TOPIC 11 KINETICS AND EQUILIBRIUM Part 1 Kinetics

TOPIC 11: KINETICS AND EQUILIBRIUM Part 1 – Kinetics Part 2 – General Equilibrium

AIM#1 : How does Kinetics affect the rate of chemical reactions? Kinetics: the branch of chemistry that deals with the rates (how fast the reaction is) of chemical reactions Collision Theory: Is measure in the number of mole reactants used or the number of mole products formed. 1. In order for a reaction to occur, reactants must collide with each other

2. An effective collision is when reactants come together with the correct amount of energy and in the correct position to form a product As the amount of effective collisions increases, the faster products are formed

FACTORS THAT AFFECT THE RATE (speed) OF CHEMICAL REACTIONS 1. Nature of Reactants 2. Concentration 3. Surface Area 4. Pressure 5. Temperature 6. Adding a Catalyst

1. NATURE OF REACTANTS: the types of substances you are reacting. This can involve the breaking of old bonds and the formation of new bonds. 1. Covalent: 2. Ionic: 3. Breaking bonds requires more energy than making bonds during collisions

2. CONCENTRATION: the amount of particles

3. SURFACE AREA: more sides are exposed for collisions

4. PRESSURE: effects gases only!

5. TEMPERATURE: average kinetic energy

6. CATALYST

AIM # 2: HOW CAN WE INTERPRET POTENTIAL ENERGY DIAGRAMS? Potential energy diagrams are graphs that depict heat transfers during the course of a reaction. These diagrams show the potential energy of reactant particles changes to chemical potential energy stored in bonds


The forward reaction is read from left to right reactants to products (PE diagram #1) For Example: Activation Energy: A+B AB

Activated Complex: Reverse reaction- reading the PE diagram from right to left (products to reactant) A + B AB

Endothermic- Energy is absorbed (PE diagram # 1, page 10) Sketch diagram! Always show energy being absorbed. Products are higher than reactants. Δ H is always positive

Exothermic- energy is released Sketch diagram Always show energy being released. Product side is lower than reactant side. Δ H is always negative

How do catalysts shorten the reaction time needed for the reaction to occur?
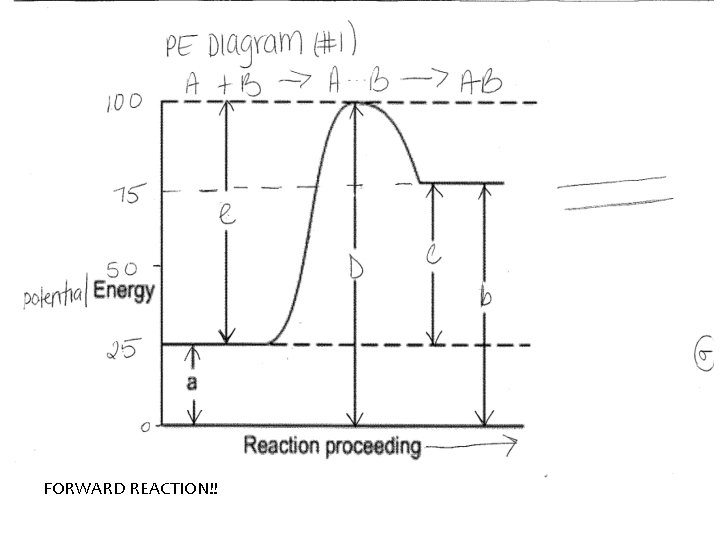
AIM: HOW CAN WE INTERPRET POTENTIAL ENERGY DIAGRAMS? FORWARD REACTION!!

Lets Label the Diagram A. PE of Reactants B. PE of Products C. H (heat of reaction) = H = PE products – PE reactants If H is positive [PE products PE reactants] – endothermic If H is negative [PE products PE reactants] – exothermic Table I – shows different chemical reactions and H for each one

Label the Diagram D. PE of Activated Complex – intermediate molecule that forms when reactants have an effective collision. It is unstable and temporary E. Activation Energy of forward reaction – amount of energy needed to start the reaction in the forward direction F. Activation Energy (with catalyst) – amount of energy needed to start the reaction if a catalyst is added * Catalysts – speed the reaction rate by lowering the activation energy needed to start a reactions (gives an alternate pathway)
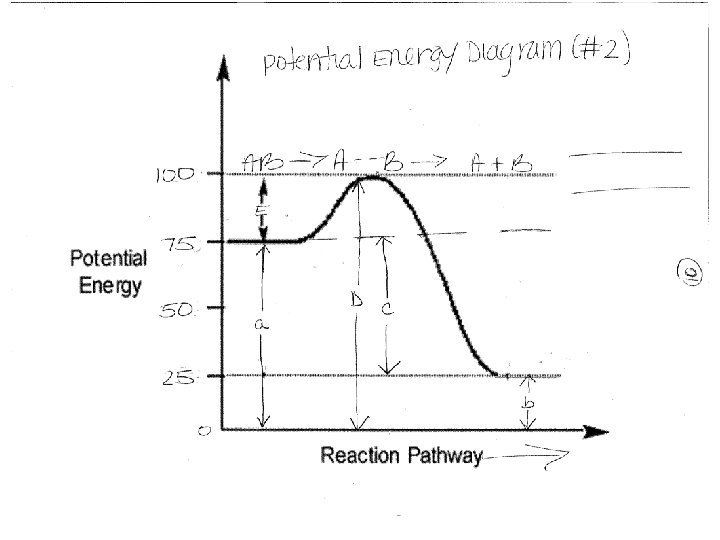

AIM #3: HOW CAN WE CLASSIFY ENERGY IN CHEMICAL REACTIONS? Enthalpy (H) – flow of energy (heat exchange) at constant pressure when two systems are in contact. Also known as heat of the reaction H = PE products – PE reactants

What does it really tell you about the changes in energy regarding a chemical reaction? Δ H = + reaction is endothermic and heat energy is added into the system Δ H = - reaction is exothermic and heat energy is lost from the system

Using Table I Heats of reaction Endothermic- energy is located in the reactant and is a + value Exothermic- energy is located in the product and is a value.

Look at the reactions (where is the energy located? ) CH 4 + 2 O 2 CO 2 + 2 H 2 O + 890. 4 k. J N 2 + O 2 + 66. 4 k. J 2 NO 2
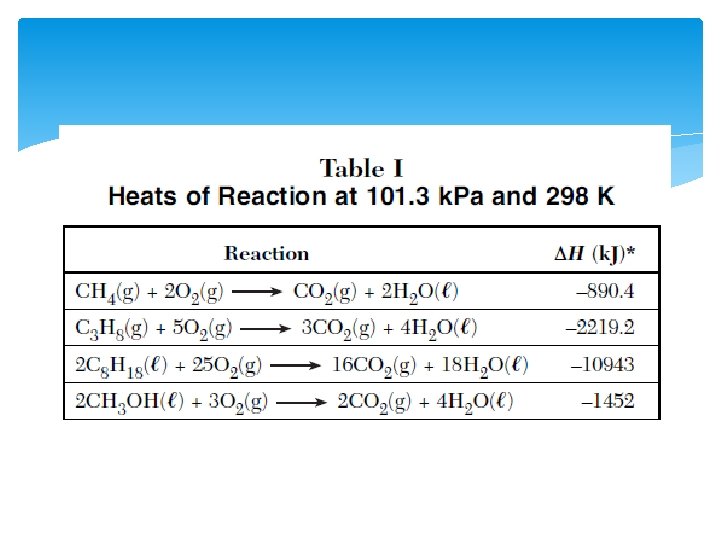

Practice using Table I 1. Write the reaction that gives off the most energy 2. Write the reaction that gives off the least energy 3. Write the reaction that requires the most energy to occur Whatever you do to a chemical reaction, you must also do to the heat of reaction. 1. if you reverse the reaction, you must reverse the sign 2. if you double the equation (coefficients) then you MUST double the change in H.

ENTROPY ΔS: disorder or randomness of the matter and energy of a system (more disordered/dispersal is favored) Gases have high entropy, solids have low entropy. Nature favors CHAOS (high entropy low energy) - Δ H + Δ S the more exothermic, the more stable

• These are spontaneous or thermodynamically favored • Increasing entropy is easy! • Phase changes s -> l -> g • Mixing gases • Dissolving a substance • If moles increase, so will the entropy

Predict which has the largest increase in entropy: CO 2(s) CO 2(g) H 2(g) + Cl 2(g) 2 HCl(g) KNO 3(s) KNO 3(l) C(diamond) C(graphite)

AIM # 4: What is chemical equilibrium? Equilibrium: when the forward and reverse reactions occur at the same rate – it is a state of balance Physical Equilibrium: the changes that take place in chemical reactions during physical processes such as changes of state or dissolving. 1. Phase Equilibrium: Equilibrium between phases Solid and liquid phase – during melting the rate of dissolving is equal to the rate of crystallization in a closed container (system) (H 20 (s) H 2 O (l))

EQUILIBRIUM 2. Liquid and Gas phase – during this phase the rate of evaporation is equal to the rate of condensation in a closed container (H 20 (l) H 2 O (g))

2. Solution Equilibrium: the rate of dissolving = the rate of crystallization only in saturated solutions 3. Chemical Equilibrium as time progress in a reaction, the concentration of the reactants can decrease causing the overall reaction to slow. As this occurs the concentration of the products can increase causing the reaction to reverse (indicated by a double arrow) ** Concentrations are constant**

Le Chateliers Principle If a stress is applied to a system at equilibrium the reaction will shift to release the effects of the stress Stressors: 1. Temperature 2. Concentration 3. Pressure

TEMPERATURE If you increase temperature, ALL reactions will speed up. The endothermic reaction will speed up the most (away from where the heat is) Increase in temperature favors the endothermic reaction Decrease in temperature will favor the exothermic reaction • *The side you shift towards will increase and the side you shift away from will decrease** • Note: Heat/ Energy/ Joules will either be in the reactant or product
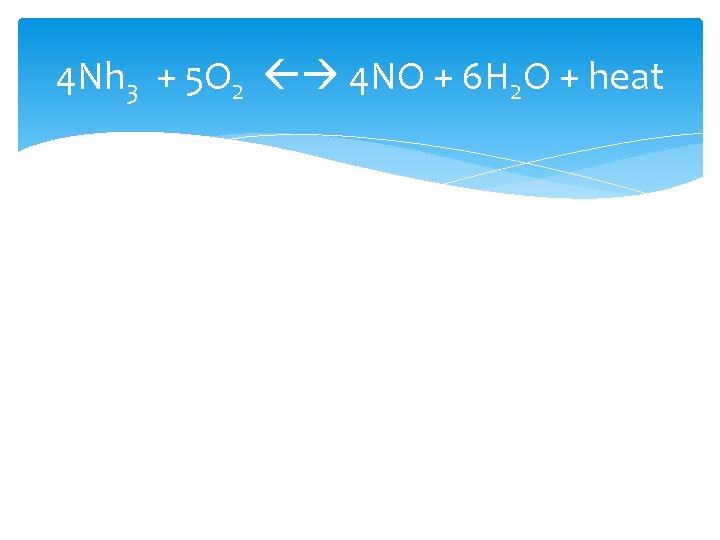
4 Nh 3 + 5 O 2 4 NO + 6 H 2 O + heat

CONCENTRATION If you increase the concentration of a substance the reaction will shift away from the substance 4 Nh 3 + 5 O 2 4 NO + 6 H 2 O + heat

Le Chateliers Principle Increase conc. of reactants Favors FORWARD reaction Speeds up the forward direction SHIFTS RIGHT Decrease conc. of reactants Favors REVERSE reaction Speeds up the REVERSE direction SHIFTS LEFT Increase conc. of products Favors REVERSE reactions Speeds up the REVERSE direction SHIFTS LEFT Decrease conc. of the products SHIFTS RIGHT Favors FORWARD reaction Speeds up the FORWARD direction

CONCENTRATION ADD AWAY (increase) TAKE TOWARDS (decrease)

PRESSURE Only affects gases!! Need to know how many gas molecules are on the reactant side and on the product side Equal # of gas molecules – pressure will have no effect!! Increase pressure – shift from more gas molecules towards less gas molecules Decrease pressure – shift from less toward more

PRESSURE 4 NH (g) + 5 O 2(g) 4 + 5 = 9 gas molecules 4 NO(g) + 6 H 2 O(g) 4+6 = 10 gas molecules Increase pressure: shift to the left (more to less) 9 Decrease pressure: shift to the right (less to more) 9 10 10

What are the conditions at equilibrium? At equilibrium the rate of the forward reaction is equal to the rate of the reverse reaction Concentrations are constant not equal Adding a catalyst would speed up the forward and reverse reactions to the same extent
- Slides: 42