Infrared Spectrometry Outline Infrared spectroscopy measures the bond

第四讲:红外光谱法 Infrared Spectrometry

Outline Infrared spectroscopy measures the bond vibration frequencies in a molecule and is used to determine the functional group n The IR region n Theory of n Instrumentation IR

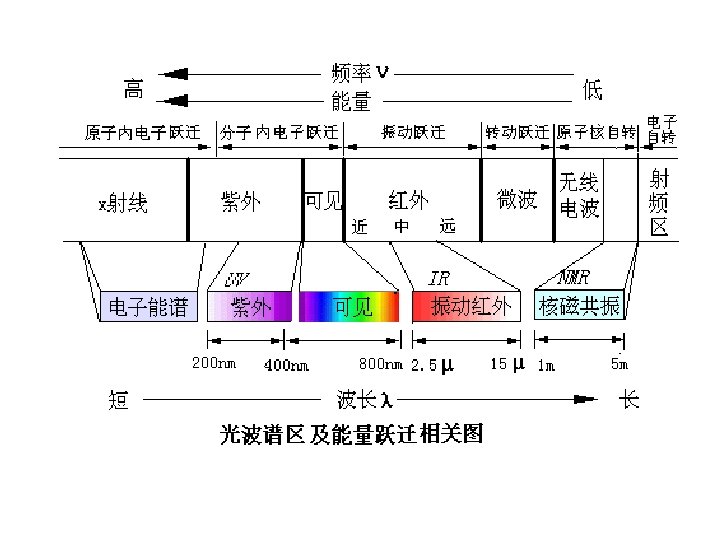
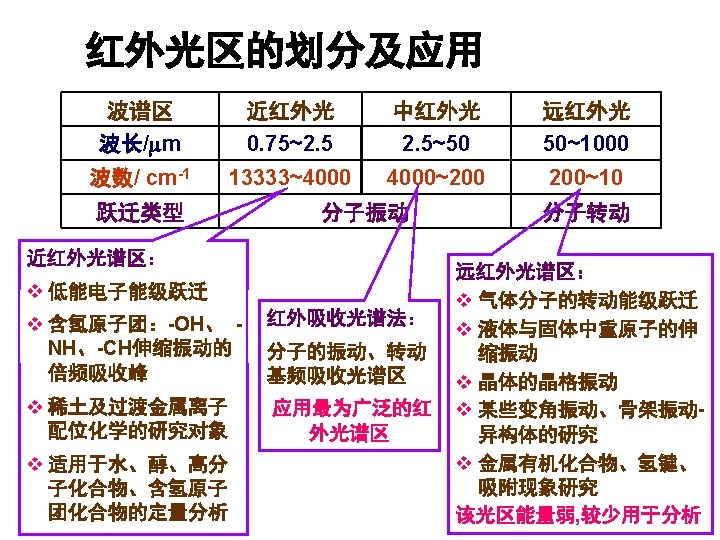
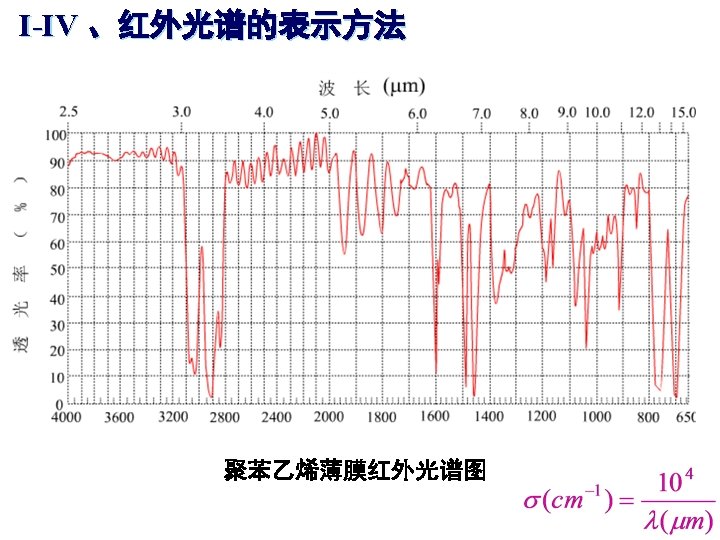
The IR Region • Just below red in the visible region • Wavelengths usually 2. 5 -25 mm • More common units are wavenumbers, or cm-1, the reciprocal of the wavelength in centimeters (104/mm = 4000 -400 cm-1) • Wavenumbers are proportional to frequency • The IR region is divided into three regions: the near, mid, and far IR. and energy The mid IR region is of greatest practical use to the organic chemist.


• In order for IR absorbance to occur two conditions must be met: 1. If the frequency of the radiation matches the natural frequency of the vibration (or rotation), the IR photon is absorbed and the amplitude of the vibration increases.

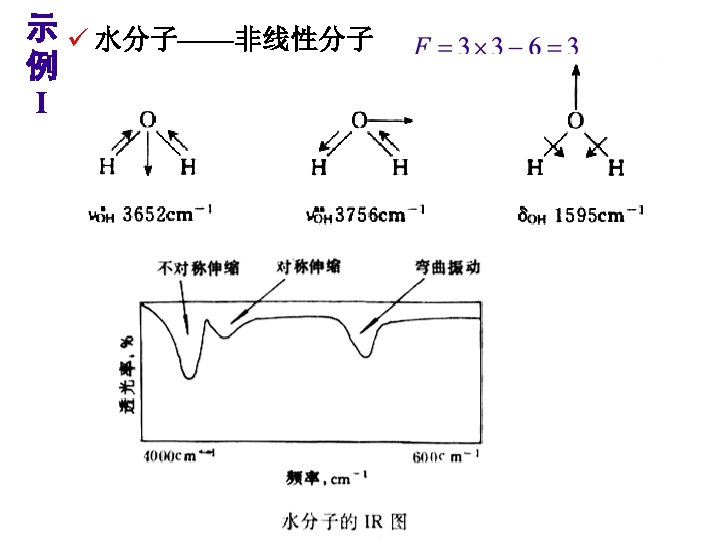
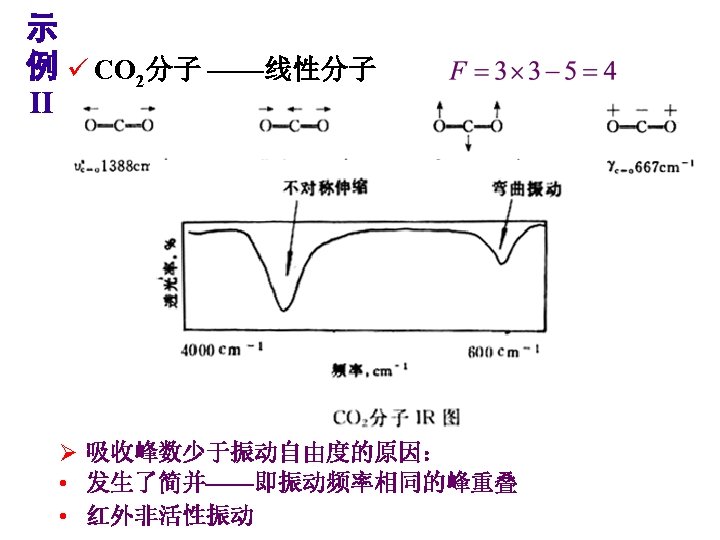
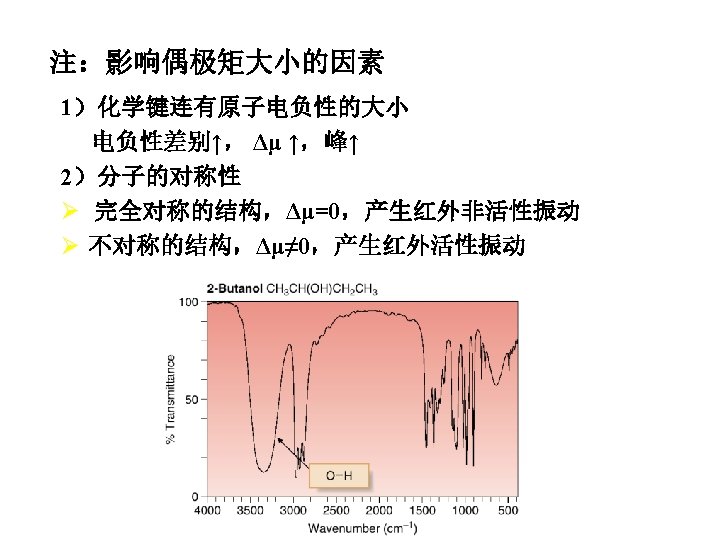
Theory of Infrared Absorption Spectroscopy 2. There must be a change in the dipole moment of the molecule as a result of a molecular vibration (or rotation). The change (or oscillation) in the dipole moment allows interaction with the alternating electrical component of the IR radiation wave. Symmetric molecules (or bonds) do not absorb IR radiation since there is no dipole moment.
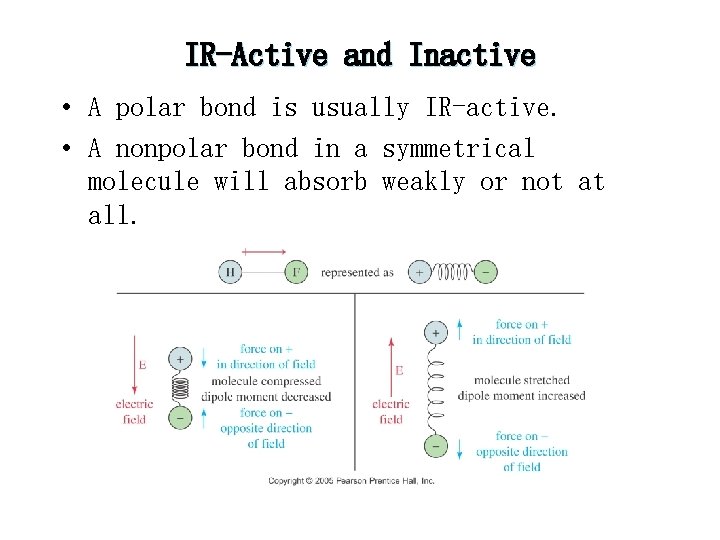
IR-Active and Inactive • A polar bond is usually IR-active. • A nonpolar bond in a symmetrical molecule will absorb weakly or not at all.

When the bond stretches, the increasing distance between the atoms increases the dipole moment. Therefore, the greater the dipole, the more intense the absorption. (i. e. , The greater the molar extinction coefficient ( ) in Beer’s law, A = bc.
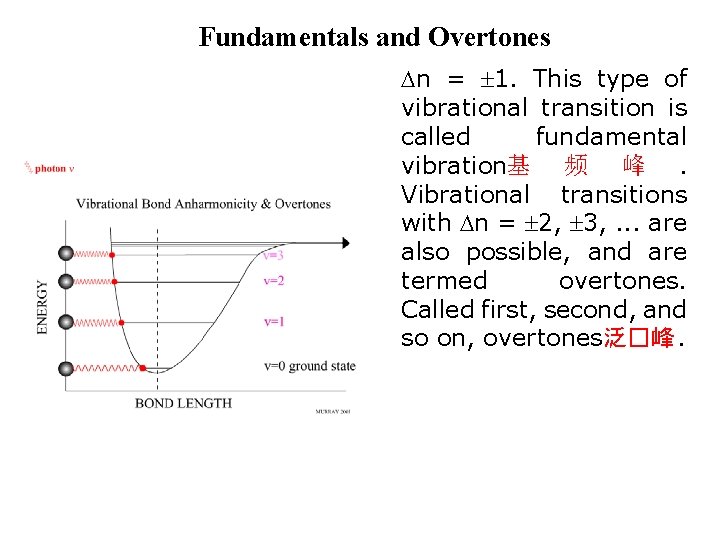
Fundamentals and Overtones n = 1. This type of vibrational transition is called fundamental vibration基 频 峰. Vibrational transitions with n = 2, 3, . . . are also possible, and are termed overtones. Called first, second, and so on, overtones泛�峰.
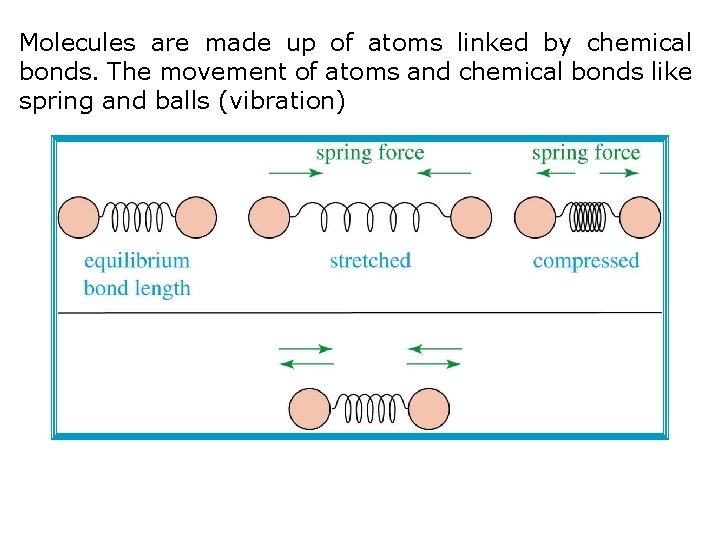
Molecules are made up of atoms linked by chemical bonds. The movement of atoms and chemical bonds like spring and balls (vibration)

Hooke’s Law ν = The vibration frequency (cm-1)振�� 率 c = Velocity of light (cm/s)光速 k= force constant of bond (dyne/cm)� 力常数 μ折合� 量,M 1 and M 2 are mass (g) of atom M 1 and M 2

§ The relative contributions of bond strength is also considered in vibrational frequencies. § In general functional groups that have a strong dipole give rise to strong absorptions in the IR.
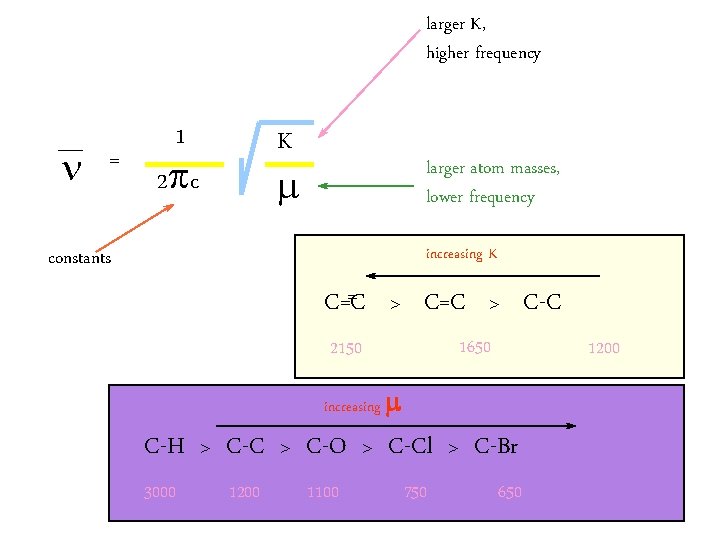
larger K, higher frequency n = 1 K 2 p c larger atom masses, lower frequency increasing K constants = > C=C > C-C C=C 1650 2150 1200 increasing C-H > C-C > C-O > C-Cl > C-Br 3000 1200 1100 750 650
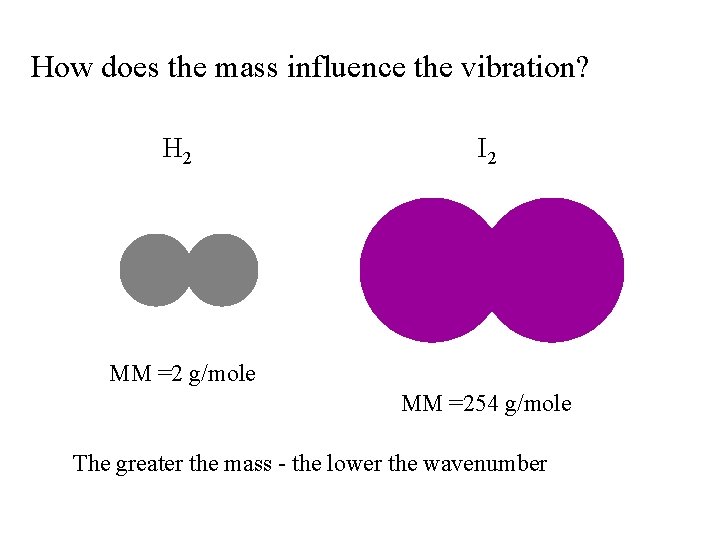
How does the mass influence the vibration? H 2 I 2 MM =2 g/mole MM =254 g/mole The greater the mass - the lower the wavenumber

Vibrations How a molecule vibrates? Any change in shape of the molecule- stretching of bonds, bending bonds, or internal rotation around single Thereofare two main vibrational modesbonds : 1. Stretching - change in bond length (higher frequency) Stretching vibration
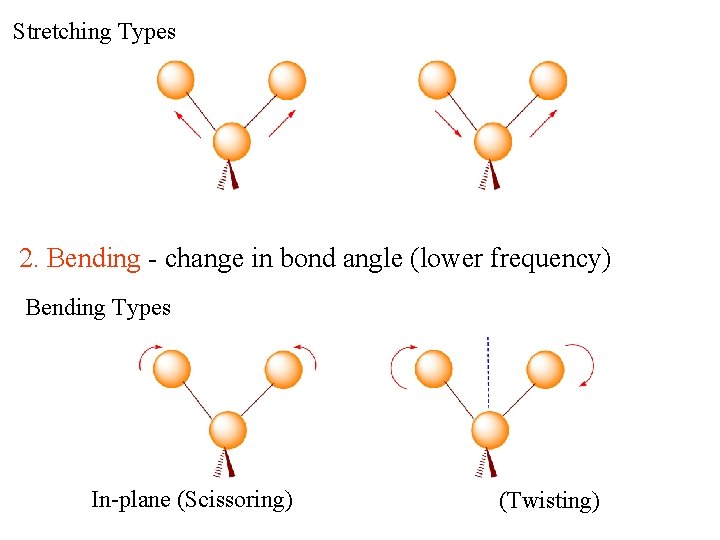
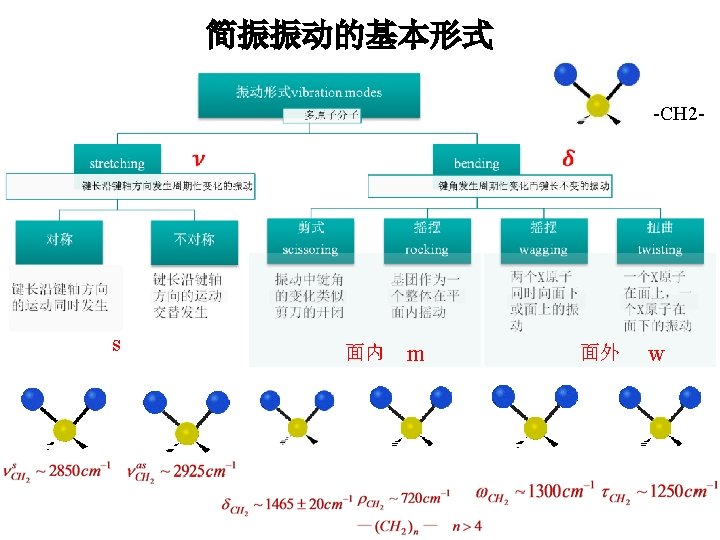
Stretching Types Symmetric Asymmetric 2. Bending - change in bond angle (lower frequency) Bending Types In-plane (Scissoring) Out-plane (Twisting)
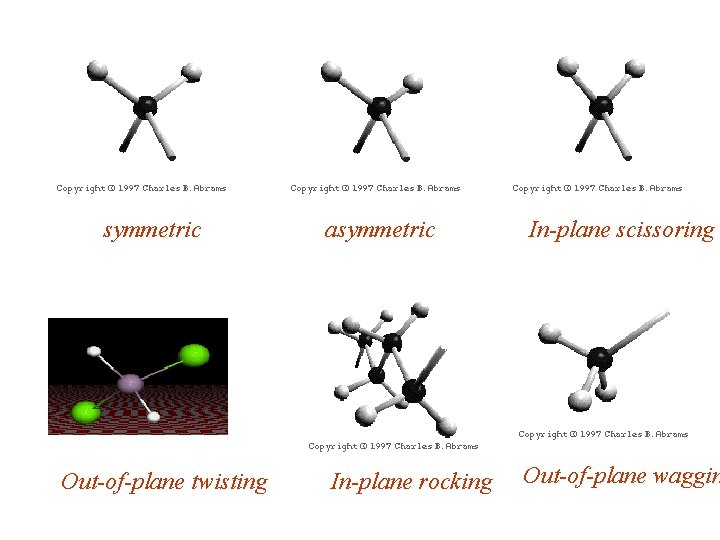
symmetric Out-of-plane twisting asymmetric In-plane scissoring In-plane rocking Out-of-plane waggin


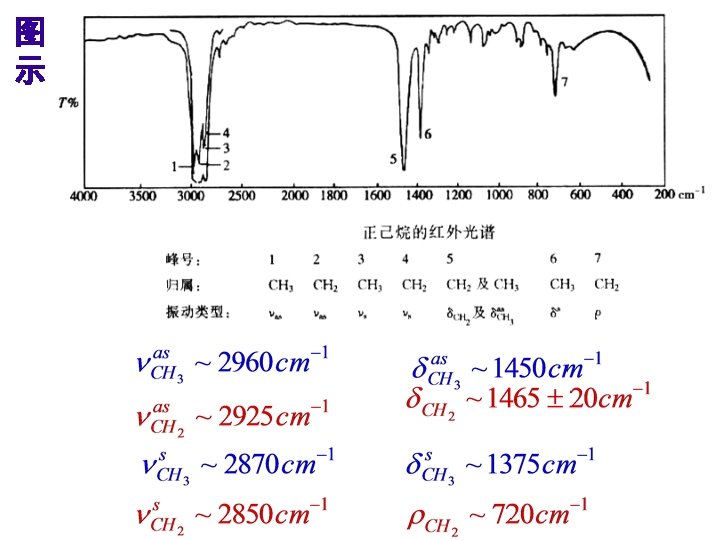


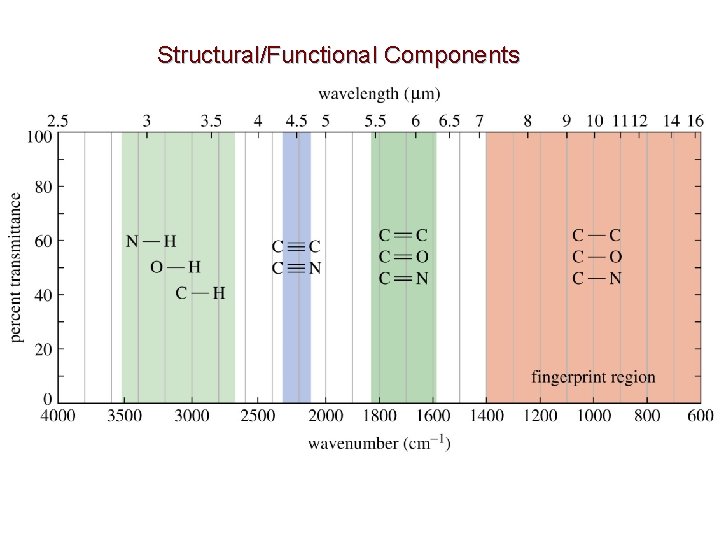
Structural/Functional Components
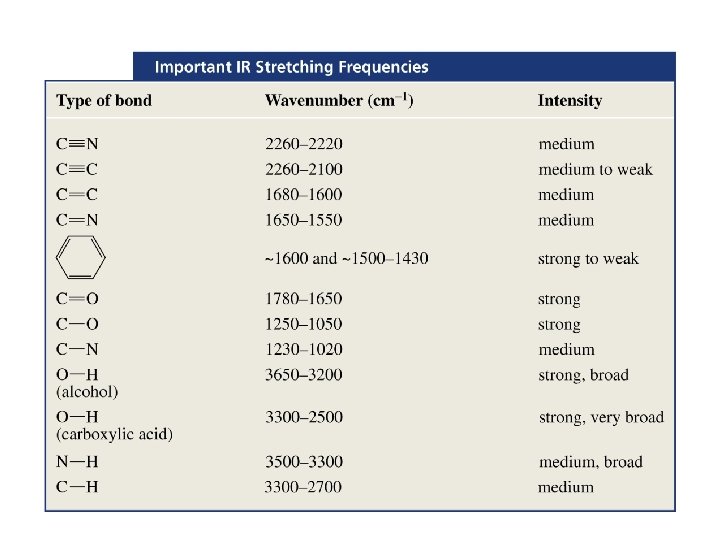
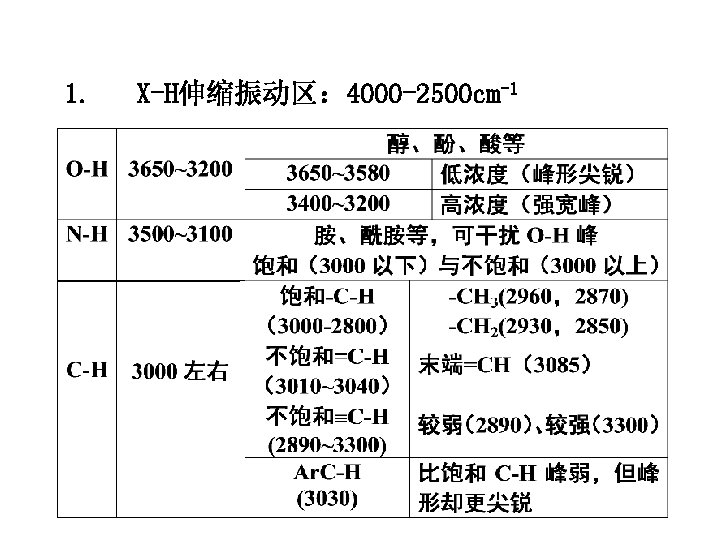
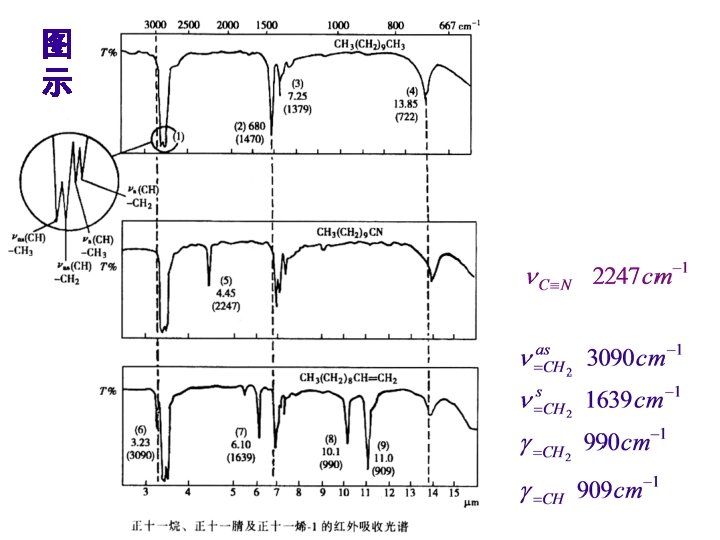
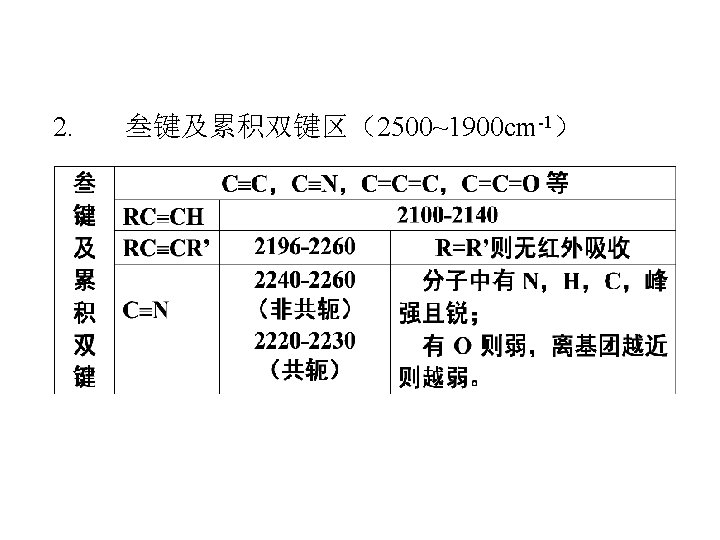
1. X-H伸缩振动区: 4000 -2500 cm-1






Instrumentation l. Dispersive Infrared spectrometer 色散型红外光谱仪 l. FT-Infrared spectrometer(FTIR) 傅里叶变换红外光谱仪
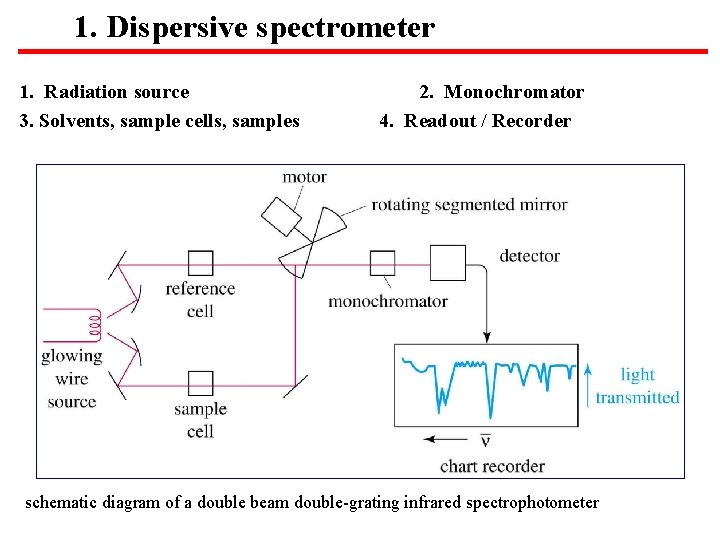
1. Dispersive spectrometer 1. Radiation source 3. Solvents, sample cells, samples 2. Monochromator 4. Readout / Recorder schematic diagram of a double beam double-grating infrared spectrophotometer
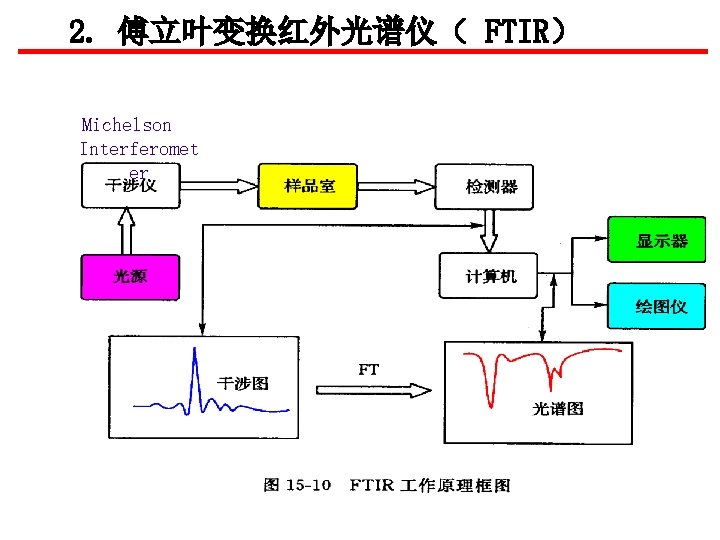
2. 傅立叶变换红外光谱仪( FTIR) Michelson Interferomet er
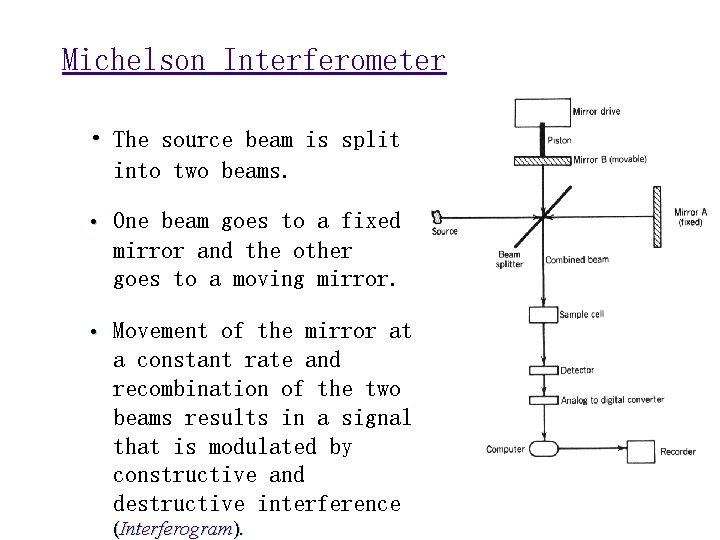
Michelson Interferometer • The source beam is split into two beams. • One beam goes to a fixed mirror and the other goes to a moving mirror. • Movement of the mirror at a constant rate and recombination of the two beams results in a signal that is modulated by constructive and destructive interference (Interferogram).
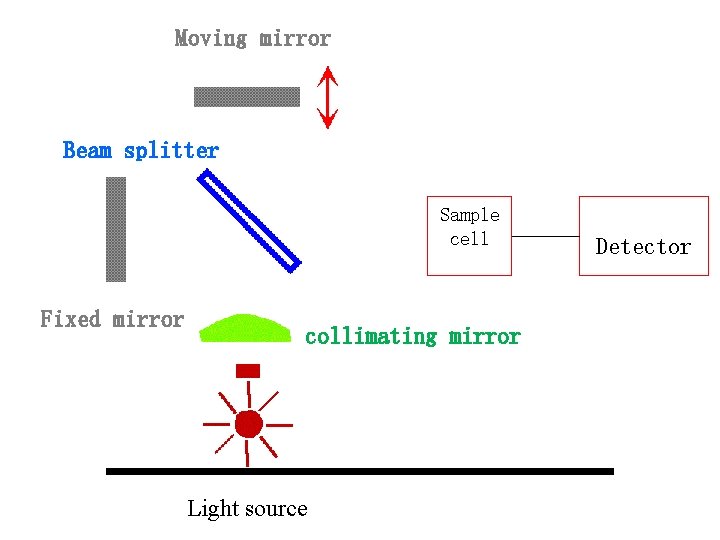
Moving mirror Beam splitter Sample cell Fixed mirror collimating mirror Light source Detector
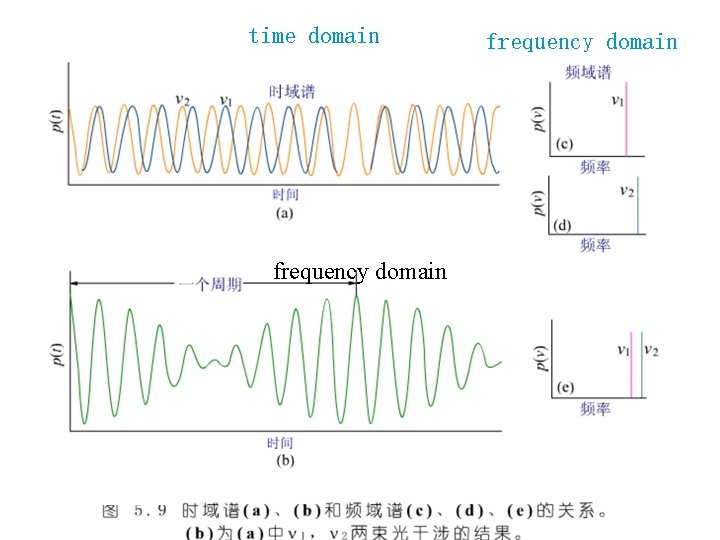
time domain frequency domain
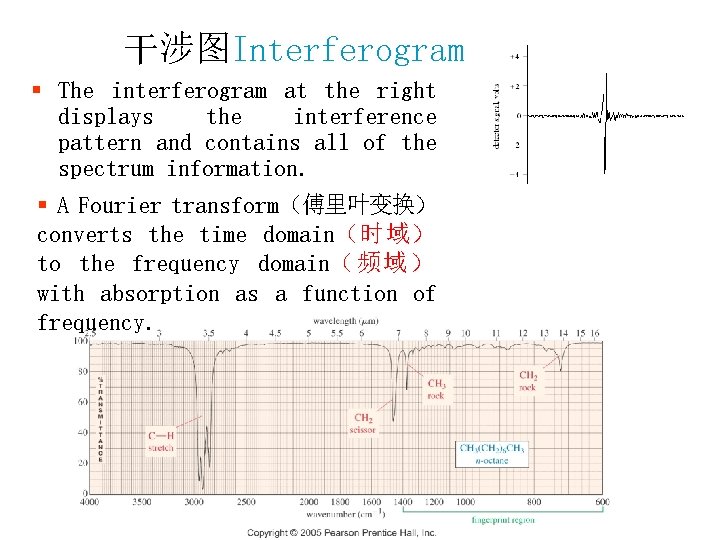
干涉图Interferogram § The interferogram at the right displays the interference pattern and contains all of the spectrum information. § A Fourier transform(傅里叶变换) converts the time domain(时域) to the frequency domain( 频 域 ) with absorption as a function of frequency.


Advantages of FT Spectrometers l S/Ns better than those of a dispersive instrument; 高信噪比 l High resolutions(<0. 1 cm-1); 分辨率高 l Accurate and reproducible frequency determinations; 波长(数)精度高 (± 0. 01 cm-1) 。重现性好 l Short response time; 扫描速度快

IR Sources and Detectors 红外光源与检测器 Sources光源 - inert solids that heat electrically to 1500 – 2200 K. • Emit blackbody radiation produced by atomic and molecular oscillations excited in the solid by thermal energy. • The inert solid “glows” when heated. • Common sources: Nernst glower(能斯特灯)、 Globar(硅碳棒)、Incandescent wire(白炽线圈)
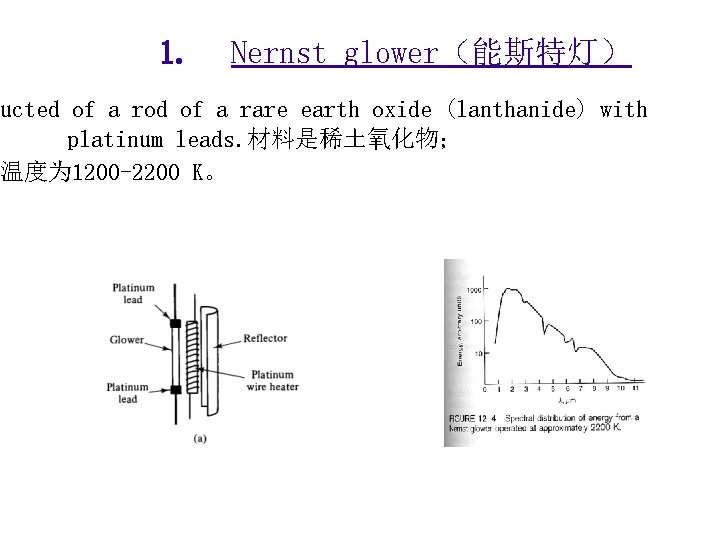
1. Nernst glower(能斯特灯) ucted of a rod of a rare earth oxide (lanthanide) with platinum leads. 材料是稀土氧化物; 温度为 1200 -2200 K。
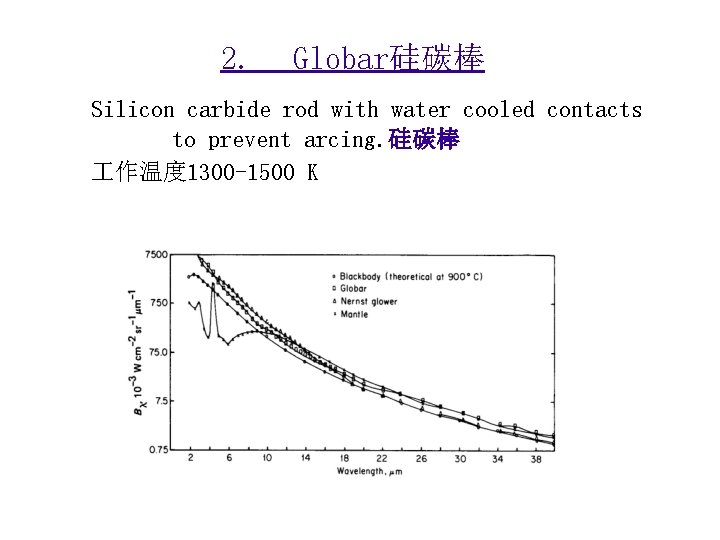
2. Globar硅碳棒 Silicon carbide rod with water cooled contacts to prevent arcing. 硅碳棒 作温度 1300 -1500 K


IR Transducers红外检测器 – measure minute changes in temperature. 1. Thermal transducer (热检测器) • the radiation is absorbed by a small blackbody and the resultant temperature rise is measured; • Have a slow response time, so they are not well suited to FT-IR; • Thermal noise from surrounding have to be minimized;

IR Transducers红外检测器 2. Pyroelectric transducer (热电检测器) • Constructed of crystalline wafers of triglycine sulfate (TGS) that have a strong temperature dependent polarization. 热电检测器使用具有特殊热 电性质的绝缘体,如硫酸三甘氨酸酯TGS。 • Have a fast response time and are well suited for FT-IR. 热电检测器的响应速率很快,可以跟踪干涉仪随 时间的变化,故多用于傅立叶变换红外光谱仪中。

3. Photoconducting transducer (光电导检测器) • Constructed of a semiconducting material (lead sulfide, mercury/cadmium telluride, or indium antimonide) deposited on a glass surface and sealed in an evacuated envelope to protect the semiconducting material from the environment. 光电导检测器采用半导体材料薄膜,如Hg-Cd-Te或Pb. S或In. Sb,将其 置于非导电的玻璃表面密闭于真空舱内。 • Absorption of radiation promotes nonconducting valence electrons to a conducting state, thus decreasing the resistance (W) of the semiconductor. 吸收辐射后非导电性的价 电子跃迁至高能量的导电带,从而降低半导体的电阻,产生信号。 • Fast response time, but require cooling by liquid N 2. 这种 检测器比热电检测器灵敏,在FT-IR及GC/FT-IR仪器中获得广泛应用。 需冷至液氮温度(77 K)以降低噪声


Solvents 1. Must be transparent in the region studied: no single solvent is transparent throughout the entire IR region 2. Water and alcohols are seldom employed to avoid O-H band of water. 3. Must be chemically inert (does not react with substance or cell holder). CCl 4, CS 2, or CHCl 3; may be used but we should consider its IR spectrum

Cells - Na. Cl or KCl cells may be used (moisture from air and sample should be avoided: even with care, their surfaces eventually become fogged due to absorption of moisture) - Very thin (path length = 0. 1 to 1. 0 mm) - Sample concentration = about 0. 1 – 10%

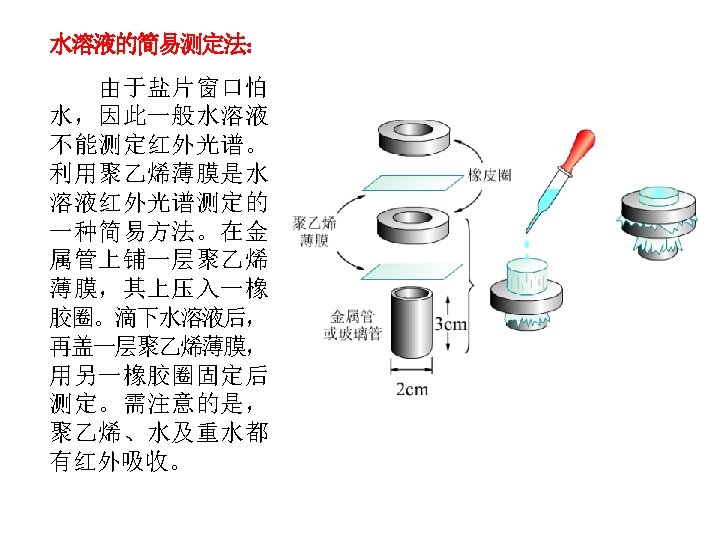


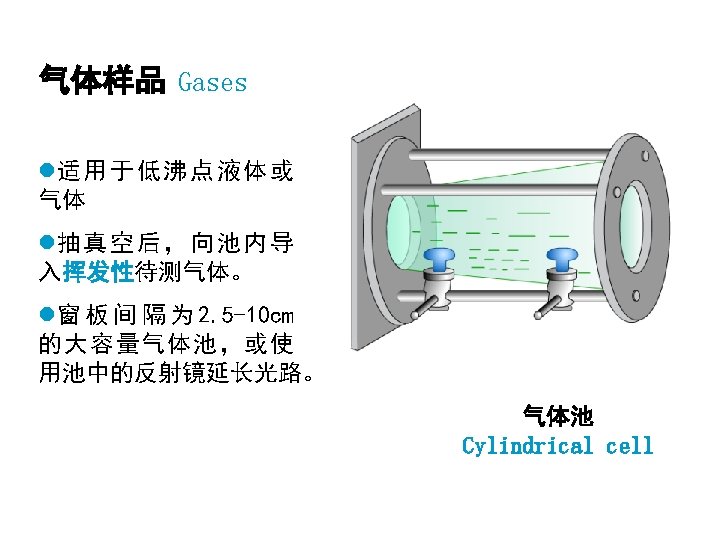

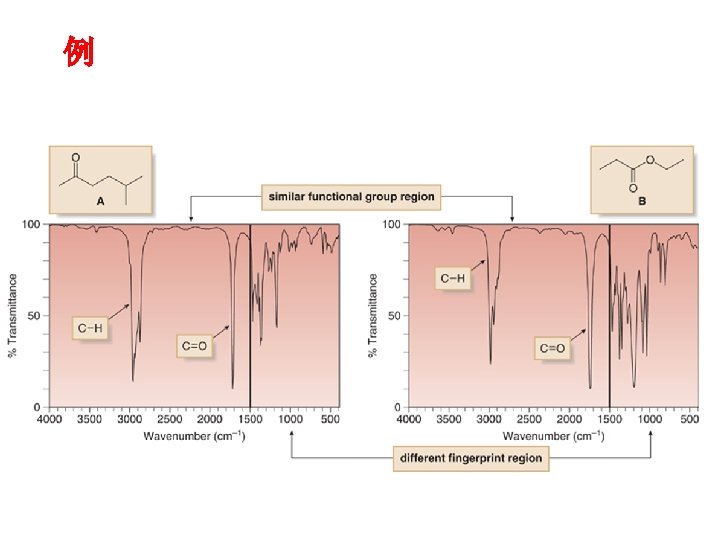
红外光谱的应用Application: Identify Organic compounds: 1. 有机化合物中官能团的鉴定,鉴定化合物结构;( group frequency);Identification of functional groups on a molecule ;Spectral matching can be done by computer software and library spectra 2. 检测纯度; Since absorbance follows Beer’s Law, can do quantitative analysis;

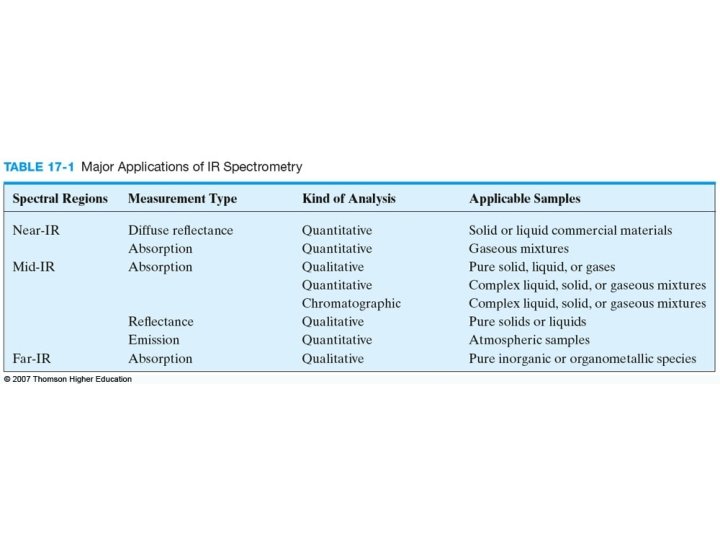
Infrared spectrometry is applied to the qualitative and quantitative determination of molecular species of all types. The most widely used region is the mid-infrared that extends from about 670 to 4000 cm-1 (2. 5 to 14. 9 m). The near-infrared region from 4000 to 14, 000 cm-1 (0. 75 to 2. 5 m) also finds considerable use for the routine quantitative determination. The far-infrared region has been for the determination of the structures of inorganic and metal-organic species.
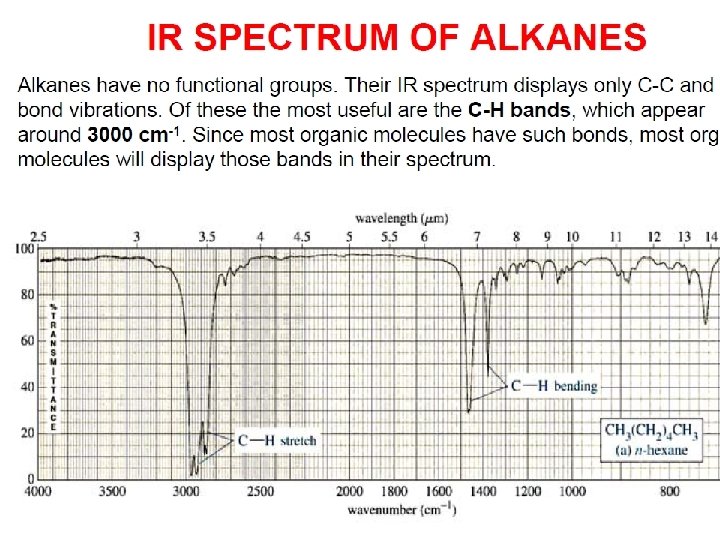
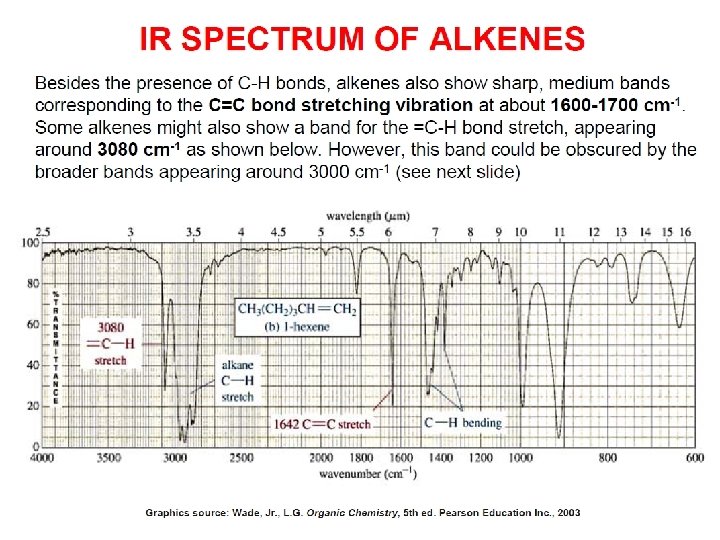
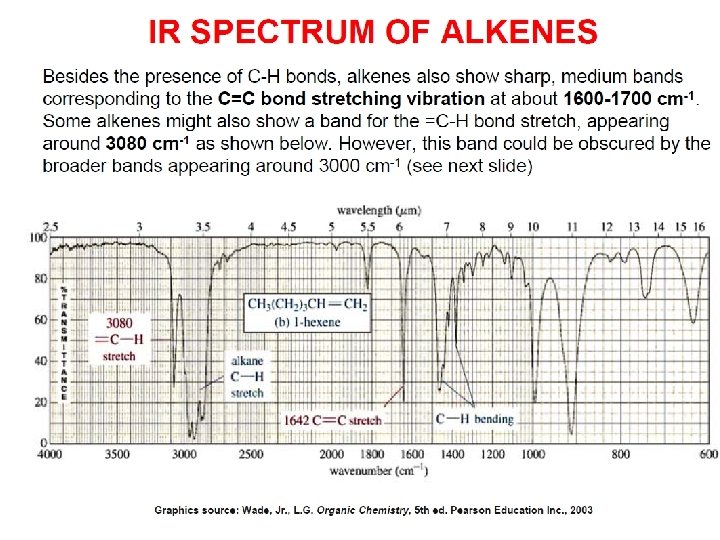
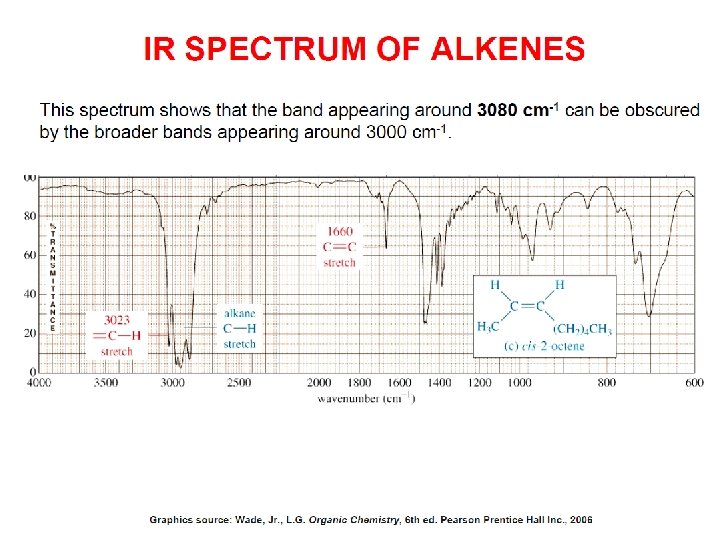

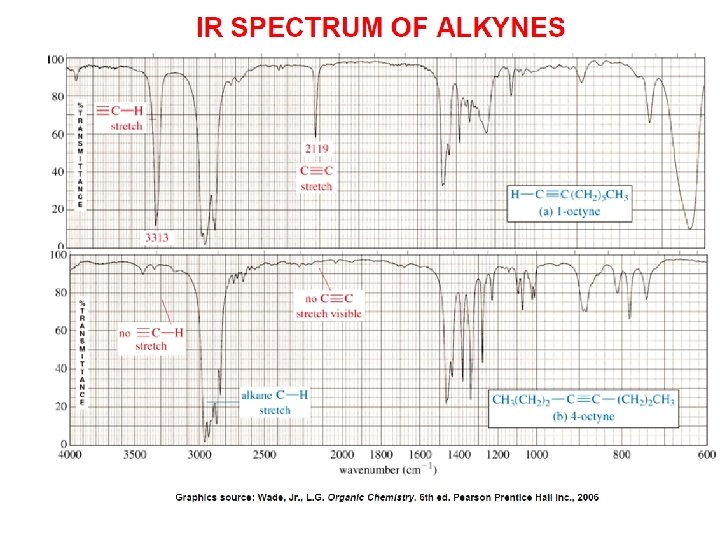
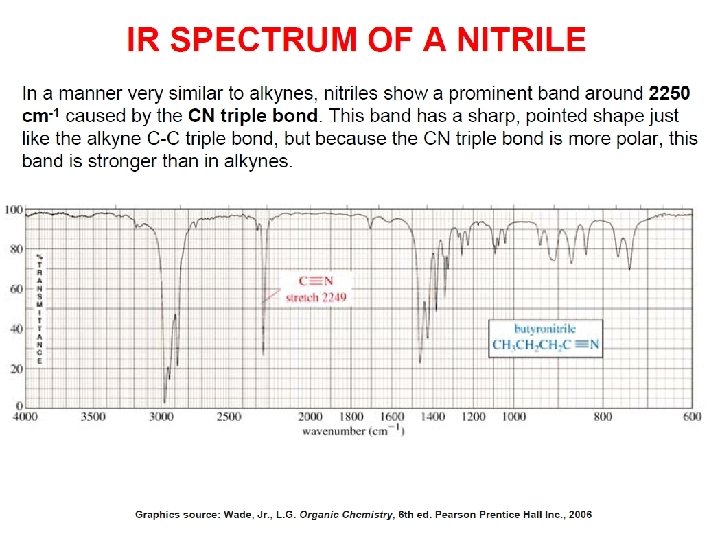
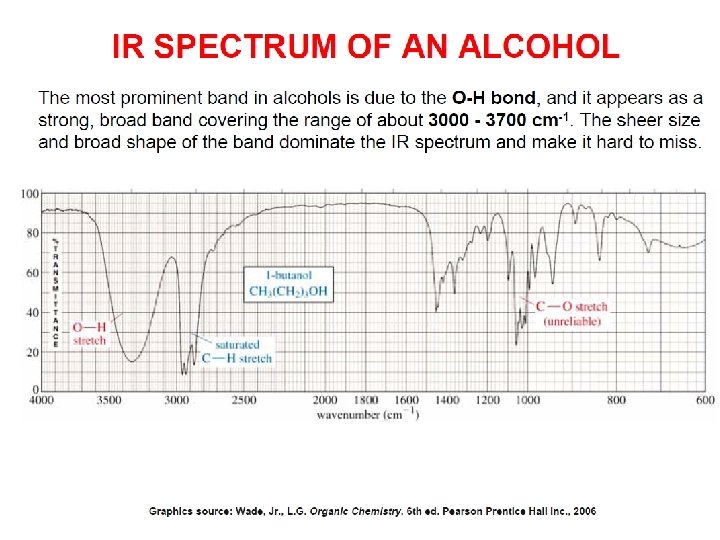

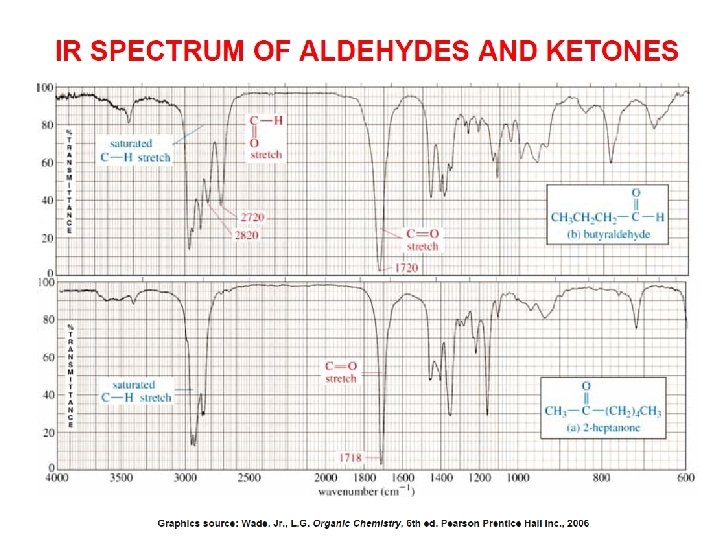

- Slides: 89