Infrared Spectroscopy and Mass Spectroscopy CH 12 Leroy










- Slides: 37

Infrared Spectroscopy and Mass Spectroscopy CH 12 Leroy Wade Mohammed Ali 1

12 -1 Introduction Absorption Spectroscopy: Measurement of the amount of light absorbed by a compound as a function of the wavelength of light. In general, a sample is irradiated by a light source, and the amount of light transmitted at various wavelenght is measured by a detector and plotted on a graph. Mohammed Ali 2

Spectroscopic Techniques: Techniques Ø Mohammed Ali 3

12 -2 The Electromagnetic Spectrum Mohammed Ali Fig 12 -1 4

12 -3 The Infrared Region The position of IR band is specified by its wavelength (l), measured in micron, one millionths of a meter. A more common unit is wave number, u , number of cycles (wavelength) of the wave in a centimeter. The wavenumber is the wavelength (in centimeter) Mohammed Ali reciprocal of the 5

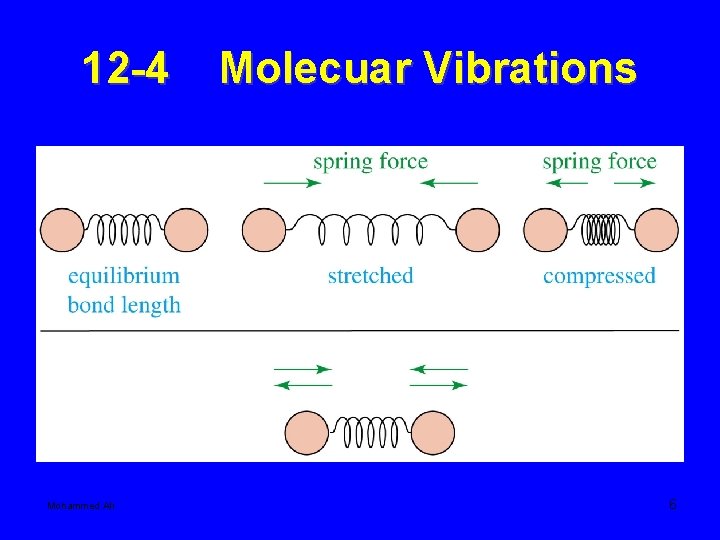
12 -4 Mohammed Ali Molecuar Vibrations 6

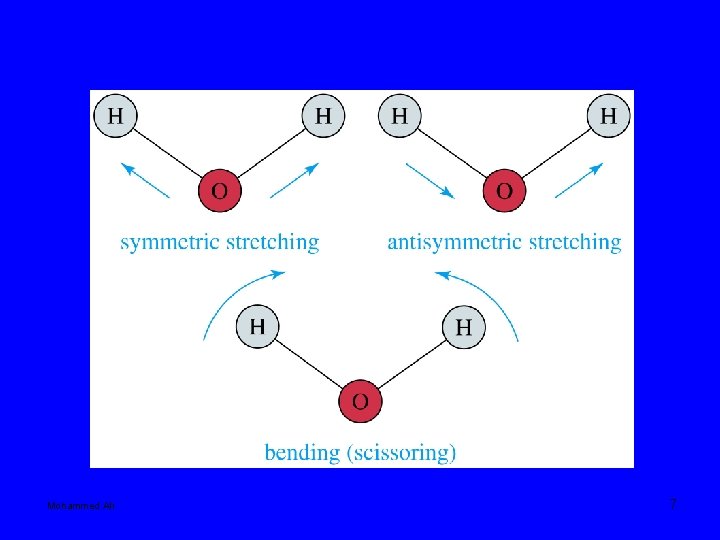
Mohammed Ali 7

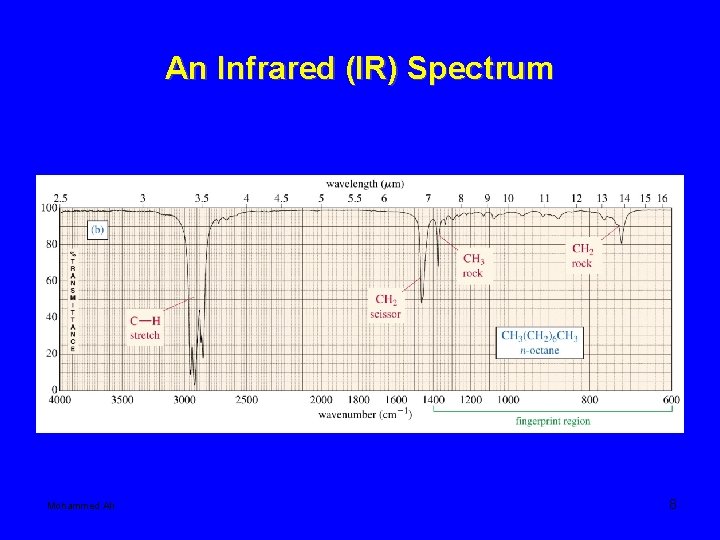
An Infrared (IR) Spectrum Mohammed Ali 8

12 -5 IR Active and IR Inactive Vibrations of bonds with dipole moment absorbs IR and are said to be IR active. Vibrations of bonds (symmetric bonds) without a dipole moment does not absorb IR and are said to be IR inactive. Mohammed Ali 9

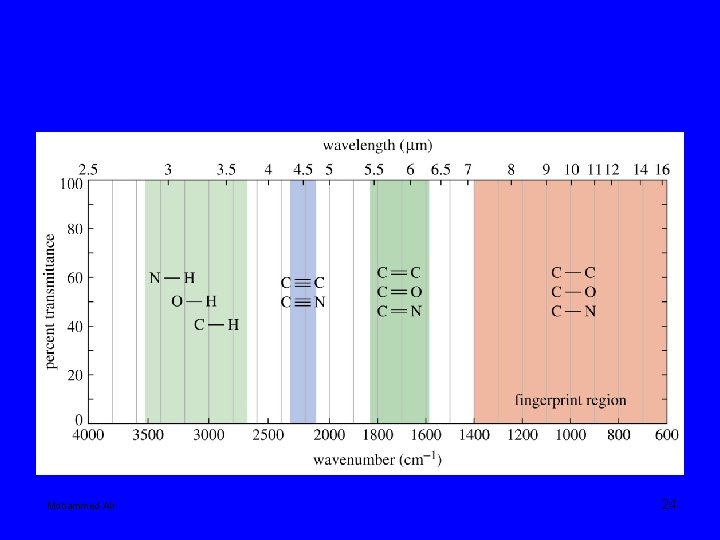
IR frequency range 600 -4000 cm-1 Fingerprint region: 600 – 1400 cm-1 frequencies of two different molecules are different in this region. 1600 -3500 cm-1 where the characteristic frequencies of various FG shows. Mohammed Ali 10

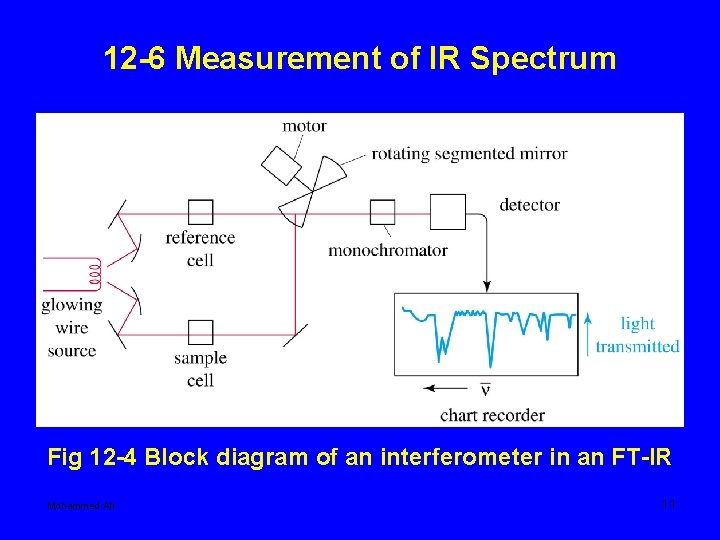
12 -6 Measurement of IR Spectrum Fig 12 -4 Block diagram of an interferometer in an FT-IR Mohammed Ali 11

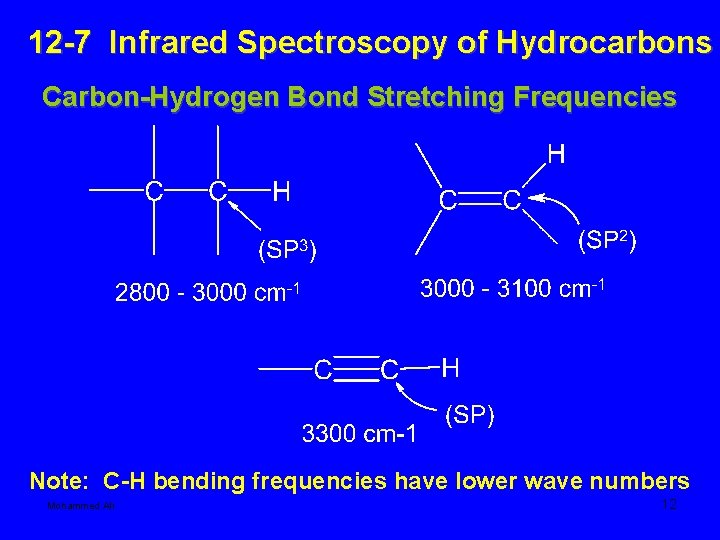
12 -7 Infrared Spectroscopy of Hydrocarbons Carbon-Hydrogen Bond Stretching Frequencies Note: C-H bending frequencies have lower wave numbers Mohammed Ali 12

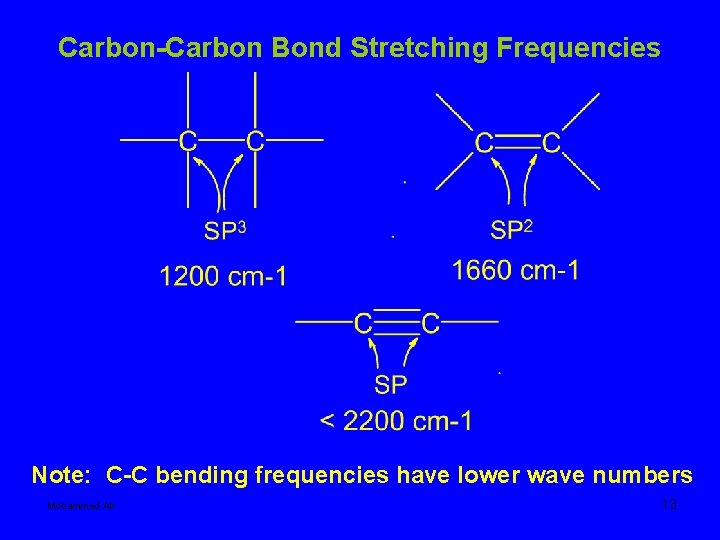
Carbon-Carbon Bond Stretching Frequencies Note: C-C bending frequencies have lower wave numbers Mohammed Ali 13

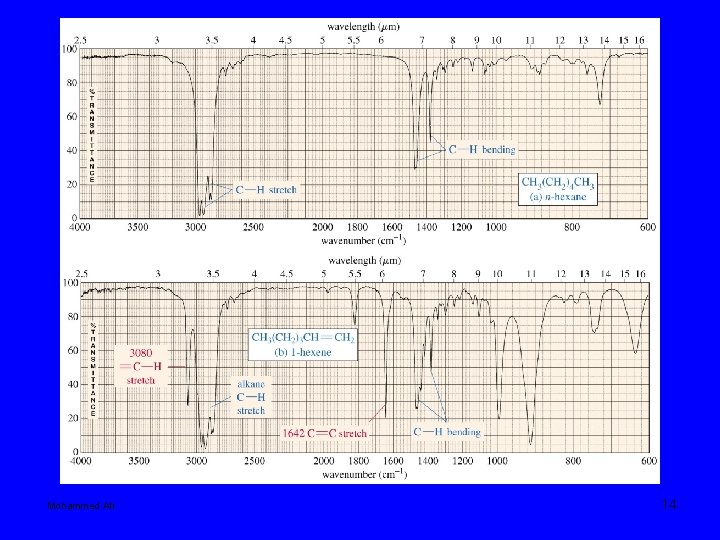
Mohammed Ali 14

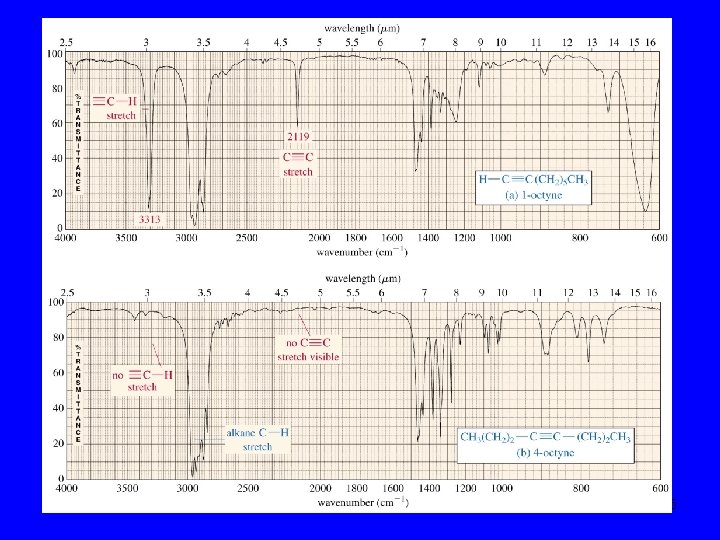
Mohammed Ali 15

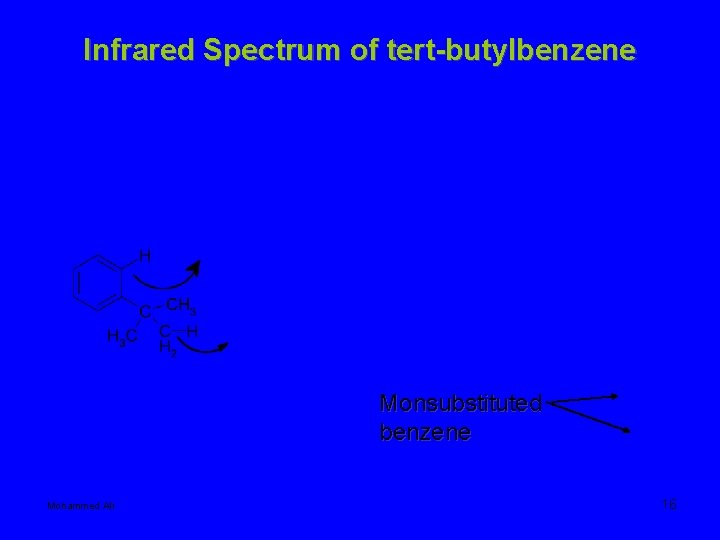
Infrared Spectrum of tert-butylbenzene Monsubstituted benzene Mohammed Ali 16

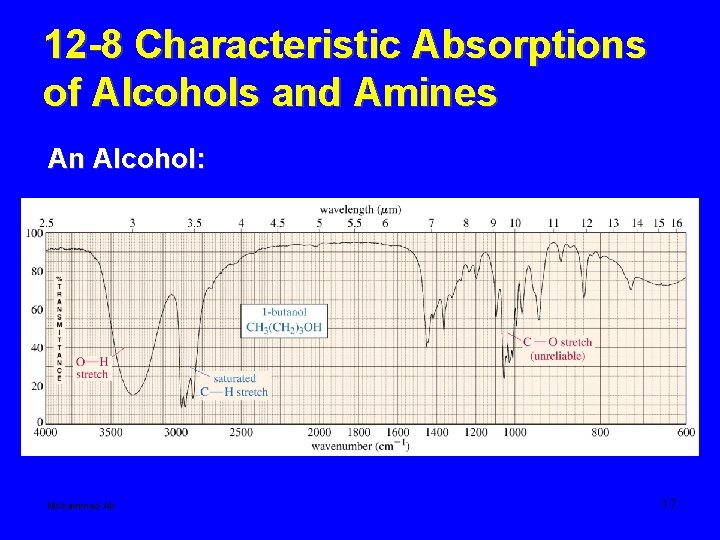
12 -8 Characteristic Absorptions of Alcohols and Amines An Alcohol: Mohammed Ali 17

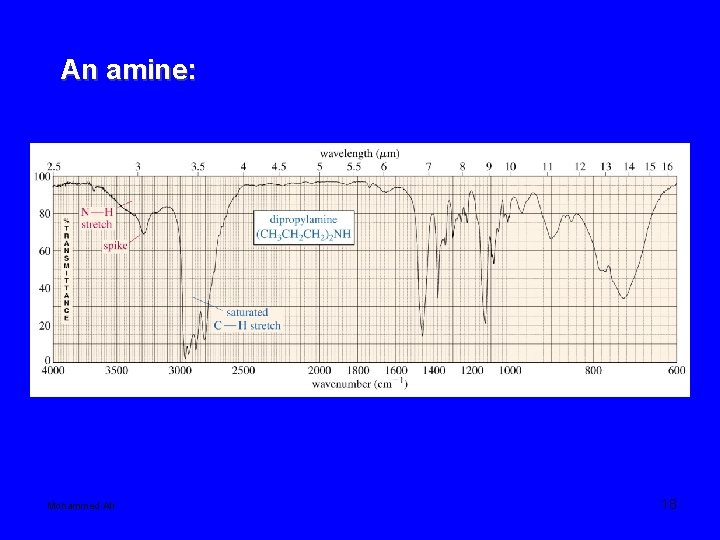
An amine: Mohammed Ali 18

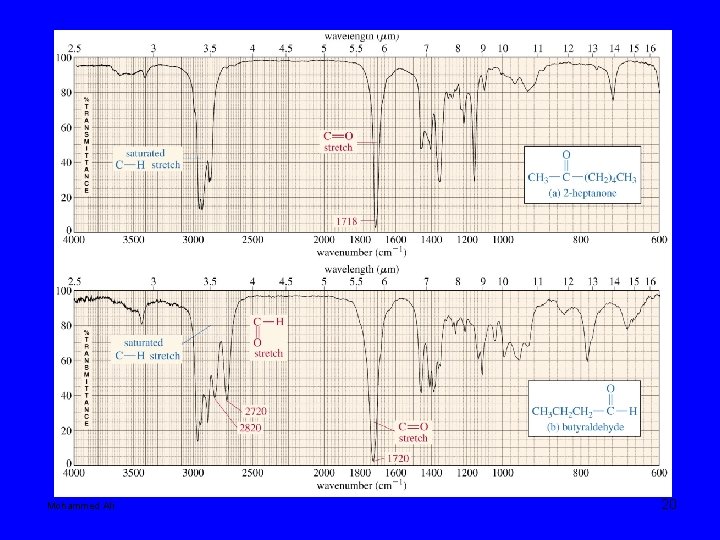
12 -9 Characteristic Absorptions of Carbonyl Compounds (Esters and carbonates have slightly higher C=O absorption value) Mohammed Ali 19

Mohammed Ali 20

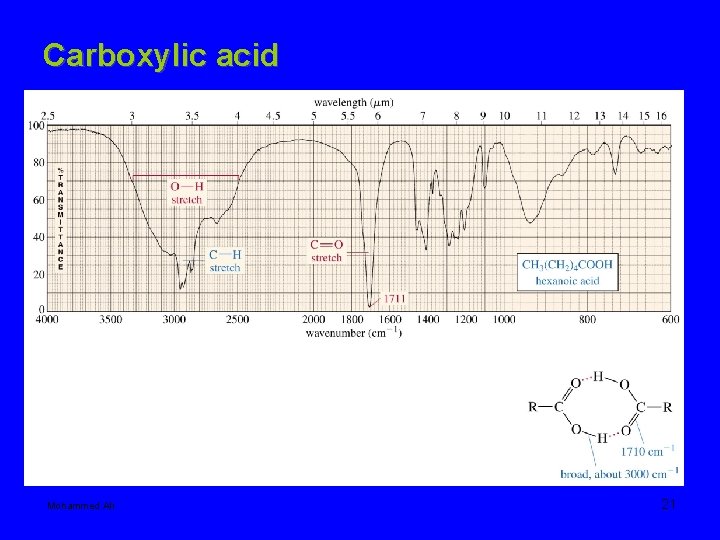
Carboxylic acid Mohammed Ali 21

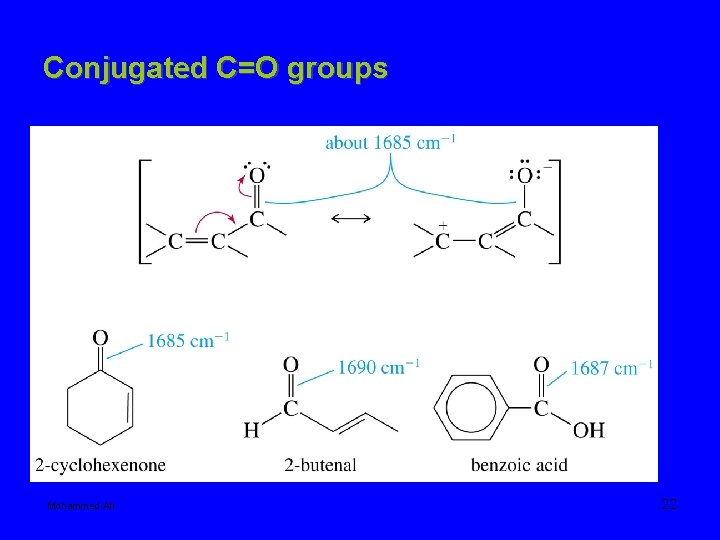
Conjugated C=O groups Mohammed Ali 22

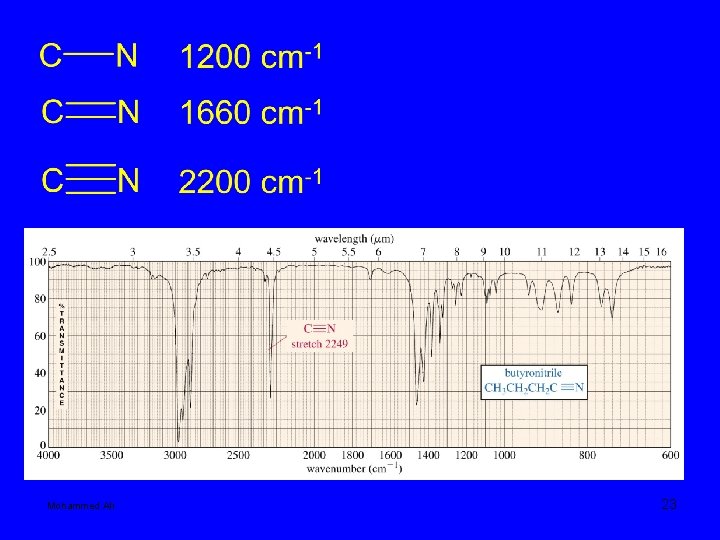
Mohammed Ali 23

Mohammed Ali 24

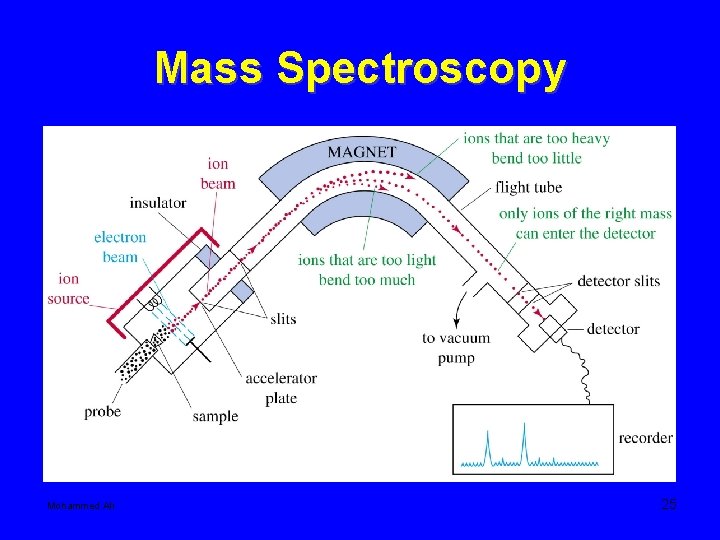
Mass Spectroscopy Mohammed Ali 25

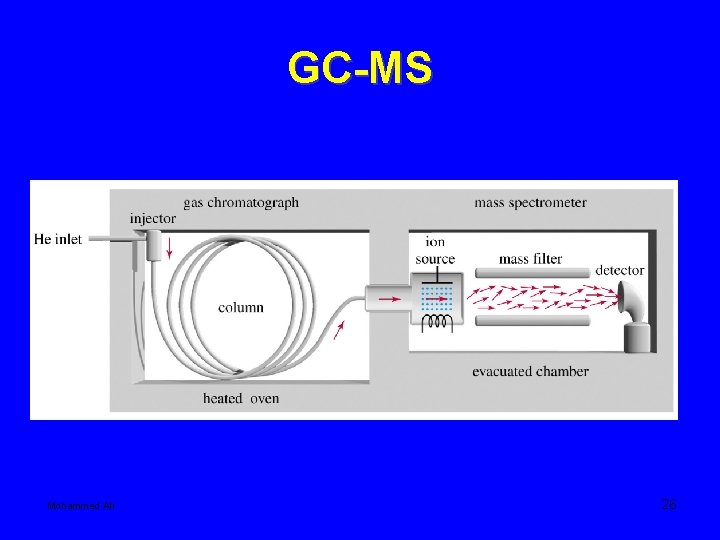
GC-MS Mohammed Ali 26

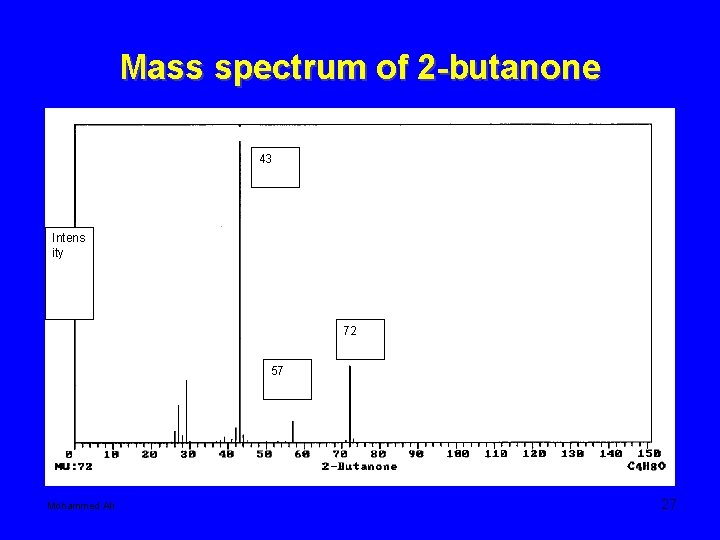
Mass spectrum of 2 -butanone 43 Intens ity 72 57 Mohammed Ali 27

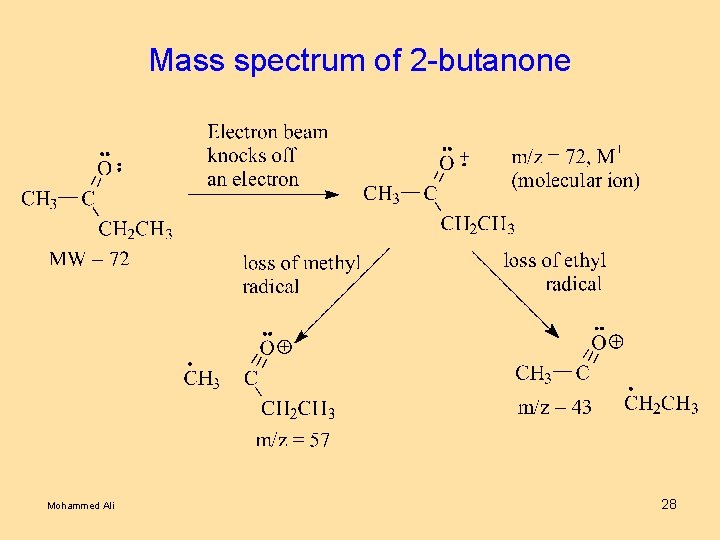
Mass spectrum of 2 -butanone Mohammed Ali 28

“m/z” means mass to charge ratio. Most ions have only one charge, so m/z equals the molecular weight of the ion. The ion produced after the first electron is removed is called the “molecular ion”, and sometimes abbreviated as M+. Other important data we can obtain from mass spectrometry. • nitrogens will have an odd molecular weight, usually. This is always true if the compound only contains C, H, N, and O. Mohammed Ali 29

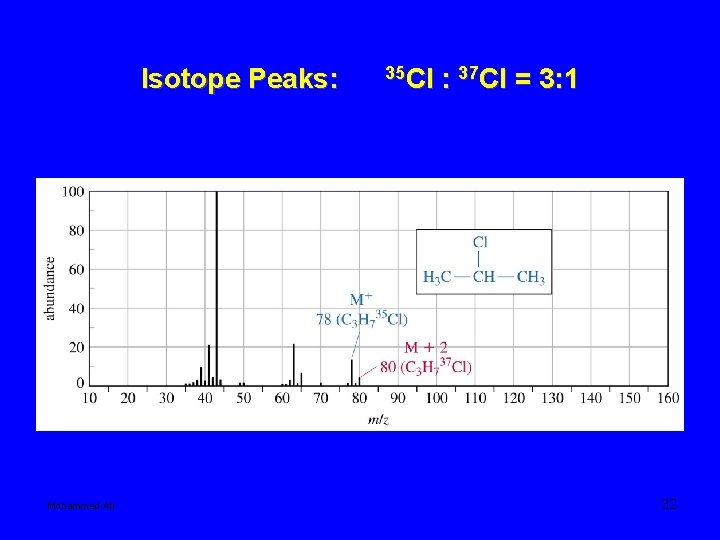
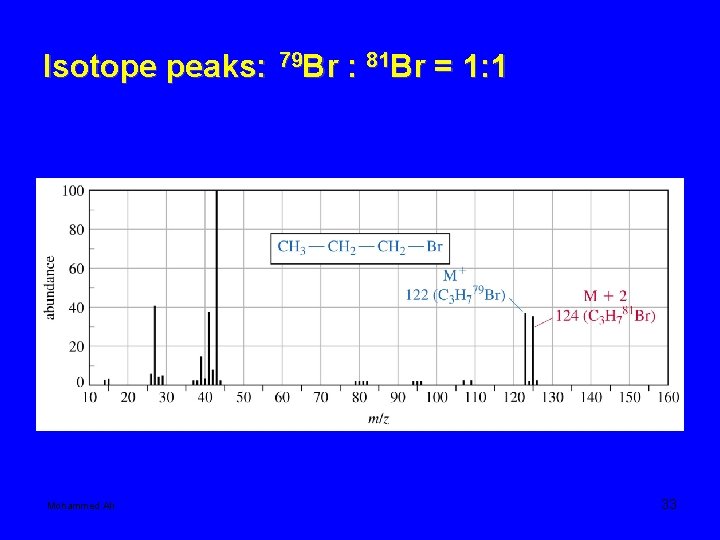
• Compounds containing chlorine or bromine show two peaks two mass units apart for fragments that contain a chlorine or bromine. This is because chlorine is composed of two major isotopes, 35 Cl and 37 Cl. About 75% of Cl is 35 Cl, and 25% is 37 Cl. Therefore, the size of the two peaks will have a ratio of about 3: 1. The two isotopes of Bromine occur in about equal amounts, so the two peaks will be about the same size. Mohammed Ali 30

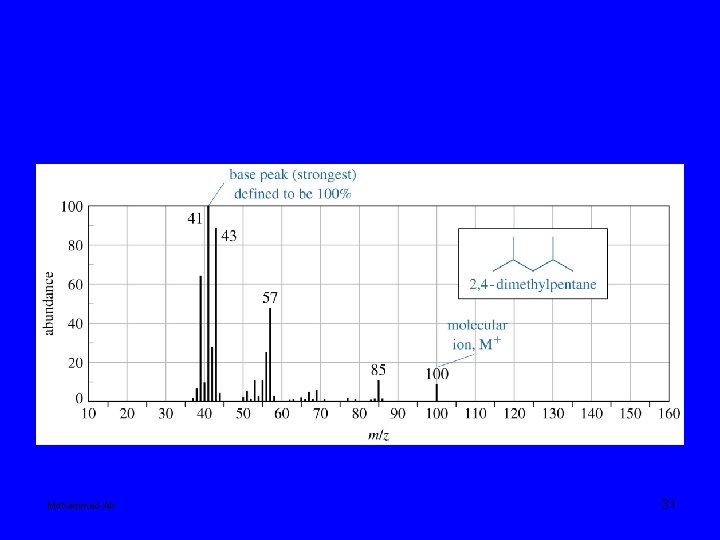
Mohammed Ali 31

Isotope Peaks: Mohammed Ali 35 Cl : 37 Cl = 3: 1 32

Isotope peaks: Mohammed Ali 79 Br : 81 Br = 1: 1 33

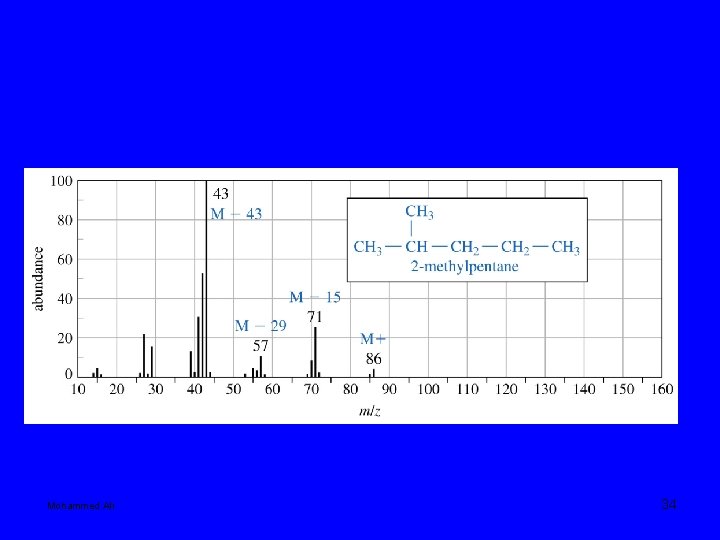
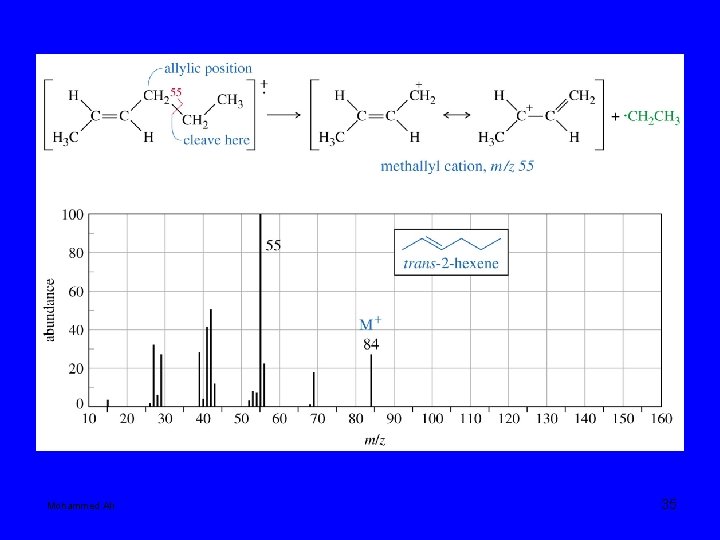
Mohammed Ali 34

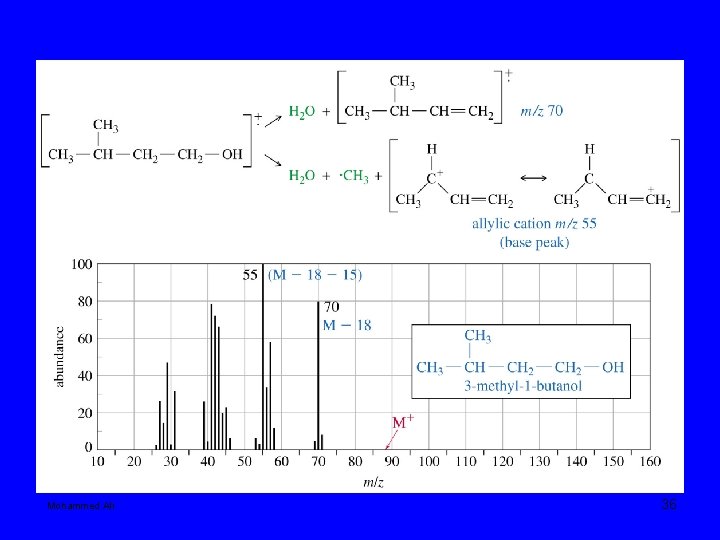
Mohammed Ali 35

Mohammed Ali 36

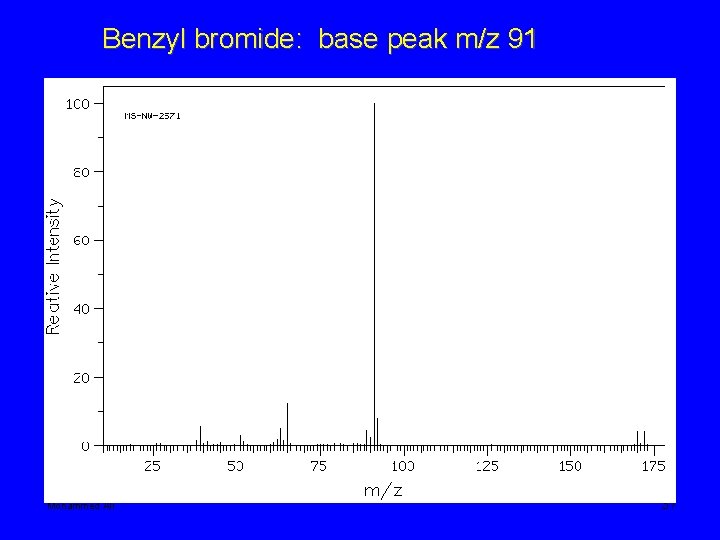
Benzyl bromide: base peak m/z 91 Mohammed Ali 37