Lecture Power Point to accompany Inquiry into Life



































- Slides: 50

Lecture Power. Point to accompany Inquiry into Life Twelfth Edition Sylvia S. Mader Chapter 6 Copyright © The Mc. Graw-Hill Companies, Inc. Permission required for reproduction or display.

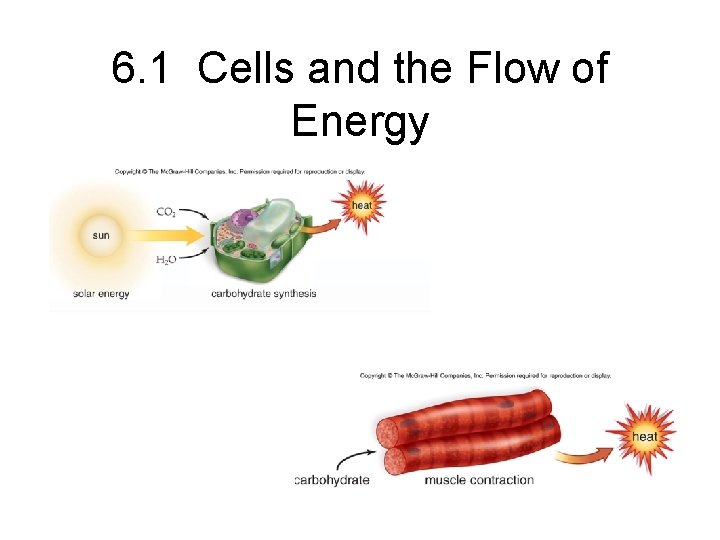
6. 1 Cells and the Flow of Energy

6. 1 Cells and the Flow of Energy • Energy is the ability to do work or bring about change.

6. 1 Cells and the Flow of Energy • Energy is the ability to do work or bring about change. • Forms of Energy

6. 1 Cells and the Flow of Energy • Energy is the ability to do work or bring about change. • Forms of Energy – Kinetic energy is the energy of motion.

6. 1 Cells and the Flow of Energy • Energy is the ability to do work or bring about change. • Forms of Energy – Kinetic energy is the energy of motion. – Potential energy is stored energy.

Flow of Energy

6. 1 Cells and the Flow of Energy • Two Laws of Thermodynamics – Energy cannot be created or destroyed, but it can be changed from one form to another. – Energy cannot be changed from one form to another without a loss of usable energy.

6. 1 Cells and the Flow of Energy

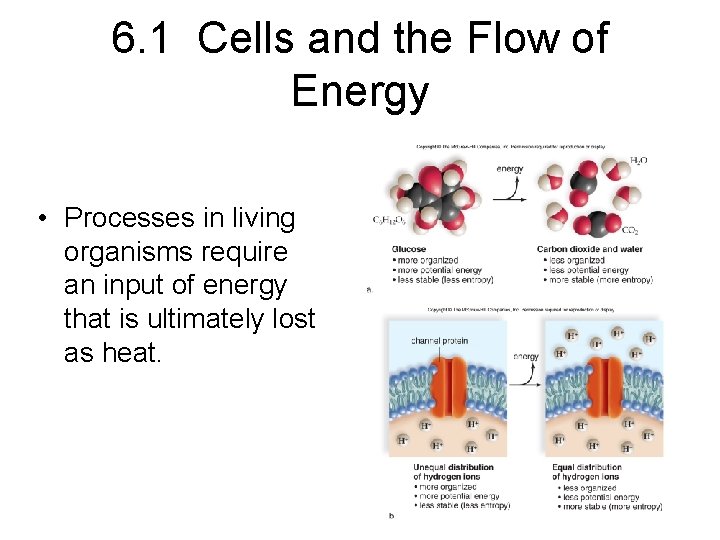
6. 1 Cells and the Flow of Energy • Cells and Entropy – Entropy refers to the relative amount of disorganization.

6. 1 Cells and the Flow of Energy • Cells and Entropy – Entropy refers to the relative amount of disorganization. – Energy transformations in cells increase the amount of entropy.

6. 1 Cells and the Flow of Energy • Processes in living organisms require an input of energy that is ultimately lost as heat.
6. 2 Metabolic Reactions and Energy Transformations

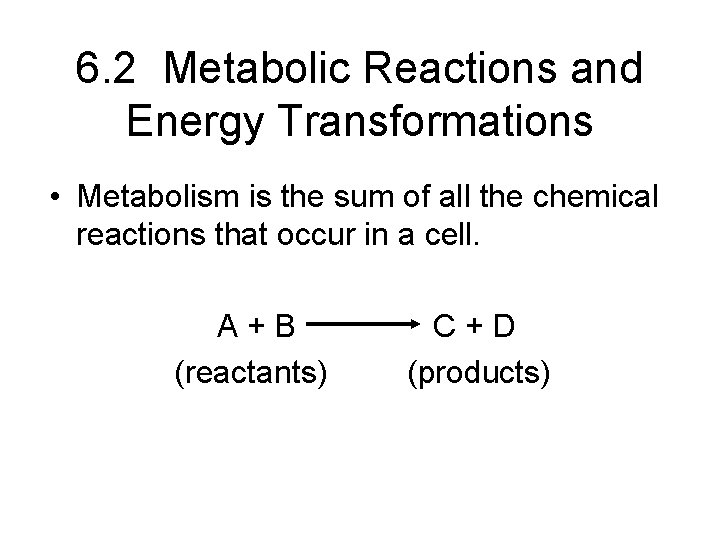
6. 2 Metabolic Reactions and Energy Transformations • Metabolism is the sum of all the chemical reactions that occur in a cell.

6. 2 Metabolic Reactions and Energy Transformations • Metabolism is the sum of all the chemical reactions that occur in a cell. A+B (reactants) C+D (products)

6. 2 Metabolic Reactions and Energy Transformations • Free energy (∆G) is the amount of energy available.

6. 2 Metabolic Reactions and Energy Transformations • Free energy (∆G) is the amount of energy available. – Exergonic reactions are ones where energy is released (∆G is negative)

6. 2 Metabolic Reactions and Energy Transformations • Free energy (∆G) is the amount of energy available. – Exergonic reactions are ones where energy is released (∆G is negative) – Endergonic reactions require an input of energy. (∆G is positive)

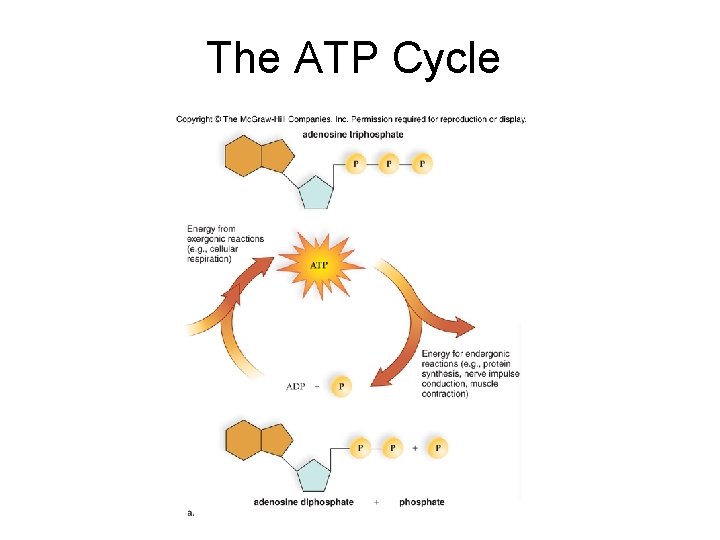
6. 2 Metabolic Reactions and Energy Transformations • ATP: Energy for Cells – ATP stands for adenosine triphosphate, the common energy currency for cells.

6. 2 Metabolic Reactions and Energy Transformations • ATP: Energy for Cells – ATP stands for adenosine triphosphate, the common energy currency for cells. – ATP is generated from ADP (adenosine diphosphate) + an inorganic phosphate molecule ( P )

The ATP Cycle

6. 2 Metabolic Reactions and Energy Transformations • Structure of ATP – ATP is a nucleotide that is composed of: • Adenine (a nitrogen-containing base) • Ribose (a 5 -carbon sugar) • Three phosphate groups

6. 2 Metabolic Reactions and Energy Transformations • Structure of ATP – ATP is a “high energy” compound because a phosphate group can easily be removed.

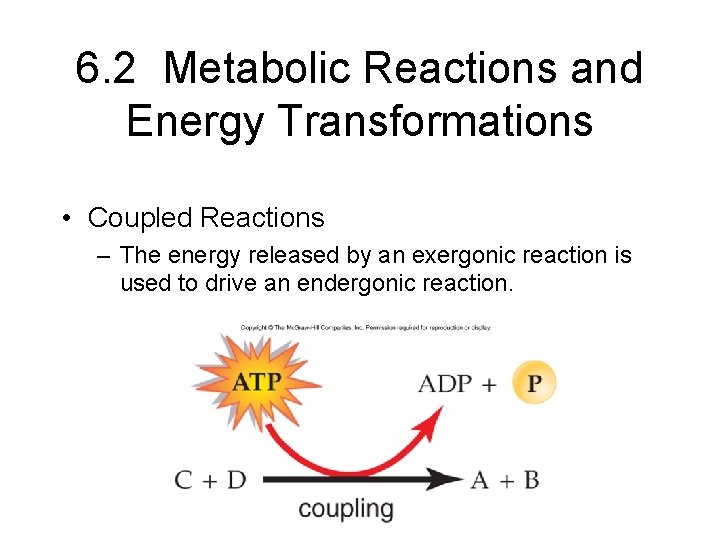
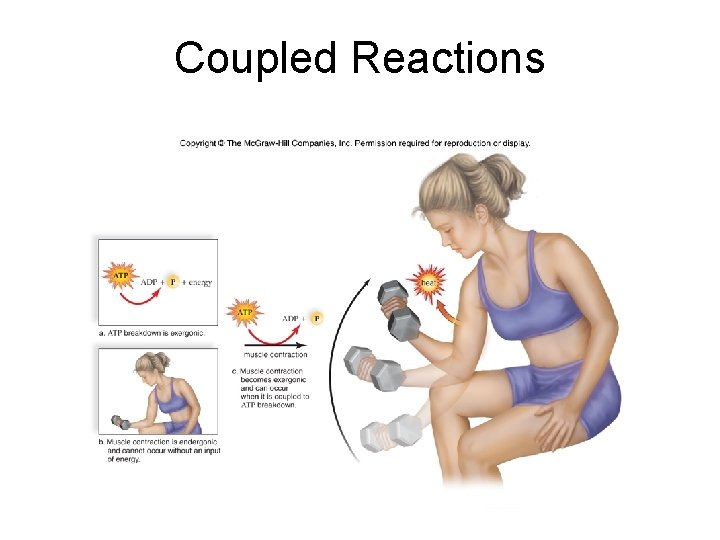
6. 2 Metabolic Reactions and Energy Transformations • Coupled Reactions – The energy released by an exergonic reaction is used to drive an endergonic reaction.

Coupled Reactions

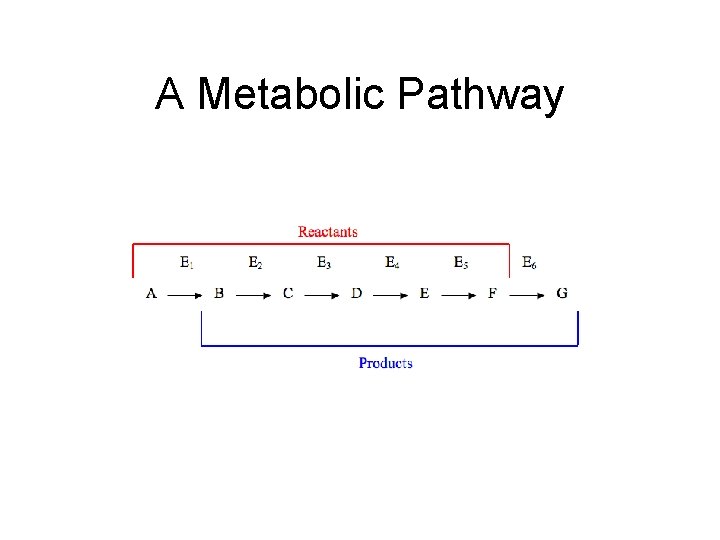
6. 3 Metabolic Pathways and Enzymes • Metabolic pathways are a series of linked reactions. – These begin with a specific reactant and produce an end product

6. 3 Metabolic Pathways and Enzymes • Enzymes are usually proteins that function to speed a chemical reaction. – Enzymes serve as catalysts

A Metabolic Pathway

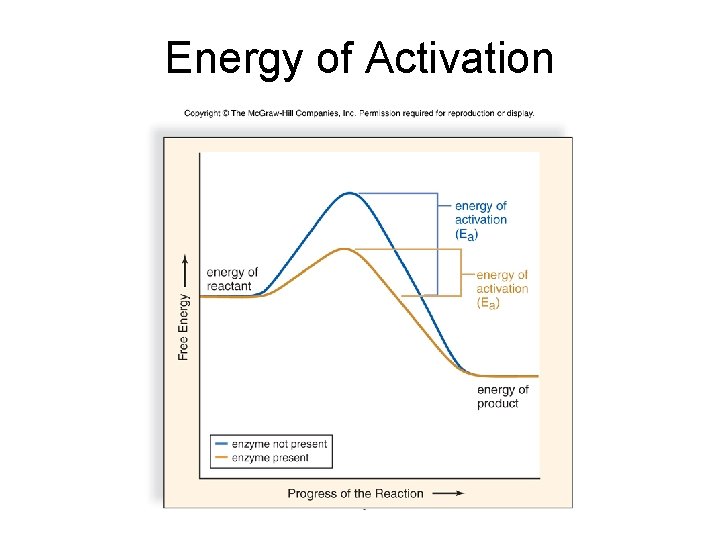
6. 3 Metabolic Pathways and Enzymes • The Energy of Activation (Ea) is the energy that must be added to cause molecules to react with one another.

Energy of Activation

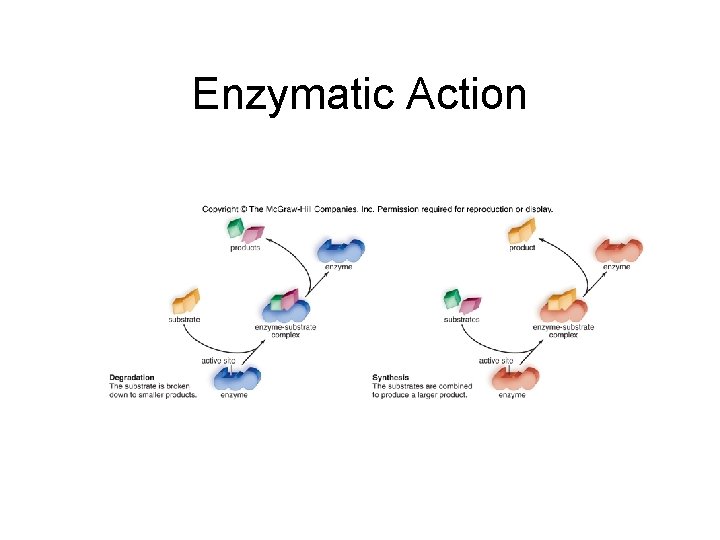
6. 3 Metabolic Pathways and Enzymes • How Enzymes Function – Enzyme binds substrate to form a complex – E + S E + P

Enzymatic Action

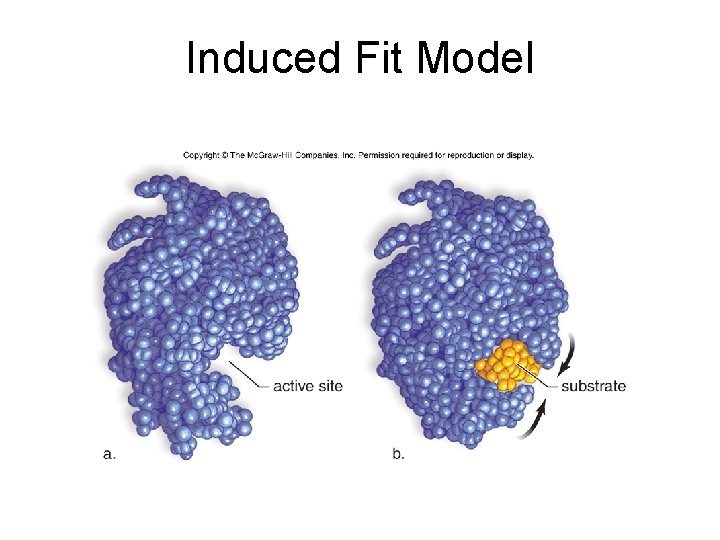
6. 3 Metabolic Pathways and Enzymes • How Enzymes Function – Enzyme binds substrate to form a complex – E + S E + P – Induced fit model • Substrate and active site shapes don’t match exactly • Active site is induced to undergo a slight change in shape to accommodate substrate binding

Induced Fit Model

6. 3 Metabolic Pathways and Enzymes • Factors Affecting Enzymatic Speed – Substrate Concentration – Temperature and p. H – Enzyme Activation – Enzyme Inhibition – Enzyme Cofactors

6. 3 Metabolic Pathways and Enzymes • Substrate Concentration • Enzyme activity increases as substrate concentration increases because there are more collisions between substrate and enzyme • Maximum rate is achieved when all active sites of an enzyme are filled continuously with substrate

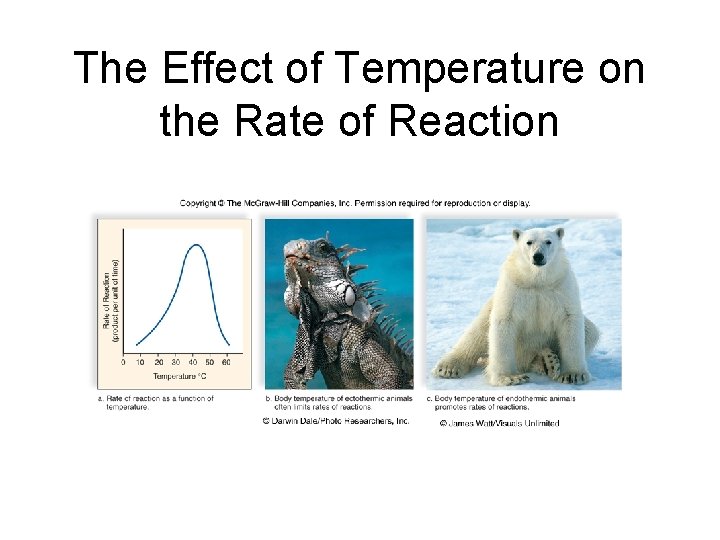
Metabolic Pathways and Enzymes • Temperature – Enzyme activity increase as temperature rises – Higher temperatures cause more effective collisions between enzymes and substrates – High temperatures may denature an enzyme, inhibiting its ability to bind to substrates

The Effect of Temperature on the Rate of Reaction

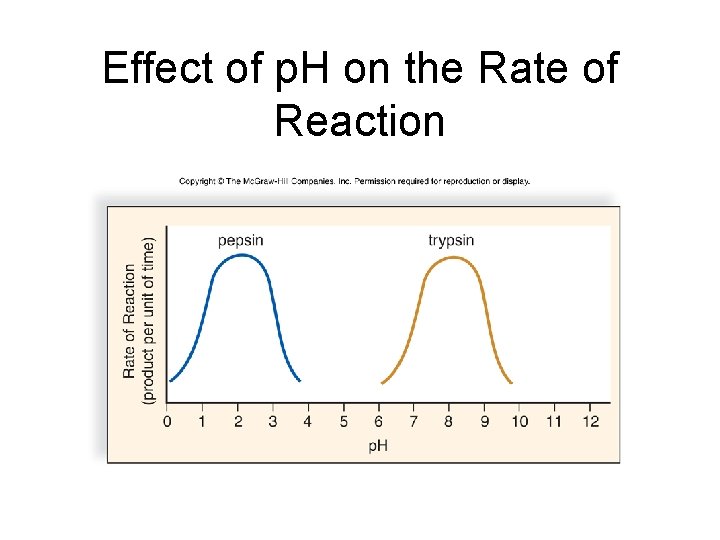
Metabolic Pathways and Enzymes • p. H • Each enzyme has an optimal p. H • Enzyme structure is p. H dependent • Extremes of p. H can denature an enzyme by altering its structure

Effect of p. H on the Rate of Reaction

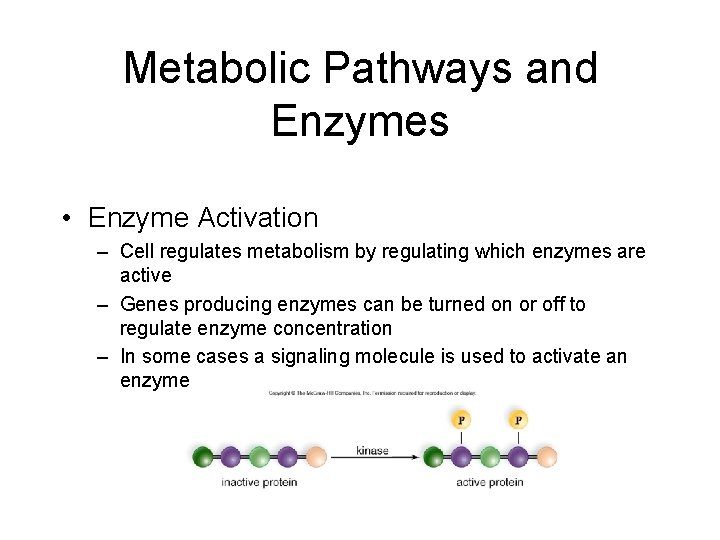
Metabolic Pathways and Enzymes • Enzyme Activation – Cell regulates metabolism by regulating which enzymes are active – Genes producing enzymes can be turned on or off to regulate enzyme concentration – In some cases a signaling molecule is used to activate an enzyme

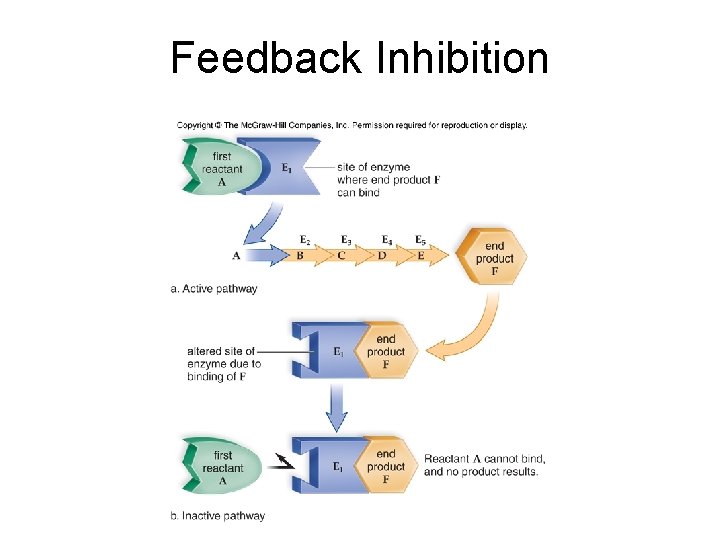
Metabolic Pathways and Enzymes • Enzyme Inhibition – Occurs when enzyme cannot bind its substrate – Activity of cell enzymes is regulated by feedback inhibition – Ex: when product is abundant it binds to the enzyme’s active site and blocks further production – When product is used up, it is removed from the active site – In a more complex type of inhibition, product binds to a site other than the active site, which changes the shape of the active site – Poisons are often enzyme inhibitors

Feedback Inhibition

Metabolic Pathways and Enzymes • Enzyme Cofactors – Molecules which help enzyme function – Copper and zinc are examples of inorganic cofactors – Organic non-protein cofactors are called coenzymes • Vitamins are often components of coenzymes

6. 4 Oxidation-Reduction and the Flow of Energy • Oxidation-Reduction – Oxidation is the loss of electrons – Reduction is the gaining of electrons – Ex: when oxygen combines with a metal like Mg, oxygen receives electrons (becomes negatively charged) and Mg loses electrons (becomes positively charged) • We say Mg has become oxidized, and oxygen is reduced (has a negative charge) when Mg. O forms

6. 4 Oxidation-Reduction and the Flow of Energy • Oxidation-Reduction – The term oxidation is used even when oxygen is not involved • Ex: Na+ + Cl- Na. Cl in which sodium is oxidized and chloride is reduced – This also applies to covalent reactions involving hydrogen atoms – Oxidation is the loss of hydrogen and reduction is the gain of hydrogen atoms

6. 4 Oxidation-Reduction and the Flow of Energy. • Photosynthesis – energy + 6 CO 2+6 H 2 O C 6 H 12 O 6 + 6 O 2 – Hydrogen atoms are transferred from water to carbon dioxide and glucose is formed – Energy is required and this comes in the form of light energy from the sun – Chloroplasts convert solar energy to ATP which is then used along with hydrogen to reduce carbon dioxide to glucose

Oxidation-reduction and the flow of energy cont’d. • Cell Respiration – – C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + energy Glucose is oxidized (lost hydrogen atoms) Oxygen is reduced to form water Complete oxidation of a mole of glucose produces 686 kcal of energy – This energy is used to form ATP – The oxidation of glucose to form ATP is done is a series of small steps to increase efficiency

6. 4 Oxidation-Reduction and the Flow of Energy. • Organelles and the flow of energy – Cycling of molecules between chloroplasts and mitochondria allows energy to flow from sun to all living things – Chloroplasts use light energy from the sun to make carbohydrates – Mitochondria break down carbohydrates to form ATP – Cell respiration produces carbon dioxide and water which are used in photosynthesis

Relationship of Chloroplasts to Mitochondria