Experiment 15 Chemical Kinetics Purpose The purpose of






![Rate = k[A]x[B]y • • • [A], [B]: molarities of A and B in Rate = k[A]x[B]y • • • [A], [B]: molarities of A and B in](https://slidetodoc.com/presentation_image_h2/0ed5a91fc5b42fa19aaced1f6eb3d1de/image-7.jpg)


















- Slides: 26

Experiment 15 Chemical Kinetics

Purpose • The purpose of this experiment is to determine the rate of a chemical reaction (potassium permanganate, KMn. O 4, + oxalic acid, H 2 C 2 O 4) as the concentrations are varied and to determine the rate law for the reaction.

Introduction • For a reaction a. A+b. B c. C+d. D


Different Rates • Average (this experiment) • Initial (Experiment 17) • Instantaneous

Rate Law • Rate law---a relationship between concentrations and rate. For a reaction a. A + b. B products the rate law often takes the form Rate = k[A]x[B]y
![Rate kAxBy A B molarities of A and B in Rate = k[A]x[B]y • • • [A], [B]: molarities of A and B in](https://slidetodoc.com/presentation_image_h2/0ed5a91fc5b42fa19aaced1f6eb3d1de/image-7.jpg)
Rate = k[A]x[B]y • • • [A], [B]: molarities of A and B in solution x, y: orders with respect to A and B, respectively. (These orders might not correspond to coefficients from the balanced equation!) k: rate constant

What We’re Running • 2 Mn. O 4 - + 5 H 2 C 2 O 4 + 6 H+ 2 Mn 2+ + 10 CO 2 + 8 H 2 O • We assume a rate law Rate = k [Mn. O 4 -]m[H 2 C 2 O 4]n • The rate law and rate constant are not affected by concentration.

Strategy • • • One pair of experiments (assignments 1 and 2): [Mn. O 4 -] is constant and [H 2 C 2 O 4] doubles. In another pair (1 and 3), [Mn. O 4 -] doubles and [H 2 C 2 O 4] is constant. For each of these pairs, divide the rate measured in one experiment by that from another.

• Comparing assignments 1 and 2: • This can be rewritten as • Since rates and concentrations are known, n (the order with respect to oxalic acid) is available.

• Similarly, comparing assignments 1 and 3 gives • And, therefore, From this, m (the order with respect to Mn. O 4 -) is available.

• Once the orders are known, we can calculate the rate constant from the rate law. • Since rates depend on temperature. we will also look at the effect of temperature on the rate of this reaction.

Safety • Aprons and glasses. • KMn. O 4 is a strong oxidant (and also stains skin and clothing); oxalic acid is poisonous. • Waste into waste bottles.

Safety 2 • If you use the Bunsen burner for heating water, keep hair, clothing, paper, and other flammable material away. • Shut off burner before mixing hightemperature samples.

Procedure • Work in pairs. • Check out pipettes and bulbs from stockroom. • Needed equipment: medium-sized test tubes; 250 - and 400 -m. L beakers. May also need ring stand, ring, wire gauze, and Bunsen burner.

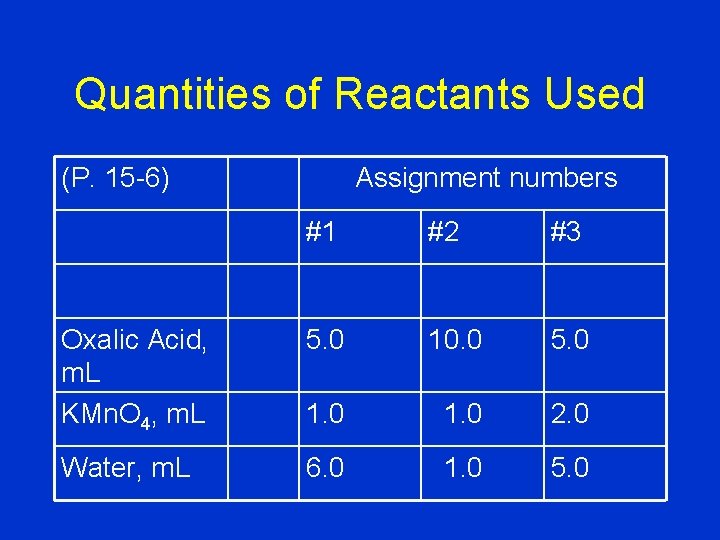
Quantities of Reactants Used Assignment numbers (P. 15 -6) #1 #2 #3 Oxalic Acid, m. L KMn. O 4, m. L 5. 0 10. 0 5. 0 1. 0 2. 0 Water, m. L 6. 0 1. 0 5. 0

• Mark an X on a piece of white paper. • Get 75 m. L oxalic acid and 15 m. L KMn. O 4 solutions; record concentrations. • Two tubes for each assignment; pipet desired volumes of oxalic acid and water into each tube. For use of volumetric pipets, review Expt. 11 from CHEM 1031.

• Prepare four additional tubes for Assignment #1; also pipet 1. 0 m. L of KMn. O 4 solution into each of four small test tubes. Save these for temperature study---last part of experiment. • Start with first oxalic acid-water tube for Assignment 1. Place the paper behind the test tube.

• Pipet KMn. O 4 solution into tube; begin timing when half the solution has been added. • When you can see the X through the test tube, record elapsed time on your data sheet. • Repeat the run you have just completed; then do duplicate runs for the other two assignments.

• Effect of temperature: Place two oxalic acid tubes and two KMn. O 4 tubes into a beaker containing warm water (10 o. C above room temperature). (If tap water is not sufficiently warm, use Bunsen burner. ) • After tubes have been in warm water for 5 minutes or so, add KMn. O 4 to an oxalic acid-water mix; record elapsed time for X to become visible. Repeat.

• Make cool water bath (ice in water) to get 10 o. C below room temperature. • Cool other two oxalic acid-water mixes and KMn. O 4 samples. Again mix, record elapsed time as before. Repeat.

Calculations • Concentrations of oxalic acid and KMn. O 4 from dilution formula: • Example: 5. 00 m. L of 0. 755 M oxalic acid diluted to 12. 00 m. L gives 0. 315 M.

• For each assignment, average the times for the two runs. • D[Mn. O 4 -] = [Mn. O 4 -]f – [Mn. O 4 -]i = -[Mn. O 4 -]i

• From measured rates, determine orders: • Round the orders to the nearest integers.

• Go back to the rate law: Rate = k [Mn. O 4 -]m[H 2 C 2 O 4]n • You now know rates, concentrations, and orders. Calculate k for each assignment and average.

The effect of temperature • Rate is proportional to DConcentration/Dtime • ---if the time decreases by a factor of 3 (say), the rate correspondingly increases by a factor of 3.