must draw all structures! * Check out Wayne Breslyn on youtube. com for help


More Practice: • Predict the most stable structure: ONC- or OCN- or NOC-

LEWIS DOT STRUCTURES Predict the most stable structure: ONC- or OCN- or NOC. . . : O: : N: : C: or : O: : C: : N: or : N: : O: : C: 1) Total electrons is: 6 e- for O + 5 e- for N + 4 e- for C + 1 e- for negative charge = 16 etotal. All structures fulfill the octet rule. 2) FC (X) = # of valence electrons - (1/2 bonding electrons + nonbonding electrons) structure#1: FC(C) = 4 - (1/2 4 + 4) = -2 FC(O) = 6 - (1/2 4 + 4) = 0 FC(N) = 5 -(1/2 8 + 0) = +1 structure#2: FC(C) = 4 - (1/2 8 + 0) = 0 FC(O) = 6 - (1/2 4 + 4) = 0 FC(N) = 5 -(1/2 4 + 4) = -1 structure #3: FC(C) = 4 - (1/2 4 + 4) = -2 FC(O) = 6 - (1/2 8 + 0) = +2 FC(N) = 5 -(1/2 4 + 4) = -1 structure #2 has the combination with the lowest formal charge. It also has the negative formal charge on one of the more electronegative atoms

Practice problems • Draw: – Si. H 4 – As. H 3 – Challenge: Cl. O 2 -
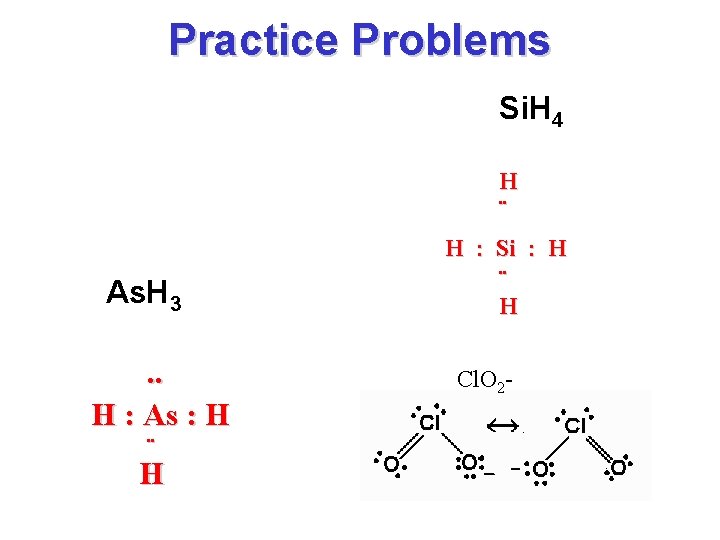
Practice Problems Si. H 4 H ¨ As. H 3. . H : As : H ¨ H H : Si : H ¨ H Cl. O 2 -

Classwork: • Do “WS Covalent Lewis Dot Structures”. You can check your answers on youtube or on kentchemistry. com.

CW- Interactive • Interactive Practice in class today: Must do a MINIMUM of 5 structures to get credit. I will be watching! – Hydrogen carbonate (bicarbonate) – Carbonic acid – 3 more- your choice! • http: //www. stolaf. edu/depts/chemistry/courses/toolkits/123/js/lewis/ (link is on blog) • Also, complete structures on next slide

More Class Practice Problems. You may work together, if helpful. Time: 30 min 1. Draw the best Lewis structure for the following a) H 2 S b) PH 3 c) CH 2 O d) NO 2 - e) H 2 CO 3 f) CBr 4 g) CH 2 FCl 2. 3. 4. h) C 2 H 2 Check the formal charges! If there are resonance structures, draw! I may call a few of you up to the board I) O 3

FYI • Nonmetals in period 3 can be tricky. They can use their d orbitals and sometimes expand their octet. I will not emphasize these “exceptions” on the test (would only ask as an Extra Credit question)

Shape of Molecules (*I have another PPt on VSEPR) • Molecular shapes can be predicted by noting how many bonds and electron pairs surround individual atoms and applying what is called the valence-shell electron-pair repulsion (VSEPR) model. • The basic idea of VSEPR model is that the negatively charged clouds of electron in bonds and lone pair repel each otherefore tends to keep apart as far as possible causing molecules to assume specific shape.

• There are three steps needed to apply the VSEPR model: • Step 1: Draw a Lewis structure of the molecule, and identify the atom whose geometry is of interest. • Step 2: Count the number of electron charge clouds surrounding the atom of interest. • Step 3: Predict molecular shape by assuming that the charge clouds orient in space so that they are as far away from one another as possible.

VSEPR Model The shape depends on the number of charged clouds surrounding the atom

Focus on Molecular Geometry • The concepts of Molecular Geometry vs. Electron Geometry can be confusing…especially the 1 st time through. • I’ve posted a VSEPR PPt on my blog to help you discern between the 2, as you’re likely to encounter both. Just know that we will mainly focus on Molecular Geometry • For the following slides, focus on steric #s 2, 3 and 4 (steric # = number of electron groups = domains) FOR THIS TEST!
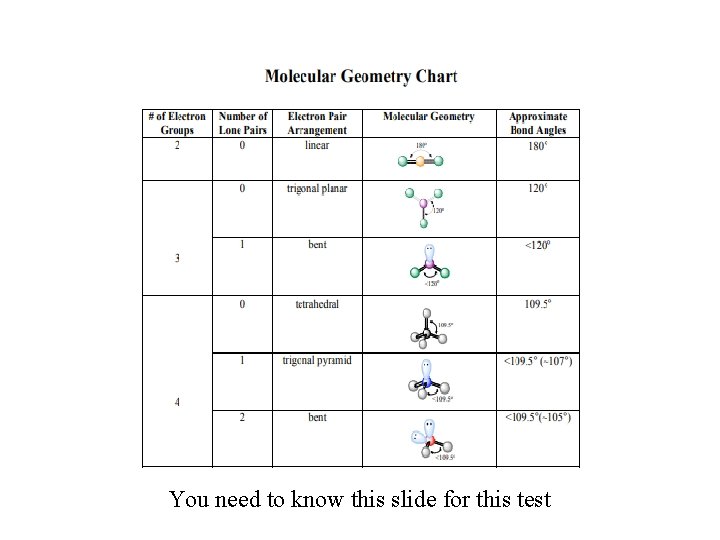
You need to know this slide for this test

This slide is for reference only (no test questions)
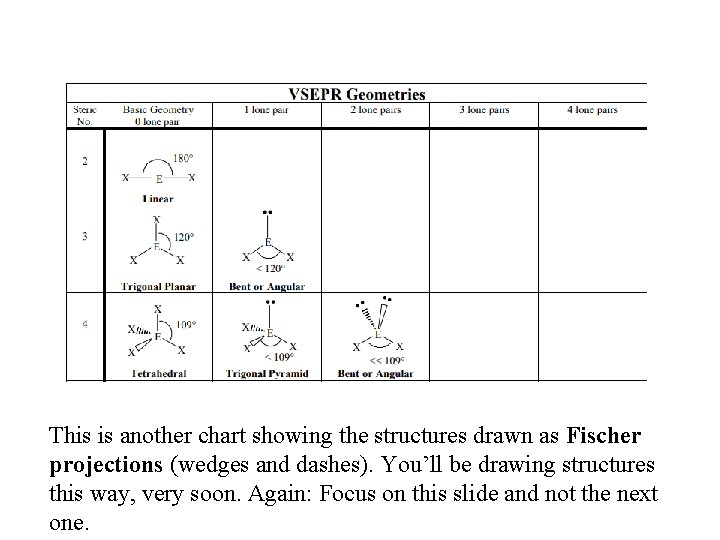
This is another chart showing the structures drawn as Fischer projections (wedges and dashes). You’ll be drawing structures this way, very soon. Again: Focus on this slide and not the next one.
For reference only

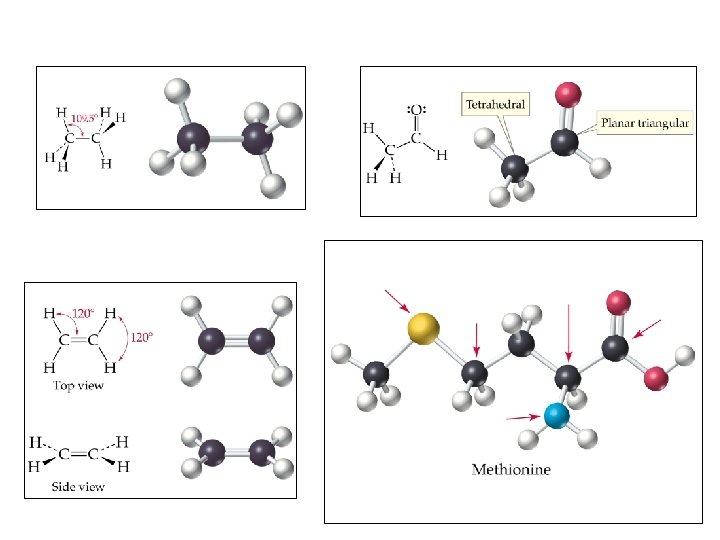
Bond angles to know: • Linear-180 • Trigonal planar-120 (If all e- are in bonded pairs like BF 3. If one pair is lone…. and thus “bent”, like SO 2 , then it’s actually less) • Tetrahedral-109. 5 • Pyramidal-107 (lone pair pushes bonded pairs closer together) • Bent (water)-104. 5 (2 lone pairs) • Practice: Likely we’ll do the Ph. ET interactive (below link no longer works) http: //www. chem. umass. edu/genchem/whelan/Class_ Handouts/111_Molecular_Geometry_Worksheet. pdf

Polar Covalent Bonds and Electronegativity • • • Electrons in a covalent bond occupy the region between the bonded atoms. If the atoms are identical, as in H 2 and Cl 2, electrons are attracted equally to both atoms and are shared equally. If the atoms are not identical, however, as in HCl, the bonding electrons may be attracted more strongly by one atom than by the other and may thus are shared unequally. Such bonds are known as polar covalent bonds.


• In HCl, electrons spend more time near the chlorine than the hydrogen. – Although the molecule is overall neutral, the chlorine is more negative than the hydrogen, resulting in partial charges on the atoms. • Partial charges are represented by placing d- on the more negative atom and d+ on the more positive atom. • Ability of an atom to attract electrons is called the atom’s electronegativity. • Fluorine, the most electronegative element, assigned a value of 4, and less electronegative atoms assigned lower values • Electronegativity differences: – > ~ 1. 7 -1. 9 is considered ionic – < ~ 1. 7 -1. 9 is considered covalent • • 0. 5 to 1. 7 is Polar Covalent 0. 0 to 0. 5 is Non-Polar Covalent
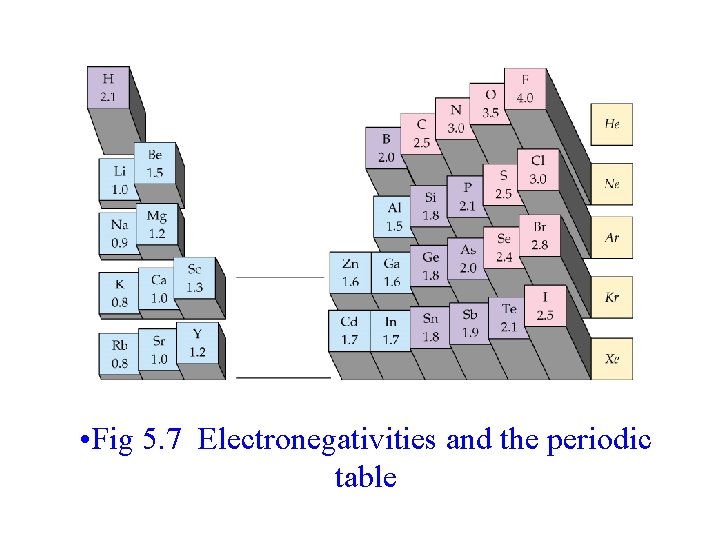
• Fig 5. 7 Electronegativities and the periodic table

Polar Molecules • • Entire molecule can be polar if electrons are attracted more strongly to one part of the molecule than to another. Molecule’s polarity is due to the sum of all individual bond polarities (sometimes called dipolar forces or dipoles) and lone -pair contribution in the molecule.
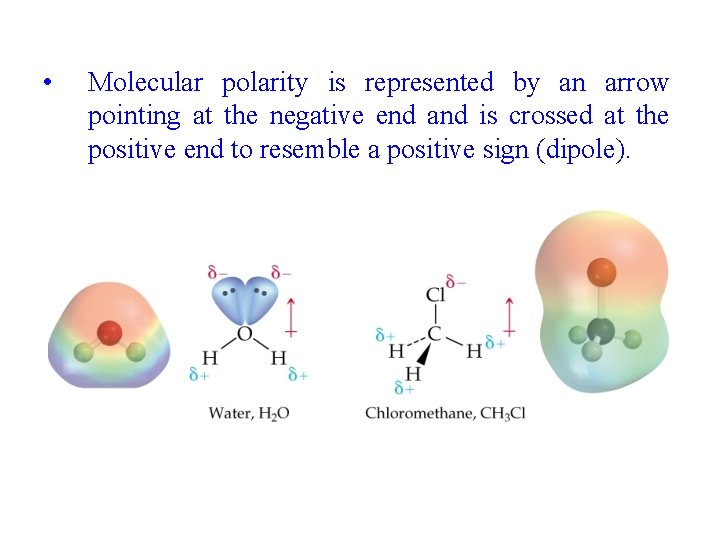
• Molecular polarity is represented by an arrow pointing at the negative end and is crossed at the positive end to resemble a positive sign (dipole).
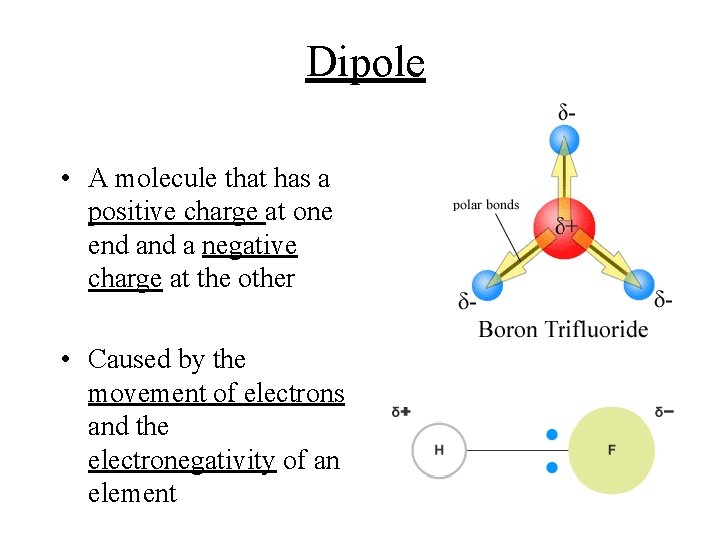
Dipole • A molecule that has a positive charge at one end a negative charge at the other • Caused by the movement of electrons and the electronegativity of an element

• • Molecular polarity depends on the shape of the molecule as well as the presence of polar covalent bonds and lone-pairs. Below shows molecules with no net polarity (dipoles cancel)

Intermolecular Forces (*there’s also a separate PPt on blog) • IMF: Forces of attraction or repulsion which act between neighboring molecules • You are responsible for knowing: – London dispersion forces – Dipole – dipole attractions – Hydrogen bonding

What is a London Dispersion Force? • Forces of attraction that exist between SMALL dipoles in non -polar molecules • Small dipoles are created by the movement of electrons in the atom • Relatively speaking, they’re ‘weak’ attractions

What is Dipole – Dipole Attraction? • Attractive forces between polar molecules Orientation of Polar Molecules in a Solid

What is Hydrogen Bonding? • Special type of dipole – dipole • VERY STRONG intermolecular force • Occurs when F, O, or N are bonded to H – “H bonding is FON”
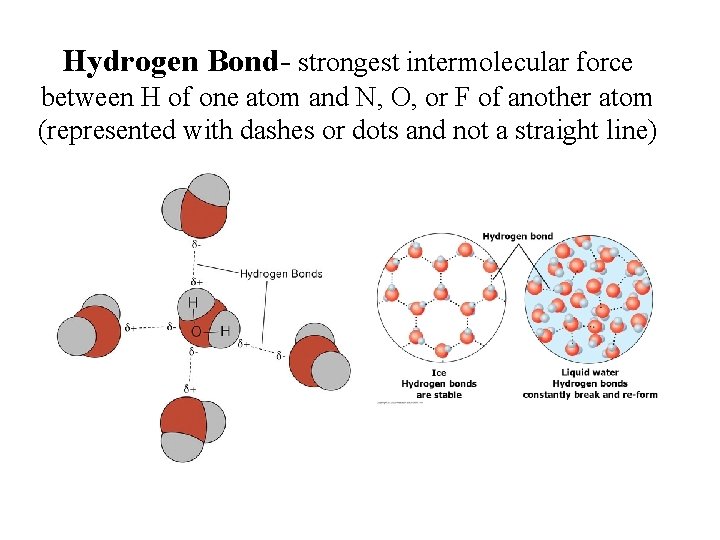
Hydrogen Bond- strongest intermolecular force between H of one atom and N, O, or F of another atom (represented with dashes or dots and not a straight line)

Strength of Forces • London dispersion << Dipole – Dipole < Hydrogen ASSIGNMENT: Complete the VSEPR, Polarity, IM Worksheet

Naming Binary Molecular Compounds • • When two different elements combines together they form binary compound. The formulas of binary compounds are usually written with the less electronegative element first. Thus, metals are always written before non-metals. Prefix such as mono, di, tri, tetra etc, are used to indicate number of atoms of each element.
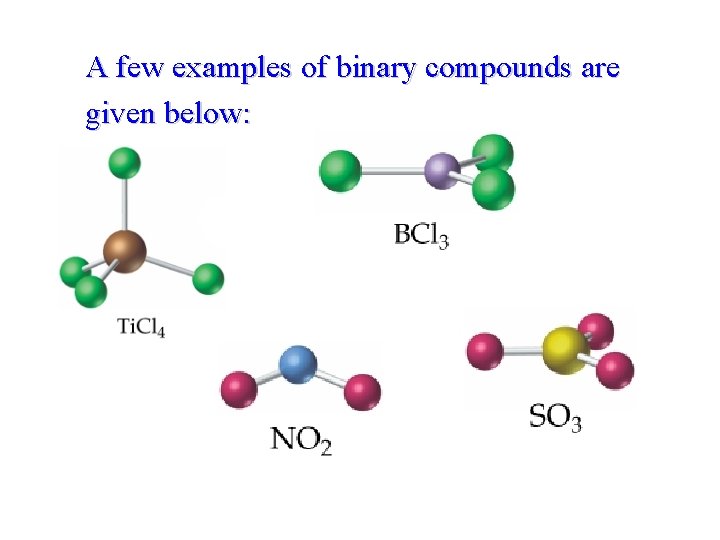
A few examples of binary compounds are given below:

• the • The following ‘two steps guide’ is helpful in naming binary compounds: Step 1: Name formula, using a prefix if needed to indicate the number of atoms. ment second • Step the 2: Name formula, using an –ide ending as for anions, along with a prefix if needed to indicate the number of atoms.

Characteristics of Molecular Compounds • Molecules are neutral as a result there is no strong electrical attractions between the molecules to hold them together. However, there are weaker forces that exist between molecules. –Gases: intermolecular forces are very weak –If the intermolecular forces are somewhat stronger, the molecules are pulled together into a liquid. –If the forces are stronger, the substance becomes a molecular solid.

• • • solids are lower than those of ionic solids. water. when melted because they have no charged particles.

Review: Remember… • Electronegativity increases across periods and decreases down groups • Ionization Energy increases across periods and decreases down groups • Atomic radius decreases across periods and increases down groups
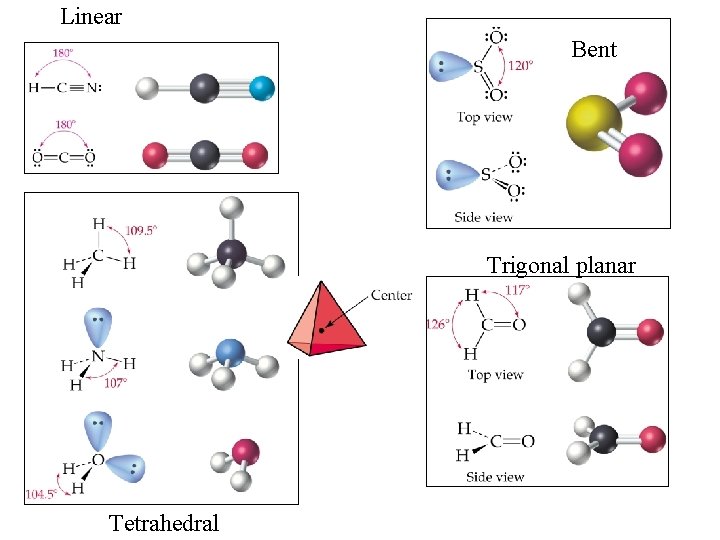
Linear Bent Trigonal planar Tetrahedral
- Slides: 99